Abstract
Background:
Older adults may be less comfortable with continuous glucose monitoring (CGM) technology or require additional education to support use. The Virtual Diabetes Specialty Clinic study provided the opportunity to understand glycemic outcomes and support needed for older versus younger adults living with diabetes and using CGM.
Methods:
Prospective, virtual study of adults with type 1 diabetes (T1D, N = 160) or type 2 diabetes (T2D, N = 74) using basal-bolus insulin injections or insulin pump therapy. Remote CGM diabetes education (3 scheduled visits over 1 month) was provided by Certified Diabetes Care and Education Specialists with additional visits as needed. CGM-measured glycemic metrics, HbA1c and visit duration were evaluated by age (<40, 40-64 and ≥65 years).
Results:
Median CGM use was ≥95% in all age groups. From baseline to 6 months, time 70 to 180 mg/dL improved from 45% ± 22 to 57% ± 16%; 50 ± 25 to 65 ± 18%; and 60 ± 28 to 69% ± 18% in the <40, 40-64, and ≥65-year groups, respectively (<40 vs 40-64 years P = 0.006). Corresponding values for HbA1c were 8.0% ± 1.6 to 7.3% ± 1.0%; 7.9 ± 1.6 to 7.0 ± 1.0%; and 7.4 ± 1.4 to 7.1% ± 0.9% (all P > 0.05). Visit duration was 41 min longer for ages ≥65 versus <40 years (P = 0.001).
Conclusions:
Adults with diabetes experience glycemic benefit after remote CGM use training, but training time for those >65 years is longer compared with younger adults. Addressing individual training-related needs, including needs that may vary by age, should be considered.
Introduction
Older adults with type 1 diabetes (T1D) and insulin-requiring type 2 diabetes (T2D) have more hypoglycemia and hypoglycemia unawareness when compared to younger adults, and this increases with longer duration of diabetes.1 -5 Hypoglycemia is associated with greater glycemic variability and increased falls, fractures, cardiovascular events, emergency room visits, hospitalizations, and mortality, as well as declines in cognition and quality of life. 4 ,6 -10 Hyperglycemia in older adults is also associated with worse outcomes, including infections, cardiovascular events, urinary incontinence, decline in physical and cognitive functioning, dehydration, falls and increased mortality.11 -15
The use of continuous glucose monitoring (CGM) has been shown to reduce hypoglycemia and improve CGM-measured time in range 70 to 180 mg/dL (TIR) and quality of life in clinical trials of older adults with T1D and T2D insulin users but is underutilized.16 -19 Although barriers to CGM use change over time, in general older adults are more likely to be more uncomfortable using technology, have cognitive impairment, perceive greater burdens associated with initiation of CGM and require more time for self-management training and support.20 -23 In addition, most adults with diabetes receive their diabetes care in primary care settings, which may not have the staff or expertise to help people with diabetes use CGM.
We previously reported benefits with Virtual Diabetes Specialty Clinic (VDiSC) remote CGM training and support provided by certified diabetes care and education specialists (CDCESs) for adults ≥18 years of age with T1D and T2D using insulin. 24 HbA1c improved from baseline to 6 months in those with T1D (7.8 to 7.1%) and T2D (8.1 to 7.1%), with increased TIR. 24 Given that older adults can benefit from CGM use but may not be as comfortable using new technologies and are less likely to use telemedicine,21 -23, 25 it was important to examine the effectiveness of the VDiSC methodology in this vulnerable age group. Here we report both glycemic outcomes and duration of remote CGM training visits in older adults and compare results to those found in younger adults.
Methods
Study methods have been described previously 24 and are summarized herein. Recruitment pathways included insurance provider (CVS Health Clinical Trial Services LLC), primary care, or endocrinology clinic referrals. Study recruitment materials included an invitation link to complete an electronic consent form approved by the Jaeb Center for Health Research Institutional Review Board. As part of the informed consent process, individuals interested in participating were able to reach out to the study team with questions. Major eligibility criteria included age ≥18 years, U.S. resident, T1D or T2D requiring insulin intervention (multiple daily injections [MDI] or insulin pump), and access to a smartphone and computer with internet access. Potential participants were excluded if pregnant, currently undergoing renal dialysis, and/or currently using an automated insulin delivery system. Current CGM users were eligible only if % TIR was <60% or time <54 mg/dL was >1%.
Current CGM users shared recent CGM data to confirm eligibility. Participants not currently using a CGM completed a run-in phase using a blinded Dexcom G6 Pro CGM sensor (Dexcom Inc., San Diego, CA). Study supplies, including Dexcom G6 CGM supplies, were shipped directly to study participants.
