Abstract
Objective:
Extended hypoglycemia (Ehypo) and extended hyperglycemia (Ehyper) are recently defined continuous glucose monitoring (CGM) metrics by the International Consensus for clinical trials as secondary endpoints for continuous outcomes. This study aims to evaluate the changes in Ehypo and Ehyper before and after automated insulin delivery (AID) initiation in adults with type 1 diabetes (T1D).
Research Methods:
This is a retrospective single-center study that evaluated Ehypo and Ehyper in addition to other CGM metrics in 154 adults that initiated an AID system. Metrics were compared before and after AID initiation by Wilcoxon signed-rank test.
Results:
Median (interquartile range) Ehypo (<70 mg/dL) events/week decreased from 0.1 (0-0.4) to 0 (0-0.1) and Ehyper (>250 mg/dL) events/week decreased from 2.2 (0.9-4.5) to 0.8 (0.3-1.7) (both P < .001) after AID initiation compared with before AID initiation. All other CGM metrics improved after AID initiation. There was a strong positive correlation between Ehyper (>250 mg/dL) and mean glucose (before AID: r = 0.947, after AID: r = 0.894), glucose management indicator (before AID: r = 0.947, after AID: r = 0.887), and time above range (TAR; >180 mg/dL) (before AID: r = 0.957, after AID: r = 0.917) and a strong positive correlation between Ehypo (<70 mg/dL) and time below range (TBR; <70 mg/dL) (before AID: r = 0.823, after AID: r = 0.608) before and after AID initiation, respectively.
Conclusion:
Automated insulin delivery initiation significantly improved Ehypo and Ehyper metrics. Ehypo and Ehyper had a strong positive correlation with TBR and TAR, respectively. Ehypo and Ehyper events can be used in addition to TBR and TAR metrics in clinical studies as secondary outcomes.
Keywords
Introduction
The use of continuous glucose monitoring (CGM) has been shown to improve HbA1c, increase time in glycemic target range, and decrease time in hypoglycemia in people with type 1 diabetes (T1D).1-3 Many randomized clinical trials and observational real-world studies validated the improvement in well-established CGM metrics with CGM and automated insulin delivery (AID) use.4-6 In 2023, The International Consensus recommended CGM-derived endpoints for clinical trials. New CGM metrics have been described such as extended hypoglycemia (Ehypo) and extended hyperglycemia (Ehyper) (also known as sustained hypoglycemia and hyperglycemia), 7 and they were suggested to be used as secondary endpoints for continuous outcomes. 7 However, there have been no studies that evaluated these new CGM metrics yet. In this study, we evaluated the changes of newly defined CGM metrics, Ehypo, and Ehyper in adults with T1D who initiated an AID system.
Methods
In this longitudinal retrospective cohort study, adults with T1D who were using CGM and initiated AID system (Tandem t:slim X2 Control IQ) at the Barbara Davis Center between February 2020 and June 2022 were recruited. Their demographic and clinical data were collected from electronic health records. Their CGM raw data were downloaded for two different time intervals from Dexcom Clarity. The first-time interval (pre-AID) starts four months prior to AID initiation and up to 90 days of CGM data was downloaded prospectively. The second time interval (post-AID) started one month after AID initiation and up to 90 days of CGM data was downloaded prospectively. People with at least 14 days of CGM data with >70% completeness in both time intervals were included in this study. This retrospective study was approved by the Colorado Multiple Institutional Review Board under the exempt category.
