Abstract
Ongoing innovation in diabetes technologies has led to the development of advanced tools such as automated insulin delivery (AID) systems that adjust insulin delivery in response to current and predicted glucose levels, residual insulin action, and other inputs (eg, meal and exercise announcements). However, infusion sets continue to be the “Achilles heel” of accurate and precise insulin delivery and continued device use. A recent study by Kalus et al (DERMIS Study) revealed higher vessel density and signals of inflammation by optical coherence tomography (OCT), in addition to increased inflammation, fat necrosis, fibrosis, and eosinophilic infiltration by histopathology. Although the study provided a comprehensive description of what was happening, the results raise important questions that require additional research. On February 29, 2024, the Leona M. and Harry B. Helmsley Charitable Trust sponsored a conference to begin addressing these issues. This article summarizes the DERMIS study findings and testing methodologies discussed at the conference and proposes the next steps for developing insulin infusion sets that reduce the variability in insulin delivery and extend wear.
Introduction
Ongoing innovation in diabetes technologies has led to the development of advanced tools such as automated insulin delivery (AID) systems that adjust insulin delivery in response to current and predicted glucose levels, residual insulin action, and other inputs (eg, meal and exercise announcements). 1 These devices have been shown to be safe and effective in helping individuals safely achieve their glycemic targets.2-7 However, infusion sets continue to be the “Achilles heel” of accurate and precise insulin delivery and continued device use.8-12
Kalus et al 13 recently reported findings from the DERMIS (DEfining, Reviewing, and Monitoring skin pathology in type 1 diabetes Study) that was conducted to better understand the skin changes that occur from chronic CSII use and try to answer why long-term insulin flow into the insertion site becomes impaired. Using a combination of non-invasive optical coherence tomography (OCT), histological analyses of skin biopsies, and metabolomics, the authors investigated changes to the epidermal, dermal, and subcutaneous tissues in 30 adults with type 1 diabetes (T1D) with 15.8 ± 11.9 years duration of insulin pump use. The analyses revealed higher vessel density and signals of inflammation by OCT, in addition to increased inflammation, fat necrosis, fibrosis, and eosinophilic infiltration by histopathology.
Although study results comprehensively describe what is happening, they raise important questions that require additional research. For example, tissue samples from patients with >20 years of insulin pump use showed significantly fewer eosinophils than those with <10 years of wear. Was this due to early discontinuation, or are there specific patient phenotypes that are less susceptible to the immune response to tissue trauma? Moreover, it remains unclear whether the response is due to the trauma itself and/or to the designs and/or materials used in current infusion set products.
On February 29, 2024, the Leona M. and Harry B. Helmsley Charitable Trust sponsored a conference to begin addressing these issues. Hosted by the University of Washington Diabetes Institute, the conference included presentations from diabetes specialists and researchers with expertise in histology, OCT technologies, metabolomics, and other related fields, who met to discuss the findings from the DERMIS study.
This article summarizes the DERMIS study findings and testing methodologies discussed at the conference and proposes the next steps for developing insulin infusion sets that reduce the variability in insulin delivery and extend wear.
Anatomy and Progression of Insulin Infusion Set Failure
Physiologic Responses to Catheter Insertion
Background
Current insulin infusion sets (IISs) remain the weak link in successful use of AID therapy. Skin reactions at the catheter insertion site, resulting in pain and infection, insulin absorption variability, and catheter/needle blockage can adversely affect patient safety and metabolic control. Although manufacturers, medical societies, and regulatory agencies recommend changing IIS every two to three days, a substantial proportion of patients discontinue using their AID device due to insertion site issues. 8
Mechanisms of insulin absorption
The lymphatics are open channels within the immune system that allow for the absorption of insulin hexamers, dimmers, and monomers. Although hexamers typically take longer to be absorbed through the lymphatic system, edema fluid in the wound area increases hydrostatic pressure, which in turn increases lymphatic absorption. It has been hypothesized that even hexamers can be absorbed lymphatically through the fenestrated capillaries of local lymph nodes to reach the bloostream, thereby eliminating the need to travel through multiple lymph nodes and the thoracic duct. The connective tissue acts as a mechanical barrier through which the insulin must diffuse to reach the healthy capillaries and lymphatics. Dynamic changes in local capillary blood flow and flow into local lymphatic vessels cause variable insulin absorption into the systemic circulation.
Effects of insulin infusion set insertion on insulin absorption
An animal study that evaluated both straight and angled commercial Teflon catheters demonstrated that when the catheter was first inserted, the connective tissue was compressed around the catheter orifice, making it difficult for the insulin to diffuse out into the surrounding tissue. 14 An acute inflammatory response was triggered by the initial piercing of the epidermis, dermis, and subcutaneous tissue with a steel introducer needle and Teflon catheter. The infusion of insulin, excipients, and insulin fibrils also triggered an acute inflammatory reaction. Needle and catheter insertion damaged epithelial cells, adipose cells, capillaries, lymphatics, connective tissue, and extracellular matrix. Damaged adipose cells released plasma membranes, cytoplasm, organelles, deoxyribonucleic acid (DNA), ribonucleic acid (RNA), triglycerides, and fatty acids into the adjacent subcutaneous tissue, which led to activation of the cellular and humoral immune systems.
The subcutaneous tissue around the catheter insertion site changed dynamically over wear-time. The thickness of inflammatory tissue surrounding the inserted catheter increased rapidly, starting with a small amount of blood around the insertion site. The accumulation of thrombus, tissue debris, inflammatory cells, and the deposition of fresh collagen required insulin to travel along the path of least resistance toward intact and functional capillaries and lymph vessels. By day 2, the surrounding layer of inflammatory tissue reached almost a millimeter thickness, with white blood cells (neutrophils, macrophages, lymphocytes, eosinophils) infiltrating the area.
Over time, collagen deposition from fibroblasts became denser, attempting to wall-off the foreign body. Pressure within the pump tubing increased over time due to the surrounding layer of inflammatory tissue becoming thicker, denser, and less compliant. Back pressure in the pump tubing provided an estimate of the inflammatory response and the resistance to the flow of fluid.
The thickness and area of inflammation around an inserted Teflon catheter plateaued after two to four days in some catheters and after seven days in others. A thick, dense, and non-compliant layer of inflammatory tissue typically produced high pump tubing back pressure and limited insulin flow into adjacent vascular tissue. It was not uncommon for the insulin to migrate upward along the IIS onto the skin surface when pump tubing pressure was very high. Fibrinolysis and the formation of cracks/fissures in the surrounding layer of inflammatory tissue produced a lower pump tubing back pressure and more diffuse spread of insulin into adjacent subcutaneous tissue.
Capillary blood flow typically increases in an area of acute inflammation due to the high metabolic needs of the macrophages, neutrophils, eosinophils, lymphocytes, and fibroblasts. Interstitial fluid flow into adjacent lymphatic vessels typically increases in an area of acute inflammation due increased capillary permeability/leakage, leading to increased hydrostatic and oncotic pressure. In summary, insulin absorption from the subcutaneous tissue into the systemic circulation changes dynamically over time due to changes in the anatomy and physiology of the tissue surrounding the inserted catheter.
Influence of insertion sites on insulin infusion set longevity
The investigators also found a significant difference in tissue response and insulin bolus distribution between straight 90-degree catheter insertions and 30-degree angled catheter insertions. Although the angled catheters were more than twice the length of the straight catheters (13 mm vs 6 mm), there was a 50% reduction in the thickness and volume of the layer of inflammatory tissue surrounding an angled catheter. This difference may have been caused by ongoing tissue trauma by the 90-degree Teflon catheter tip hitting underlying muscle facia during body movement, while angled catheters tended to cause less ongoing mechanical tissue damage. Investigators also observed differences in the degree of inflammation, capillary density, and the spread of insulin based on the site of catheter insertion.
