Abstract
Background:
Diabetes technology is a fundamental part of modern diabetes therapy. Its widespread usage is associated with an increasing amount of “diabetes technology waste.” The aim of this study was to quantify this waste in a real-world situation in a specialized diabetes practice in Germany.
Methods:
Eighty patients with diabetes and insulin treatment participated and collected all of their therapy-associated waste for three months. Their attitude toward sustainability of antidiabetic therapy, waste generation, and their own waste reduction/separation behavior was surveyed.
Results:
In total, 23 707 pieces of therapy-associated waste were collected. They comprised 5362 test strips, 630 glucose sensors, 14 619 needles, 519 insulin cartridges, 599 pens, and 1463 pieces of aids for insulin pump therapy. Type and quantity of the collected waste depended on the type of diabetes and the respective therapy, ie, multiple daily injections, usage of glucose sensors, or pump therapy. Most participants (92%) were surprised by the amounts of waste and reported an increased awareness toward the resource consumption of their therapy (87%). The survey indicated an enhanced interest in waste separation (94%) and a demand for the reduction and recycling of devices/aids (93%).
Conclusions:
Our data revealed the amount and complexity of the waste generated by modern diabetes therapy. Extrapolating these data, it can be estimated that around 1.2 billion pieces of diabetes technology waste are generated in Germany per year. The major concern of the study participants was the limited number of recycling options. A clear demand for improved sustainability of the medical products was expressed.
Introduction
Massive environmental challenges around the globe push for sustainability also in the medical field.1,2 Diabetes mellitus is one of the most common chronic medical conditions globally. 3 As technological options for the treatment of people with diabetes (PwD) continue to develop, they are increasingly using medical products in their care: with >400 million PwD worldwide, the amount of waste generated using diabetes devices and their packaging each day is considerable.4-7 However, our knowledge about the amount of waste generated by PwD during daily diabetes therapy remains fairly unknown. A single product may contain many different components, each constructed from a different material requiring thought and effort to disassemble (if possible, at all) and sort into the appropriate recycling bins. As a result, diabetes devices are normally left assembled and thrown away with the general household waste.
As sustainability concerns are growing in the face of limited resources and the climate crisis, which have a reciprocal impact on each other, 8 the environmental impact of diabetes technology and the aim for sustainable products were increasingly addressed in recent years, 4 shaping the term “Green Diabetes”. 6
There is a clear need for further research aiming to reduce the environmental impact of antidiabetic therapies to ensure state-of-the-art medical care for PwD while reducing the environmental impact associated at the same time. Although this is more related toward the manufacturer of such products, an option for PwD is appropriate recycling of these aids. The manufacturer can support this by guiding and support PwD when it comes to the disposal of these products, by, eg, labeling the different parts in a way that eases waste separation.
However, there are little, if any, data on the real-world amount of waste produced as a by-product of antidiabetic therapy and the attitude of PwD toward the sustainability of their daily diabetes management. Therefore, our study aimed to assess quantitatively how much waste PwD on insulin therapy are disposing of various products and devices and at what rates at an individual level in a single specialized diabetes practice in a mid-size town in Germany. In addition, the awareness of PwD for this topic was evaluated.
Methods
Participants and Study Procedure
Eighty patients regularly treated in a specialized diabetes practice in a mid-size town volunteered to take part in the study. Patients were included if their antidiabetic therapy comprised multiple daily insulin injections (MDIs) or if they used an insulin pump for continuous subcutaneous insulin infusion (CSII) therapy solely or as part of an automated insulin delivery (AID) system. There were no further formal inclusion or exclusion criteria. Patients were enrolled if they were willing and able to collect and record their diabetes technology-related waste for three months. They were asked to collect every single piece of this waste between two routine visits.
Ethics
The study was approved by the local Ethics Committee of the Faculty of Medicine, Justus Liebig University, Giessen, Germany, without formal review as patient data were processed anonymized and there was no potential study-related harm.
Study Design
At baseline, the patients’ diabetes type, treatment, age, body weight, body height, body mass index (BMI), years since diabetes diagnosis, antidiabetic therapy, and HbA1c were determined as part of their routine visit. All volunteers collected their diabetes technology-related waste for three months until the next routine visit to the practice.
All participants received written instructions on which type of waste to collect and how. The pieces of waste considered are listed in Supplemental Table S1. Inserter for glucose sensors of continuous glucose monitoring (CGM) systems were not collected; however, as all systems relied on single-use devices, a count equal to the employed sensors was assumed.