The participant’s primary contact for care during the study was an assigned CDCES. Each participant was scheduled for three remote training sessions with their CDCES within the first month (Day 0, Day 7-14, and Day 21-28). Additional participant contacts were scheduled as needed and were individualized to increase comfort and understanding of system use and participant goals. Certified diabetes care and education specialists interactions included training sessions through videoconference, and communication through phone calls, texts, and/or emails.
If the participant was currently not using a CGM device, the first training session was designed to cover the basics of CGM use, such as sensor insertion, alerts, alarms, and how to upload and visualize the data. For those already using a CGM device, the first training session provided an opportunity to answer any initial questions about device use. In the second visit, participants reviewed data with their CDCES and discussed how to use data visualization tools to better manage insulin dosing, meals, and exercise. The third training visit presented another opportunity to provide education to support individualized diabetes management goals and address any lingering concerns.
The CDCES, while there to provide support on how to use the CGM to better manage blood glucose, also had the ability to adjust the insulin dosing (≤20% change for basal, ≤30% change for boluses). In addition, the virtual clinic structure included endocrinologist consultation on an as needed basis for both participants and CDCES. A behavioral health team was also available to provide support for management of diabetes-related stress.
Participants were asked to provide fingerstick samples through a capillary blood collection kit at baseline, 12, and 26 weeks for HbA1c measurement. The participants mailed their samples directly to the Advanced Research and Diagnostic Laboratory at the University of Minnesota for analyses (Tosch Automated Glycohemoglobin Analyzer HLC-723G8).
Self-reported questionnaires completed by the participants at baseline, 12 weeks, and 24 weeks included the Personal Health Questionnaire Depression (PHQ-8), 26 Diabetes Distress, 27 Hypoglycemia Fear, 28 Hypoglycemia Confidence Scale, 29 Glucose Monitoring Satisfaction Survey, 30 and a one-item sleep quality survey (SQS). 31
Outcome Measures
Outcomes included HbA1c, TIR, time <70 mg/dL, time >250 mg/dL, amount of CGM use, training visit time, and participant-reported psychosocial and diabetes treatment satisfaction questionnaire scores. The results reported here include data through 6 months of follow-up.
Statistical Methods
Analysis was restricted to participants who initiated use of real-time CGM, completed at least one training visit, and had ≥168 hours of CGM data (about 4% of the 6-month follow-up period). Participants were analyzed in three categories <40, 40 to 64, and ≥65 years according to their age at baseline. Analyses were performed separately for T1D and T2D participants as well as combined. Terms for an age by diabetes type interaction were not statistically significant when added to the model. Missing HbA1c values were imputed from the CGM data as described previously. 32 A linear model was used to perform pairwise comparisons of these three age groups for the change from baseline to 6 months adjusting for the baseline value as a covariate. The time <70 mg/dL, time <54 mg/dL, and visit length had skewed distributions and were analyzed using robust regression with an M-estimator. Multiple comparisons in these analyses were handled by controlling the false discovery rate using the Benjamini–Hochberg adaptive two-stage method. 33 Analyses were performed using SAS 9.4.
Results
There were 234 total participants (160 with T1D and 74 with T2D); 81 (35%) were 20 to 40 years, 126 (54%) were 40 to 64 years and 27 (12%) were ≥65 years old. Median (IQR) duration of diabetes was 20 (13, 30) years. Mean (SD) age of the older adults (≥65 years) was 71 ± 4 years; 48% were female, 96% non-Hispanic White, and 30% had annual household income <$50,000 (Table 1). Supplemental Table 1 provides baseline characteristics according to type of diabetes.
Baseline Participant Characteristics by Age Group (T1D and T2D).