Summary
The wear time of an IIS may be prolonged by overcoming the initial acute inflammatory response and, thus, eliminate the associated variability in insulin absorption and premature catheter failure. An ideal IIS would produce rapid and consistent absorption of insulin from the subcutaneous tissue into the bloodstream from dose-to-dose and day-to-day for more than seven days and minimize the formation of scar tissue that may adversely affect insulin absorption and future pump use.
DERMIS Study: Methods and Results
Histopathology and Immunohistochemistry
Background
The DERMIS study enrolled 30 patients with T1D using CSII. Most patients who participated in the DERMIS study reported experiencing itching at the site. 13 Others experienced varying levels of discomfort, redness on their skin at the insertion site, and some patients described feeling a nodule under the skin at the pump site. Approximately one quarter of patients described the skin opening up at the pump site.
Methods and results
The assessments performed in the DERMIS study were conducted on three specific skin sites: current site, recovery site, and control site. The current site was where the infusion set was in place for approximately 72 hours before being removed for the biopsy. The recovery infusion site was marked by the patient when the infusion set had been removed 72 hours prior to the biopsy. The control site was located 15 cm below the axilla on the side of the rib cage, where no IIS or insulin injection had been performed.
The histologic analysis involved examining the thickness of the skin, fibrosis, fibrin deposition, inflammation, vasculature, and fat necrosis. Immunohistochemistry stains were then used to analyze the expression of specific proteins in the skin samples, which allowed for the visualization of key proteins (eg, insulin-like growth factor [IGF], transforming growth factor-beta [TGF-β], and interferon) in the different cell types. Statistical analysis was performed to compare the findings between current, recovery, and control sites (Figure 1).
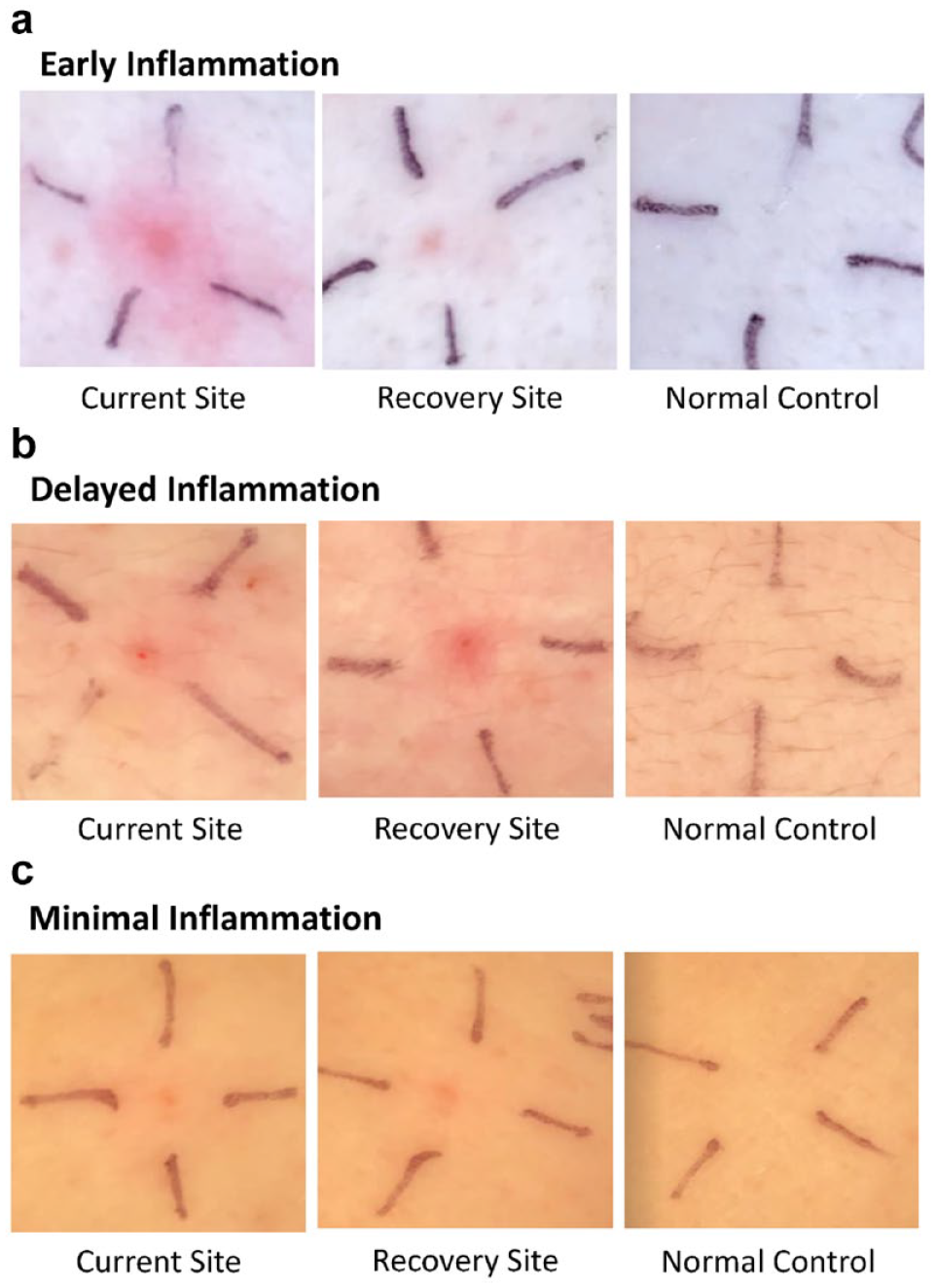
Assessment of infusion sites found varying degrees of inflammation in time and patients.
Images showed a range of visible changes at the infusion sites, which varied between patients. Some patients exhibited inflammation early (Figure 1A), which lessened over time, while others experienced delayed inflammation that was evident in the recovery infusion site (Figure 1B). Other patients showed minimal inflammation at both sites compared with the control site (Figure 1C).
Histology assessments
A significant change was observed among the inflammatory cells, with a high number of eosinophils seen in 74% of the current infusion sites and 75% of the recovery sites. There were no eosinophils present in the control sites. Eosinophils are not normally present in the skin, so their presence suggests an abnormal immune response. Interestingly, patients who were using an insulin pump >20 years had significantly fewer eosinophils than those with <10 years of insulin pump use. This may suggest that long-term users develop a tolerance to the infusion sets and that those with shorter duration of insulin pump therapy have not yet achieved that level of tolerance and stopped using pumps due to skin reactions driven by eosinophils. However, this may also be due to something specific about the people who have been using an insulin pump for a long time. The histology evaluation in all patients also showed that the infusion sites had more dermal thickness, more fibrosis, more fibrin deposition, fat necrosis, and increased vascularity.
Immunohistochemistry assessments
The immunohistochemical analysis confirmed that the infusion sites had significant changes compared with control skin. Transforming growth factor-beta was heavily expressed in fibroblasts and keratinocytes, which can contribute to fibrosis and scarring. The IGF, which was expressed mostly in fibroblasts, promotes keratinocyte migration and tissue repair. The TGF-β3 was found to be present in fibroblasts and keratinocytes.
