Abstract
Background:
A smartphone-based automated insulin delivery (AID) controller device can facilitate use of interoperable components and acceptance in adolescents and children.
Methods:
Pediatric participants (N = 20, 8F) with type 1 diabetes were enrolled in three sequential age-based cohorts: adolescents (12-<18 years, n = 8, 5F), school-age (8-<12 years, n = 7, 2F), and young children (2-<8 years, n = 5, 1F). Participants used the interoperable artificial pancreas system (iAPS) and zone model predictive control (MPC) on an unlocked smartphone for 48 hours, consumed unrestricted meals of their choice, and engaged in various unannounced exercises. Primary outcomes and stopping criteria were defined using fingerstick blood glucose (BG) data; secondary outcomes compared continuous glucose monitoring (CGM) data with preceding sensor augmented pump (SAP) therapy.
Results:
During AID, there was no more than one BG <50 mg/dL except in one young child participant; no instance of more than two episodes of BG ≥300 mg/dL lasting longer than 2 hours; and no adverse events. Despite large meals (total of 404.9 grams of carbs) and unannounced exercise (total of 182 minutes), overall CGM percent time in range (TIR) of 70 to 180 mg/dL during AID was statistically similar to SAP (63.5% vs 57.3%, respectively, P = .145). Overnight glucose standard deviation was 43 mg/dL (vs SAP 57.9 mg/dL, P = .009) and coefficient of variation was 25.7% (vs SAP 34.9%, P < .001). The percent time in closed-loop mode and connected to the CGM was 92.7% and 99.6%, respectively. Surveys indicated that participants and parents/guardians were satisfied with the system.
Conclusions:
The smartphone-based AID was feasible and safe in sequentially younger cohorts of adolescents and children.
ClinicalTrials.gov:
NCT04255381 (https://clinicaltrials.gov/ct2/show/NCT04255381)
Introduction
The management of type 1 diabetes (T1D) is challenging during childhood due to changing insulin requirements and sensitivity with growth and puberty, unpredictable meals and bouts of physical activity, and limited self-care behaviors. 1 There is potential for adverse neurological effects on the developing brain from hypoglycemia, 2 hyperglycemia, 3 and glycemic variability. 4 Given the increasing evidence of the safety and efficacy of closed-loop glucose control using automated insulin delivery (AID) systems, it is important to evaluate innovative AID approaches in the pediatric population.5,6 Indeed, AID in youth has been evaluated in recent studies.7-13
Herein, we report a pilot evaluation of a smartphone-based AID utilizing the interoperable artificial pancreas system (iAPS) 14 platform and zone model predictive control (MPC)15,16 algorithm in sequentially younger age-based cohorts of adolescents and children with T1D. The study design incorporated purposeful challenges of high-carbohydrate meals and unannounced exercise to evaluate the feasibility and preliminary safety of an unlocked smartphone as the AID controller device.
Methods
Study Design and Procedures
The single-arm, 48-hour study was performed sequentially across three age-based cohorts, starting with adolescents 12-<18 years old. If no stopping criteria (as noted below) were met upon review by the investigative team and the Data and Safety Monitoring Board (DSMB), enrollment of school-aged children 8-<12 years old occurred, who were then followed by enrollment of the youngest cohort 2-<8 years old, again after data safety review (Supplementary Figure 1). The primary outcomes, stopping criteria, and continuation criteria were designed for safety to assess severe hypoglycemia and sustained hyperglycemia using fingerstick blood glucose (BG) values, while secondary outcomes included efficacy related to device performance assessed using continuous glucose monitoring (CGM) data.
The investigational plan was approved by the Food and Drug Administration (FDA) and the Yale University Institutional Review Board and registered on clinicaltrials.gov (NCT04255381). After informed consent/assent and confirmation of eligibility (Section 1, Supplementary Material), participants used their own insulin pumps along with a Dexcom G5/G6 CGM (Dexcom, San Diego, CA) for up to two weeks to provide baseline sensor augmented pump (SAP) therapy data. They could use the predictive low glucose suspend (PLGS) feature, if available, on their pump.
Twelve to 24 hours prior to AID initiation, a new CGM sensor was inserted at home. Next, participants, along with a parent/guardian arrived at the study hotel around noon on Day 1 (Supplementary Figure 2). Over the next 48 hours, participants chose meals and engaged in various activities, such as walking, painting and pottery classes, trips to local museums, and exercise activities such as visits to an escape room (adolescents), trampoline park (school-age and young children), and climbing/ropes course (all ages). Meals were selected by participants, without restrictions, at fast food restaurants and hotel breakfast buffets. Some participants opted for late-evening desserts of cookies or ice-cream. After two overnight periods, the study concluded on Day 3, around noon following lunch.
During the study, insulin boluses for all meals were given at mealtime based on carbohydrates counted by participants and/or parents/guardians and the participants’ usual insulin-to-carbohydrate ratios; no advice or “coaching” on insulin dosing for meals was provided by study staff. Participants engaged in multiple unannounced exercises with pre-treatment as per their usual diabetes care. They were instructed to refrain from initiating correction doses for hyperglycemia unless instructed by study staff to allow the zone MPC to autocorrect via insulin microboluses.