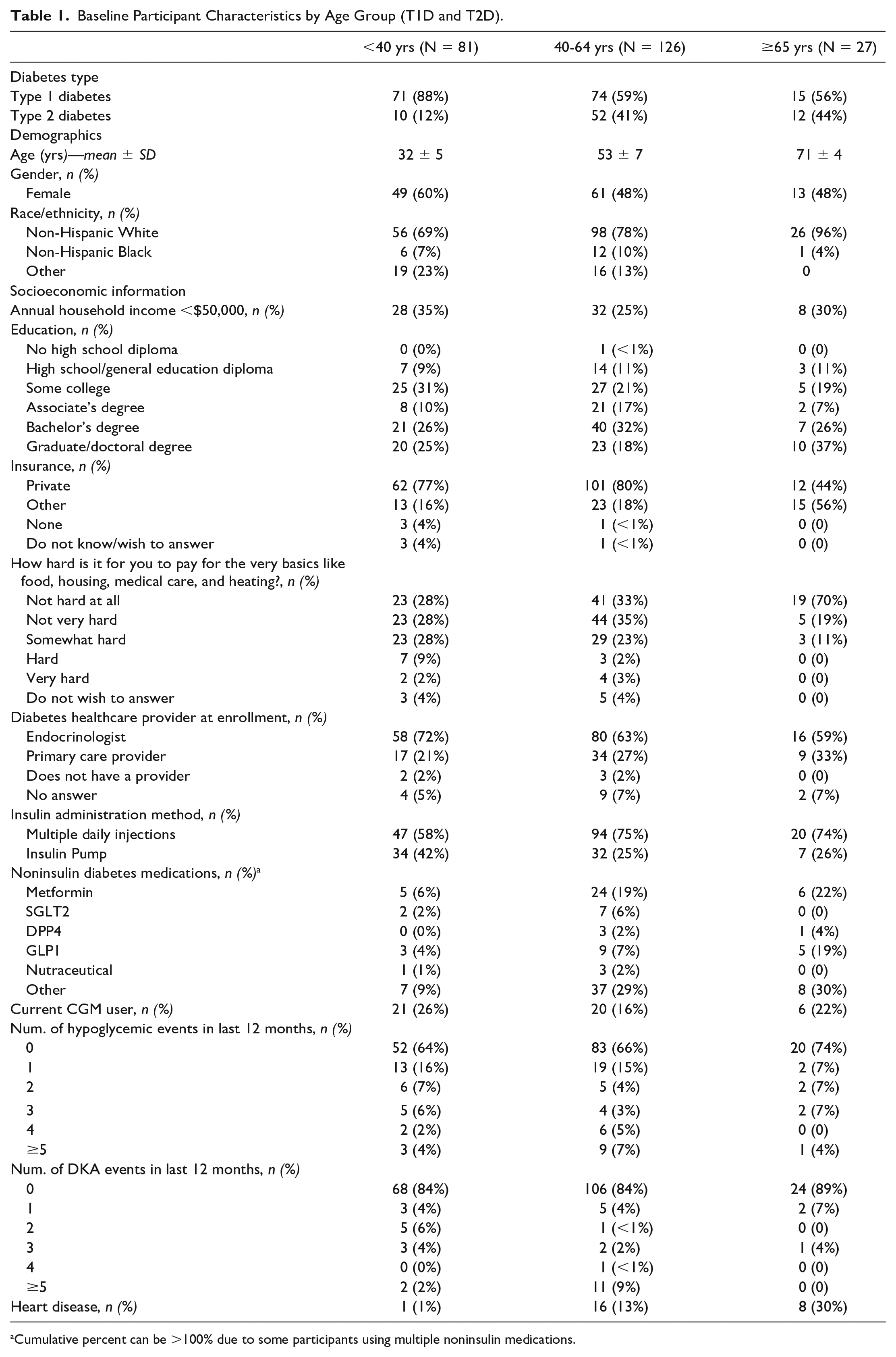
Cumulative percent can be >100% due to some participants using multiple noninsulin medications.
Median CGM use was 95% for both those <40 years and those 40 to 64 years, while the median CGM use was 97% for those ≥65 years (Table 2) and was similarly high in all age groups for participants with T1D and T2D (Supplemental Table 2).
Glycemic Results by Age Group for All Participants (T1D and T2D).
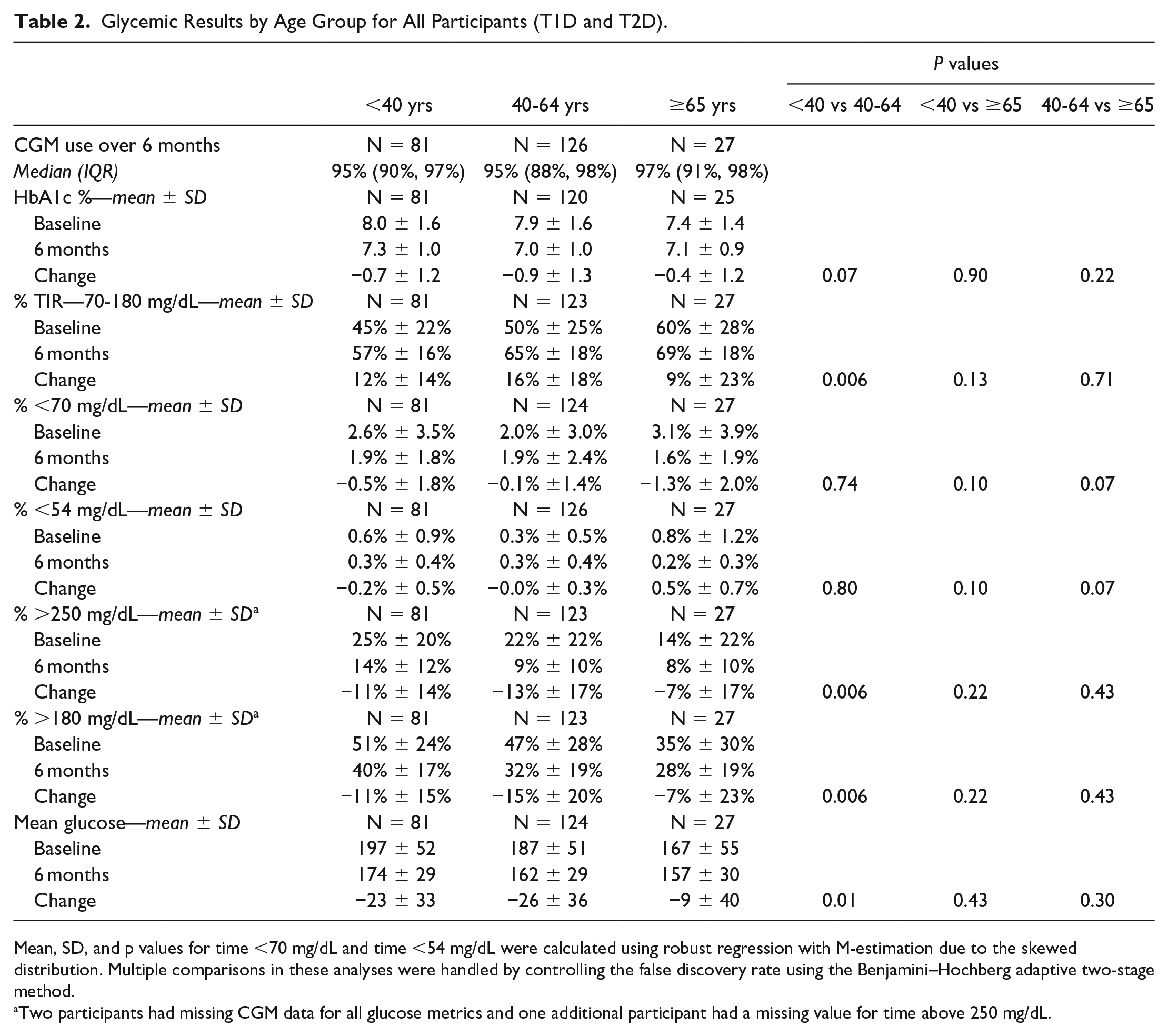
Mean, SD, and p values for time <70 mg/dL and time <54 mg/dL were calculated using robust regression with M-estimation due to the skewed distribution. Multiple comparisons in these analyses were handled by controlling the false discovery rate using the Benjamini–Hochberg adaptive two-stage method.
Two participants had missing CGM data for all glucose metrics and one additional participant had a missing value for time above 250 mg/dL.
HbA1c improved from baseline to 6 months in all three age groups (<40 years −0.7% ± 1.2%; 40-64 years −0.9% ± 1.3%; ≥65 years −0.4% ± 1.2; Table 2).TIR also improved in all three groups (<40 years 12% ± 14%; 40-64 years 16% ± 18%; ≥65 years 9% ± 23%; Table 2) with significantly greater change