Abstract
Artificial intelligence (AI) is increasingly being used to diagnose complications of diabetes. Artificial intelligence is technology that enables computers and machines to simulate human intelligence and solve complicated problems. In this article, we address current and likely future applications for AI to be applied to diabetes and its complications, including pharmacoadherence to therapy, diagnosis of hypoglycemia, diabetic eye disease, diabetic kidney diseases, diabetic neuropathy, diabetic foot ulcers, and heart failure in diabetes.
Artificial intelligence is advantageous because it can handle large and complex datasets from a variety of sources. With each additional type of data incorporated into a clinical picture of a patient, the calculation becomes increasingly complex and specific. Artificial intelligence is the foundation of emerging medical technologies; it will power the future of diagnosing diabetes complications.
Introduction
Diabetes mellitus represents a significant global health challenge, with an increasing prevalence and profound impact on individuals and health care systems worldwide. Despite advancements in treatment modalities and therapeutic interventions, the management of diabetes remains complex, particularly concerning the diagnosis and monitoring of its associated complications. 1
Recently, the integration of artificial intelligence (AI) into health care has emerged as a promising paradigm to enhance the diagnosis, prognosis, and management of various medical conditions, including diabetes and its complications. 2 Machine learning (ML) algorithms, in particular, offer the potential to analyze vast amounts of heterogeneous data types, ranging from electronic health records (EHRs) and imaging studies to genomic information and wearable sensors data. These methods comprise a variety from supervised, unsupervised, reinforced learning, generalization optimization, reduction algorithms, ensemble learning, transfer learning, and meta-learning techniques. A selection of frequently employed types of AI algorithms used in the creation of diagnostic and prognostic frameworks is illustrated in Figure 1. Machine learning techniques, such as deep neural networks and tree-based ensemble models, have demonstrated notable efficacy in predicting complications associated with diabetes such as retinopathy development 3 and risk of future hypoglycemia during daily living 4 and in hospital patients. 4
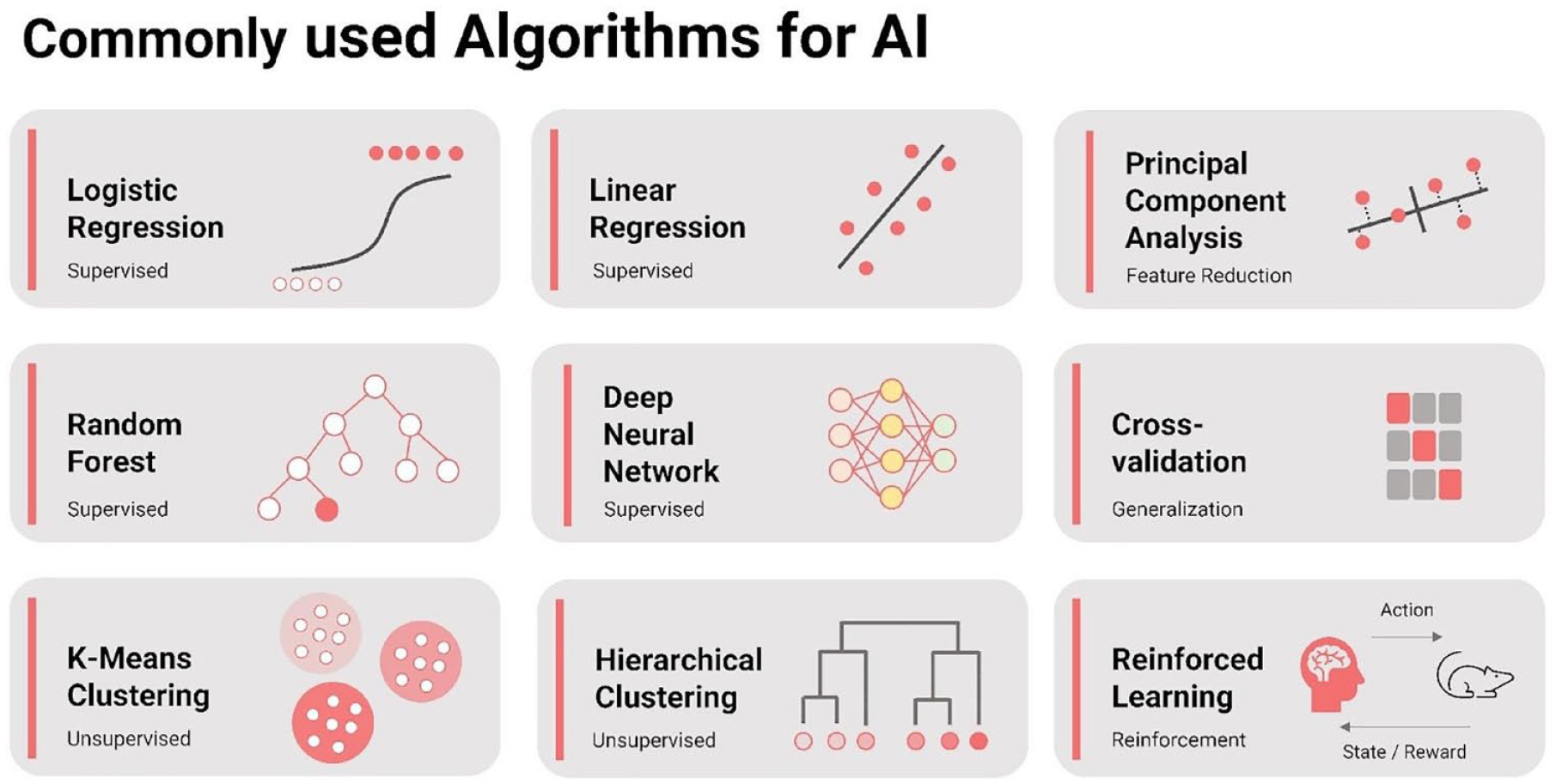
Frequently employed types of algorithms for AI. This figure illustrates a variety of algorithms frequently employed in the field of AI, categorized by their type and purpose (supervised, unsupervised, reinforcement, feature reduction, generalization).
