Abstract
Insulin infusion sets (IISs) are an integral and intricate part of continuous subcutaneous insulin infusion for subjects with type 1 diabetes, infusing insulin from pump to the subcutaneous space. Insulin infusion sets interface with the skin surface, the dermis, and the subcutaneous space and may be the cause of infusion failure due to biological events or mechanical problems. Novel IISs with extended wear time and anti-inflammatory properties to mitigate these issues are described in the literature although material-tissue interactions are poorly understood. This rapid review focuses on the impact of IIS materials and designs on the subcutaneous response in people with diabetes and includes literature identified in PubMed, Embase, and Cochrane databases. Twenty-one studies were identified for qualitative synthesis that encompassed a limited and heterogenic body of evidence including 10 clinical reports, six reviews, one case report, two abstracts, and two communications. Two clinical reports were randomized crossover studies. Reports on materials mostly compared steel versus polytetrafluoroethylene (Teflon) cannulas and suggested no substantial difference in tissue response to these materials. Reports on designs focused mostly on the angle of cannula insertion. To drive and improve research on extended wear and nonimmunogenic IISs, future studies should focus on material-tissue interaction as dedicated outcome measures, quantified with punch biopsy and imaging techniques such as ultrasound, optical coherence tomography, and confocal reflectance microscopy. Original studies are required to further a field too young for a systematic meta-analysis.
Keywords
Introduction
Type 1 diabetes, affecting more than 20 million people worldwide, is an autoimmune disease causing a gradual loss of pancreatic beta cells leading to complete insulin deficiency and need for life-long treatment.1-3 Type 2 diabetes can be characterized by progressively inadequate insulin secretion and insulin resistance.4,5 Insulin therapy is commonly given by multiple daily injections (MDIs) or by continuous infusion in subcutaneous tissue.2,6 Continuous subcutaneous insulin infusion (CSII) is a modern approach to mitigating insulin deficiency or insulin resistance by using an insulin pump with an insulin infusion set (IIS) or a tubeless pump situated directly on the skin. An IIS catheter essentially infuses insulin from a pump reservoir to the subcutaneous space.5,7-9 CSII can be an alternative to MDIs and can result in good and reliable glycemic control.10-12 Moreover, CSII enables patients to combine insulin infusion and glucose monitoring in a closed-loop feedback system, potentially improving glycemic control and quality of life.13,14 Infusion sets constitute an indispensable interface between pump mechanisms and skin and are often regarded as the most intricate and delicate elements in the drug delivery chain.3,8,15 Similarly, tubeless pumps infuse directly from reservoir into the skin via a cannula. Common mechanical issues associated with IISs are occlusion, kinking, and adhesive detachment.8,16,17 Interfacing with skin, IISs can also lead to biological issues such as local skin reactions and subcutaneous immunological responses,3,15 potentially exacerbating the adverse effects of insulin and its preservatives, compounds known to affect IIS wear time. 18 Several advancements in IIS research have been made to mitigate mechanical issues including extending wear time and reducing the risk of occlusion and kinking.15,19,20 In contrast, tissue responses to IIS biomaterials and designs have not been subject to extensive research in humans despite the key role IISs play in the drug delivery chain by acting as the main foreign body interfacing with skin. 3 Not only are adverse tissue responses to IIS biomaterials and designs potential barriers for predictable pharmacokinetics and pharmacodynamics, but they also limit factors in skin tolerability and extended wear time, factors hugely impacting patients’ quality of life, willingness to continue CSII, and treatment outcome.8,21-23
The objective of this study was to rapidly review the literature that describes the impact IIS biomaterials and designs may have on subcutaneous tissue responses.
Methods
Design
A rapid review of the literature24,25 was conducted based on a systematic literature search executed in July 2022 to identify articles on the topic of infusion set materials and designs and their impact on the subcutaneous response in people with diabetes. The PROSPERO register was checked for any ongoing reviews of similar nature before executing database searches. 26 The systematic search strategy was documented in a PRISMA flowchart 27 (Preferred Reporting Items for Systematic Reviews and Meta-Analysis) and included searches in EMBASE, PubMed (MEDLINE), and Cochrane databases. Inclusion criteria were papers reporting on and/or discussing the impact of infusion set materials and designs on the subcutaneous response in people with diabetes. Nonhuman studies were not included.