in the <40 years group when compared to the 40 to 64 years group (<40 vs. 40-64, P = 0.006; Table 2). Time in hypoglycemia <70 mg/dL was highest for those ≥65 years at baseline (3.1% ± 3.9%) and changed by −1.3% ± 2.0% at 6 months. Time <70 mg/dL also improved for those <40 years (−0.5% ± 1.8%), and those 40 to 64 years (−0.1% ± 1.4%). Hyperglycemia (>250 mg/dL) changed by −11% ± 14%, −13% ± 17%, and −7% ± 17%, respectively in the three age groups (Table 2). Results further categorized diabetes type (type 1 vs type 2) as shown in Supplemental Table 2 and Supplemental Figure 1.
There was an increase in mean total training contact time for all participants with age (176 ± 44 for those <40 years, 195 ± 49 for those 40 to 59 years, and 217 ± 71 for those ≥60 years; <40 vs. 40-64 years P = 0.009; <40 vs. ≥65 years P = 0.001; 40-64 vs. ≥65 years P = 0.05; Table 3). Similar trends were seen in both T1D and T2D participants (Supplemental Table 3). There were improvements in all participants reported outcomes and measures were comparable across age groups at baseline and 6 months (Table 4 and Supplemental Table 4).
Training and Education Visit Duration by Age Group for All Participants (T1D and T2D).