By leveraging advanced computational techniques, AI models can uncover intricate patterns, identify novel biomarkers, and generate personalized risk profiles for individuals with diabetes. The application of AI in diabetes care holds the promise of more accurate and timely detection of complications, enabling proactive interventions to mitigate risks and improve patient outcomes. 5
However, the deployment of AI in clinical practice raises important considerations regarding model interpretability, generalizability, scientific reporting, and ethical implications. Addressing these challenges is paramount to ensure the responsible and equitable integration of AI technologies into diabetes management.6,7
In this article, we review the current landscape of AI-based approaches for diagnosing complications of diabetes within seven areas: pharmacoadherence to therapy, hypoglycemia, diabetic eye disease, diabetic kidney disease (DKD), diabetic neuropathy (DN), diabetic foot ulcers (DFUs), and heart failure (HF) in diabetes. We discuss the current status of AI and expected future use of AI in relation to diagnostics.
AI and Pharmacoadherence
The use of AI to improve pharmacoadherence in diabetes care is in its infancy.
The potential value of AI to improve pharmacoadherence will be multifaceted.
Most of the benefits of AI will be achieved in conjunction with human-to-human interactions rather than AI in isolation.
“The real power of AI, when designed appropriately, is delivering highly personalized nudges and intelligent support that boost behavioral change and adherence.” 8
Current Status of AI to Diagnose Lack of Adherence to Therapy
The requirement for continuous use of oral and injectable medications is a daily requirement for most people living with diabetes. However, lack of adherence to a prescribed medication is common and may account for the gap between the hemoglobin A1c (HbA1c) lowering seen in clinical trials compared with what occurs in routine clinical practice where the glucose lowering effect of new medicines may be somewhat less than anticipated. 9 As an example, the introduction of smart insulin pens has highlighted the frequency and adverse consequences of insulin omission. 10 Challenges to adhering to a prescribed regimen include the sheer number of medications being prescribed, their mode of delivery (eg, injections instead of oral medications), cost, and concerns about side effects such as hypoglycemia or weight gain. Other factors influencing pharmacoadherence include psychosocial factors, such as peer influences, culture, myths, and stigma. 11 Although pharmacist-led interventions appear to be effective, previous approaches have not been shown to provide sustained benefits. 12 Going forward, digital health technologies supported by ML and AI are potential approaches to improve pharmacoadherence within diabetes care. 13
Artificial intelligence has already been used to predict the risk of nonadherence to prescribed medications by leveraging data from multiple sources, including electronic medical records, and pharmacy dispensing data, including requests for refills and insurance claims. 14 Additional approaches have included the combination of digital pill counters, telehealth-based interventions, ingestible sensors attached to oral medications, and motion sensors. 15 However, using AI to improve pharmacoadherence is in its infancy.