Monitoring and Treatment Procedures
A summary of monitoring, rescue treatment procedures, and criteria for temporary suspension of AID appear in Supplementary Table 1. Remote monitoring alerts were enabled to signal for CGM levels < 70 and < 55 mg/dL, levels ≥ 300 and ≥ 350 mg/dL, and for device connectivity issues. Fingerstick BGs were checked using the study meter when prompted by the iAPS predictive hypoglycemia alarm,
17
for any CGM readings < 70 mg/dL or with symptomatic hypoglycemia. For any documented BG < 70 mg/dL or in the setting of symptomatic hypoglycemia, participants were treated with 15 to 30 grams of fast-acting carbohydrate as per their usual diabetes care, and BG was rechecked in 15 minutes to confirm resolution of hypoglycemia or need for additional treatment. For CGM ≥ 300 mg/dL for > 1 hour, participants performed confirmatory BG check and, for BG ≥ 300 mg/dL, blood ketones (B-OHB) levels were also checked; if B-OHB > 1 mmol/L, infusion set was changed, and correction dose of insulin was given. In addition, all participants checked their BG levels prior to exercise. Finally, all participants and parents/guardians returned to the study hotel by 9
Stopping Criteria
Stopping criteria for a participant included either two episodes of a confirmed BG < 50 mg/dL (separated by at least 30 minutes), or B-OHB > 1.0 mmol/L for over 2 hours, or any B-OHB > 3.0 mmol/L. If more than two participants met individual stopping criteria, then the study would be stopped for that age-based cohort, and no further participants would be enrolled until DSMB review. If any single participant experienced seizure, loss of consciousness, or diabetic ketoacidosis (CO2 < 15 mEq/L or venous pH < 7.3), then the study would be stopped for that participant and no additional participants would be enrolled until DSMB review.
Study Outcomes
Primary outcomes for an individual participant using fingerstick BG data were defined for safety: (1) no more than one confirmed BG < 50 mg/dL; (2) no more than two episodes of confirmed BG ≥300 mg/dL lasting longer than 2 hours, unless due to infusion site failure; (3) no BG ≥ 400 mg/dL, unless due to site failure; (4) no B-OHB > 1.0 mmol/L for longer than 2 hours, or any B-OHB > 3.0 mmol/L, unless due to site failure; and (5) no significant adverse events (ie, severe hypoglycemia with seizure or loss of consciousness, diabetic ketoacidosis). Secondary outcomes related to AID efficacy included CGM metrics according to published criteria.18,19 After completion of the study, parents/guardians and participants (except for young children) completed the 21-item and 17-item INSPIRE questionnaires,20,21 respectively.
Zone MPC
The zone MPC algorithm was initialized using the participant’s baseline SAP profiles, including total daily insulin (TDI), insulin-to-carbohydrate ratios, correction factors, and basal rates. There were no modifications or optimizations made to baseline profiles for AID. For safety reasons, only participants receiving a minimum TDI of 10 U were included, although this was not an algorithm limitation. There were no limitations imposed on the magnitude of other profile variables. With meal announcement, AID operated in a hybrid configuration.16,22 The zone MPC algorithm penalized deviation of glucose from a zone of 90 to 120 mg/dL during the day and 100 to 120 mg/dL overnight (12
Statistical Analysis
Statistical variables appear as mean (standard deviation) unless specified as median (interquartile range [IQR]), based on their distributions. Overnight analysis included data from 12
Daily carbohydrate intakes were extracted from participants’ own pump downloads for the baseline SAP period and from data entered into the iAPS app during the AID period. Due to the unequal length of SAP and AID, the meal/snack carbohydrate intakes were compared using 24-hour bins. For SAP, 24-hour carbohydrate intakes were calculated as a median of 24-hour carbohydrate intakes for up to two weeks. For AID, since the study started on Day 1 noon till Day 3 noon, 24-hour carbohydrate intakes were calculated as half of the total 48-hour carbohydrate intake, where lunch on Day 3 was excluded for a balanced comparison. TDI during AID was calculated similarly by excluding insulin for and following lunch on Day 3.
The INSPIRE surveys were tallied according to published recommendations,20,21 with each item response scored on a 0- to 4-point scale, and converted to a total score range of 0 to 100 by multiplying the mean item scores by 25. Data were analyzed using MATLAB 2021a (The MathWorks, Natick, MA).
Results
Baseline Participant Characteristics
A summary of baseline characteristics is shown in Table 1. In total, 20 participants with T1D (8 female, mean age of 11.3 years, mean HbA1c 7.6%) completed the study between October 2018 and January 2020. Three sequential age-based cohorts consisted of eight adolescents (five female, mean age of 15 years, mean HbA1c 7.6%), seven school-aged children (two female, mean age of 10.2 years, mean HbA1c 7.3%), and five young children (one female, mean age of 6.9 years, mean HbA1c 7.9%). The body mass index (BMI) was 21.2 kg/m2 with a BMI standard score of 0.9 and a TDI/kilogram of 0.9 U/kg. Four participants (20%) used the PLGS feature on their pump during baseline SAP therapy. The first three adolescents used the Dexcom G5 CGM with the iAPS app and followed the daily requirements for fingerstick glucose calibrations twice daily, while the remainder used the factory calibrated Dexcom G6 CGM.
Baseline Characteristics for All Participants (2-<18 Years), and for the Three Age-Based Cohorts: Adolescent (12-<18 Years), School-Age Children (8-<12 Years) and Young Children (2-<8 Years).