Study participants answered a survey about their attitude toward the sustainability of antidiabetic therapy, waste generation, and their own waste reduction/separation behavior at the beginning and the end of the study (Supplemental Table S2).
Measurements
HbA1c was measured employing an immunoturbidimetric assay (Bioscientia, Ingelheim, Germany). People with diabetes collected their therapy-associated waste in envelopes, paper bags, glass jars, or original packaging. Interestingly, the majority of patients delivered their collected waste carefully sorted in cardboard boxes; however, some used bags (Figure 1a-c). In addition to the patient’s records, every single piece of waste collected was counted to obtain the exact number. All waste collected was thoroughly checked, sorted, and counted for this purpose, ie, collection containers were emptied, and even single needles and glucose test strips were checked carefully (Figure 1d). This procedure was conducted by two people of the study team with one counting and the other taking notes.

Examples of waste deliveries. Representative images of (a-c) different methods of packaging and (d) an example of waste items after counting. Needles were collected by the patients in glass jars, other pieces of waste in envelopes, paper bags, or original packaging. Most patients delivered their collected waste carefully sorted in cardboard boxes, others in bags. Each image shows the collected waste of a single patient.
Statistics
The statistical analysis was performed using GraphPad Prism 9 (GraphPad Software, San Diego, California). Data were tested for normal distribution by the Shapiro-Wilk test and analyzed by Student’s t-test, Mann-Whitney test, one-way analysis of variance (ANOVA), or Kruskal-Wallis test, as appropriate. Data are given as means (standard deviation [SD]) unless otherwise stated. A P-value of <.05 was considered significant.
Results
People with Diabetes Characteristics and Subgroups