Future Use of AI to Diagnose Lack of Adherence to Therapy
At present, AI is being used to understand the link between medicine administration (eg, insulin dosing) and achieved glucose profiles from continuous glucose monitoring (CGM) data. 16 However, the opportunities for AI in diabetes care are much wider. For example, the use of large language models (LLMs) to capture doctor-patient interactions has the potential to offload mundane tasks, thus freeing up clinician time to interact with a person with diabetes on the topic of pharmacoadherence. 17 By freeing up a doctor’s time, this can allow greater use of empathy as a therapeutic tool as part of the traditional doctor-patient relationship. An illustration of the components of compassion in health care settings is illustrated in Figure 2. 18 Artificial intelligence can also be used to record, assess, and monitor diabetes complications including retinopathy and foot ulcers. From the perspective of pharmacoadherence, AI systems can also respond authoritatively (based on current and future evidence) to questions related to drug treatment; to stratify populations into subgroups to support personalization of drug therapies; to better understand the impact of multiple therapies, lifestyle changes, and comorbidities on treatment response 19 ; and to create opportunities to simulate patient-level reasoning (ie, counterfactual thinking) to the decision-making association with adherence. 20
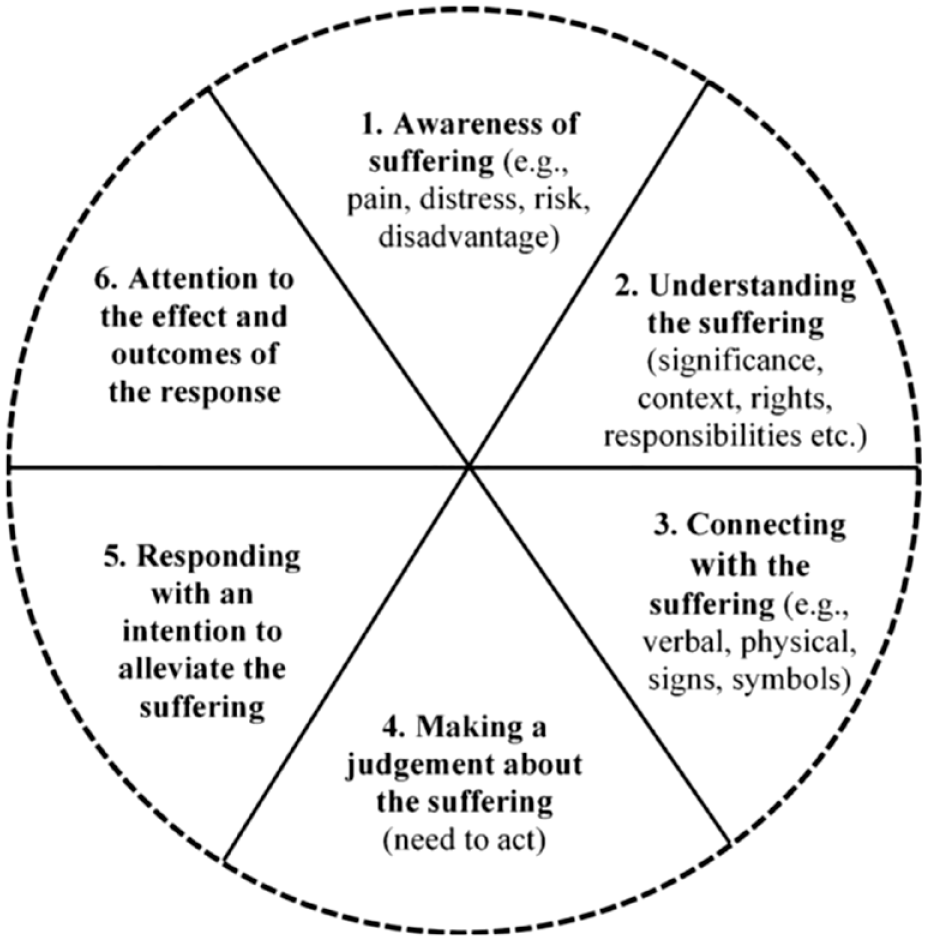
Conceptualizing compassion. Reproduced from Morrow et al 18 under the CC-BY license (https://creativecommons.org/licenses/by/4.0/).
The decision to take (or not) a prescribed medication has several external influences, including access to the medicines, the time burden, concerns about adverse events, or a lack of understanding of the potential benefits. The application of multimodal monitoring could be combined with AI to create just-in-time adaptive interventions (JITAIs) to facilitate pharmacoadherence albeit taking into consideration the pharmacokinetics and pharmacodynamics of the medication being considered. A JITAI takes data from multiple sensors to monitor an individual’s physiology and behavior in real time. The data are analyzed to detect moments when an intervention (eg, taking additional exercise) is most likely to be accepted and effective by an individual. The type and timing of the intervention is usually delivered through a connected device or phone with the system continuously learning about the effectiveness of this approach. 21 The use of JITAIs is already being employed in diabetes care to improve physical activity behaviors. 22
For AI to be successful in improving pharmacoadherence, there will be a need to educate both prescribers of AI systems and users of the system, that is, people with diabetes. In addition to knowledge, trust in technology is especially important for communities facing a disproportionate burden of diabetes and health disparities. To improve pharmacoadherence through the application of AI, it will be essential for the AI model to have included data and testing from these marginalized and historically excluded populations—to use only ethical AI.
