Abstract
Background:
The number of older adults with type 1 diabetes (T1D) is increasing. Use of automated insulin delivery (AID) may influence nutrition and eating behaviors. We explored how three eating styles (restrained, external, emotional) differ between older adults with T1D who use and do not use AID.
Methods:
We administered a one-time electronic survey from September to November 2023 to adults ≥65 years with T1D receiving care through a university-affiliated hospital system. Clinical and demographic information was collected from medical records. Eating styles were characterized with the Dutch Eating Behavior Questionnaire.
Results:
Our sample (n = 77, 95% non-Hispanic white) had mean (SD) age: 71.8 (4.1) years, diabetes duration: 33 (18) years, hemoglobin A1c (HbA1c): 6.83 (1.12%), and body mass index (BMI): 27.3 (4.7) kg/m2. Respondents reported variable eating styles, with the highest median scores for external and restrained eating and lower scores for emotional eating. Older adults using AID systems had higher median scores for emotional and external eating, and more varied restrained eating scores compared to those not using AID systems. Weak correlations were found between eating styles and HbA1c (restrained: r = −0.14; external: r = 0.08; emotional: r = 0.15), as well as between restrained (r = 0.09) and external (r = 0.04) eating with BMI, with a small correlation between emotional eating and BMI (r = 0.27).
Conclusions:
Eating styles may vary between older adult AID users and non-users. To our knowledge, this is the first study to characterize eating styles in this population, though generalizability is limited by a non-diverse and small sample with high technology use overall (eg, continuous glucose monitoring, insulin pumps).
Introduction
As treatments for type 1 diabetes (T1D) have improved, more adults with T1D are living well beyond the age of 65 years.1-3 For this older population, diabetes technologies such as automated insulin delivery (AID) systems hold great promise for enhancing safety and providing solutions for the complex challenges of managing glucose levels alongside age-related changes in older adulthood.4,5
Integrating insulin delivery technology into T1D self-management may impact multiple aspects of a person’s life. For example, previous studies have shown that use of AID systems is associated with positive patient-reported outcomes in older populations (ie, reduced diabetes distress6 and psychological burden).7,8 However, diabetes technology may also influence nutrition, and more specifically, eating behaviors.9 Eating behaviors are clinically important because they can influence overall dietary intake and meal timing, which may in turn drive clinical outcomes such as weight status, hemoglobin A1c (HbA1c), and psychological well-being.7,8,10,11 In older adults specifically, who are more susceptible to hyperglycemia and hypoglycemia,12 eating behaviors that disrupt glycemic management may increase the risk of both short-term and long-term complications.12 These behaviors also contribute to psychological distress, which can negatively affect relationships, self-esteem, and everyday functioning.13 Finally, behaviors that result in excess caloric intake or deficiency may exacerbate weight management concerns.14 Yet, few studies have explored associations between technology use and eating behaviors in older adults with T1D.
Characterizing eating “styles”—the distinct eating patterns and tendencies that an individual exhibits—may help identify modifiable factors that influence differences in sustained eating behaviors.15 Eating styles include restrained eating, which refers to the conscious restriction of food intake, often for weight control, which can sometimes lead to cycles of overeating. External eating is characterized by eating in response to food-related cues, like the sight or smell of food, independent of hunger or fullness. Emotional eating comprises two dimensions: eating in response to diffuse emotions and eating in response to clearly labeled emotions, both in the absence of hunger.15 Emotional and external eating styles are associated with overeating, a driver of weight gain and obesity.10 There have been inconsistencies in findings regarding the relationship between the restrained eating style and obesity or weight gain.10 In a study of adults with T1D, those who reported binge eating were more likely to engage in restrained, emotional, and external eating styles compared to those who do not binge eat.16
Therefore, our aim was to explore how eating styles (restrained, external, and emotional eating) may differ between subgroups of older adults with T1D who use and do not use AID systems. The cross-sectional nature of our study prohibited us from developing or testing any hypotheses related to causal links between AID use and eating styles.