Eighty PwD took part in the study. 67% (n = 31) of the PwD with type 1 diabetes (T1D) were treated with MDI, and 33% (n = 15) used an insulin pump alone or as part of an AID system. All PwD with type 2 diabetes (T2D) used MDI. The HbA1c at baseline and the end of the study did not differ( 6.9 [0.9]% vs 6.7 [0.7]%; P = .1). The participants were separated into subgroups based on the type of diabetes and their antidiabetic therapy, ie, T1D MDI, T1D CSII/AID, or T2D MDI (Table 1). People with diabetes with T2D were older, had a higher BMI, and were diagnosed at an older age.
Patient Characteristics and Subgroups.
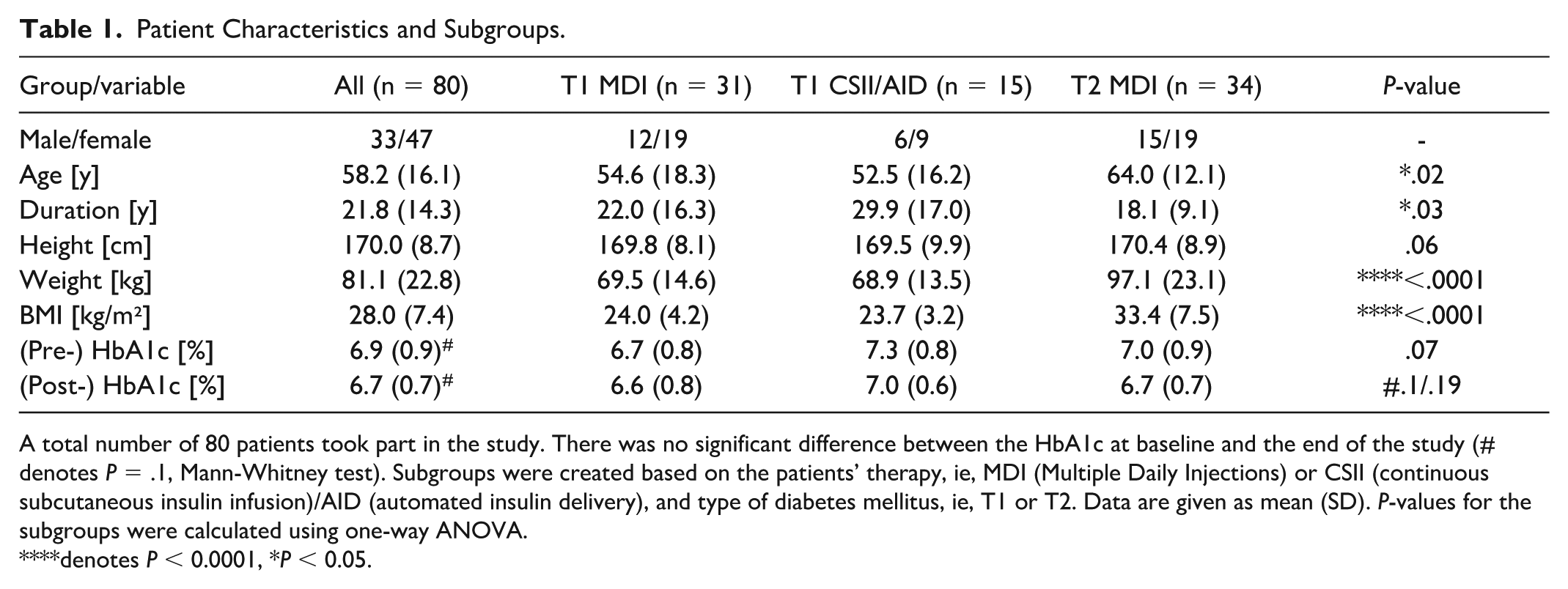
A total number of 80 patients took part in the study. There was no significant difference between the HbA1c at baseline and the end of the study (# denotes P = .1, Mann-Whitney test). Subgroups were created based on the patients’ therapy, ie, MDI (Multiple Daily Injections) or CSII (continuous subcutaneous insulin infusion)/AID (automated insulin delivery), and type of diabetes mellitus, ie, T1 or T2. Data are given as mean (SD). P-values for the subgroups were calculated using one-way ANOVA.
denotes P < 0.0001, *P < 0.05.
Diabetes Technology Waste
In total, 23 707 pieces of waste were collected throughout the study period (T1D MDI: 9569, T2D MDI: 11 196, T1D CSII/AID 2942). These figures correspond to an average of 309 pieces of waste for T1D MDI, 329 for T2 MDI, and 209 for T1D CSII/AID over three months and a mean daily disposal of 263 items. This mean count was significantly different between the three subgroups (**P = .0085) with T2D MDI patients having generated the highest amount of waste items and T1D CSII/AID the lowest (T1D MDI: 9569, T2D MDI: 11 196, T1D CSII/AID: 2942 pieces). More than half of the collected waste were needles (14 619 in total) and blood glucose test strips (5362 in total).