Hypoglycemia
An updated literature review from 2015 to June 2024 identified 70 studies on hypoglycemia ML models reporting performance metrics, highlighting advancements in predictive performance.
Most ambulatory prediction models targeted patients with type 1 diabetes (T1D) using small sample sizes with CGM data and short prediction horizons, while inpatient models used larger EHR data sets, included mixed populations, and had longer prediction horizons.
Although many models reported good to excellent predictive performance in internal validation, few have been externally validated, or deployed and evaluated prospectively for usability or clinical utility.
Current Status of AI to Diagnose Hypoglycemia
Hypoglycemia is a significant complication of diabetes management and ranges from mild to severe in clinical impact. 23 Artificial intelligence continues to advance in hypoglycemia prediction and detection, as highlighted by comprehensive reviews from Zhang et al 24 and Tsichlaki et al. 25 We conducted an updated literature review through May 2024 and identified 70 studies with sufficient information on the predictive performance of ML models.4,26-93
Among these studies, general hypoglycemia (mild or severe) was the most common scenario (40.0%), followed by hypoglycemia in the hospital (24.3%), and nocturnal hypoglycemia (22.9%) (Figure 3a). Less commonly, studies addressed postprandial hypoglycemia, exercise-induced hypoglycemia, and hypoglycemia during driving simulation. 51 Most ambulatory prediction models targeted patients with T1D (60.4%), whereas inpatient models primarily included mixed populations (70.6%) (Figure 3b). Ambulatory studies often used small sample sizes (<100 participants) and integrated data from CGMs and insulin pumps. In contrast, inpatient models typically involved larger EHR data (Figure 3c). Consequently, prediction horizons were shorter in ambulatory settings compared with inpatient settings (Figure 3d).
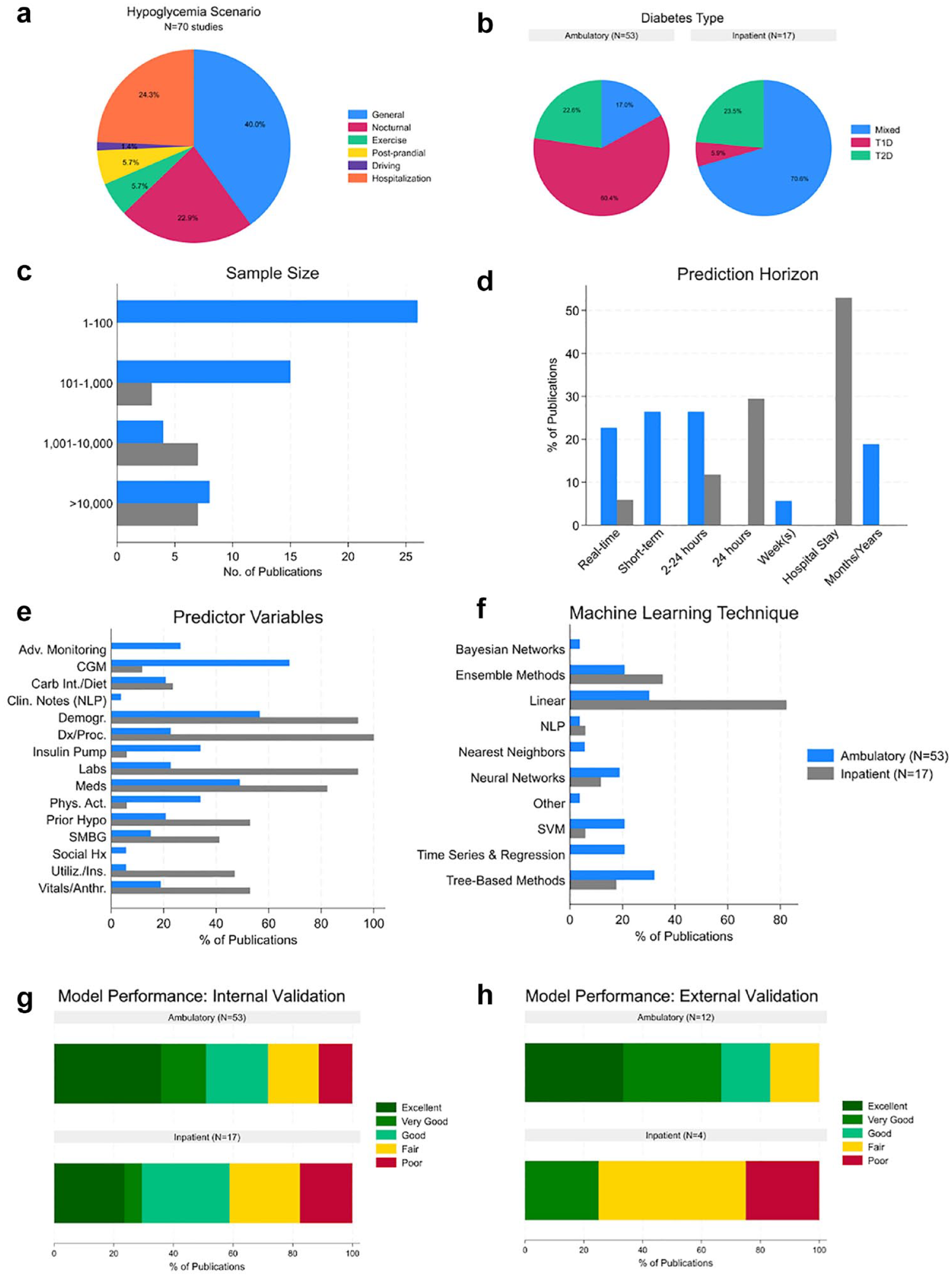
Key findings in AI/ML-based prediction models for hypoglycemia from 70 studies reporting sufficient performance metrics. These findings include a scenario surrounding hypoglycemia (a), whether T1D, T2D, or both were studied (b), sample size (c), length of prediction horizon (d), data inputs (e), ML technique (f), internal validation model performance (g), and external validation model performance (h).
Data inputs varied by setting: ambulatory models used advanced monitoring technologies (eg, smartwatches, fitness trackers, electrocardiogram), CGMs, and insulin pump data, whereas inpatient models used information widely available in the EHR (Figure 3e). Common ML techniques included linear models (eg, logistic regression), tree-based methods (eg, random forest), and ensemble methods (eg, XGBoost) (Figure 3f). Ambulatory ML models, with access to robust CGM data, also employed neural networks, support vector machines, and time series methods.