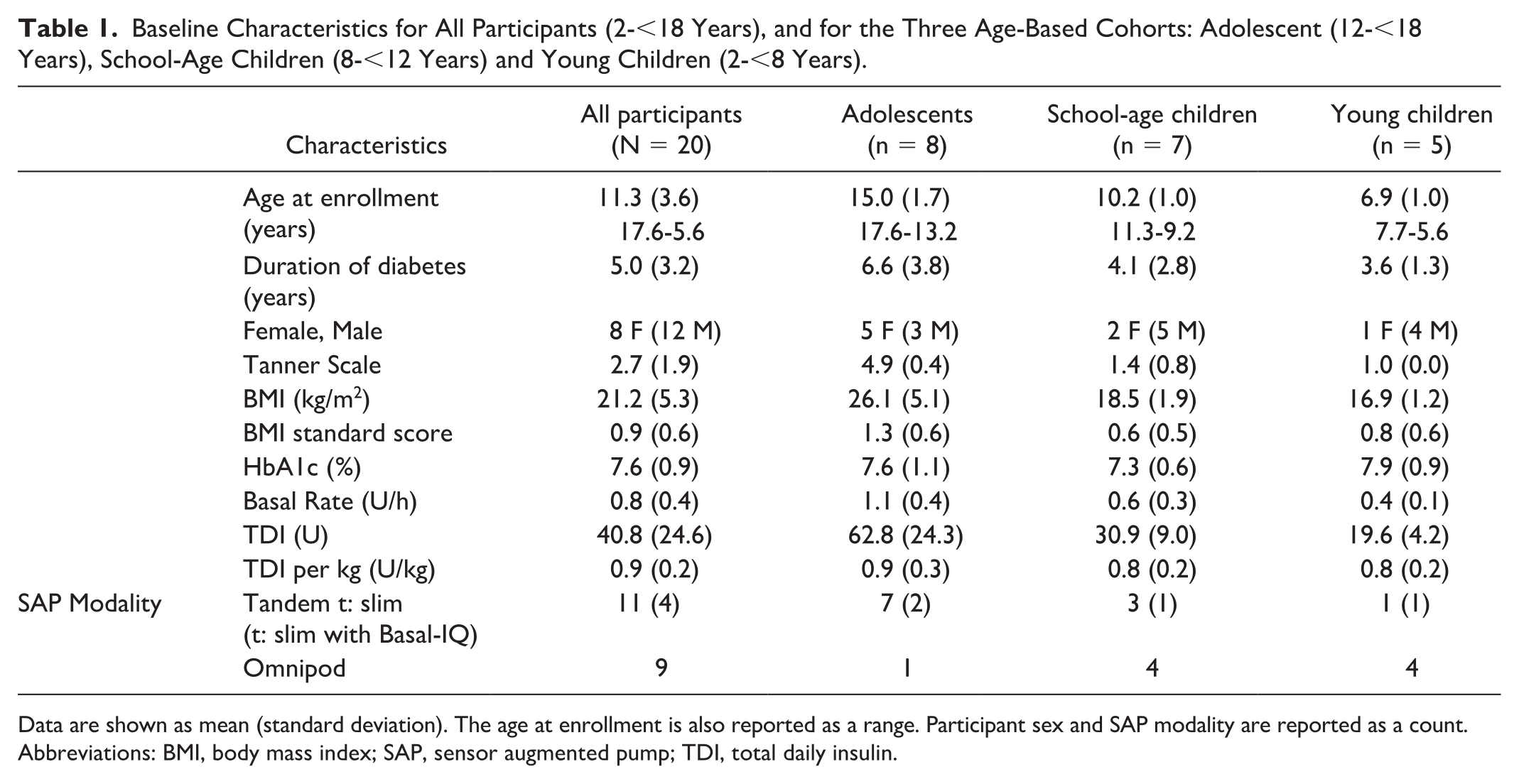
Data are shown as mean (standard deviation). The age at enrollment is also reported as a range. Participant sex and SAP modality are reported as a count.
Abbreviations: BMI, body mass index; SAP, sensor augmented pump; TDI, total daily insulin.
Glycemic Outcomes
The primary outcomes were broadly satisfied for all three age-based cohorts (Figure 1 and Supplementary Table 2). There were no instances of more than one confirmed BG <50 mg/dL except for one young child participant with two recorded instances of BG of 47 mg/dL (Day 1 after dinner) and 48 mg/dL (Day 2 after breakfast). There were no participants with more than two confirmed episodes of BG ≥ 300 mg/dL lasting longer than 2 hours not due to site failure. One school-age participant had a BG ≥ 400 mg/dL after breakfast during the ropes course on Day 3. There was no B-OHB > 1.0 mmol/L for longer than 2 hours, nor any B-OHB > 3.0 mmol/L. The mean of the recorded minimum BG values was 72.2 mg/dL and the maximum BG was 296.6 mg/dL.