Summary
Itch and eosinophilic inflammation was very common at pump sites in our study. Images showed significant variability between patients at the infusion site, ranging from minimal inflammation to more severe inflammation. Although the histology evaluation showed increased dermal thickness, fibrosis, fibrin deposition, vascularity, and inflammation, the high number of eosinophils at the infusion sites was notable because it indicated an abnormal immune response. The immunohistochemical stains revealed the expression of cytokines involved in wound healing, such as IGF and TGF-ß. Although it is speculated that long-term insulin pump users may develop a tolerance for infusion sets over time, it is possible that this tolerance is only achieved in specific patients.
Optical Coherence Tomography
Background
Optical coherence tomography and optical coherence tomography angiography (OCTA) were used to extract information about the vascular density, surface roughness, epidermis thickness at the patients’ current, recovery, and normal control sites. 13 The procedures were performed prior to the histopathologic analyses.
OCT/OCTA
The OCT is a non-invasive technique that uses near infrared (NIR) light waves to create high-resolution, three-dimensional (3D) images of human tissue. 15 The scanning procedure is fast, and the images can be obtained in seconds. The OCTA is a functional extension of OCT that enables blood flow mapping, utilizing the flowing blood cells within vessels as the intrinsic contrast agent to enable visualization of vascular networks without the need for dye injection. The resulting OCTA image provides information about the vascular network, vessel density, and any abnormalities or changes in blood flow.
Results
The OCT/OCTA analyses showed increased blood flow and vascularization at the current and recovery sites compared with the control site and between the current and recovery sites. These findings suggest that there may be angiogenesis and inflammation occurring at the pump site. The images reveal increased blood flow and reduced optical attenuation at the current and recovery sites compared with the normal control site. (Figure 2) Although epidermis thickness was enhanced in the recovery site, it did not show a significant difference between the current site and the normal site.
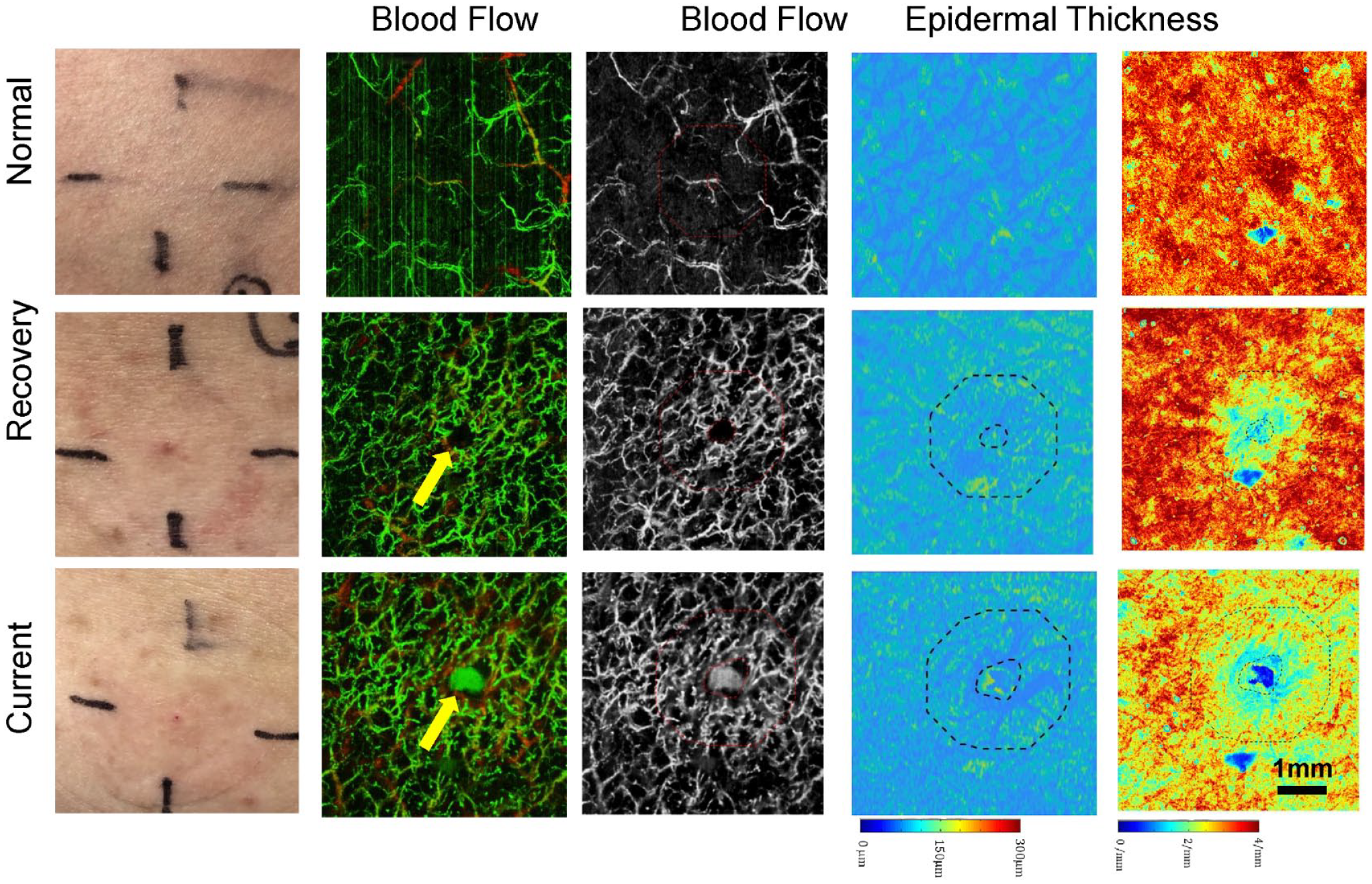
OCT/OCTA images of changes in blood flow, vascularization, and epidermal thickness at the test sites.
Left column shows the photographs taken at the skin sites for imaging. The second column shows the OCTA blood flow images with depth-color coded vessel information where green color indicates more superficial vessels and red color indicates more deeper vessels. The third column shows the corresponding OCTA enface images (ie, depth-collapsed projection) for quantification purpose. The fourth column shows the corresponding epidermal thickness maps. And the last column shows the corresponding optical attenuation maps of the scanned skin tissue.
Summary
Results from the OCT/OCTA analyses confirmed the findings from the histopathologic studies, which also showed increased inflammation at the pump sites. Importantly, the results demonstrated the usefulness of OCT/OCTA in non-invasive imaging and its potential for studying changes in vascularization and blood flow.
Potential Causes of Glycemic Variability and Insulin Infusion Set Failure
Timing of Catheter Changes
Background
A notable drawback to CSII is the systematic shift in the pharmacokinetics (PK) and pharmacodynamics (PD) of insulin as infusion sets over two to four days of use.16-18 The variation in insulin absorption and subsequent action is one of the factors contributing to glucose variability, 19 potentially leading to both hyperglycemia and hypoglycemia. Investigators recently reported findings from two studies that utilized a combination of ex-vivo analyses 20 and data from a clinical trial 21 to investigate insulin absorption in the subcutaneous depot.
Methodologies and findings
Ex-vivo
The ex-vivo study investigated the utility of a new, real-time, ex-vivo method to obtain real-time follow-up of insulin subcutaneous propagation. 20 Investigators utilized explants from post-bariatric surgery patients, which were imaged a micro-CT scanner to study the morphology of the insulin depot. 20 The images were assessed using an investigator-developed index of dispersion (IoD) to measure the surface-to-volume ratio of the insulin depot (Figure 3).