Most models reported internal validation results (Figure 3g), with only 16 studies providing external validation performance (Figure 3h). In internal validation, the majority showed excellent to good predictive performance across settings, with some fair and poor performance in inpatient settings. In external validation, ambulatory models had a high proportion of excellent and very good ratings, whereas inpatient models showed significant fair and poor performance.
Certain factors and demographic characteristics are associated with a higher risk of hypoglycemia: age, ethnicity, educational level, lab test results, pre-existing conditions, medications, and more. 24 For instance, patients treated with insulin may be at a higher risk for hypoglycemia. 94 On an individual level, AI algorithms can include these and other factors to effectively prevent hypoglycemia and severe hypoglycemia in high-risk individuals and avoid the corresponding complications associated with hypoglycemia.
Future Use of AI to Diagnose Hypoglycemia
Despite the high predictive accuracy of validated models, few studies have prospectively evaluated ML-based models for their ability to predict hypoglycemia and reduce its incidence or severity. 95 In the inpatient setting, practical challenges remain in configuring ML models within EHR systems. Another challenge is the lack of interoperability, particularly accessing discrete data from CGMs or insulin pumps. The EHR vendors in the United States are working on this issue, and once discrete data are accessible within EHRs, prediction models combining CGM and EHR data will likely become more accurate. Some EHR systems have cognitive computing platforms to enable ML model deployment, but configuring these models is resource-intensive and requires identifying integration points, features, software to execute the model, and model scheduling.
We anticipate that future models will integrate multiple data sources (noninvasive sensors and EHR data) to enhance predictive accuracy. Key factors for successful adoption will include having accurate models with a relevant prediction horizon for timely interventions. In addition, future models that integrate automated medication adjustments are needed for insulin-treated patients with type 2 diabetes (T2D).
Large language models represent another potential area of development. These could be more useful for predicting long-term hypoglycemia risk or identifying at-risk individuals from EHR notes rather than for real-time, personalized risk prediction. Predictive accuracy for postprandial and exercise-related hypoglycemia remains relatively low, necessitating further research. Future research should focus on understanding acceptable thresholds for false positives/negatives for hypoglycemia prediction in various contexts and evaluating the usability and clinical utility of AI/ML-based prediction tools for hypoglycemia.
Eye Disease
Artificial intelligence in ophthalmology shows high diagnostic accuracy for diabetic retinopathy (DR) and glaucoma.
Three FDA-approved AI systems enable autonomous screening for early detection of more-than-mild diabetic retinopathy (mtmDR) by primary care providers, without the need of specialists.
Future AI advancements aim to enhance real-time monitoring of ophthalmological abnormality through the integration of wearable devices.