As defined by the International Consensus, Ehypo event is number of events with sensor glucose <70 mg/dL (<3.9 mmol/L) lasting at least 120 minutes and event ends when glucose returns to ≥70 mg/dL (≥3.9 mmol/L) for ≥15 minutes. 7 Ehyper event is number of events with sensor glucose >250 mg/dL (>13.9 mmol/L) lasting at least 120 minutes and event ends when glucose returns to ≤180 mg/dL (≤10.0 mmol/L) for ≥15 minutes. 7
Statistical Analysis
To produce CGM metrics, CGM raw data were processed in cgmanalysis R package, which is a validated package to analyze CGM raw data. 8 Ehypo metrics were generated with three different cutoffs: <70 mg/dL, <60 mg/dL, and <54 mg/dL. Ehyper metrics were generated for >180 mg/dL, >200 mg/dL, and >250 mg/dL cutoffs. Glucose excursions lasting at least 120 minutes below Ehypo cutoffs and above Ehyper cutoffs were included as Ehypo or Ehyper events, respectively. In addition to newly defined Ehypo and Ehyper metrics, well-established CGM metrics previously defined by the International Consensus were also collected (percent time in 70-180 mg/dL [time in range, TIR], time above range 180 mg/dL [TAR], above 250 mg/dL [TAR2], time below range 70 mg/dL [TBR], and below 54 mg/dL [TBR2], mean glucose, standard deviation [SD], coefficient of variation [CV], glycemia risk index [GRI], and glucose management indicator [GMI]).9,10 