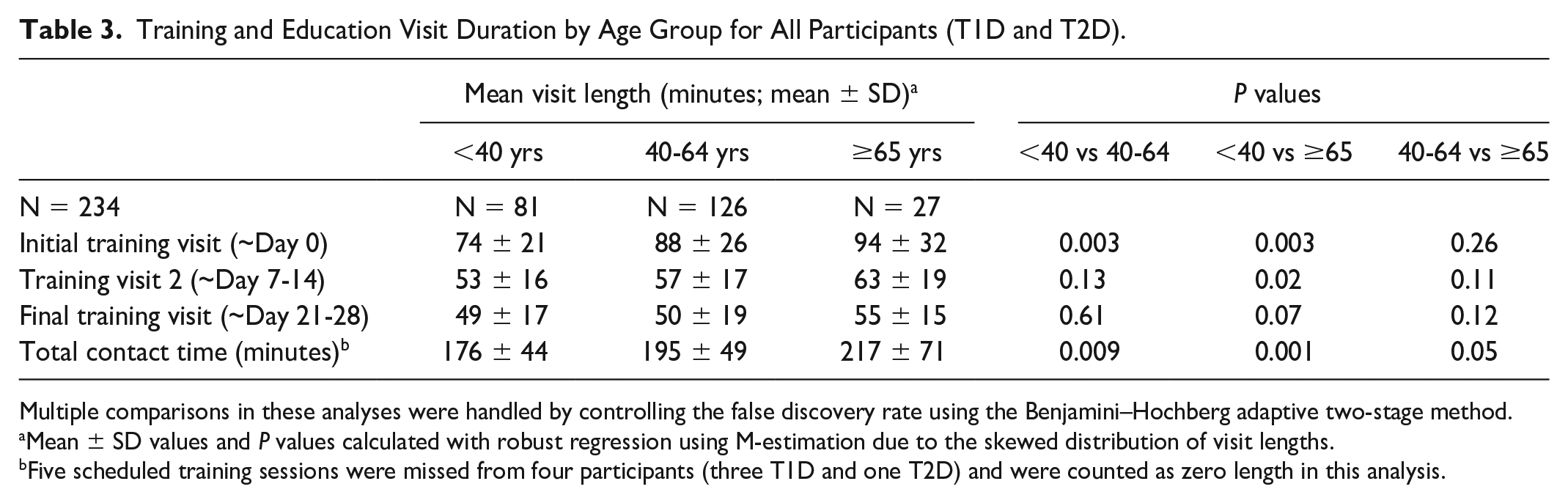
Multiple comparisons in these analyses were handled by controlling the false discovery rate using the Benjamini–Hochberg adaptive two-stage method.
Mean ± SD values and P values calculated with robust regression using M-estimation due to the skewed distribution of visit lengths.
Five scheduled training sessions were missed from four participants (three T1D and one T2D) and were counted as zero length in this analysis.
Participant-Reported Outcome Benefits by Age Group for All Participants (T1D and T2D).
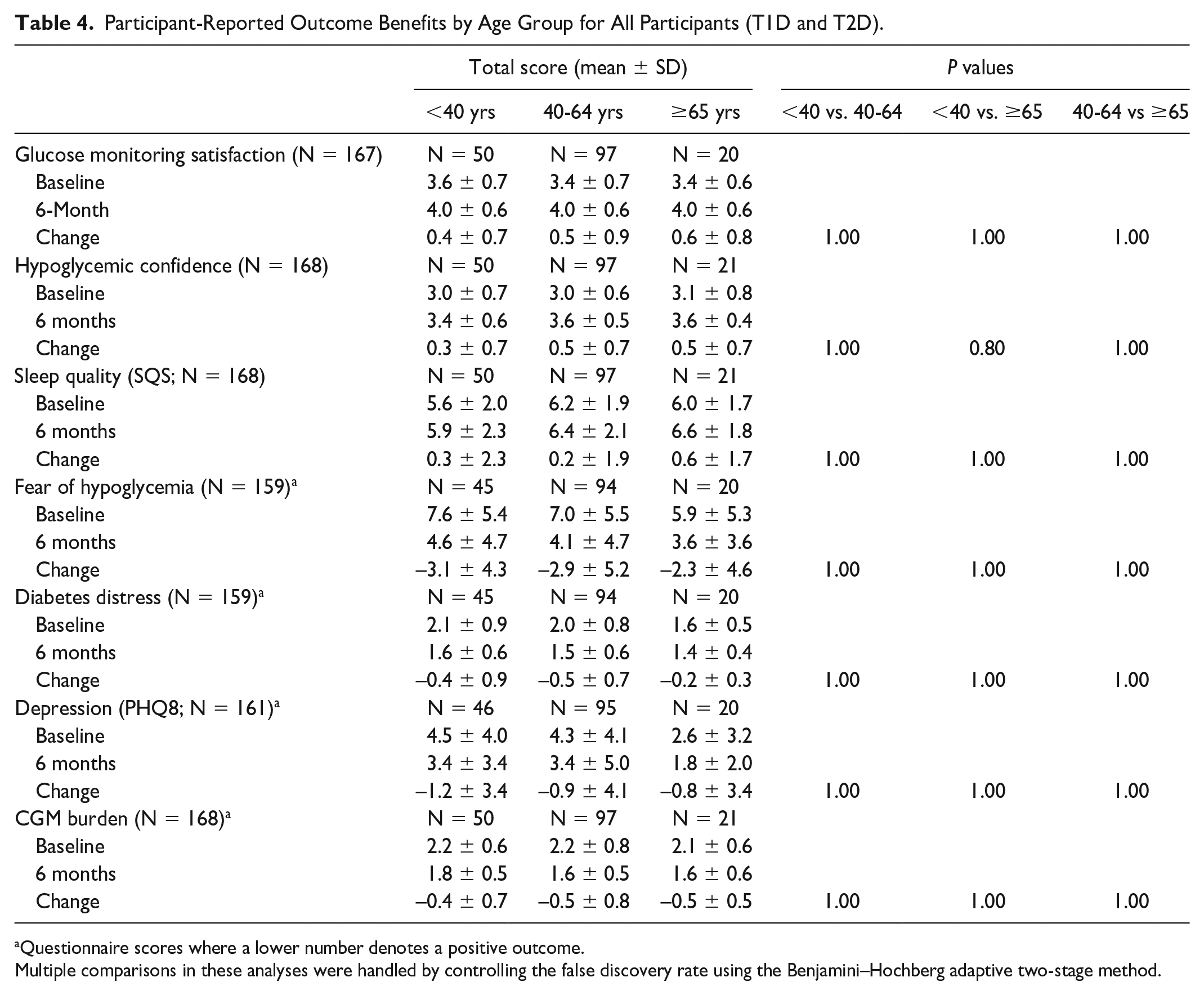
Questionnaire scores where a lower number denotes a positive outcome.
Multiple comparisons in these analyses were handled by controlling the false discovery rate using the Benjamini–Hochberg adaptive two-stage method.
Discussion
These results demonstrate that adults with both T1D and insulin requiring T2D ≥65 years of age experience glycemic benefits that are similar to or better than younger adults after receiving remote training and support in the use of CGM. This important finding adds to outcomes reported from clinical trials in older adults in which CGM was started in-person and glycemic benefits were demonstrated.16,18,19 Reductions in hypoglycemia were observed in the older adults with T1D (who had greater percent hypoglycemia at baseline than other age groups or those with T2D). Given the morbidity associated with hypoglycemia in this age group, this is a notable finding. In all adults with T2D regardless of age group, there was a significant reduction in hyperglycemia. Overall, older adults had improvements in HbA1c, TIR and time in hypoglycemia that were either similar to or better than that observed in younger adults. At 6 months, older adults had less fear of hypoglycemia, less diabetes distress, and more satisfaction with glucose