Methods
We administered a one-time electronic survey to adults 65 years and older with T1D receiving care through a university-affiliated hospital system (UNC Health System, NC, USA). Participants were recruited through flyers, MyChart messaging, and e-mails to complete an online survey about eating behaviors, usual dietary intake, and diabetes management routines. Clinical and demographic information was collected from the electronic medical record. Survey data were collected between September and November 2023. Responses were monitored and following a sustained lapse in new responses, the survey was closed. All participants signed informed consent and a HIPAA authorization prior to completing the survey and were incentivized for survey completion. The study was approved by the Institutional Review Board of the University of North Carolina at Chapel Hill.
Eating styles were characterized with the Dutch Eating Behavior Questionnaire (DEBQ), a 33-item, 5-point Likert-style scale (range 1-5) with three subscales.15 The Emotional Eating subscale assesses the tendency to eat in response to emotions. The External Eating subscale measures the propensity to eat in response to external food cues (eg, sight, smell). The Restrained Eating subscale evaluates the extent to which individuals consciously restrict food to control weight or shape.15 The scales exhibit high internal consistency and factorial validity.15 We summarized individual subscale scores and compared distributions of scores between older adults using and not using AID technology. We also tested correlations with clinical outcomes including HbA1c and body mass index (BMI). To our knowledge, there are no established clinical cutoffs or thresholds to reflect clinically significant eating behaviors.
Results
The survey response rate was 11.6%. The characteristics of the study participants are described in Table 1. Our sample (n = 77) had a mean age of 71.8 years (SD 4.1) and was 95% non-Hispanic white. Mean (SD) diabetes duration was 3317 years, mean (SD) HbA1c and BMI were 6.83 (1.12%) and 27.3 (4.7) kg/m2, respectively. The mean Diabetes Eating Problem Survey–Revised (DEPS-R) was 11.5 (6.0), which is below the threshold for clinically significant disordered eating behaviors (score of 20 or greater).
Respondent Characteristics.

Mean (SD) reported for continuous variables; n (%) reported for categorical variables, unless otherwise noted.
Percent among participants who are current users of CGM.
Older adults who used AID systems (n = 41; 53%) were more likely to live alone (98% vs 81%; P = .02), to have a bachelor’s degree or higher (83% vs 61%; P = .03), and to have used continuous glucose monitoring (CGM) for longer (8.5 vs 4 years; P = .002). HbA1c (%) was lower in AID users, median (IQR) 6.3 (5.9, 7.0) vs 7.1 (6.1, 8.1); P = .06, and BMI was not significantly different between groups (P = .2). There were no significant differences in the most recent receipt of nutrition counseling between the AID users and non-users.
Respondents reported a range of eating styles, with highest median scores on the external and restrained eating subscales and lower median scores on the emotional eating subscale (Table 2). Compared to those not using AID systems, older adults using AID systems had a higher median score on each of the subscales (Table 2). Figure 1 depicts the distribution of responses for each subscale stratified by AID user groups, suggesting a separation across technology use groups.
Eating Styles Among Older Adults With Type 1 Diabetes (N = 77), Stratified by AID System Use.

Range for each item = 1-5. Higher scores indicate a greater propensity for the respective eating behavior (ie, emotional eating, external eating, or restrained eating), while lower scores suggest less involvement in that behavior.
Median (IQR).