Search Strategy
A conservative search strategy was developed by authors K.H., T.S., and K.O. Emtree and MeSH terms in Embase and PubMed were combined with keywords with no limits to constitute conservative broad searches. The Cochrane database search was limited to systematic reviews and meta-analyses. Search documentation including exact terms and dates are available in supporting information S1. A PRISMA flowchart depicting the systematic search is shown in Figure 1.
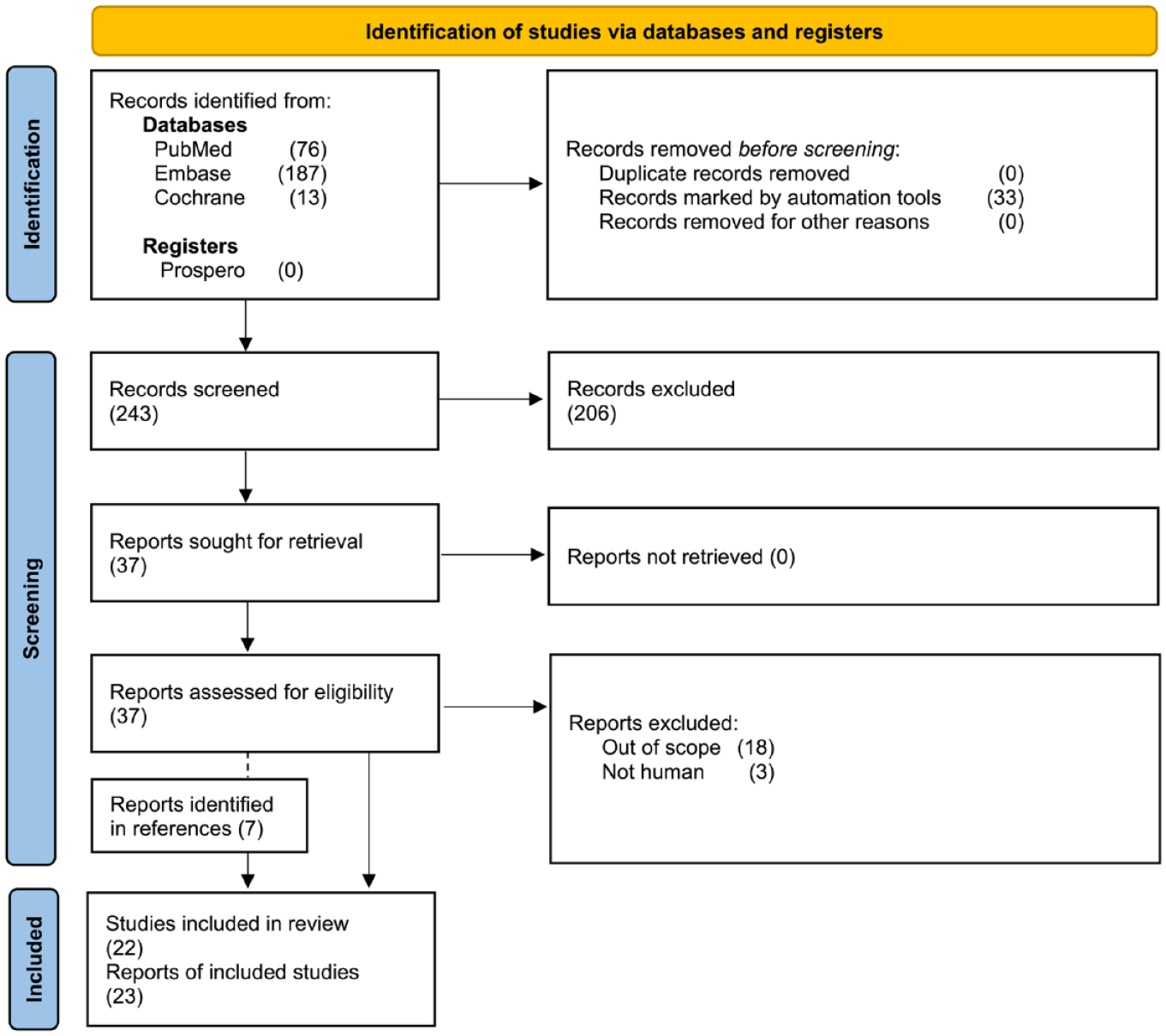
PRISMA-based flowchart of the systematic search for the rapid review.