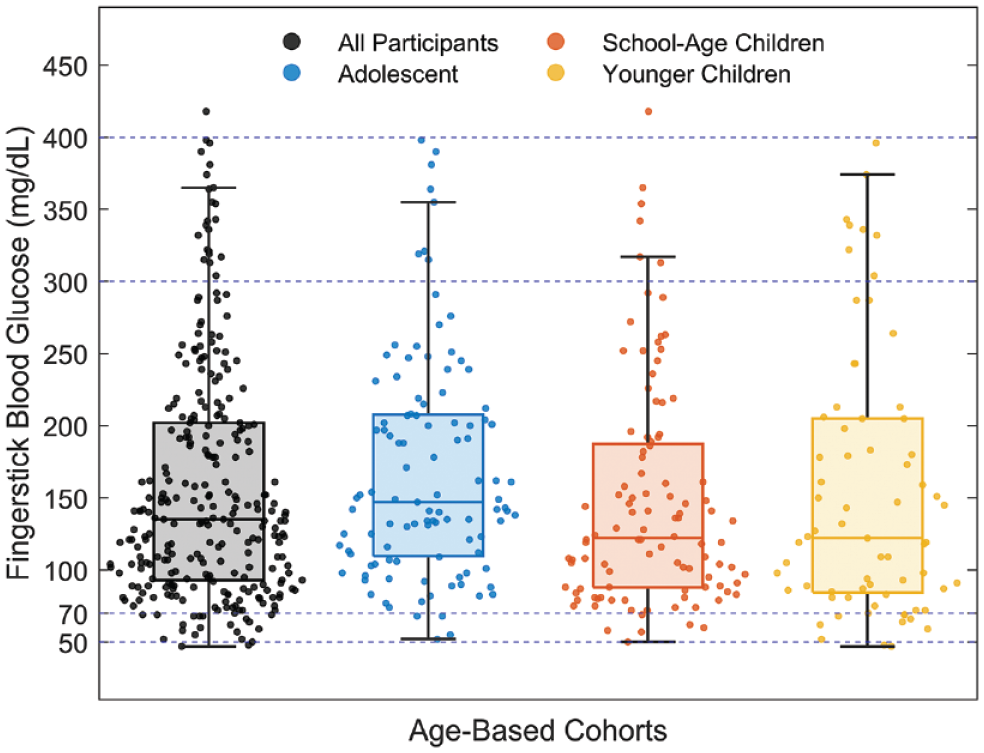
Distribution of fingerstick blood glucose (BG) values during automated insulin delivery (AID) for all participants and for the three age-based cohorts. In each box, the central line indicates the median, the bottom and top edges indicate the 25th and 75th percentiles, respectively, and the whiskers extend to the extreme data points, excluding the outliers. Superimposed on the box plots are the raw BG data jittered using the kernel density estimate. Dashed horizontal lines indicate the BG thresholds used for monitoring and stopping criteria.
Table 2 displays secondary glycemic outcomes using CGM data during SAP compared with AID for all participants and for the three age-based cohorts. Despite the significant glycemic disturbances from large unrestricted meals and extensive unannounced exercises, overall CGM outcomes were modestly and uniformly more favorable, albeit statistically similar, during AID compared with SAP for the entire sample. Percent time in range (TIR) 70 to 180 mg/dL during AID was 63.5% versus 57.3% during SAP (P = .145), percent time >180 mg/dL was 34.7% during AID versus 40.4% during SAP (P = .2), and percent time <70 mg/dL was 1.8% during AID versus 2.3% during SAP (P = .28). During the overnight period, the low blood glucose index was 0.3 during AID versus 0.6 during SAP (P = .007), glucose variability by glucose standard deviation was 43.0 mg/dL during AID versus 57.9 mg/dL during SAP (P = .009), and glucose variability by coefficient of variation was 25.7% during AID versus 34.9% during SAP (P < .001). The number of 5-min CGM data points during AID was a mean of 566 versus a mean of 3529 during SAP. During SAP, one young child participant had a percent time < 70 mg/dL of 10.6% where the number of CGM data points was 755.