Categorical variables are shown as absolute numbers and percentages while continuous variables are shown as mean ± standard deviation or median (interquartile range). Metrics before and after AID initiation were compared with Wilcoxon signed-rank test. Correlation between metrics was analyzed with Spearman correlation. IBM’s SPSS v29 was used for statistical analysis and P values <.05 considered significant.
Results
A total of 154 people (78% non-Hispanic White, 65% female, 79% private insurance) had CGM data with at least 14 days of data with >70% completeness before and after AID initiation. At baseline, mean age was 40.2 ± 14.6 years, with 21.7 ± 14.4 years diabetes duration, and A1c 7.3% ± 1.2%. Before AID initiation, 38% were using multiple daily injections (MDI) and 62% were using non-AID insulin pumps. Median CGM wear duration was 89 (81-90) days, with 93% (87%-97%) completeness before AID and 90 (86-90) days with 95% (91%-97%) completeness after AID. All CGM metrics, including mean glucose, SD, CV, GMI, GRI, and percent time metrics, were improved after AID initiation (Table 1). Ehypo (<70 mg/dL) events/week decreased from 0.1 (0-0.4) to 0 (0-0.1) and Ehyper (>250 mg/dL) events/week decreased from 2.2 (0.9-4.5) to 0.8 (0.3-1.7) (both P < .001) after AID initiation compared with before AID initiation. Percent time below and above ranges decreased as cutoffs changed to more extreme values, Ehypo and Ehyper metrics also followed a similar pattern (Figure 1). The number of events was less than percent values of time above/below ranges for all cutoffs.