Screening and Selection
A manual screening was conducted by authors K.H. and T.S. using Covidence software (Veritas Health Innovation, Melbourne, Australia). Titles, abstracts, and keywords were screened by the authors separately for criteria: human, diabetes-related, infusion set–related, and subcutaneous response–related. Disagreements in votes were resolved by discussion (K.H. and T.S.). Selection following full-text review was finalized by K.H. and T.S.
Data Extraction
All data were extracted by K.H. and reviewed by T.S. Disagreements were jointly reviewed by the authors before inclusion for discussion. Data include any description, results reported, and/or discussion on the impact of infusion set material and designs on the subcutaneous response in people with diabetes. A literature map (Figure 2) was created with LitMaps software (Litmap Ltd, Wellington, New Zealand). 28

Literature map of the included literature. Bubbles: individual publications. Bubble size: arbitrary size based on within-relative relative number of citations. The smallest bubble represents zero citations. Bubble colors: blue, clinical investigations; orange, reviews; and green, conference proceedings. Lines: citation chain. X-axis: publication year. Y-axis: citation cluster, that is, arbitrary spatial placement in relation to citation chain. The map does not include conference proceedings published in a conference proceedings publication.
Quality Appraisal and Risk Assessments
There were no systematic reviews, meta-analyses, or randomized controlled trials identified in the search. Two clinical reports were randomized crossover studies29,30 and were appraised for risk of bias according to the RoB 2 Tool for crossover studies 31 by K.H.
Rapid Review Synthesis
Final qualitative synthesis and discussion of included abstract were conducted by K.H. and K.O.
Results
Report and Study Characteristics
The PROSPERO database did not identify any protocols for a systematic review on the present topic. The MeSH and Emtree searches in PubMed, Embase, and Cochrane databases returned 276 reports, of which 33 were duplicates, resulting in 243 unique reports included for screening (Figure 1). A total of 37 reports were sought for retrieval following screening of titles, abstracts, and keywords. Full-text analysis of 37 reports was conducted. An additional 7 references were identified from the citations in the articles included for full-text review. Twenty-two reports were included for the rapid review. One report was a conference abstract preceding data in a journal article, leading to the final number of 22 studies included for synthesis (Table 1). There was a high degree of citation flow interconnectivity between the included studies (Figure 2).
Overview of the Literature.
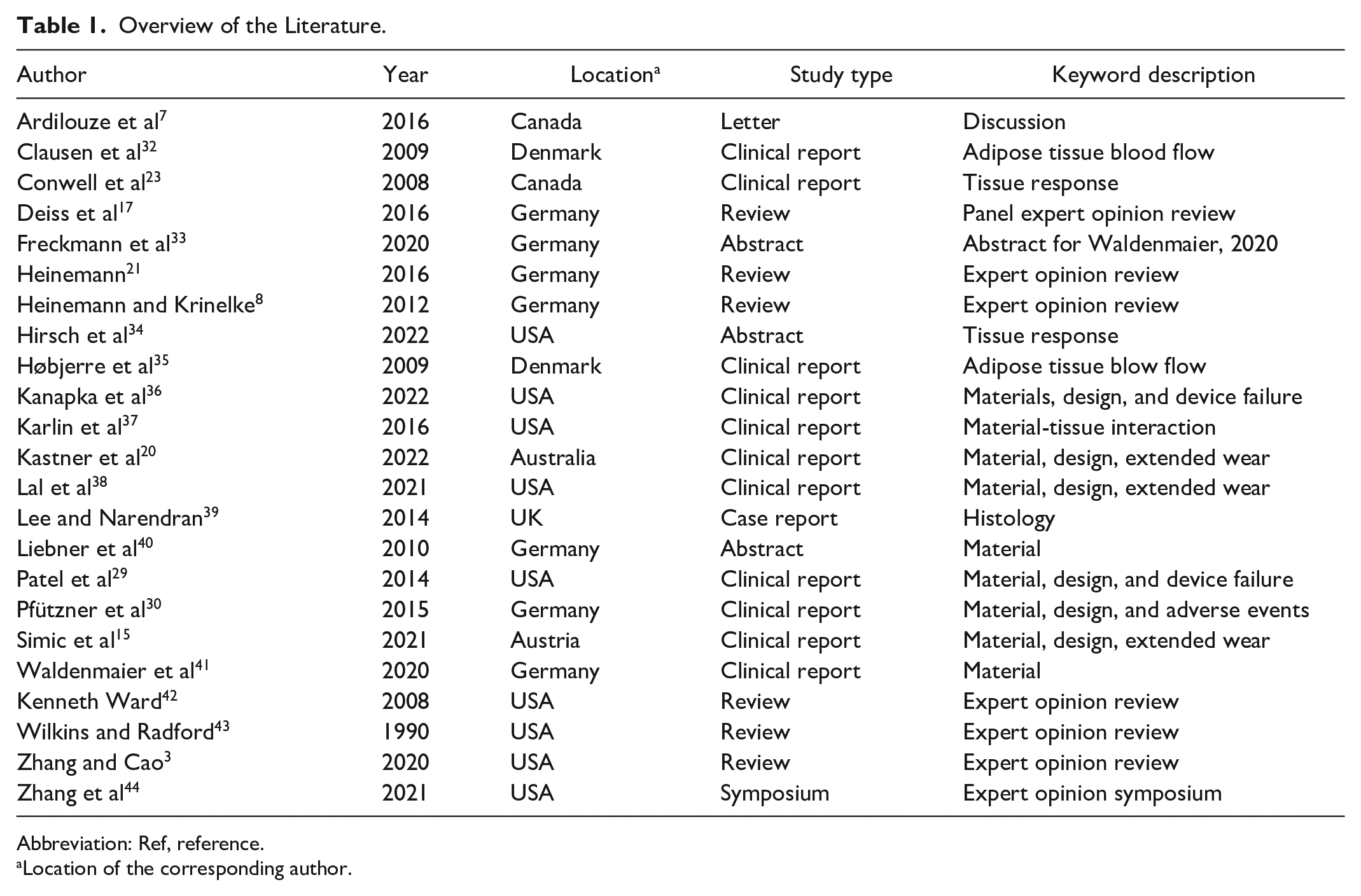
Abbreviation: Ref, reference.
Location of the corresponding author.
Methodological Quality
A wide variety of articles with a high level of heterogeneity were included for review synthesis. Two randomized crossover studies29,30 presented an overall low risk of bias by RoB 2–based appraisal 31 despite blinding being inherently infeasible. No systematic reviews or larger meta-analyses were identified, whereas several expert opinion and/or comprehensive reviews were included.
Materials