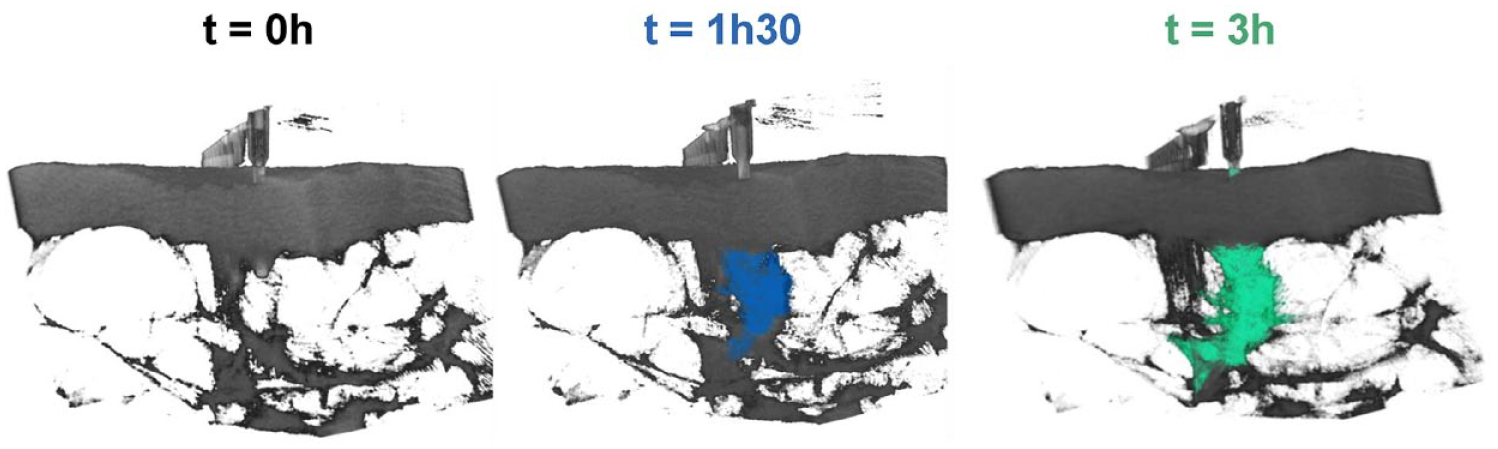
Example of 3D images of insulin diffusion over time.
This example presents a 3D aspect of insulin depots in an explant at 0 hour, 1 hour 30 minutes, and 3 hours of insulin infusion. Investigators reported that IoD increases with time for all injections. Several minor bubbles, one occlusion, and pressure elevations were also observed.
Clinical approach
In the clinical approach, investigators analyzed data from the IMPLIQUE Study 21 to investigate the effects of catheter change on glucose excursions, both in standard insulin pump delivery (in open-loop mode) and while using a closed-loop system, in real-life settings. The system collected continuous glucose monitoring (CGM) data, pump memory data (eg, basal rates, insulin needs, etc), and information about catheter changes. The goal was to observe the glucose excursion after catheter changes in open-loop, in real-life settings and determine whether the closed-loop system could mitigate this phenomenon. Investigators observed a significant increase in the percentage of time spent above the glucose threshold on the day of catheter change compared with the following days, both in the open-loop and closed-loop settings.
Investigators also analyzed the timing of bolus administration in relation to catheter changes. Patients were categorized into groups based on whether they administered bolus insulin before catheter change, after catheter change, or both before and after catheter change (Figure 4).
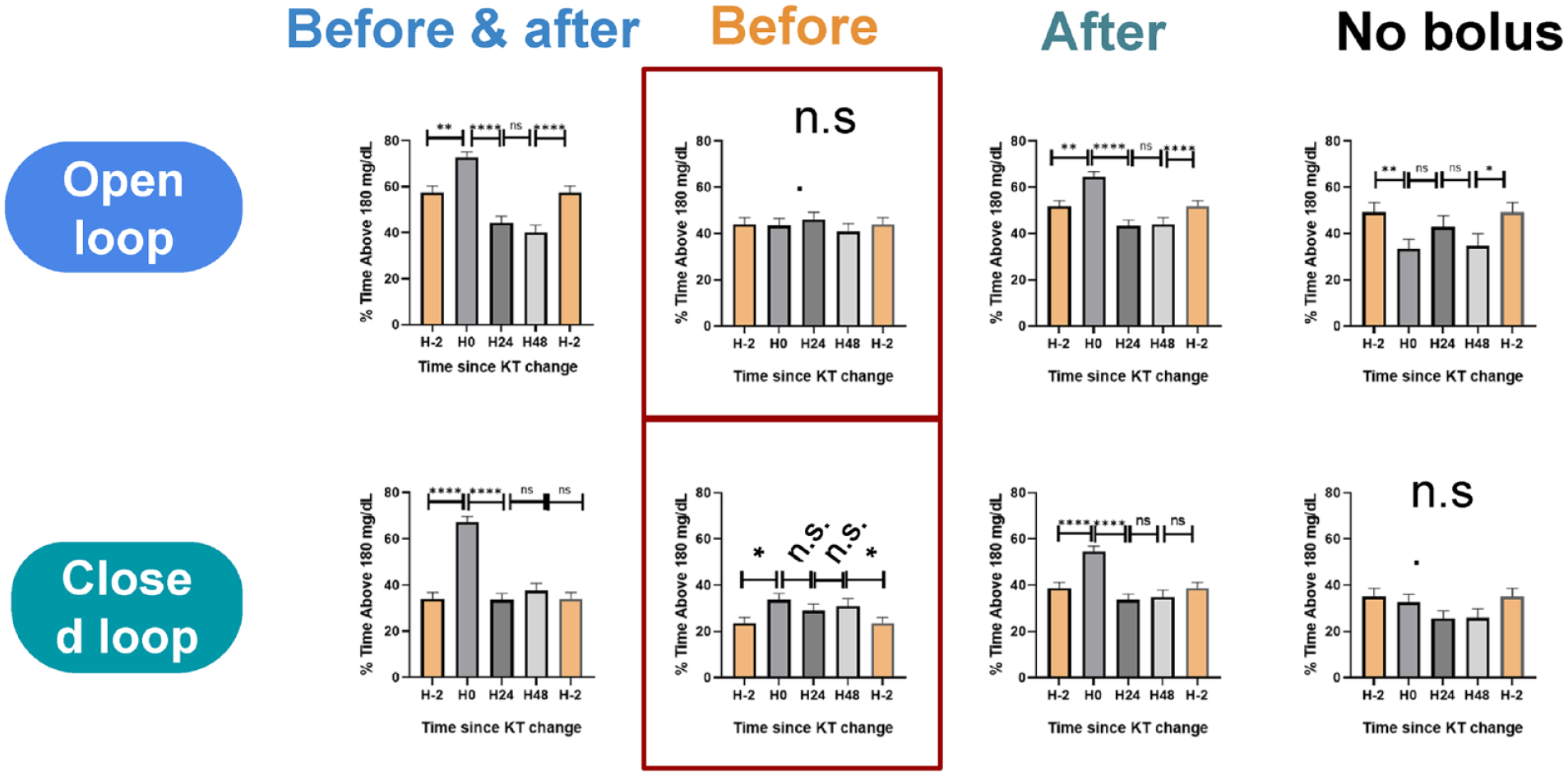
Timing of bolus insulin administration relative to catheter changes.
No significant between-group difference in glucose excursions were observed when patients administered bolus insulin before a catheter change. However, when bolus insulin was administered after or before and after catheter change, there was an increase in glucose excursion compared with other days.