Comparison of Secondary Glycemic Outcomes, Carbohydrate Intakes, and Total Daily Insulin During AID, Utilizing Interoperable Artificial Pancreas System (iAPS) and Zone Model Predictive Control (MPC), Versus SAP.
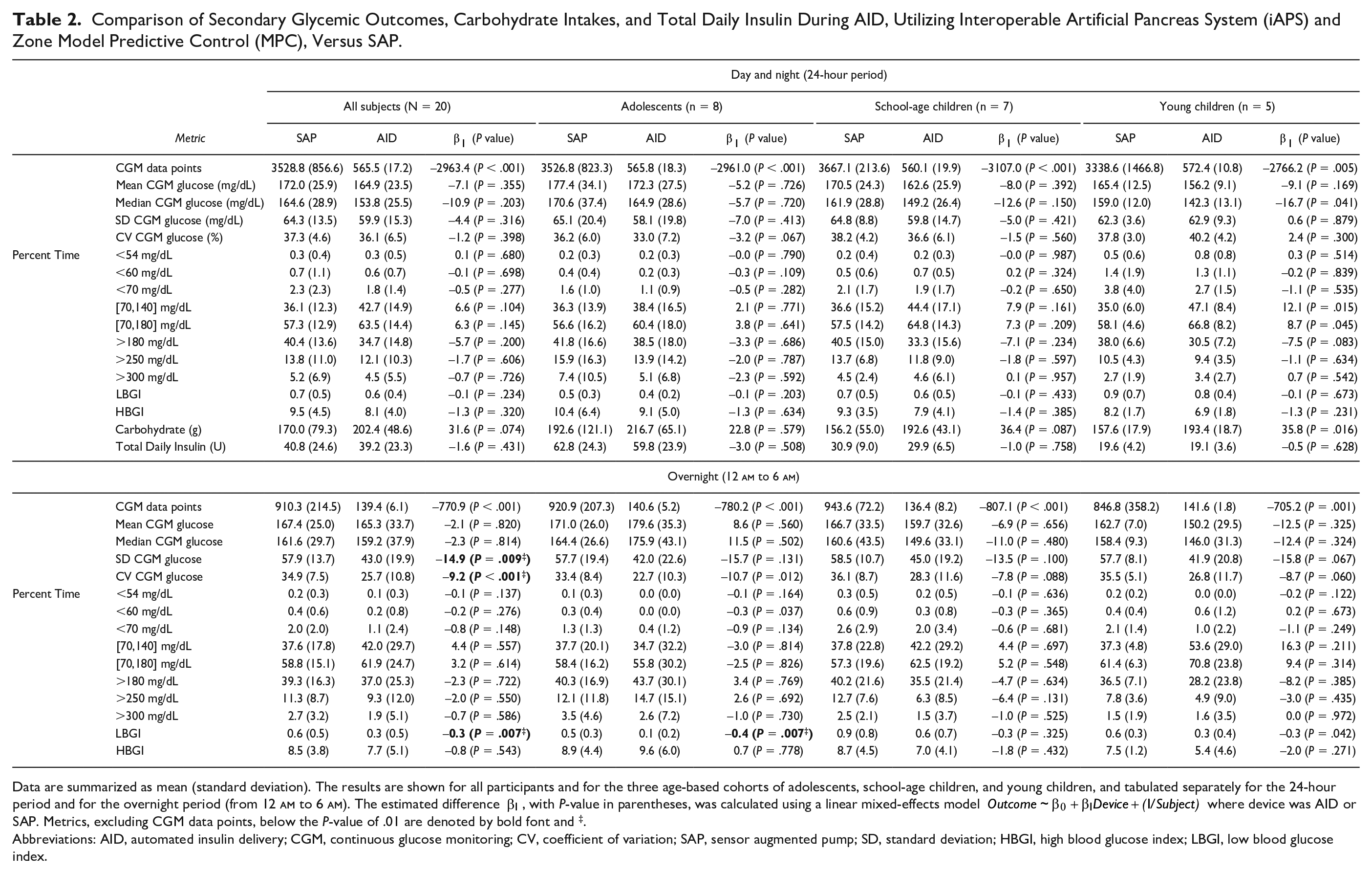
Data are summarized as mean (standard deviation). The results are shown for all participants and for the three age-based cohorts of adolescents, school-age children, and young children, and tabulated separately for the 24-hour period and for the overnight period (from 12
Abbreviations: AID, automated insulin delivery; CGM, continuous glucose monitoring; CV, coefficient of variation; SAP, sensor augmented pump; SD, standard deviation; HBGI, high blood glucose index; LBGI, low blood glucose index.
The 24-hour glucose profiles (Figure 2) corroborate tighter glucose control, reduced glucose variability overnight, and lower early morning glucose levels before breakfast during AID compared with SAP. The scatter plot of percent time below (TB) 70 mg/dL and percent TIR (Figure 3) highlights the improvement in group mean and the decrease in group standard deviation during AID. Figure 4 and Supplementary Figure 3 display paired within-participant comparisons for mean glucose and percent time in the 70-180 mg/dL range for each age-based cohort. Overall, AID improved mean glucose and time in range compared with baseline SAP, particularly in younger cohorts.
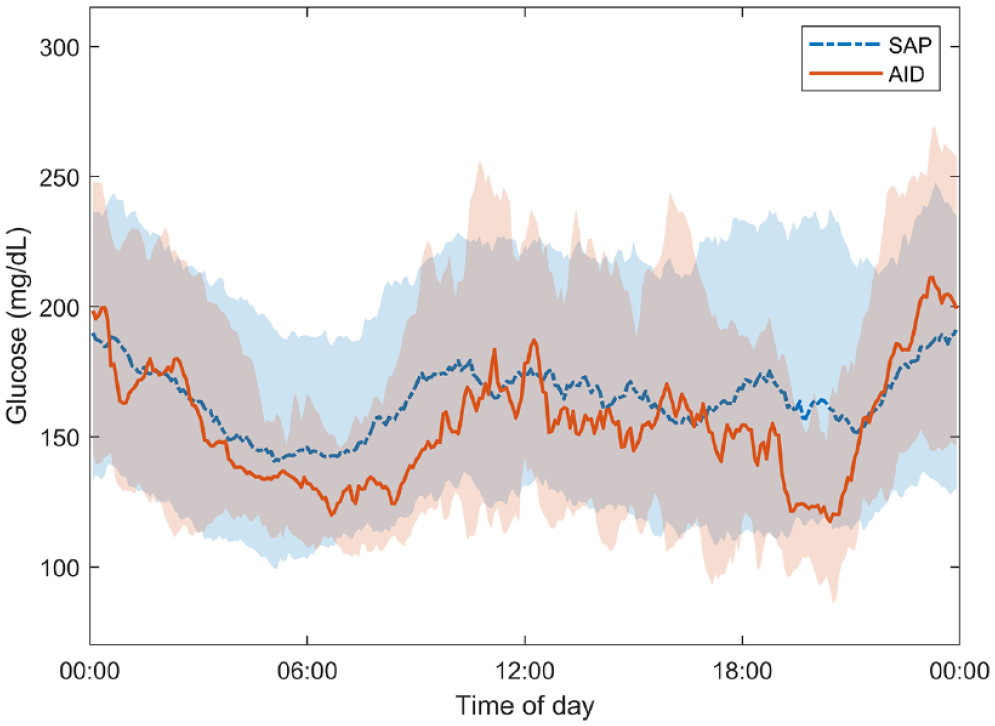
Comparison of 24-hour glucose profiles from AID versus preceding SAP therapy. The median glucose, shown using the dash-dot and the solid lines, and the interquartile range glucose, shown using the shaded region, were calculated for each time point across all participants. The overnight glucose variability decreased during AID compared with SAP. Abbreviations: AID, automated insulin delivery; SAP, sensor augmented pump.