monitoring, similar to results found in the younger adults. Although the virtual diabetes clinic study did not have any in-person visits, and enrolled participants through insurance providers, primary care physicians and endocrinology practices with whom they did not have a prior relationship, the positive results demonstrate the potential for virtual care models to facilitate use of CGM and improve glycemic control for many individuals with diabetes, including those from far-reaching under-resourced areas, by greatly increasing access.
Older adults may be less likely to use technology routinely than younger adults. They are more likely to have physical and cognitive impairments that can interfere with the use of new technologies compared with those who are younger. 17 Other studies have reported barriers in T1D adults not using CGM, with older adults perceiving greater burdens associated with CGM use compared with younger individuals. 21 Older adults reported concerns that readings could not be trusted and that CGM caused worry, takes too much time to use, and was too hard to understand. 21 Kahkoska et al 23 studied factors associated with CGM use in older adults. Her work emphasized the need for and importance of external emotional and technical support and encouragement. Technical and personalized support from the care team and caregivers, family and friends, as well as individualized education and re-education are often needed and are associated with increased safety (less hypoglycemia) and growing trust in the use of technology over time. 17 The VDiSC study demonstrated that virtual access to CDCESs can successfully provide the needed support and CGM training to enable adults with diabetes of all ages, including older adults, to benefit from the use of this technology irrespective of their geographic location.