Change in Continuous Glucose Monitoring Metrics After Automated Insulin Delivery Initiation.
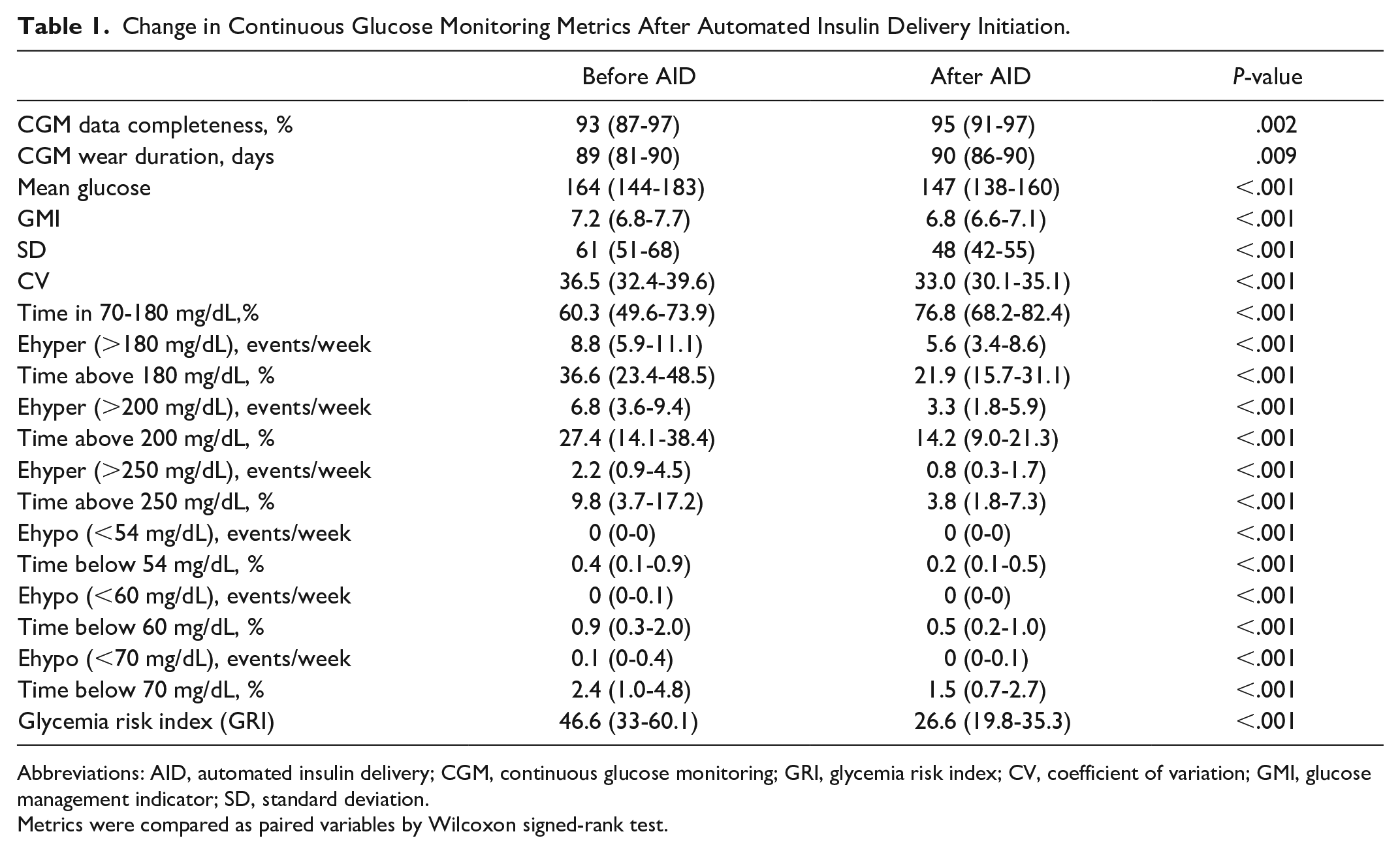
Abbreviations: AID, automated insulin delivery; CGM, continuous glucose monitoring; GRI, glycemia risk index; CV, coefficient of variation; GMI, glucose management indicator; SD, standard deviation.
Metrics were compared as paired variables by Wilcoxon signed-rank test.
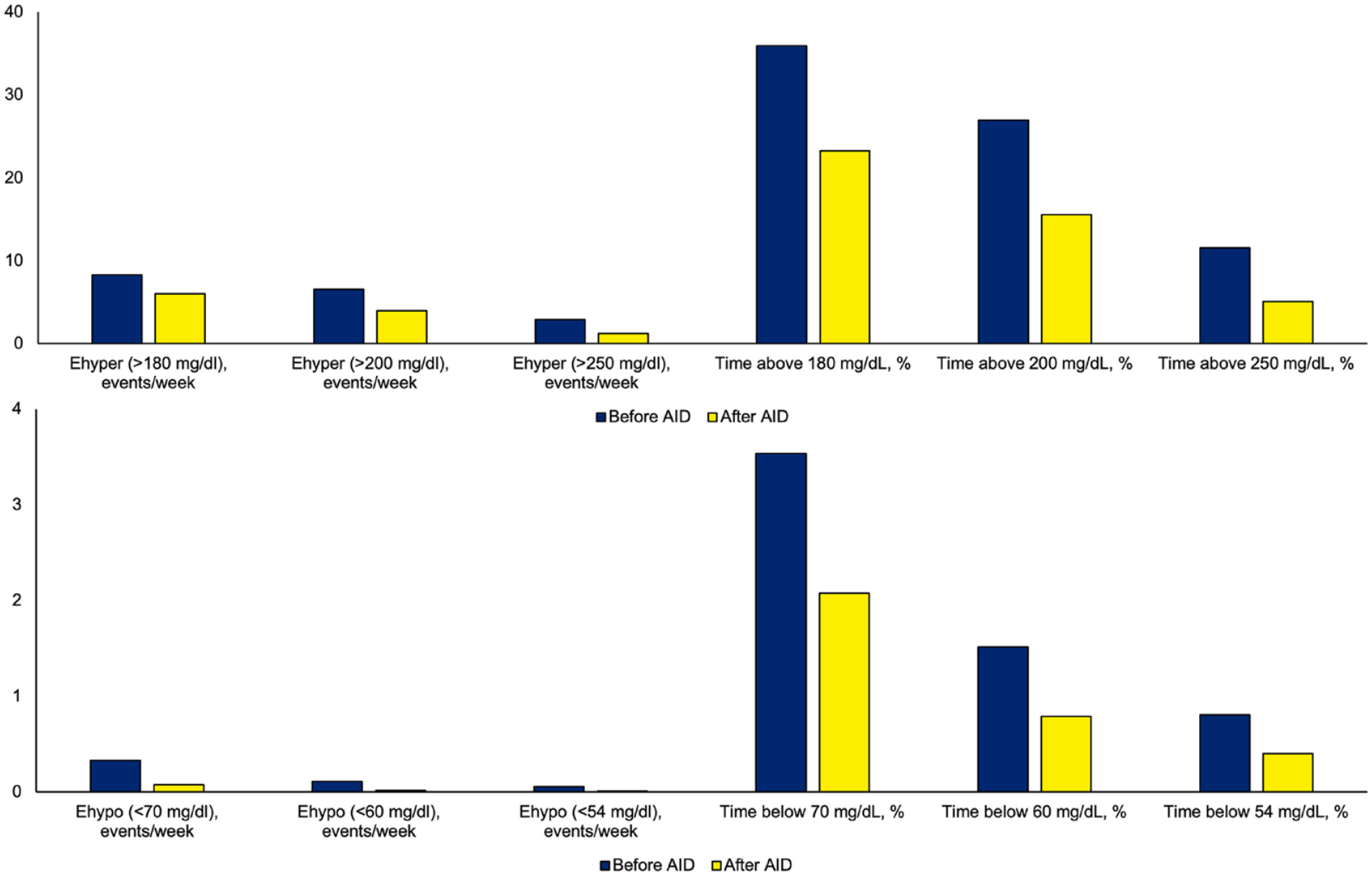
Extended hypoglycemia (Ehypo), extended hyperglycemia (Ehyper), time below, and time above range metrics before and after automated insulin delivery initiation. Columns represent mean values of metrics, and all metrics reduced significantly after AID initiation (P < .001).
Correlation between metrics was analyzed separately for before AID and after AID. In both time intervals, Ehypo and Ehyper metrics correlated to mean glucose and GMI similar to time below and above range metrics; however, time below range metrics had higher correlation than Ehypo metrics (Figure 2). Similarly, CV correlated better with percent time below ranges than Ehypo; however, Ehyper and time above range metrics were comparable (Figure 2).