Current Status of AI to Diagnose Eye Disease
Artificial intelligence is revolutionizing the field of ophthalmology by detecting eye diseases from images with accuracy comparable with, or surpassing, that of a human physician in real time,96-98 even beyond the utility of already-established automated eye scans. The proficiency of AI in interpreting complex retinal images, such as fundus photographs and optical coherence tomography (OCT) scans, is illustrated in Figure 4. The AI models show promise in diagnosing eye conditions such as DR, glaucoma, and visually significant cataracts (VSC) which typically require ophthalmologists to identify. 99 A recent study by Kubin et al 100 evaluated 21 algorithms for DR screening and revealed that the top five algorithms achieved high accuracy (89.2%), sensitivity (84.3%), and specificity (91.3%) at identifying referable DR. A three-dimensional deep learning system for glaucoma using spectral-domain OCT images achieved an impressive sensitivity of 89% and specificity of 96%, outperforming traditional two-dimensional (2D) imaging–based AI algorithms. 100 For VSC, Young et al 101 reported an XGBoost model, trained using Optum EHR data from nearly 1.5 million patients, that can triage patients for referral to eye care specialists with a sensitivity of 76.8% and a specificity of 82.4%. These developing AI models highlight the potential clinical applications in aiding clinicians in early detection and monitoring of eye diseases with efficient, consistent, and accurate assessments of complex retinal images and EHR data.
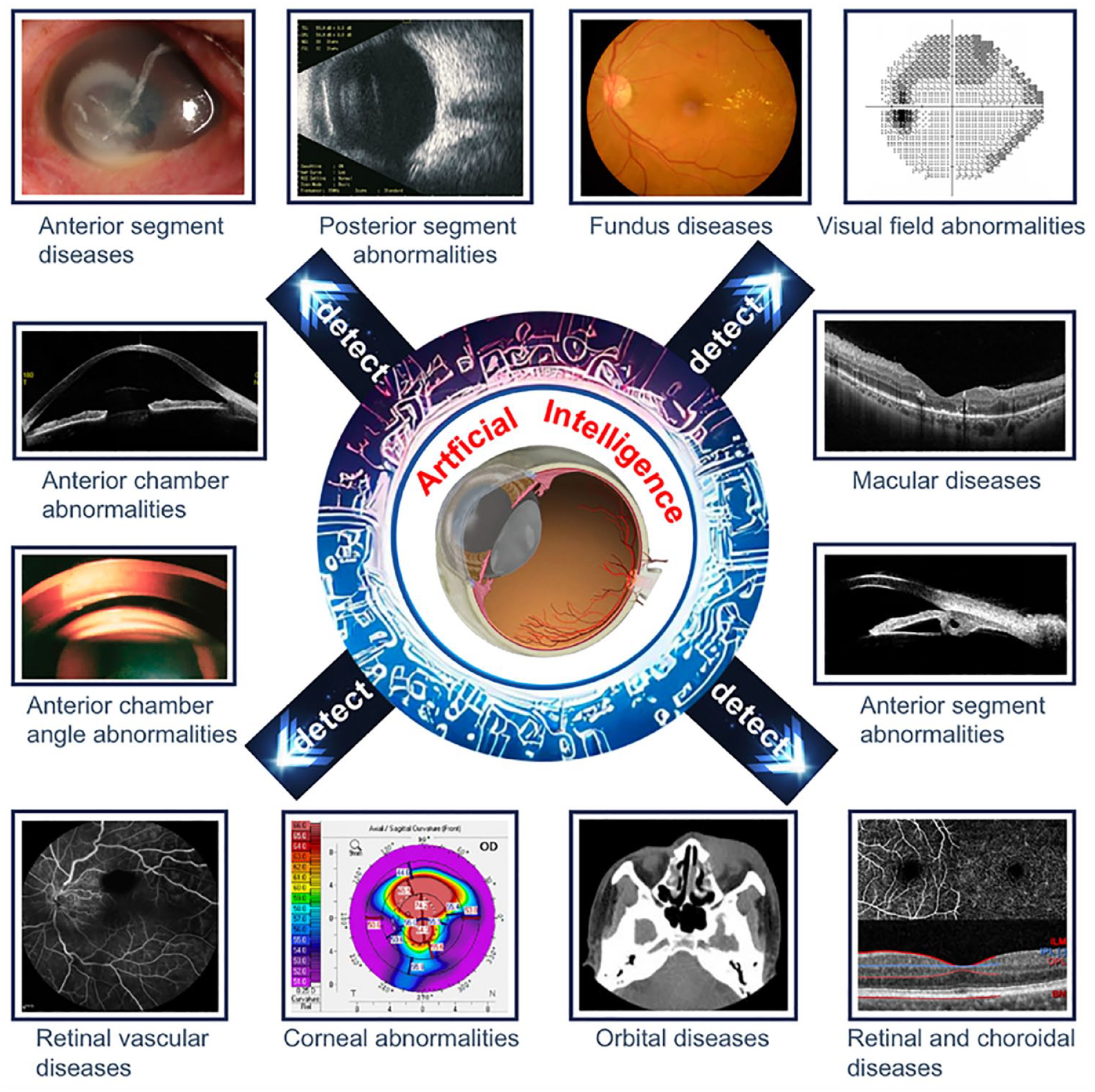
Overall schematic diagram describing the practical application of AI in all common ophthalmic imaging modalities. Figure reproduced from Li et al 99 under the CC BY-NC-ND license (http://creativecommons.org/licenses/by-nc-nd/4.0/).