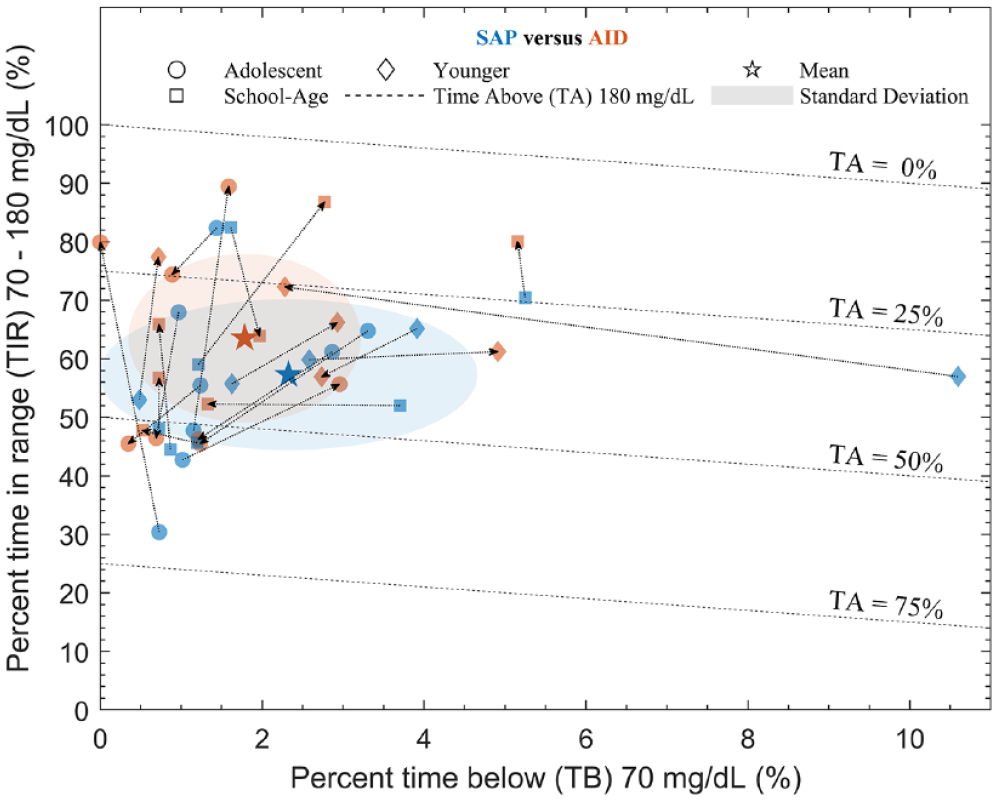
Scatter plot of percent time below (TB) 70 mg/dL and percent time in range (TIR) 70 to 180 mg/dL for AID versus preceding SAP therapy. The parallel dashed isolines enclose a region with a percent time above (TA) of 180 mg/dL. Dotted lines connect individual participants, highlighted using markers for each age-based cohorts, with an arrow pointing from SAP (blue) to AID (red). Compared with SAP, AID moved the group mean toward the top left of the chart (higher TIR and lower TB) while reducing group standard deviation (smaller shaded areas). Abbreviations: AID, automated insulin delivery; SAP, sensor augmented pump.
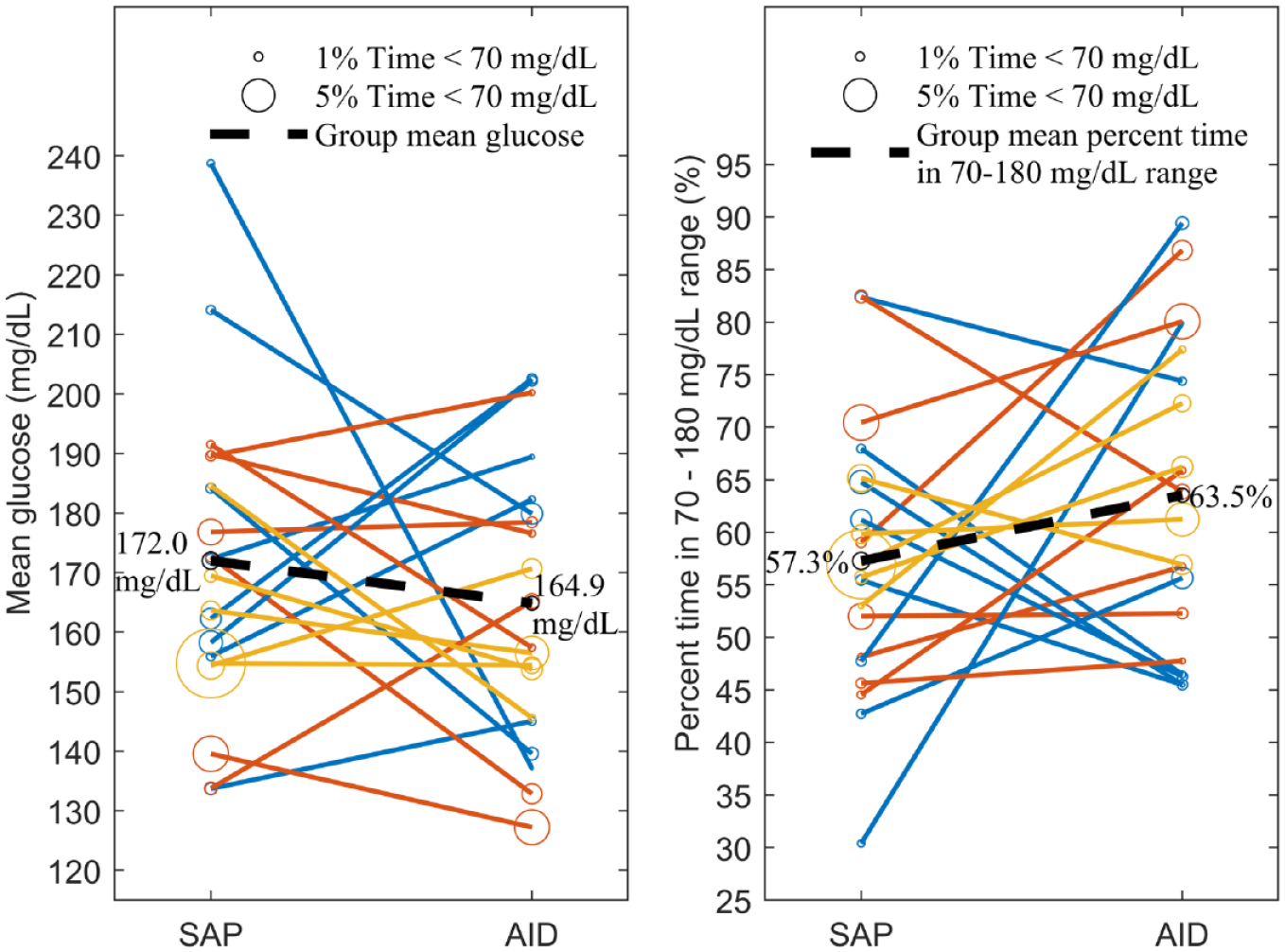
Paired comparison of mean glucose (left) and percent time in 70 to 180 mg/dL range (right) for the three age-based cohorts. The adolescent cohort is shown in blue, the school-age cohort in red, and the young children’s cohort in orange. In each plot, the bubble size is proportional to time below 70 mg/dL and the mean response for the entire sample is shown as a black dashed line. Abbreviations: AID, automated insulin delivery; SAP, sensor augmented pump.