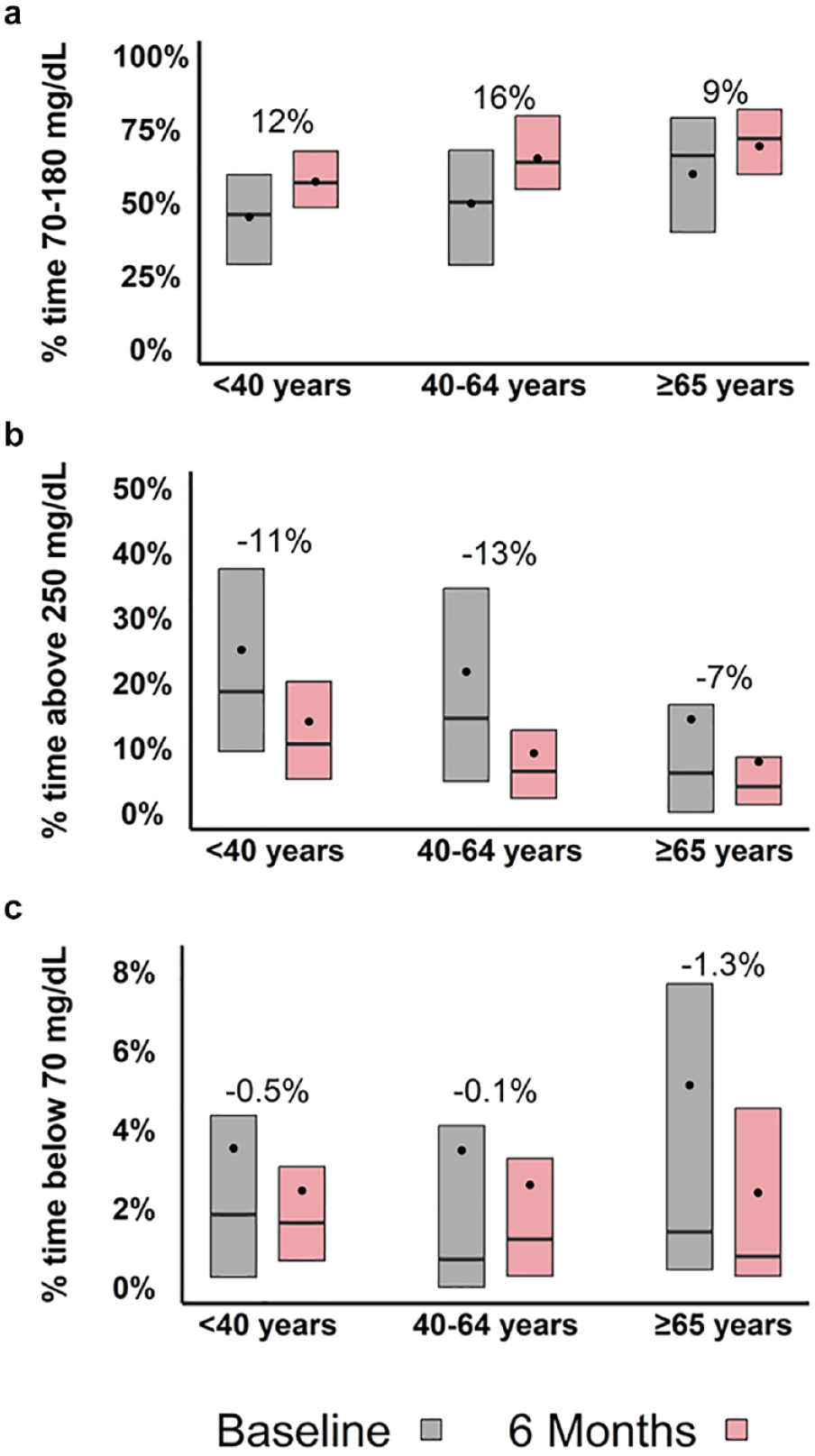
CGM metrics in all participants across age groups at baseline and 6 months. Dots represent mean values, and the line inside the box denotes the median. Top and bottom of the box denote the 75th and 25th percentiles, respectively.
In this study, focused on initiating and supporting CGM use, we tracked the time needed by the remote CDCESs to provide CGM training and support, and not surprisingly, older adults require more time for training than those <40 years of age. We found that older insulin-requiring adults needed more and longer remote education visits at the beginning of the COVID-19 pandemic as well. 22 There are many possible reasons for this finding. In addition to the barriers discussed above, in this study, proficiency in the use of smartphones or computers was not assessed. We do not know how much of the increased training time was related to more difficulties in the use of technology in general versus use of CGM specifically. It is also possible that some older adults asked more detailed questions or felt less pressure to complete training sessions quickly. A challenge facing primary care and diabetes clinics is inadequate staffing to provide this support and inadequate reimbursement from insurers to cover these services. Remote CDCESs can help deliver these services, but this is only sustainable with sufficient compensation. As newer CGM devices become available, with better usability, training time may be able to be reduced.
A limitation of this study is that participants were mostly privately insured, White, and were required to have a smartphone and access to a computer with internet. Further studies are needed to assess needs in diverse populations.