The AI-based screening tools, approved by the Food and Drug Administration (FDA), are increasingly being implemented in primary care and remote settings, enabling timely detection and interventions of eye diseases. In 2018, IDx-DR was the first AI software approved by the FDA to aid health care providers in detecting mtmDR with high sensitivity (85.0%) and specificity (82.5%). Such high performances lead to a widespread use in primary care settings and may particularly benefit underserved populations and communities.102-104 Today, there are not enough well-trained ophthalmologists to identify DR. Artificial intelligence would address this urgent need. The EyeArt Automated DR Detection System, approved in 2020, demonstrated even higher sensitivity (95.5%) and specificity (85.0%) for detecting mtmDR compared with IDx-DR.96,105 The most recent new AI software for the diagnosis of mtmDR, AEYE-DS, which was approved in 2024, achieved high sensitivity between 93% and 94.7% and a specificity between 88.6% and 91.4%. 12 Postapproval monitoring in real-world settings is crucial in ensuring the safety, effectiveness, and reliability of these novel AI-based tools. Despite the recent proliferating development of AI-based screening and diagnosing tools for eye diseases, only 3 algorithms successfully gained FDA’s approval while the majority remained in the research development stage.99-101,106 This small number highlights both the potential and the cautious approach taken toward integrating AI into clinical practice for the field of ophthalmology. 107
Future Use of AI to Diagnose Eye Disease
Ongoing research is focused on improving the reliability of AI models. This includes optimizing accuracy in challenging cases and complex images. Future AI systems will train on larger, more diverse data sets to improve their performance across various populations, addressing current limitations related to biases in training data. Collaborative initiatives between technology companies and medical institutions are vital to accelerate the development and integration of AI technologies into ophthalmology clinics.
The integration of AI with wearable technology—such as devices including smart contact lenses, augmented reality glasses equipped with AI algorithms, and vision tracking cameras—will continuously monitor eye health, detecting early signs of disease and prompting timely medical consultations. 108 Remote monitoring systems will provide real-time data to ophthalmologists, facilitating ongoing patient management and reducing the necessity for frequent in-person visits. 108
Early detection of diabetes-driven eye diseases, such as DR, is especially critical since timely intervention and ongoing management are essential to prevent disease progression.
Ongoing efforts in AI research and development not only promise to reduce the burden on health care professionals by streamlining clinical workflow, but also offer the potential to improve patient outcomes through early detection facilitated by AI-powered technology and integrated solutions that pair AI technology with appropriate treatment protocols.
Kidney Disease
AI/ML can be used in conjunction with electronic medical records to help diagnose or predict the likelihood of a patient developing chronic kidney disease (CKD)/ DKD as well as associated mortality/morbidity.
AI/ML can be used to analyze renal ultrasound (US) and magnetic resonance imaging (MRI) images to identify and diagnose CKD/DKD.
In the future, we will see even more advanced AI/ML tools to assist with the diagnosis of kidney disease, down to the specific biomolecules implicated.
Current Status of AI to Diagnose Kidney Disease
Currently, AI can be used as a diagnostic tool for kidney disease by providing alternatives to kidney biopsy, analyzing kidney images, and even predicting the development of kidney disease before it occurs. Patients with both CKD and diabetes may have DKD (which is a complication resulting from diabetes), nondiabetic kidney disease (NDKD), or both. 109 Both DKD and NDKD can be differentiated through various pathological features and indicators such as the patient’s duration of DM, presence or absence of retinopathy, red blood cell count, and urinary glucose excretion. 110
Kidney biopsy is the “gold standard” method used to diagnose DKD versus NDKD in patients with DM, but the procedure can be risky and many patients may be unable or unwilling to undergo a biopsy, hence kidney biopsies are uncommon in patients with DM.110,111 To provide an alternative to biopsies as a diagnostic tool for DKD, a group of researchers created an ML model that can diagnose DKD in an effective, noninvasive manner using five key indicators: duration of DM, systolic blood pressure, HbA1c levels, presence of hematuria, and DR. This model has relatively high sensitivity (80%) and specificity (100%), indicating the usefulness of this model, instead of kidney biopsy, for diagnosis of DKD. 111 It is important to diagnose DKD versus NDKD as early as possible, as different strategies are used to treat the two diseases. 110 In this way, AI could help to establish a diagnosis, identify a best treatment, and provide a better prognosis for patients with DKD and NDKD.