Carbohydrate Intake
Participants consumed a mean of 404.9 grams of carbohydrates over the 48 hours of AID. In terms of 24-hour grams of carbohydrates, there was 19% more during AID (202.4 grams) than SAP (170.0 grams), a difference of 31.6 grams (P = .074) for the entire sample (Table 2 and Supplementary Figure 4). Notably, over the 48 hours of AID, there was a median (IQR) of 3(2) rescue treatments. There was no documentation of rescue treatments given during SAP.
Exercise Activities During AID
Over the 48 hours of AID, participants engaged in various exercise activities for a mean of 182 minutes (Supplementary Figure 5) over two exercise sessions (Day 1 afternoon or Day 2 afternoon and Day 3 morning, Supplementary Figure 2), with each session lasting a mean of 91 minutes. During the activity periods, across all participants, the median of the minimum BG was 102 mg/dL and the maximum BG was 142 mg/dL. There was a median (IQR) of 1 (1) rescue treatment during the exercise period. There was no capture of exercise activities during SAP.
Insulin Outcomes
The TDI during AID did not differ from that during SAP (39.2 U vs 40.8 U, respectively, P = .43, Table 2). During AID, 54.5% of commanded insulin microboluses differed from the basal rates, and 20% of microboluses were of zero units, equivalent to basal suspension (Supplementary Figure 6). There was one instance of an overnight user-requested correction bolus when glucose was above 300 mg/dL and B-OHB was 0.1 mmol/L (a mean of 0.025 correction bolus per overnight period). During SAP, there were a mean of 0.3 correction bolus per overnight period.
iAPS Use, Adverse Events and Survey Results
iAPS was in closed-loop mode connected to the pump and CGM for 92.7% and connected to the CGM for 99.6% of the time (Supplementary Table 3). There were no adverse events, serious adverse events, or adverse device effects. One adolescent participant arrived at the study site with a BG of 527 mg/dL and a B-OHB of 0.2 mmol/L. Notably, this participant had not received a bolus dose for breakfast and also required a set change for low insulin in the pump reservoir. The infusion site was changed, a correction bolus was given, and, per protocol, the initiation of the start of AID was delayed for six hours, when the BG was 144 mg/dL and B-OHB < 0.1 mmol/L.
The INSPIRE surveys revealed overall satisfaction with the AID system, with a mean parents/guardians score of 84 (range: 62-100) and a participant score of 81 (range: 59-100). Parents/guardians most favorably endorsed items relating to staying in target, worrying less, and improved quality of life, while youth most favorably endorsed items relating to greater ease and improved social life (Supplementary Table 4).
Discussion
Use of zone MPC integrated into the iAPS was feasible and safe in age-based cohorts of adolescents and children. There were no instances of severe hypoglycemia or sustained hyperglycemia, no adverse events or adverse device effects, and excellent connectivity to devices. The day and night glycemic outcomes during AID did not change when compared with baseline SAP therapy despite higher carbohydrate intake, which would likely increase the area under the glucose curve, along with the unannounced exercise, which would likely increase time in hypoglycemia and glycemic variability. In addition, glycemic variability and risk of hypoglycemia were reduced during the overnight period, with the potential to reduce parental stress of fear of hypoglycemia and the need for nighttime surveillance. 24 The overnight time in 70 to 180 mg/dL range was lower than target for many participants for several potential reasons: inclusion of evening snacks; pre-study non-optimized user profiles leading to controller action saturation by safety constraints; and no intervention per the clinical protocol if glucose level was below 300 mg/dL (as evidenced by only one instance of overnight correction bolus), whereas participants received more overnight correction boluses during SAP.
Glycemic outcomes during AID presented herein are comparable with other AID studies in children and adolescents. 13 In Sherr et al, 8 carbohydrates consumed (199 grams for adolescents and 214 grams for children) and exercise performed (66 minutes per session) were lower than in our study. In Ekhlaspour et al, 11 carbohydrates consumed (123 grams) and exercise performed (at least 30 min per session) were also lower than in our study. In both comparisons, although the TIR was higher than our study, it is not entirely comparable due to the purposeful meal and activity challenges and the absence of adjustments to user profiles before and during our study. Although iAPS is designed for research use, its percent time in closed-loop mode was similar to commercial systems.10,12 It is notable that following a short hands-on training, participants and parents of the youngest group could operate the iAPS app with little to no professional supervision.