People with diabetes with T1D used significantly more CGM sensors than those with T2D (T1D MDI: 8 ± 3 vs T2D MDI: 6 ± 3 vs T1D CSII/AID: 13 ± 6, ****P < .0001). People with diabetes with T1D CSII/AID required more batteries than PwD and T1D or T2D with MDI, but almost no needles (both ****P < .0001). Prefilled insulin pens were mostly used by PwD and T2D on MDI (396 vs 62 pens), whereas insulin cartridges were used by both groups of PwD on MDI. Only one reusable pen was disposed of. An overview of all collected pieces of waste per patient is given in Figure 2. The disposed items per subgroup are displayed in Supplemental Figure S1, and detailed data are given in Supplemental Table S3.
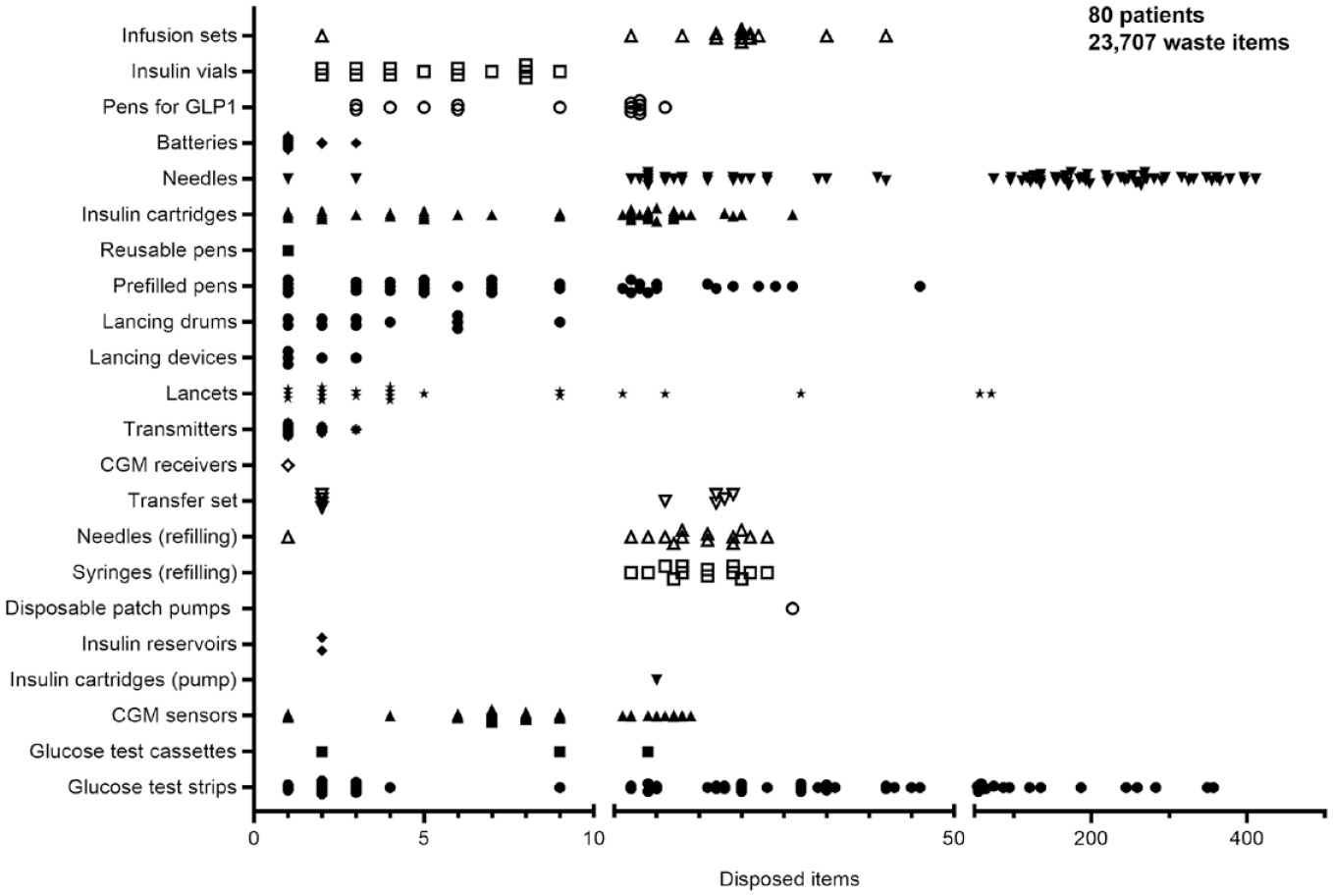
Overview of all collected pieces of therapy-related waste per patient. The 80 patients enrolled collected a total of 23 707 pieces of waste. Each symbol indicates the disposed items per category of waste for a single patient. Symbols for zero collected items are not displayed. Detailed data are given in Supplemental Table S3.
Survey Results
A total of 78 (98%) questionnaires were completed. At baseline, 28% of respondents stated that the waste generated by their therapy is or rather is an issue for them in contrast to 55% who said that this statement is false or rather false. After the waste collection, 81% of patients stated that the waste is or rather is an issue for them, and only 16% said that it is or rather is not. This was similar to the personal concern with the generated waste: at baseline, 35% of participants replied that they are or rather are concerned. The percentage increased to 92% after the study. Interestingly, the percentage of patients who stated that they separate the generated waste rose from 32% to 94%, and patients who reported willingness to contribute to waste reduction increased from 69% to 93%. Furthermore, the number of participants who said to dispose of their glucose sensors in collection sites for batteries grew by 71% (13% at baseline, 84% after the study). Data are displayed in Figure 3 and Supplemental Table S4.