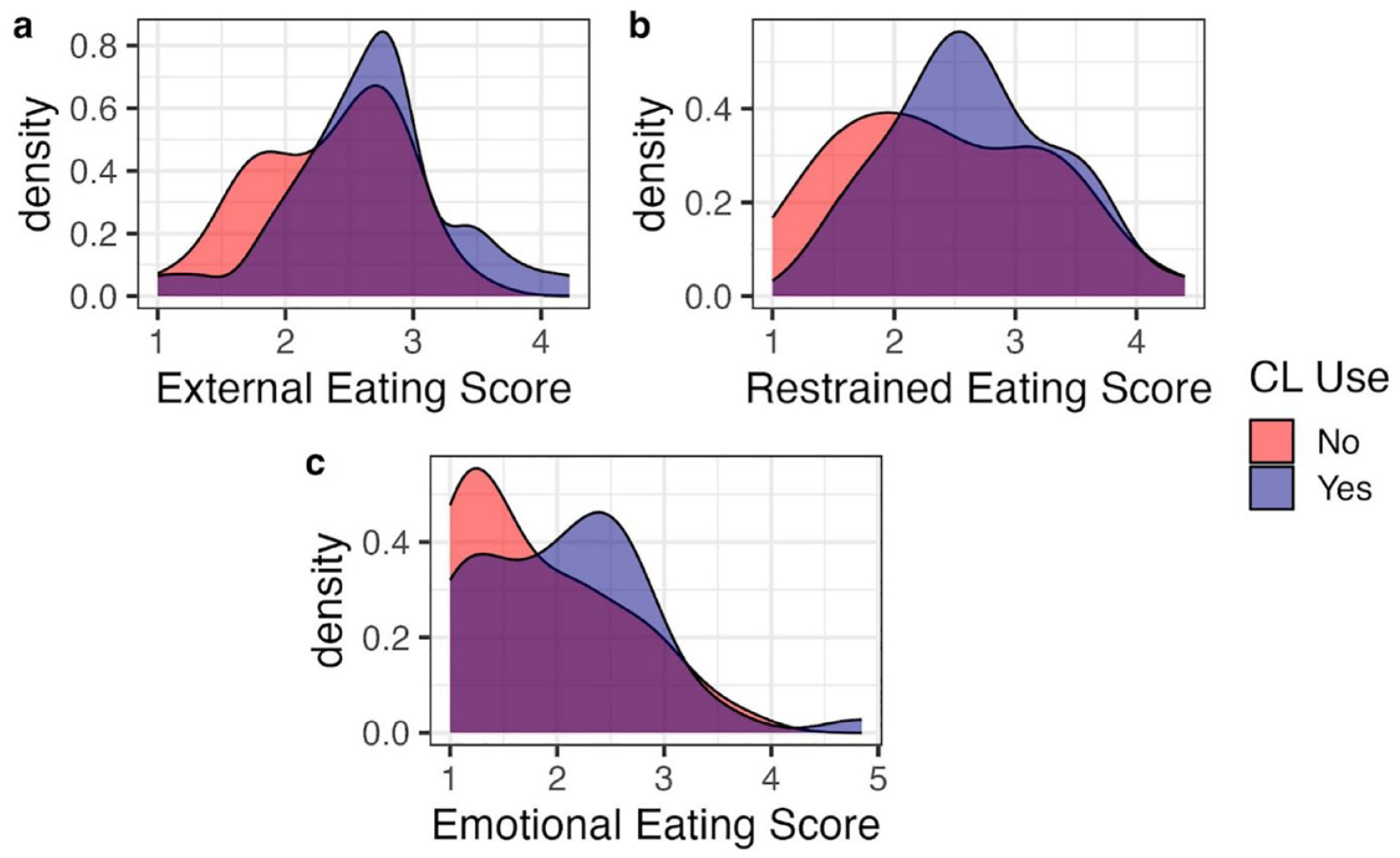
Distributions of DEBQ subscale scores stratified by AID system use (users N = 41, non-users N = 36). Panel (a) shows external eating; Panel (b) shows restrained eating; and Panel (c) shows emotional eating. Range for each item = 1-5. Higher scores indicate a greater propensity for the respective eating behavior (ie, emotional eating, external eating, or restrained eating), while lower scores suggest less involvement in that behavior.
There were weak correlations between eating styles and HbA1c (restrained eating: r = −0.14; external eating: r = 0.08; emotional eating: r = 0.15). There were similarly weak correlations between restrained eating (r = 0.09) and external eating (r = 0.04) with BMI, and a small correlation between emotional eating and BMI (r = 0.27).