Summary
Findings from the ex-vivo study support the use of this method to compare infusion parameters such as pump model, basal rate, catheter characteristics, infusion site characteristics, or patient phenotype. The clinical approach provided insights into the impact of catheter changes on glucose excursion in real-life settings. These results highlight the importance of considering the timing of bolus insulin administration and suggest that administering the bolus before catheter change may help prevent glucose excursions.
Phenol Preservatives
Background
Phenol preservatives (PPs) such as m-creosol and phenol used in commercial insulin preparations induce tissue reactions and cellular cytotoxicity, which may contribute to variability in blood glucose and permanent loss of injection or infusion site architecture and function.22-25 However, because PPs are required to stabilize the insulin protein and promote shelf-life, eliminating these formulations is not a viable solution.26,27
Current findings
In a study that used human peripheral blood mononuclear cells (PBMCs), Kesserwan et al 24 showed the cytotoxic effects on the PBMCs after three days of exposure to concentrations of rapid-acting and pure insulin, demonstrating that preservatives play a role in tissue inflammation (Figure 5).
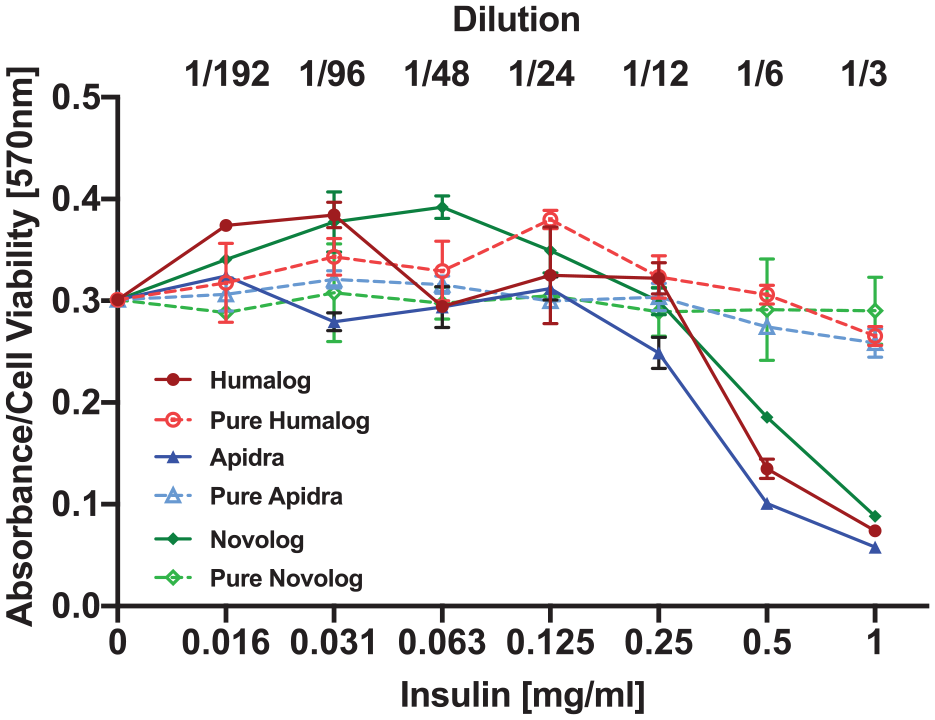
Decreasing cell viability in response to increasing insulin concentrations.
Investigators also utilized streptozotocin (STZ) induced diabetic mice to evaluate the tissue and the effects of saline, PP, insulin lispro, and filtered insulin lispro on inflammation in a modified air pouch which allows for the collection of tissue, cell, and fluid contents at injection sites. 25 In vivo data showed that insulin fibrils, which are formed when insulin is agitated or shaken, are cytotoxic in vitro and induced inflammation in the murine air-pouch model, which leads to macrophage-mediated uptake of fibrils both in vitro and in vivo. 28 These data also indicated fibril cytotoxicity is highly cell type-dependent. 28 Both insulin-derived fibrils and phenolic preservatives can lead to dermatopathological changes, resulting in poor glycemic management and maladaptive wound healing. 28
Summary
Insulin preservatives significantly impact tissue inflammation and netosis, which refers to the formation of neutrophil extracellular traps. When delivering 100% insulin-derived fibrils, it was found to be cytotoxic, specifically in macrophages. Insulin fibrils will most likely exacerbate the tissue reaction at insulin delivery sites in the presence of PP.
Allergens in Medical Devices
Background
Allergic contact dermatitis is a delayed-type hypersensitivity reaction that occurs when small (<500 daltons) molecular weight organic chemicals or metal ions penetrate the skin and complex with proteins, triggering an immune response and the development of a rash. The eosinophilic inflammation reported in the DERMIS study raises concern about possible occurrence of delayed-type hypersensitivity reaction.
Allergens identified in insulin pumps
Medical devices comprise a complex composition of plastics, glues, and metals, which can potentially contain allergens that may trigger delayed-type hypersensitivity reactions in susceptible individuals. However, the chemical substances used in insulin pumps are difficult to determine due to lack of legislation requiring companies to disclose inclusion of these substances. 29 Nevertheless, several allergens have been identified in insulin pumps (Table 1).
Contained in some older insulin pumps.
The deeply situated nature of the eosinophils in the skin biopsies and the lack of eczema on the surface of the infusion site suggests that the potential allergen is being delivered at the tip of the catheter. Colophonium-related substances, such as methyldehydroabietate, have been detected in extracts acquired from the insulin reservoir and sensor adhesive of the Medtrum A6 Touchcare System. 32 Contact dermatitis to dipropylene glycol diacrylate (DPGDA) has been reported in three Omnipod insulin pumps with the chemical found the adhesive patches and the pumps themselves. 31 Isobornyl acrylate (IBOA) is found in the adhesives used to assemble some insulin pumps and may also be introduced as a manufacturing impurity. 30 Chemical analyses of several devices identified IBOA in a variety of components including the delivery pump, electronics within the unit, cannula connection, adhesive patch that connects the device to the skin, and the plastic unit.32-35 Because these devices have numerous constituent parts and are manufactured in an era of complex medical device supply chains, it is difficult to know all of the chemicals that are used in their production. There are likely other chemicals yet undefined within these devices that could elicit a delayed type of hypersensitivity response.
Methods for allergen testing in medical devices
Various diagnostic techniques can be used to identify common allergens found in medical devices. Ultrasonic bath extraction involves subjecting various parts of the insulin pump to an ultrasonic bath extraction. The extracted material can then be used for traditional patch testing. Further chemical analysis of the extract then can be performed using gas chromatography-mass spectrometry (GC-MS).36,37
Thin-layer chromatography (TLC) is another chromatographic technique that separates and analyzes mixtures. In this method, the extract obtained from the ultrasonic bath extraction is applied to a thin layer of adsorbent material. The extract is then allowed to migrate on the plate, and different components separate based on their affinity for the adsorbent material. The separated components can be transferred onto patches and used for patch testing. 36
Summary
Infusion site failures may be due, in part, to known and yet to be identified allergens in the chemicals and components used in insulin pumps, Further research and testing are required to identify these potential allergens.
Tissue Flow Resistance
Background
Tissue flow resistance (TFR) is a parameter that measures the counter pressure created by the tissue when insulin is infused into it. The TFR can be calculated by comparing the baseline pressure with the pressure required to infuse insulin into the tissue. Recent studies show that TFR may be related to insulin absorption and can potentially be used as a measure of insulin absorption efficiency.