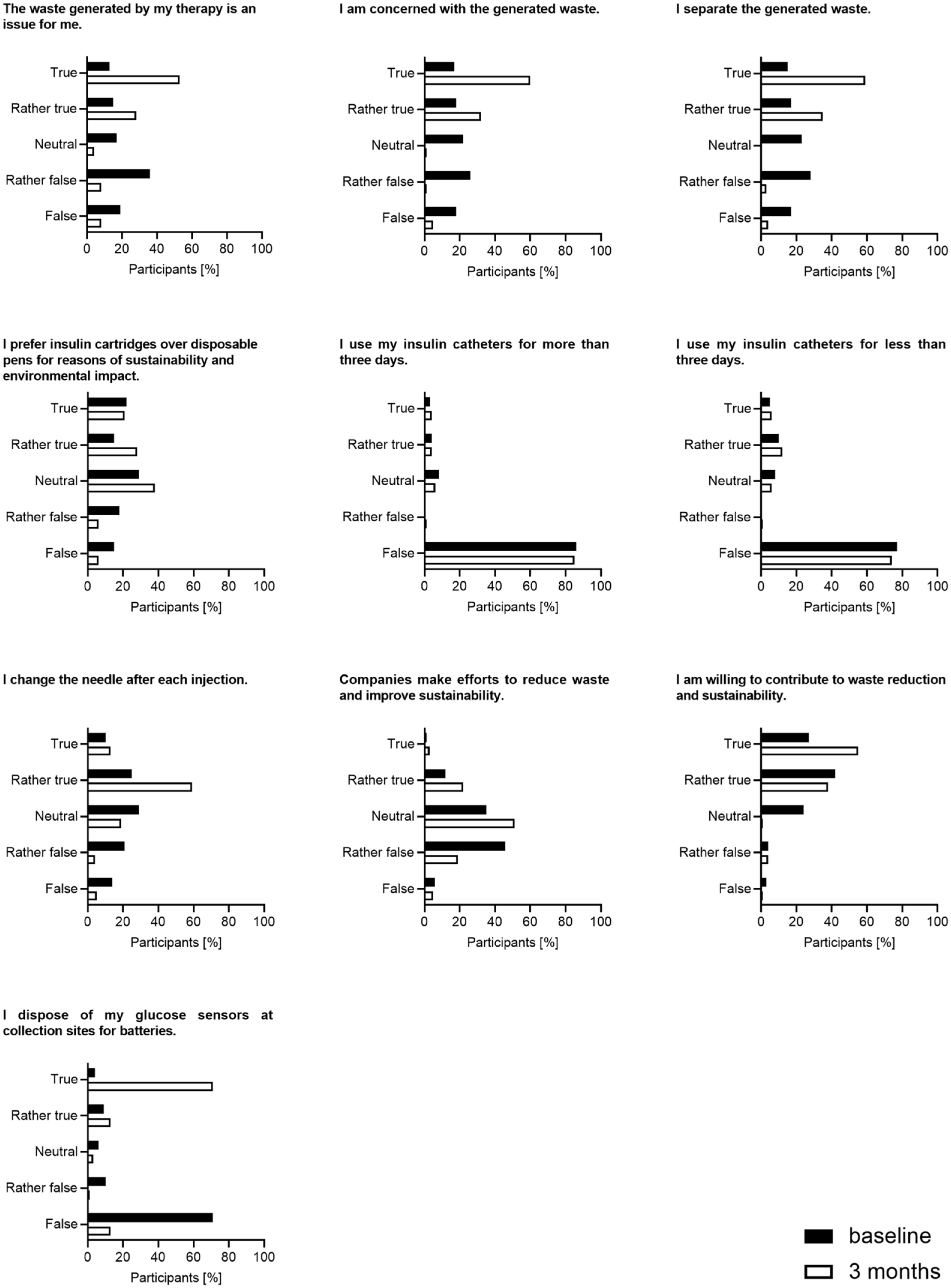
Results of survey set 1. Study participants were surveyed about their attitude toward the waste generation of their therapy and personal actions to reduce waste at baseline and at the end of the study. The questions could be answered with “true,” “rather true,” “neutral,” “rather false,” or “false.” Detailed data are given in Supplemental Table S4.
An additional set of questions was surveyed after the study. Here, 92% of respondents replied that they were surprised or rather surprised by the amount of waste generated by their therapy, 94% stated that they separate their waste now, and 95% would support projects for sustainable waste reduction. Remarkably, 87% reported that their attitude toward waste has changed by participating in the study. A clear majority of patients supported demands for waste reduction and recycling: 91% would like companies to offer options for disposal and/or recycling of glucose sensors and pens, 94% demand a reduction of packaging, and 93% request collection points where therapy-associated waste can be separately collected in medical practices, clinics, and pharmacies (Figure 4 and Supplemental Table S5).