Remote training and support using the VDiSC model can help older adults successfully adapt to using CGM and improve glycemia, thereby improving patient safety. Support for approaches that address the individual needs of older adults with diabetes, including the greater time needed for education and re-education, is required.
Supplemental Material
sj-docx-1-dst-10.1177_19322968241294250 – Supplemental material for Older Adults Benefit From Virtual Support for Continuous Glucose Monitor Use But Require Longer Visits
Supplemental material, sj-docx-1-dst-10.1177_19322968241294250 for Older Adults Benefit From Virtual Support for Continuous Glucose Monitor Use But Require Longer Visits by Ruth S. Weinstock, Dan Raghinaru, Robin L. Gal, Richard M. Bergenstal, Amy Bradshaw, Terra Cushman, Craig Kollman, Davida Kruger, Mary L. Johnson, Teresa McArthur, Beth A. Olson, Sean M. Oser, Tamara K. Oser, Roy W. Beck, Korey Hood and Grazia Aleppo in Journal of Diabetes Science and Technology
Footnotes
Abbreviations
CDCES, Certified Diabetes Care and Education Specialist; CGM, continuous glucose monitoring; DKA, diabetic ketoacidosis; HbA1c, hemoglobin A1c; IQR, interquartile range; MDI, multiple daily injections; PHQ-8, Personal Health Questionnaire Depression; SAS, Statistical Analyses System; SD, standard deviation; SQS, sleep quality survey; T1D, type 1 diabetes; T2D, type 2 diabetes; TIR, time in range (70-180 mg/dL); VDiSC, Virtual Diabetes Specialty Clinic.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RSW reported personal fees from The Leona M. and Harry B. Helmsley Charitable Trust and Jaeb Center for Health Research during the conduct of the study; grants for participation in multicenter clinical trial through her institution from Eli Lilly, Novo Nordisk, Insulet, Tandem, Amgen, and MannKind outside the submitted work; and discounted devices for clinical research from Dexcom outside the submitted work. RLG reported grants from The Leona M. and Harry B. Helmsley Charitable Trust during the conduct of the study. RMB reported grants from Abbott Diabetes Care, Eli Lilly, Hygieia, Dexcom, Sanofi, Tandem Diabetes Care, Insulet, and Medtronic and consulting and/or advisory board fees paid to his institution from Abbott Diabetes Care, Dexcom, Onduo, Sanofi, Roche, Embecta, and Medtronic during the conduct of the study. CK reported grants from The Leona M. and Harry B. Helmsley Charitable Trust and nonfinancial support from Dexcom during the conduct of the study; grants from JDRF, Diasome, and MannKind outside the submitted work; and nonfinancial support from Tandem, Dexcom, and Insulet outside the submitted work. DK reported grants from Abbott and advisor and/or speaking fees from Abbott, Dexcom, and Jaeb Center for Health Research outside the submitted work. MLJ reported grants from Jaeb Center for Health Research paid to HealthPartners Institute during the conduct of the study and grants from Dexcom given directly to HealthPartners Institute outside the submitted work. BAO reported personal fees from Lagoon Health during the conduct of the study and stock ownership in Abbott Laboratories. SMO reported grants from The Leona M. and Harry B. Helmsley Charitable Trust during the conduct of the study and consulting fees from Cecelia Health and advisory board fees from Dexcom outside the submitted work. TKO reported grants from The Leona M. and Harry B. Helmsley Charitable Trust during the conduct of the study and consulting fees from Cecelia Health, advisory board fees from Dexcom, and an investigator-initiated grant from Abbott Diabetes Care outside the submitted work. RWB reported grants from The Leona M. and Harry B. Helmsley Charitable Trust during the conduct of the study and grants from Insulet, Tandem Diabetes Care, Beta Bionics, Dexcom, and Bigfoot Biomedical; nonfinancial support from Insulet, Tandem Diabetes Care, Beta Bionics, Dexcom, Medtronic, Ascenia, Roche, Eli Lilly, and Novo Nordisk; and consulting fees paid to his institution from Insulet, Tandem Diabetes Care, Beta Bionics, Eli Lilly, Novo Nordisk, Embecta, Hagar, and Ypsomed outside the submitted work. KH reported behavioral expertise consulting fees from Cecelia Health during the conduct of the study. GA reported grants from The Leona M. and Harry B. Helmsley Charitable Trust during the conduct of the study and personal fees from Bayer, Dexcom, and Insulet; nonfinancial support from Eli Lilly; and grants from Dexcom, Eli Lilly, Fractyl Health, Emmes, MannKind, Tandem Diabetes Care, and Welldoc outside the submitted work. No other disclosures were reported.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Study funding was provided by The Leona M. and Harry B. Helmsley Charitable Trust. Dexcom provided continuous glucose monitors for the study at a discounted rate.
Trial Registration
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