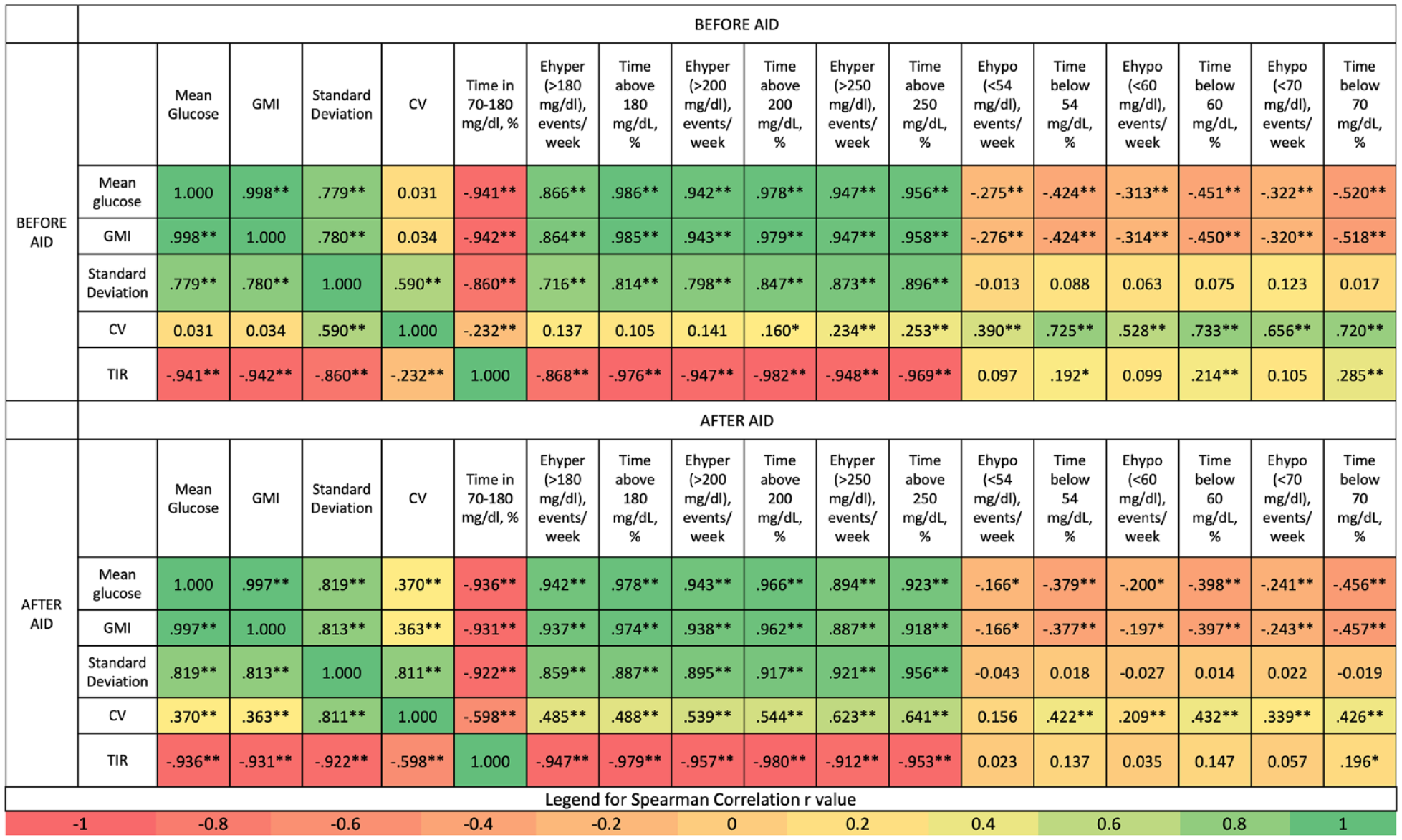
Correlation between CGM metrics. Correlation of metrics analyzed separately for before AID and after AID initiation. Spearman correlation r values were shown in cells with a heatmap. The significance of correlation is *P < .05, and **P < .01.
Correlation between percent time range metrics and extended metrics was compared (Figure 3). Ehypo metrics correlated better with other Ehypo metrics with different cutoffs than any time below range metrics. Similarly, time below range metrics correlated better with other time below range metrics with different cutoffs than any Ehypo. As expected, time below range metrics had stronger inverse correlation with hyperglycemia metrics (time above range and Ehyper) than Ehypo metrics (Figure 3). There was a strong positive correlation between Ehyper (>250 mg/dL) and mean glucose (before AID: r = 0.947, after AID: r = 0.894), GMI (before AID: r = 0.947, after AID: r = 0.887), and TAR (>180 mg/dL) (before AID: r = 0.957, after AID: r = 0.917) and a strong positive correlation between Ehypo (<70 mg/dL) and TBR (<70 mg/dL) (before AID: r = 0.823, after AID: r = 0.608) before and after AID use, respectively (Figures 2 and 3).