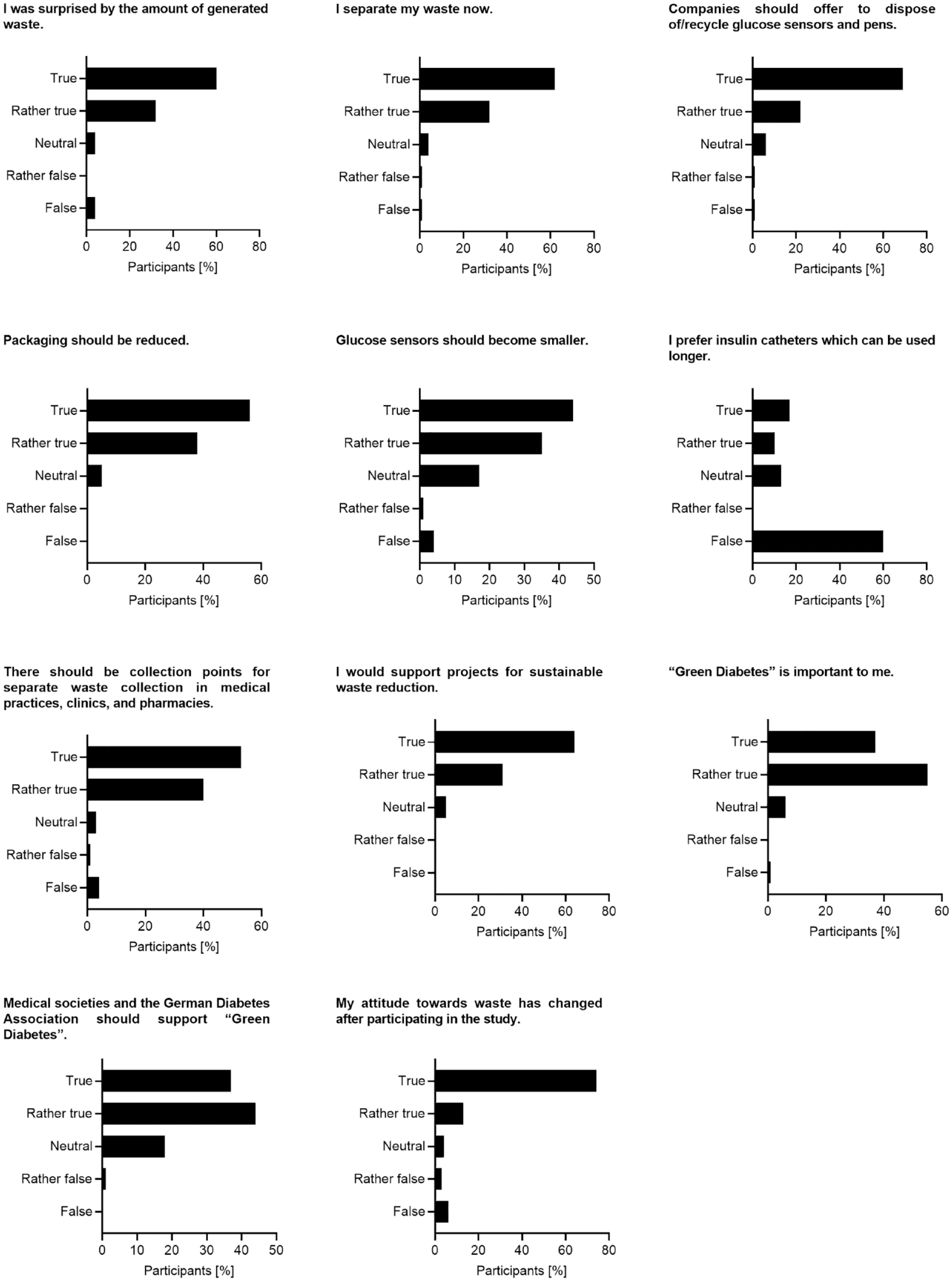
Results of survey set 2. Study participants were surveyed about their experiences during the waste collection period, their personal attitude toward their therapy-associated waste, and their demands for waste reduction. The questions could be answered with “true,” “rather true,” “neutral,” “rather false,” or “false.” Detailed data are given in Supplemental Table S5.
Discussion
Health care systems worldwide account for approximately 5% of global CO2 emissions. 9 Diabetology is not considered as extensively resource-consuming indication compared with other specialties; however, in view of the high number of PwD and the fact that current diabetes technology products are mostly single-use products, the proportion of diabetes in the amount of CO2 emissions/waste generation is high. As both the prevalence of diabetes mellitus as well as the usage of respective products has increased in the last decades a lot, more diabetes therapy-associated waste is generated year by year, a trend which most probably will even increase. However, our data are the first ones presented that quantify the amount of diabetes-related waste.
In a highly developed country like Germany, in which the costs for the antidiabetic treatment are covered by the health care system, this is associated with an impressive amount of waste. Clearly, the volume each type of waste represents differs a lot between an infusion set or a test strip, as a small piece, and the packaging of an insulin pump or the inserter of a glucose sensor, as a relatively large piece. In our study, we did not consider cardboard packaging, inserts, or blister packaging, the focus was on the products used for therapy. In this sense, our study was conservative and underestimated the total amount of waste generated.