Infusion set materials polytetrafluoroethylene (henceforth referred to as Teflon in the present text) and steel were reported on in several journal articles, in both clinical studies and reviews. A study was conducted in 10 healthy male volunteers to compare tissue response in the form of adipose tissue blood flow following application of Teflon or steel-based cannulas. 35 Over the 48-hour wear time, blood flow increased significantly with Teflon cannulas, whereas steel-based cannulas resulted in only a minor increase. Teflon cannulas also required an increasingly higher flow pressure than steel. 35 A similar study conducted in 10 healthy male volunteers demonstrated that while Teflon cannulas result in blood flow increases and slightly faster absorption of insulin in the first two days of catheter wear time, there was no impact on peak concentrations of total amount of insulin absorbed following bolus doses. 32 These results indicate that the increased adipose tissue blood flow seen with Teflon cannulas does not impact the efficacy of bolus insulin infusions. In an extended wear trial, Teflon and steel-based cannulas were found to be performing similarly over an extended wear period, with 33% Teflon and 32% steel cannulas functioning after seven days of wear time. 29 In an abstract describing a questionnaire in Germany including 432 children where 56% used Teflon and 44% used steel-based cannulas, no differences in complications related to materials were described. 40 In a larger study including 40 subjects each wearing two steel and two Teflon cannulas, only mild infusion site reactions were documented, and there were no differences in skin reactions. Furthermore, there were no differences in wear time between Teflon and steel (mean 6.2 ± 1.5 days).33,41 A clinical study comparing the use of Teflon cannulas in both lipohypertrophic skin areas and unaffected areas found that there was no reduced longevity in lipohypertrophic areas nor any effect on insulin pharmacokinetics. 37 Teflon versus steel cannula were discussed in several reviews with expert opinions.3,8,17,21 These reflect the original studies also identified in this present rapid review. Two expert opinion comprehensive reviews describe and discuss tissue-material interactions including polyethylene, polypropylene, and biomimicry materials but make no clinical conclusions.42,43 A study on a novel extended wear infusion featuring an improved immunological profile reported an infusion set survival rate of 88% at 7 days with a mean wear time of 6.6 days (±1.4). 20 Another novel extended wear infusion set coated with an anti-inflammatory compound is described in two pilot studies. One study found that all tested infusion sets survived for the full seven-day study duration. 15 In the second study, the same novel infusion set led to a median wear time of 9.1 days (interquartile range, 7.1-10.0). 38
Design: Cannula Angles and Insertion
A recent meta-analysis of two clinical trials included data on 25 154 infusion sets (mean 96 ± 44 infusion sets per subject) using a closed-loop system and grouped results by type of infusion set used: 90° Teflon, angled Teflon, 90° steel, or interchangeable use. 36 In these data based on subjects using a closed-loop system, subjects wearing 90° Teflon sets were more likely to experience hyperglycemia within 72 hours than those wearing angled Teflon or steel sets although results were generally similar during the first 24 to 48 hours. 36 Infusion sets were less likely to fail early due to prolonged hyperglycemia when worn by adults than when worn by children and adolescents. 36 A cross-sectional study evaluated 50 adolescent subjects at a single time point and found that 90° Teflon infusion sets resulted in lower mean local skin reaction severity scores than sets with smaller angle. 23 None of these studies investigating steel and Teflon cannula directly elaborate on specific subcutaneous parameters. Expert opinion reviews discuss angled sets:8,17,21 One review describes 30° to 45° angled sets as better if the subjects are prone to infections, lean, and/or pregnant. 