RHEO study
Regittnig et al 38 assessed the association between TFR and insulin absorption in individuals living with T1D during prolonged use of insulin infusion sites. Participants wore two insulin pumps during the seven-day observation period. One pump was filled with insulin aspart, the other with an insulin diluting medium (IDM). The TFR for both infusion sites were assessed daily. Results are presented in Figure 6.
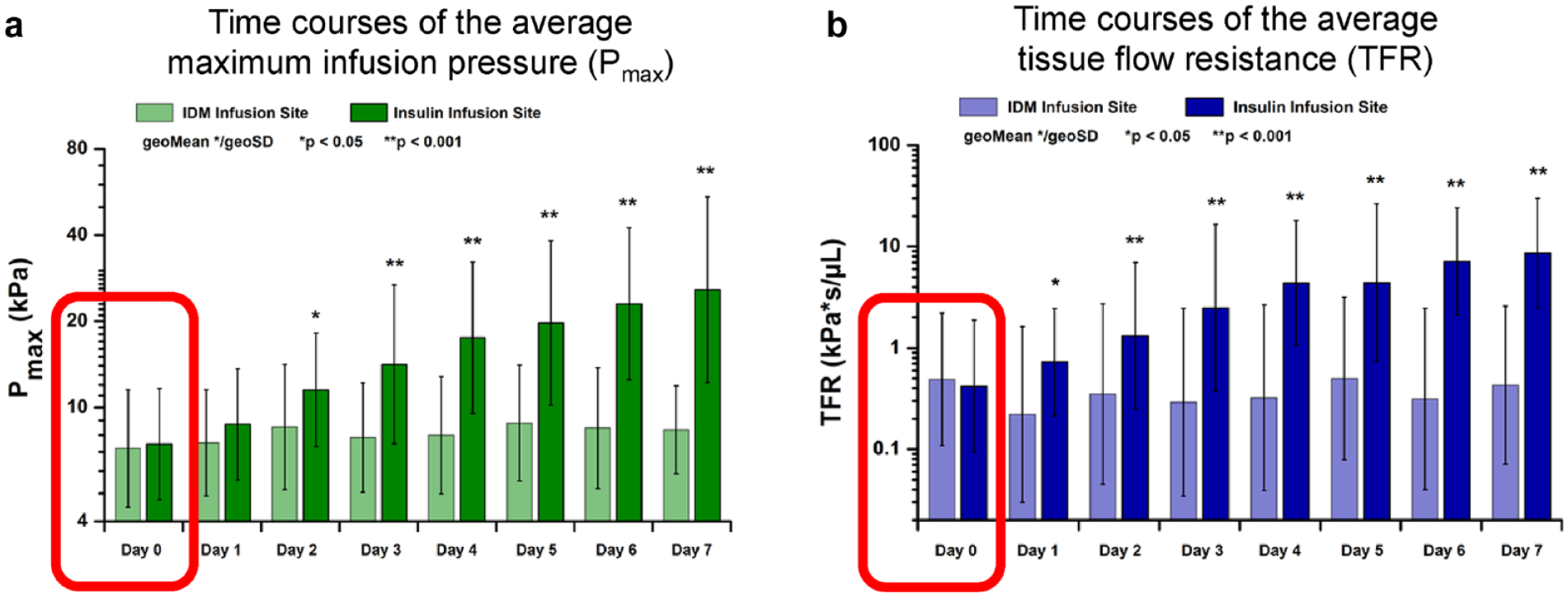
Time courses of the average Pmax (A) and TFR (B). 38
On day 1, the average maximum pressure values and TFR observed during bolus delivery into tissues (Pmax) for both infusion sites were similar. Over the subsequent days, basal levels at the IDM site remained level, whereas the (Pmax) and TFR at the insulin infusion site progressively increased and fluid leakage was detected at 7 of 30 insulin infusion sites (23.3%); no fluid leakage at the 30 IDM infusion sites.
HYDRA study
Investigators assessed the TFR at the insulin infusion site to determine its relationship to insulin absorption. The study included 10 individuals with T1D who used an insulin pump for up to 13 days without an infusion set change. The TFR was measured daily until TFR exceeded 10 × TFRbasal. The PK and PD during a 75 g oral glucose tolerance test (OGTT) on the first and last study day were assessed (Figure 7).
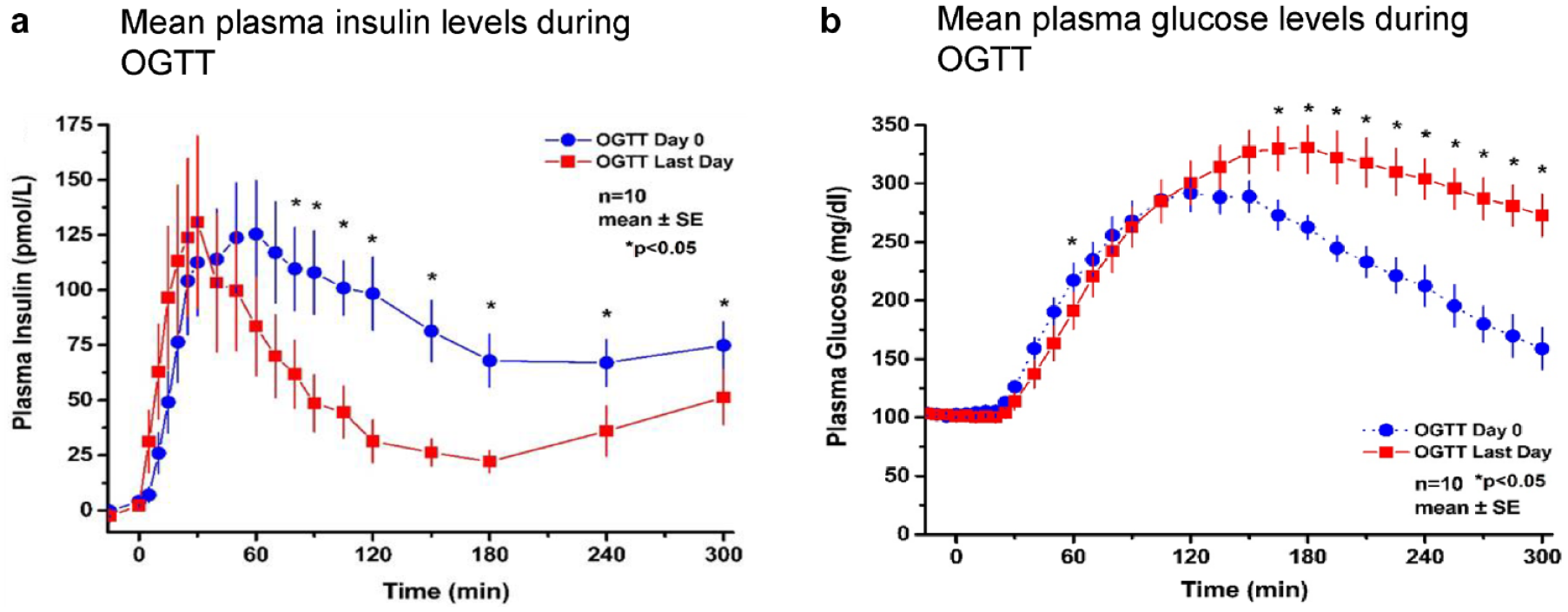
Mean plasma insulin and glucose levels during OGTT (unpublished data).
The average TFR values progressively increased over the observation period. The OGTT results showed a significant decrease from baseline in plasma insulin at two hours with a significantly increased glucose excursion (Figure 7A). Regression analysis showed a strong negative relationship between TFR and insulin area under the curve (AUC), r = −0.666, and a strong positive relationship between TFR and mean glucose during OCTA at 3 to 5 hours, r = 0.815, both P < .001 (Figure 7B).