This study had several notable strengths. First, AID was sequentially tested across a wide age range of pediatric participants with varying insulin sensitivities and glycemic outcome data included both fingerstick BG monitoring and CGM. Furthermore, we did not perform any modifications to user profiles before “closing the loop,” which might have negatively biased the performance of the AID. Finally, this is the first FDA-approved study in adolescents and children, to the best of our knowledge, with the control algorithm running on an unlocked smartphone. The methodological limitations of this study include small sample size, short study duration, lack of a supervised control arm with similar challenges, and non-randomized intervention, thus limiting generalizability. Furthermore, participants were remotely monitored, and assistance was provided for any device issues. Thus, the level of supervision in this study was higher than for at-home studies.
Conclusions
In conclusion, zone MPC integrated into the iAPS demonstrated feasibility and preliminary safety in a sample of adolescents and children with T1D, even in the combination of unrestricted meals and unannounced exercise. The smartphone-centric architecture can facilitate use of interoperable components and individual customization, and thus has potential to greatly improve acceptance and satisfaction with AID systems, particularly in adolescents and children, who might perceive lower physical and emotional burdens of diabetes management with an easy-to-carry, on-body, and socially acceptable consumer device platform compared with a medical device. 25 Larger outpatient randomized studies are required to further validate the safety and efficacy of this smartphone-based AID.
Supplemental Material
sj-pdf-1-dst-10.1177_19322968221116384 – Supplemental material for Feasibility and Preliminary Safety of Smartphone-Based Automated Insulin Delivery in Adolescents and Children With Type 1 Diabetes
Supplemental material, sj-pdf-1-dst-10.1177_19322968221116384 for Feasibility and Preliminary Safety of Smartphone-Based Automated Insulin Delivery in Adolescents and Children With Type 1 Diabetes by Sunil Deshpande, Stuart A. Weinzimer, Kathryn Gibbons, Laura M. Nally, Kate Weyman, Lori Carria, Melinda Zgorski, Lori M. Laffel, Francis J. Doyle and Eyal Dassau in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors express their gratitude to the participants and their parents and guardians for their contributions to this study. We also acknowledge additional personnel at the clinical site (Amy Steffen, Eileen Tichy, Michelle Van Name, Jennifer Sherr) and engineering center (Claire Van Strien, Jesse Rizutko) who helped support this study. The authors also acknowledge Randy Tompot, Chris Rogers and Judit Flo Gaya for contributions to the iAPS platform and Kelilah Wolkowicz for engineering support during one of the study sessions.
Abbreviations
AID, automated insulin delivery; BG, blood glucose; BMI, body mass index; CGM, continuous glucose monitoring; DSMB, Data and Safety Monitoring Board; FDA, Food and Drug Administration; iAPS, interoperable artificial pancreas system; IOB, insulin-on-board; IQR, interquartile range; MPC, model predictive control; PLGS, predictive low glucose suspend; SAP, sensor augmented pump; T1D, type 1 diabetes; TB, time below; TDI, total daily insulin; TIR, time in range.
Authors’ Note
Author Contributions
S.D. contributed to the design of the study protocol, designed the zone MPC algorithm and the iAPS platform, ensured regulatory approval, provided engineering support during the study, analyzed the data, and conceptualized and wrote the article. S.A.W. designed the study protocol, ensured regulatory approval, conducted the clinical study, collected data, assisted with writing the article, and reviewed the article. K.G. conducted the clinical study and reviewed the article. L.M.N. conducted the clinical study and reviewed the article. K.W. conducted the clinical study and reviewed the article. L.C. conducted the clinical study, collected data, and reviewed the article. M.Z. conducted the clinical study, collected data, and reviewed the article. L.M.L. participated in discussion of study procedures, reviewed data, contributed to manuscript writing, and reviewed the article. F.J.D. contributed to the design of the study protocol, designed the zone MPC algorithm and the iAPS platform, and reviewed the article. E.D. designed the study protocol, designed the zone MPC algorithm and the iAPS platform, ensured regulatory approval, provided engineering support during the study, assisted with writing the article, and reviewed the article. E.D. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: S.A.W. has received honoraria for serving as a speaker for Abbott and Dexcom, has served as consultant for Zealand, and receives grant support from NIH and Abbott. L.M.N. receives grant support from the NIH and product support from Dexcom. L.M.L. participates in consulting and on advisory boards for Medtronic, Insulet, Dexcom, Roche, Janssen, Boehringer Ingelheim, Eli Lilly, Provention, and Dompe, and receives grant support from NIH, JDRF, and Helmsley Charitable Trust. F.J.D. reports equity, licensed IP and is a member of the Scientific Advisory Board of Mode AGC. E.D. reports receiving grants from JDRF, NIH, and Helmsley Charitable Trust, personal fees from Roche and Eli Lilly, patents on artificial pancreas technology, and product support from Dexcom, Insulet, Tandem, and Roche. E.D. is currently an employee and shareholder of Eli Lilly and Company. The work presented in this manuscript was performed as part of his academic appointment and is independent of his employment with Eli Lilly and Company. All other authors report no conflict of interest related to this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was made possible by grants from the National Institutes of Health (DP3DK104057, DP3DK113511, and UC4DK108483), and the Michael and Rosemary Ryan Diabetes Research Fund. CGM sensors and transmitters and Tandem t:AP insulin pumps were purchased from Dexcom, Inc. and Tandem Diabetes Care, respectively, at full price. The funding agencies and device manufacturers had no influence on the design or conduct of the study and were not involved in data collection or analysis, the writing of the manuscript, or the decision to submit it for publication.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