17 Other reviews describe 30° and 90° angled sets but mostly indicate that it is up to patients’ preference upon considering comfort although 90° sets are described to be associated with slightly fewer insertion problems and angled sets to be associated with more occlusion events.8,21 A study on a novel extended wear-set describes an angled design based on previous in vivo data showing a better immunogenic profile. 20 A recent expert opinion symposium emphasizes the importance of extended wear technology, anti-inflammatory properties, and research in the biological process leading to individual variability. 44
Tissue-Material Interaction
One case report described a patient abandoning CSII in favor of the intraperitoneal therapy with an infusion catheter. 39 The case report includes documentation of a human skin biopsy taken from a CSII infusion site with histology showing scarring and mild chronic inflammation but no eosinophil involvement. The patient was reported not to be allergic to any typical insulin formulations. 39 One report included conference proceedings of an oral presentation where data on skin characterization of infusion sites were presented. Thirty-one subjects with diabetes had punch biopsy and optical coherence tomography (OCT) scans conducted at current, prior, and control skin sites. The CSII sites showed significant differences compared with controls (no unit types given) in epidermal thickness (0.075 vs 0.071), inflammation (1.77 vs 1.75), fibrin (1 vs 1.53), fat necrosis (1.30 vs 1.04), and vascularity (1.43 vs 1.36). Optical coherence tomography scans showed increased vascular density (0.476 vs 0.343) and reduced optical attenuation coefficient (1.23 vs 1.48) in CSII sites compared with controls. 34 Other studies discuss the paucity of histological evidence and scarcity of tissue-material interaction.3,7,17 A larger cross-sectional study on a pediatric population included a smaller subgroup for ultrasound analysis of CSII infusion sites compared with control regions and describes significantly increased echogenicity of the dermis and hypodermis compared with control regions. There were no reported differences in dermal thickness or vascularity. Overall, the sample size was limited. 23
Discussion
We have rapidly reviewed the literature on the impact of infusion set designs and materials on the subcutaneous response in patients with diabetes. The results are presented in a qualitative descriptive format due to the heterogeneity and scarcity of studies. There is a remarkable paucity in studies investigating the technical designs and tissue-material interactions of infusion sets in humans. Despite IISs’ role as a key element in the drug delivery chain and status as a foreign body interfacing with skin, there is no solid evidence to support the designs and materials chosen from the skin surface downward in continuation of general biocompatibility testing.
The primary difference between modern infusion sets is the cannula material (steel, Teflon, and polypropylene).8,17,21,30,35 Steel and soft-polymer cannulas are used worldwide, with some populations favoring one over the other. 17 There is, however, no solid evidence to suggest that one material is significantly better than the other on glycemic control, adverse events, or local skin reactions.3,8,17,21,32,35,36,41 Interestingly, some studies report that adipose tissue blood flow is affected more by polymer materials than by steel, but these findings have yet to be confirmed in larger studies evaluating a range of basal insulin infusion doses or considering other tissue reactions.32,35 None of the identified studies provided direct data on material-tissue interactions with polymers, steel, or other components of IISs.