Pathophysiological correlate of increased tissue flow resistance and reduced insulin absorption kinetics
Regittnig et al are also assessing the relationship between TFR and insulin absorption through a biopsy study using an OGTT. The study includes 22 individuals with T1D who were asked to wear the same infusion set until they experienced clinical symptoms of site failure. The TFR was measured daily using a pressure sensor, which showed increasing pressure over time. Participants underwent OGTT on days 1, 6, and the last day when infusion site failure occurred. On the last day, a biopsy was taken from the tissue surrounding the insulin infusion catheter. Preliminary results showed a strong relationship between the log-transformed TFR, decreased plasma insulin AUC, and increased plasma glucose AUC during the OGTT; plasma glucose levels were the highest on the last day.
Summary
Insulin induces a progressive increase in subcutaneous tissue resistance to the introduction of fluid. 38 Insulin absorption is decreased with increasing duration of infusion site use. Regression analyses showed that TFR is strongly correlated with the plasma insulin levels and the mean plasma glucose concentration during the OGTT. Monitoring of TFR using a pressure sensor may allow the estimation of the prevailing insulin absorption efficiency and, in turn, determination of the optimal usage duration of an insulin infusion site. Results from these studies have important implications for the future design of insulin pumps and infusion sets.
Minimizing the Effects of Scarring Through Tension Off-Loading
Background
When a wound occurs, fibroblasts migrate to the site and produce collagen, which forms the structural framework of the healing wound. Although this process facilitates wound closure, deposition of excess, abnormal extracellular matrix (ECM), driven in part by the interaction between fibroblasts and injury-responsive inflammatory cells (including lymphoid cells, eosinophils, and other myeloid cells), ultimately leads to the development of fibrosis (scar tissue). Fibrosis at the site of an insulin pump is caused by three broad insults: (1) insertion of the trocar creates a response to injury that initiates inflammation and fibrosis; (2) repeated deliveries of insulin are a second pro-inflammatory insult; and (3) during the course of the time that the cannula is in the skin, the body begins to develop a foreign body response. The formation of fibrosis from repeated insulin injections or infusion set insertions, due to these three insults, can affect the absorption and bioavailability of insulin administered at affected sites.14,39,40
Increased mechanical stress across wounds has been shown to promote hypertrophic scarring and increased fibrosis via activation of various mechanotransduction pathways. 41 This understanding of the mechanical forces in the wound environment has led to the development of the Embrace patch (Neodyne Biosciences, Inc., Fremont, California), a tension-off-loading elastomeric dressing that has been clinically shown to limit fibrosis and minimize scar formation. 42 Cleared by the US Food and Drug Administration (FDA), the patch has significant potential to reduce fibrosis/scarring and improve insulin absorption at the infusion site.
Embrace tension off-loading patch
The silicone Embrace patch is pre-stretched and then shrinks back to its original size after being placed on the skin, thereby applying a compressive force across the skin and reducing tension across the wound. Randomized studies have demonstrated significant reductions in scarring following surgery or scar revision.43,44 In animal studies, the patch has also been shown to improve the extracellular matrix ultrastructure and mechanical properties of the tissue, making it more similar to normal skin. 45
Proof of concept/feasibility studies
To determine whether tension off-loading could prevent skin fibrosis in the setting of pro-fibrotic injections, investigators developed a model in red Duroc pigs using repeated local injections of bleomycin (a profoundly fibrotic chemotherapy agent) to replicate key features of insulin-induced skin fibrosis. 42 Using histologic, tissue ultrastructural, and biomechanical analyses, investigators showed that application of the patch reduces injection-induced fibrosis. The patch was highly effective in preventing fibrosis when bleomycin was injected through the patch. In contrast, when the patch was not used at the injection site, significant fibrosis developed. However, the patch placed on existing fibrosis promoted remodeling/reversal of the existing fibrosis. These findings support the potential use of the Embrace patch both to prevent fibrosis from developing at an active pump or injection site and to rescue/remodel pre-existing fibrosis from prior pump use/injections.
Investigators are currently enrolling adults with T1D in a 16-week feasibility study (ClinicalTrials.gov NCT06025513) to determine whether this same protective effect would apply to insulin-using patients with diabetes. 46 This trial will compare insulin absorption before and after wearing the patch for 16 weeks on a lipohypertrophy (LH) lesion. Analysis will include ultrasound measurements of lesions and histology including ECM algorithm analysis of matrix fibrosis.
Summary
The Embrace tension off-loading patch is an FDA-cleared medical device that can be incorporated into IISs to promote a more regenerative healing response and potentially improve the longevity and effectiveness of the infusion set. In addition, it could be used in a follow-on fashion when a pump is moved to a new site, to prevent/treat fibrosis at the old/exhausted site and thereby maintain it for future use. This, in turn, may minimize the complications associated with insulin pump therapy and ultimately reduce variability in insulin absorption and improve patient comfort.
Moving Forward
Learnings From the Research
Results from the DERMIS study and subsequent evaluations have provided valuable insights regarding the tissue damage and infection that occurs at the time of insertion and throughout the duration of infusion set wear. These findings lay a foundation for future research efforts (Table 2).
Summary of Key Findings.

For example, both histologic and OCT analyses demonstrated significant variability between patients in visible changes at the infusion site along with increased dermal thickness, fibrosis, fibrin deposition, vascularity, and inflammation, as well as the high number of eosinophils at the infusion sites, all of which indicate pronounced and enduring inflammation. Confirmation of these findings through use of OCT demonstrated the utility of this fast, non-invasive technique in additional research programs.
The investigations by Jacquemier et al 20 provided actionable guidance for the timing of infusion site changes. Their analyses also demonstrated the viability of a method that can be used to compare infusion parameters such as pump model, basal rate, catheter characteristics, and infusion site characteristics.
For example, researchers at Johanneum Research Health (Graz, Austria) have proposed a more “ideal” insulin delivery system with all functions integrated for individuals with T1D. The pump system would be designed as a small, patch pump with integrated glucose sensor, offering convenience and discretion for users (Figure 8).