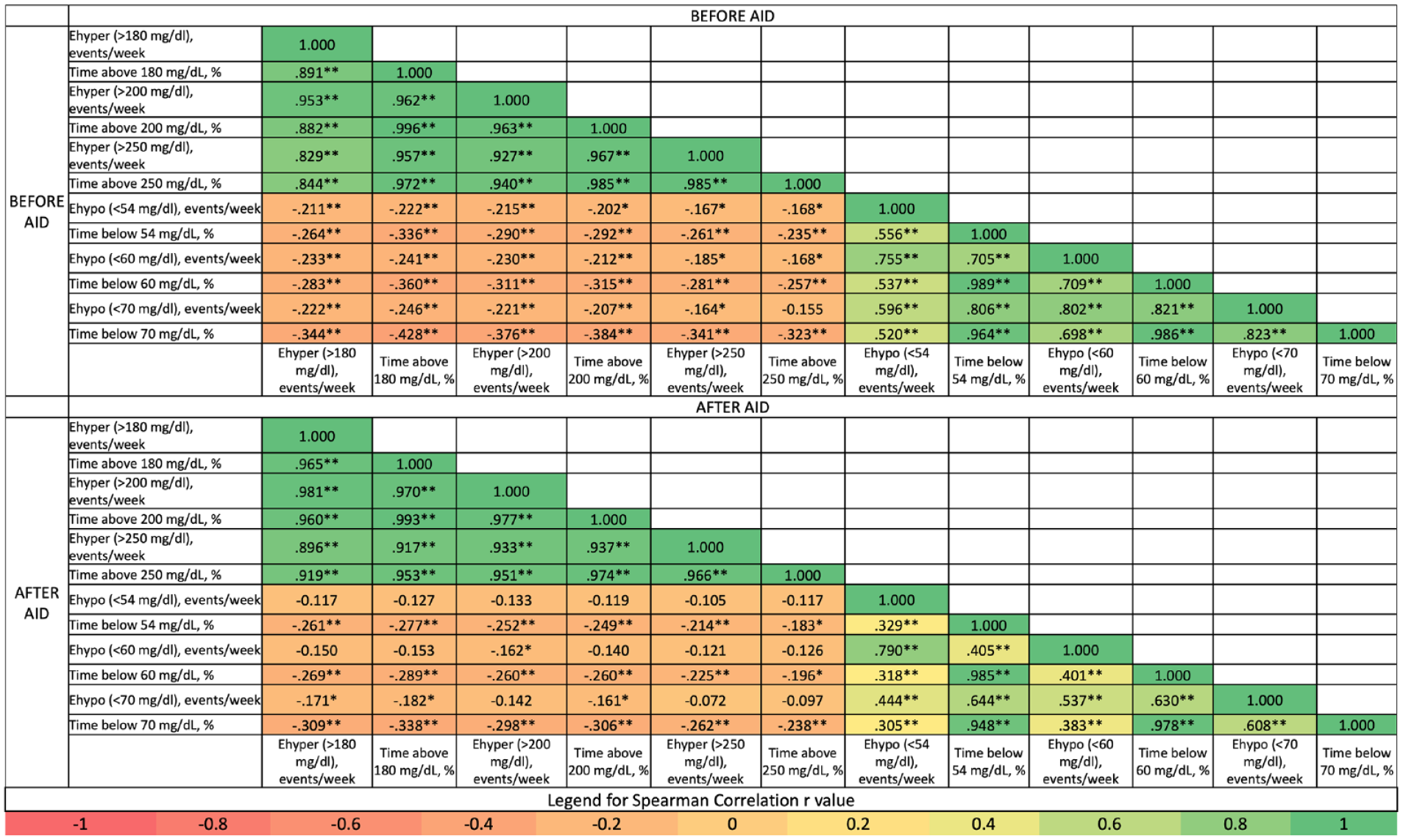
Correlation of hypoglycemia and hyperglycemia metrics. Spearman correlation r values were shown in cells with a heatmap. The significance of correlation is *P < .05, and **P < .01.