A wide variety of IISs enable subjects, caregivers, and health care professionals to identify the best individualized options through trial and error. 45 Options are well received, but the paucity of clinical research as the basis of meaningful evidence-based recommendations is alarming in a setting where an expectation of evidence-based recommendations is warranted. Moreover, expectations of future research on better biocompatibility, fewer adverse events, and extended wear based on the material-tissue interaction in humans are justified.3,7,8,21,30
People with diabetes must protect their skin real estate, which would lead to limited opportunities for biopsies and histological investigations in research on tissue-material interaction. Noninvasive techniques with conventional ultrasound and algorithm-assisted ultrasound have been used in IIS research for small sample sizes, but these and other techniques have yet to see applications on a larger scale.23,46 Light-based imaging modalities such as OCT and reflectance confocal microscopy (RCM) may be relevant additions to ultrasound in future research where skin-sparing techniques are paramount. Optical coherence tomography offers high-resolution images both en face and cross-sectional with precise angiographic visualizations but is limited to approximately 2 mm of scan depth.47,48 Reflectance confocal microscopy is highly specific offering near-histological resolution but is limited to a scan depth of approximately 0.2 mm.49,50 Limited scan depth can reduce individual usability of the imaging modality in relation to infusion set research and is certainly a limitation for subcutaneous investigations. 48 Nonetheless, combining imaging modalities can potentially be invaluable when punch biopsies are contraindicated and tissue reactions to IIS materials may be relevant even in the mid and superficial dermis. Interestingly, an abstract included in this rapid review reports data on 31 subjects with diabetes where at least 93 punch biopsies were taken, all following OCT scans. 34 The OCT results reported are limited but include an increased vascular density in CSII infusion sites compared with control sites. A significantly increased epidermal thickness was described in their histological findings, but the authors do not report on any attempt to reproduce these findings with OCT although the modality is uniquely suited for this task.34,51,52
Animal trial reports were not included in this rapid review. Results from animal trials can provide insight into histological findings and drug delivery patterns by scans and mass spectrometry when such evidence in humans is scarce. Several animal trial articles on the topic can serve as comparison guidance for optimizing outcome measures in human trials. Investigations in swine have shown that angled infusion sets at 35° may result in less-severe inflammatory reactions than perpendicular sets.53,54 Reports show that Teflon may result in less inflammation than steel in swine55,56 although an earlier study reported minimal differences in keeping with data reported in humans. 57 A meta-analysis of such results warrants a dedicated systematic review.
This rapid review is not without limitations. In addition to inherent drawbacks to the rapid approach,24,25 this systematic search was limited by several factors. The field of research on materials and designs for infusion sets lacks formalization in terms of MeSH headers on PubMed and Emtree headers on Embase. Moreover, there are no clear trends in the use of keywording for published studies. These factors lead to a risk of not detecting reports on the topic. The qualitative synthesis lacks strict outcome measures and meta-analysis but provides an indispensable overview of reports in the field. Most reviews described are based on expert opinion rather than adding any clinical evidence to the literature for the purpose of improving the data basis for clinical decision-making.
Conclusion
Despite millions of IISs used worldwide, the scientific literature on the impact of infusion set biomaterials and designs on the subcutaneous tissue response is scarce. The present rapid review identified only a small heterogenous pool of studies. Original human studies assessing the tissue-material interaction between the infusion set and skin are needed for further research to not only improve infusion set–based drug delivery but also, more importantly, to provide patients with evidence-based recommendations. The high reliability and safety of infusion sets are offset by the sheer number of millions of infusion sets used. We highly recommend a sharpened focus on skin reactions and the subcutaneous tissue responses in subjects with diabetes using CSII with clear and dedicated outcome measures in dedicated research on tissue-material interactions.
Supplemental Material
sj-docx-1-dst-10.1177_19322968221138076 – Supplemental material for Impact of Infusion Set Materials and Designs on the Subcutaneous Response in People With Diabetes: A Rapid Review of the Literature
Supplemental material, sj-docx-1-dst-10.1177_19322968221138076 for Impact of Infusion Set Materials and Designs on the Subcutaneous Response in People With Diabetes: A Rapid Review of the Literature by Kristoffer Hendel, Tobias Stumpe and Kerem Ozer in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors wish to thank Lorraine Ralph, Publication Manager at Convatec, for editing and reviewing the manuscript.
Abbreviations
CSII, continuous subcutaneous insulin infusion; IIS, insulin infusion set; MDI, multiple daily injections; OCT, optical coherence tomography.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Kristoffer Hendel, Tobias Stumpe, and Kerem Ozer are full-time employees of Convatec Infusion Care (Convatec Group plc).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