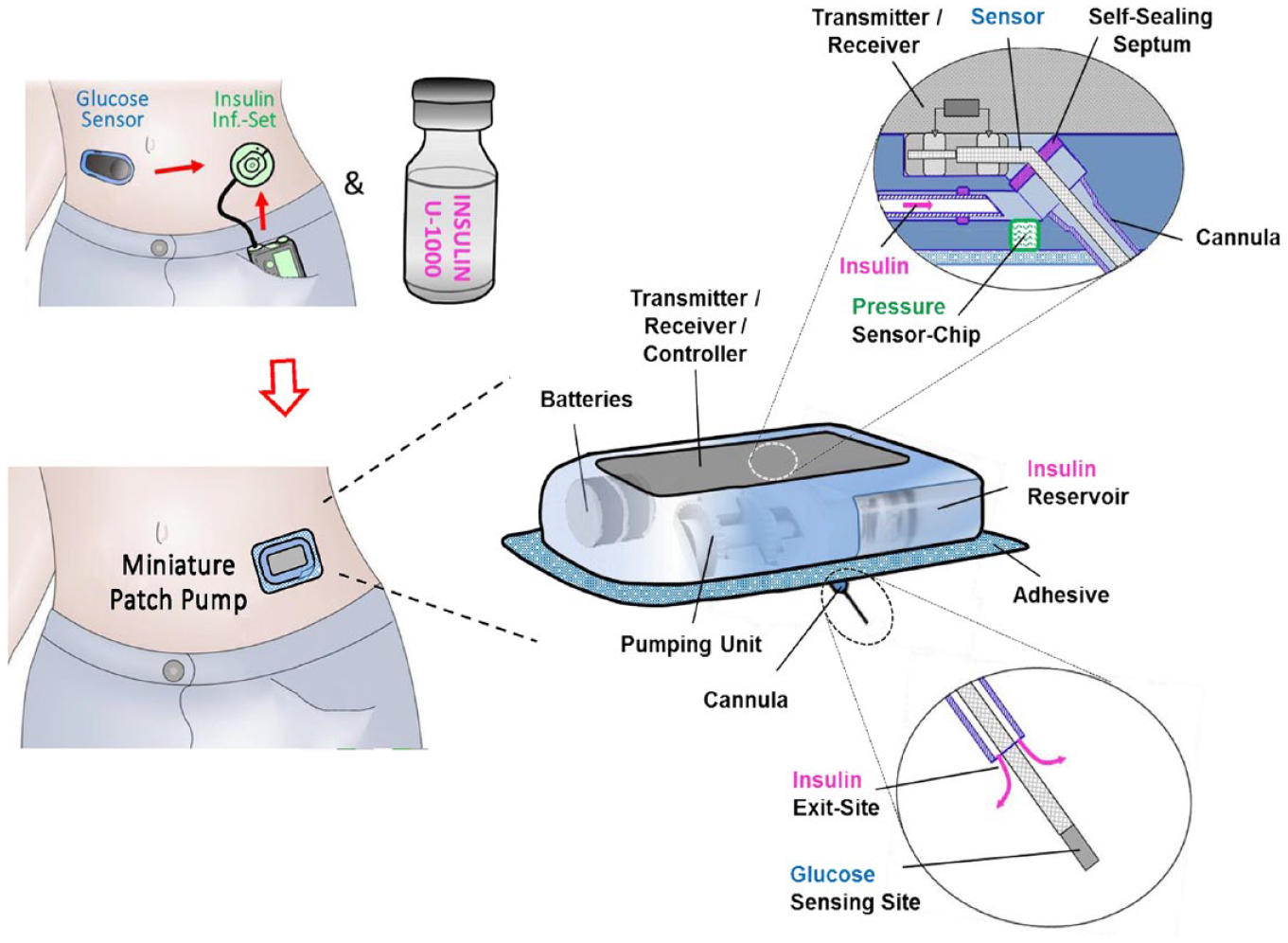
Schematic of an “ideal” insulin pump system.
The pump system would be compatible with ultrafast insulin formulations, offering improved insulin absorption kinetics and a more physiologic insulin profile. The system would include a pressure sensor to measure TFR to provide real-time monitoring of TFR. Feedback from the sensor would trigger pressure-reducing mechanism to mitigate the risk of increased TFR and insulin leakage. The system would also include an integrated wear-time sensor to accurately determine the duration of infusion catheter usage and intelligent alerts and notifications that inform patients when it is time to change the catheter based on measure TFR. Adaptations to the pump to minimize fibrosis, such as integrating a tension-off-loading dressing (Embrace device) into the adhesive, could further decrease fibrosis and increase pump efficacy. Although this approach could be considered “futuristic,” developments in combined infusion/glucose sensor designs and ultrafast insulins are already under development.
However, many questions remain unanswered. Although studies suggest an association between tissue reactions (eg, inflammation, netosis) and phenolic compounds, specifically m-creosol and phenol, which are necessary preservatives in today’s commercial insulin formulations, findings from other studies have identified insulin fibrils as more detrimental to the tissue site than the phenolic preservatives. Is it the insulin, the preservatives, both, or neither?
In addition, the difference in eosinophil volume between patients with >20 years of insulin pump use and those with <10 years of wear, has yet to be explained. Do all insulin pump users eventually develop a tolerance with long duration of wear, or is this tolerance only achieved in specific patient phenotypes?
Future Research Needs
The DERMIS study reported an increased presence of eosinophils, which implies an allergic reaction to these devices. Thus, it is imperative to identify the reasons for initiating eosinophilic infiltration. What specific triggers lead to eosinophil recruitment and activation? Is recruitment specific to insulin infusion pump sets, or can other dermal diabetes management devices, such as glucose monitoring, equally trigger eosinophil activation?
As insulin may induce a delayed hypersensitivity response, we also recommend pursuing intradermal testing with the patient’s insulin and with insulin that has been housed in the pump. Thus, additional studies should be directed to investigate the initiation of the influx of inflammatory cells, including the activation of molecular signaling pathways affected by phenolic preservatives vs insulin fibrils. Ideally the study subjects would also undergo biopsy of the pump insertion site to assess for presence of eosinophils to compare with findings in the DERMIS study. It is important to note a recent study by Zhou that reported the feasibility of incorporating a polyelectrolyte-modified carbon electrode into an infusion set for electrooxidative removal of phenol from the insulin. 47 Finally, any ultrasound extract that is found to trigger a positive patch test reaction should be followed up with GC-MS and possibly thin-layer chromatograms to help identify the contact allergen in the device.
Neodyne Biosciences, Inc is performing a rigorous trial in Germany with the company Profil to determine whether wearing Embrace patch for 16 weeks over an existing LH lesion leads to remodeling and diminished lesion size with less fibrosis. Performing detailed studies on insulin absorption and glucose control before and after wearing Embrace will be important to demonstrate that this device can be used to remodel existing fibrosis/LH sites in humans to rescue them for future use.
By substituting the Embrace patch for the current adhesive, these devices could theoretically actively minimize fibrosis while in use. In addition, as Embrace is FDA cleared for minimizing fibrosis/scarring, a very exciting indication could be use by patients as a “follow-on” treatment once a pump, injection, or CGM site is rotated out of use. Maintaining that previous site in a fibrosis-free state could prevent site exhaustion and thus facilitate preservation of that site for future use.
Conclusion
Additional studies should be directed to investigate the initiation of the influx of inflammatory cells. This knowledge should provide insights into the role of inflammatory cell interactions and how they may contribute to the tissue reaction at the insulin infusion site. This understanding should be beneficial in identifying therapeutic targets in the future (eg, cells, agents, cytokines), including usage of topical application by addressing underlying skin pathology.
Footnotes
Abbreviations
3D, three-dimensional; AUC, area under the curve; CGM, continuous glucose monitoring; CSII, continuous subcutaneous insulin infusion; DERMIS, DEfining, Reviewing, and Monitoring skin pathology in type 1 diabetes Study; DPGD, Dipropylene Glycol Diacrylate; DNA, deoxyribonucleic acid; FDA, US Food & Drug Administration; GC-MS, gas chromatography-mass spectrometry; IBOA, isobornyl acrylate; IoD, index of dispersion; IDM, insulin diluting medium; IGF, insulin-like growth hormone; IIS, insulin infusion set; LH, lateral hypothalamus; OCT, optical coherence tomography; OCTA, optical coherence tomography and angiography; PBMCs, peripheral blood mononuclear cells; PEEA, phenoxypoly (ethylenoxy) ethylacrylate; PK, pharmacokinetics; PP, phenol preservatives; RNA, ribonucleic acid; STZ, streptozotocin; T1D, type 1 diabetes; TLC, thin-layer chromatography; TFR, tissue flow resistance; TGF-β, transforming growth factor-beta.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for the meeting and manuscript development was provided by the Leona M. and Harry B. Helmsley Charitable Trust.