There were—as to be expected—significant differences depending on the type of diabetes and the related antidiabetic therapy. Blood glucose test strips as a basic tool for glucose measurements as well as needles for insulin injection accounted for the majority of single-use items collected by the PwD. Interestingly, there were no significant differences between the usages of test strips between the three groups of PwD on different treatment modalities, despite a higher usage of CGM systems by PwD with T1D. Most of them used reusable insulin pens in contrast to PwD with T2D, which commonly used prefilled pens. Strikingly, PwD with T1D with CSII/AID treatment accounted for a lower count of disposed waste items despite using more different types of products for their therapy. The subgroup of PwD with T2D and MDI disposed of most items, which was mainly driven by the number of needles.
Assuming that our data reflect the “typical people with diabetes” in Germany, we used the data obtained to estimate the number of waste items collected in total for our country. For this, we multiplied the number of items collected in three months by four to project the item count for a whole year of treatment and in the next step multiplied these numbers for the patients in the subgroups T1D MDI, T2D MDI, and T1D CSII/AID by the assumed number of PwD in these groups in Germany: 240 000 PwD with T1D treated with MDI, 130 000 PwD using an insulin pump/an AID system; 25% of the 2.5 million PwD with T2D treated with insulin (MDI), equating 600 000 cases in total. This calculation sums up to approximately 1.2 billion pieces of diabetes technology–related waste in Germany per year (Table 2).
Extrapolation of Data.
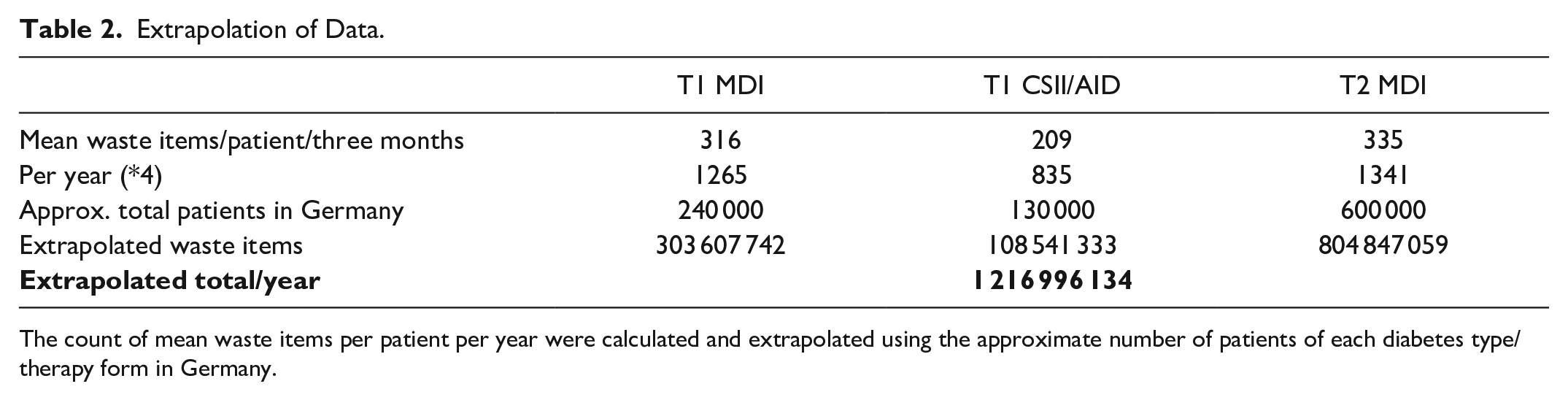
The count of mean waste items per patient per year were calculated and extrapolated using the approximate number of patients of each diabetes type/therapy form in Germany.
This study has certain limitations: the disposal of several types of products might be underreported because of the relatively short study time of three months. Products such as reusable pens and receivers of CGM systems as well as lancing devices are durable and commonly disposed only in an interval of some years. Our data fully rely on the participants collecting all their therapy-associated waste. Despite written information and pre-printed protocols for waste collection, one can assume that not every single piece of waste was collected by every participant. However, practically all patients delivered their waste thoroughly packed and with great care, indicating a high level of reliability. This was also documented by the fact that 98% of the participating PwD completed the surveys. The amount of waste generated by diabetes therapy that can be assessed by counting the recommended count of usages, ie, how many blood glucose measurements, insulin injections, sensor replacements, and so on are prescribed to the patients. However, as our data demonstrate, there are pronounced inter-individual differences between PwD in a given treatment group, which indicates at least some gap between theory and real-world data. Of note, the patients enrolled in this study represent a selected group with a high standard of care, ie, only a minority did not employ CGM, leading to an underrepresentation of, eg, the amount of blood glucose test strips and lancets. In turn, this accounts for the reported amount of the daily disposed items, which appears to be relatively low.