Discussion
This is the first study that evaluated newly defined CGM metrics, Ehypo and Ehyper. We showed improvement of Ehypo and Ehyper after AID initiation in parallel with other CGM metrics. As expected, Ehypo and Ehyper have a strong positive correlation with hypoglycemia and hyperglycemia CGM metrics, respectively. Our results are in parallel with our previous research where we showed improvement of glycemia risk index and other CGM metrics with AID initiation. 11 We can speculate that since AID systems have the feature of suspending insulin in response to predicted hypoglycemia and increasing insulin in response to predicted hyperglycemia, Ehypo and Ehyper events significantly decreased respectively. Studies with longer duration and larger samples are necessary to validate our observations in this study and assess the relationship of Ehypo and Ehyper with the risk of severe hypoglycemia and diabetic ketoacidosis, respectively. Especially, further evaluation of Ehypo in relationship to severe hypoglycemic events may be helpful to find CGM metrics that correlate better than TBR since recently some concerns were raised about detecting severe hypoglycemic events with current cut offs of and duration of analysis for TBR.12,13 We believe clinical value and usefulness of Ehyper and Ehypo can be determined better after these studies.
As these new metrics have been evolving, target cutoffs for these metrics, minimum duration to count events are unknown and 120 minutes required duration for extended events has not been validated or shown in previous studies. The International Consensus suggested that Ehypo should have no maximum agreed duration, during periods of extended hypoglycemia, and any periods of hypoglycemia <54 mg/dL should be reported separately. 7 We noted a limitation of these metrics that if they are calculated per week, event numbers are very low, and comparison of the events may be harder in clinical practice. The International Consensus did not have any suggestion on how often these metrics should be calculated (weekly, bimonthly, monthly, etc). In addition to the defined cut offs for Ehypo (<70 mg/dL) and Ehyper (> 250 mg/dL) by the International Consensus, we also evaluated different cut offs such as Ehypo (<54 and <60 mg/dL) and Ehyper (>180 and >200 mg/dL) for exploration. These exploratory cut offs were well correlated with the defined cut offs.
Strengths of this study include a large sample of people with T1D using the same CGM and AID after AID initiation and being the first study in the literature to evaluate newly defined CGM metrics of Ehypo and Ehyper. Limitations of this study include retrospective nature of the study, not having any information of participants’ diabetes management skills, and acute diabetes complication rates (severe hypoglycemia and diabetic ketoacidosis).
In summary, Ehypo and Ehyper are newly defined CGM metrics for clinical trials as secondary endpoints for continuous outcomes. Automated insulin delivery use has significantly decreased Ehypo and Ehyper events compared with MDI and non-AID insulin pump use in people with T1D. Ehypo and Ehyper have strong positive correlations with CGM metrics of hypoglycemia and hyperglycemia, respectively. Further research is required to establish the clinical value of these metrics.
Footnotes
Acknowledgements
We appreciate our patients who shared their CGM and AID data with our clinic.
Abbreviations
AID, automated insulin delivery; CGM, continuous glucose monitor; GMI, glycemic management indicator; HbA1c, hemoglobin A1c; MDI, multiple daily injections; T1D, type 1 diabetes; TAR, time above range; TBR, time below range; TIR, time in range; Ehypo, extended hypoglycemia; Ehyper, extended hyperglycemia.
Author Contributions
Conception and design of the study was done by KEK and HKA, acquisition was done by KEK, EM, HKA, analysis and interpretation of data was done by KEK, HKA, and JS-B, and drafting the paper and final approval of the paper are done by all authors. HKA has full access to all study data and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: HKA reports receiving research support and consultation fees from Tandem Diabetes Care, Medtronic, and Dexcom. KEK, EM, JS-B disclose no conflict of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