Discussion
In a small sample of older adults with T1D with high rates of technology use, we found that eating styles may vary between AID users and users of other diabetes technology, including CGM and pumps. To our knowledge, this is the first study to characterize eating styles among older adults with T1D, including how styles may differ by AID technology use. Participants in our sample reported lower emotional and external eating tendencies and roughly equivalent restrained eating tendencies compared to those previously reported in the literature among younger populations with and without diabetes.17-24 However, relatively few studies have explored eating styles in people with diabetes.25,26
Of importance, the mean level of disordered eating behaviors as determined by aggregate DEPS-R scores did not suggest a high prevalence of clinically significant disordered eating in our sample. The greater emotional and external eating tendencies that were observed among older adults who use AID systems, relative to those using even other technology such as CGM and pumps, may reflect increased dietary flexibility conferred by the technology27; however, this subgroup also displayed a range of restrained eating scores. Greater emotional and external eating tendencies may contribute to overeating; lower HbA1c levels among AID system users despite increased emotional and external eating scores may suggest technology can facilitate eating behaviors with potential negative health impacts without negatively impacting HbA1c. Looking ahead, divergences in eating styles related to technology use may have implications for how to deliver nutrition counseling and support for this age group.28
Previous evidence suggests that children and adults with T1D may benefit from the dietary flexibility conferred by insulin pump use, and that AID use improves glycemic control and psychosocial factors.29 Evidence on the relationship between AID use and eating behaviors is comparatively more limited. Existing studies are limited, but have found evidence for changes in food choices and eating practices with AID use9,30 and called for additional nutrition support and education to promote healthful eating throughout and beyond the transition to using a closed loop system.30
The study is limited in its generalizability by a small sample size recruited from a single academic specialty center in which the majority of responders self-reported non-Hispanic white race and ethnicity. Further, the sample reflects a high prevalence of technology use that is aligned with the prevalence of technology use in specialty care settings, but not primary care settings. For example, most older adults in our sample who were not AID system users were CGM and pump technology users. More work is needed to explore the eating styles among older adults who don’t use technology. Other sources of selection bias in this study, as reflected by the low response rate, include that our data comes only from older adults who were more comfortable with the clinical messaging system, QR code utilization, and the electronic survey. These individuals are also more likely to be the ones using CGM and pump technology for diabetes management. To that end, focused recruitment efforts will be needed to collect information from the high proportion of older adults who are not seen in specialty care settings31 and those who may be less comfortable with electronic data collection methods.
Finally, the cross-sectional nature of our study also precludes understanding the extent to which differences in eating styles may have influenced the decision to use AID systems, or how eating styles changed after beginning use of these systems. The DEBQ has also not been validated in this population. Future work is needed to reproduce these findings in larger, more diverse samples, especially with more variable levels of technology use, and investigate the temporality of the associations between technology use and eating styles. Future research should also further evaluate how eating behaviors are associated with dietary intake and key clinical metrics, such as weight and markers of glycemic management. Ultimately, it should translate these insights into nutrition interventions and strategies to support healthful eating in this population.
Footnotes
Acknowledgements
The authors wish to thank the survey respondents for taking the time to contribute to the study.
Abbreviations
AID, automated insulin delivery; BMI, body mass index; CGM, continuous glucose monitoring; DEBQ, Dutch Eating Behavior Questionnaire; DEPS-R, Diabetes Eating Problem Survey–Revised; HbA1c, hemoglobin A1c; IQR, interquartile range; SD, standard deviation; T1D, type 1 diabetes.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this publication was supported by the National Institute of Diabetes and Digestive and Kidney Diseases of the National Institutes of Health under Award Number P30DK056350. The project described was supported by the National Center for Advancing Translational Sciences (NCATS), National Institutes of Health, through Grant Award Number UM1TR004406. ARK is supported by the National Institutes on Aging, National Institutes of Health, through Grant K01-AG084971-01 and was formerly supported by the National Center for Advancing Translational Sciences, National Institutes of Health, through Grant K12TR004416. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