Despite these limitations, relevant conclusions can be drawn from our data. Most remarkably, a change in mindset was observed for the study participants, they reported an increased interest in waste reduction and recycling, along with an elevated concern in waste generation, after having collected their therapy-associated waste items for three months. Judging from the survey data, a clear majority of these PwD is in favor of enhanced sustainability in diabetes care.
Blood glucose test strips, needles, glucose sensors, single-use pens, and insulin cartridges contributed the most to the total count of disposed items, indicating a clear group of items for multiuse and/or recyclable alternatives. Despite requiring several different products for their therapy, the total number of disposed items was the lowest for those PwD using CSII/AID systems for their therapy. It is intriguing to speculate about a potential lower environmental impact of these complex forms of therapies; however, the plain number of disposed waste items does not necessarily indicate a high environmental impact or resource consumption, ie, disposable CGM sensor inserters are only required a few times per month but consist of 80 to 100 g of plastics. Accordingly, the composition of the waste generated, which is complex and heterogeneous in this case, must be considered. Medical devices and aids contain many valuable raw materials, such as plastics, iron, and other metals, which are crucial in terms of a circular economy. 10 Sustainable waste management starts with separate waste collection in households and surgeries. This is followed by mechanical shredding by crushers and shredders in the waste management companies. Optical and magnetic detectors separate the waste stream by type so that individual fractions can subsequently be used. 11 However, disposed items comprise sharps, plastics, and batteries which present risks to public health in addition to environmental issues. Used syringes, lancets, and test strips are among the sharp waste items that pose an enormous challenge because of their volume and potential to injure people and spread infections if not properly disposed. 12 As most aids are disposed in regular household waste, improved disposal guidance and management strategies are critically required. Such comprehensive strategies will not only mitigate the environmental impact but also safeguard public health and ensure sustainability.
Conclusions
In conclusion, this systematic evaluation of the amount of diabetes technology-associated waste generated by PwD under real-world conditions documented an enormous amount of disposed items used for antidiabetic therapy. This highlights the need for reducing the use of disposable products and for offering recycling options. The surveyed PwD express a demand for a more sustainable antidiabetic therapy, ie, multiuse aids, options for collection and recycling of items, and increased efforts of industry and medical associations to support “Green Diabetes.”
Supplemental Material
sj-docx-1-dst-10.1177_19322968241257004 – Supplemental material for Diabetes Technology and Waste: A Real-World Study in a Specialized Practice in Germany
Supplemental material, sj-docx-1-dst-10.1177_19322968241257004 for Diabetes Technology and Waste: A Real-World Study in a Specialized Practice in Germany by Sebastian F. Petry, Friedrich W. Petry, Johannes K. Petry, Stefan Gäth and Lutz Heinemann in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors thank all people with diabetes who participated in collecting their therapy-related waste for three months. The authors are grateful for the support of all members of the diabetes team of the Medicum Wetzlar for helping to store, sort, and count the pieces of waste, especially Tina Jatzlauk, Yeliz Salk, and Benjamin Apel. The authors also thank Gudrun Weitzel from Diabetologen Hessen eG for her support in carrying out the survey.
Abbreviations
AID, automated insulin delivery; CSII, continuous subcutaneous insulin infusion; ICT, intensified conventional insulin therapy; MDI, multiple daily insulin injections; PwD, people with diabetes.
Author Contributions
FWP, SG, and LH contributed to conceptualization. SFP, FWP, and JKP contributed to data curation. SFP contributed to formal analysis. FWP contributed to investigation. FWP contributed to methodology. SFP and FWP contributed to project administration. SFP and FWP contributed to resources. SFP, FWP, and JKP contributed to software. FWP and LH contributed to supervision. FWP and SFP contributed to validation. SFP contributed to visualization. SFP contributed to writing—original draft. SFP, FWP, JKP, SG, and LH contributed to writing—review & editing. All authors approved the final version of the manuscript. SFP is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