Various AI/ML tools have also been developed to diagnose CKD by analyzing kidney images from US or MRI. Convolutional neural networks (CNNs) are particularly adept at finding patterns in images 112 ; CNNs and similar ML tools can identify microscopic structural changes, such as interstitial fibrosis, tubular atrophy, and glomerulosclerosis that are indicative of CKD.112,113 For instance, one CNN that analyzed renal US images was able to noninvasively predict estimated glomerular filtration rate, the main biomarker for diagnosis of CKD, with 85.6% accuracy. 114 According to Zhao et al, 113 this model was able to identify CKD more accurately than other ML models and even nephrologists.
Researchers have also used AI/ML to analyze retinal images to predict the development of CKD/DKD, because similar pathophysiological changes may occur both in the retina and kidney of a person with diabetes. 113 This type of retinal screening may increase the likelihood of an early diagnosis and more effective intervention for patients with CKD/DKD. 113 In a study conducted by Sabanayagam et al, the researchers developed a deep learning algorithm (DLA) to diagnose CKD based on retinal images, classic risk factors for CKD, or both. They found that DLA was similarly successful at predicting CKD using either retinal images or risk factors. 115
Artificial intelligence has also been used both to predict the likelihood of a person developing diabetes, and, once they have already been diagnosed with diabetes, to predict diabetic complications. For instance, one group of researchers used ML alongside patient data to “predict the onset of DKD with high accuracy, approximately two to three months before clinical diagnosis.” 111 The ML algorithms that use genetic information and biomarkers, together, have been found to be the most accurate method for predicting the development of DKD. 111 Current uses of AI/ML to diagnose kidney disease, as well as potential future uses of AI/ML to diagnose kidney disease, are summarized in Figure 5.
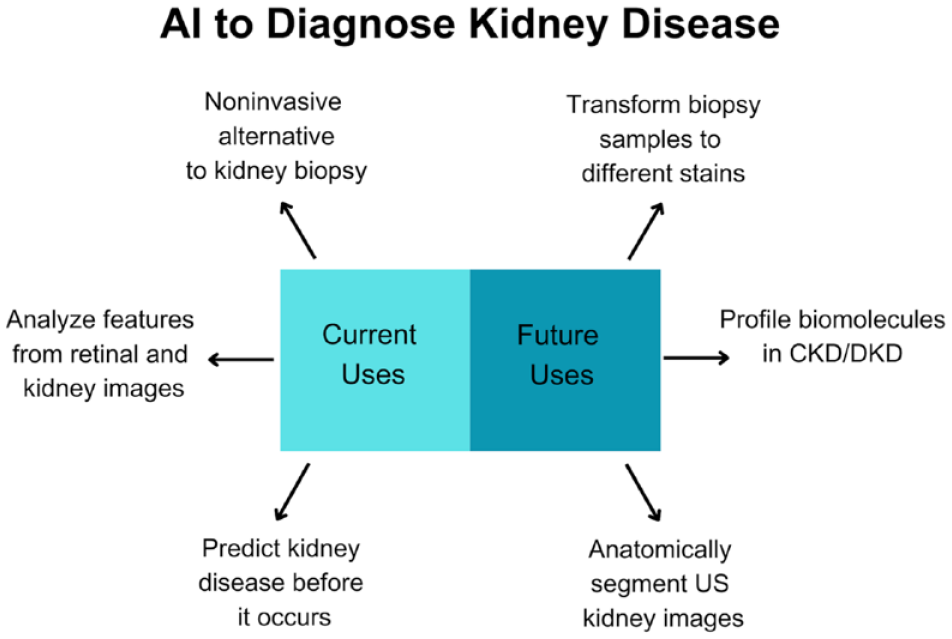
Summary of current and future uses of AI to diagnose kidney disease as a complication of diabetes.
Future Use of AI to Diagnose Kidney Disease
In the future, AI may be used to transform different stains on kidney biopsy samples, profile biomolecules implicated in CKD/DKD, and segment renal US images anatomically for further analysis and diagnosis. According to Goodman et al, 112 it may soon be possible for a person to perform just one stain on a kidney biopsy sample, and then “digitally transform . . . slides into other routine stains utilized in renal pathology.” Furthermore, other research groups are trying to use AI to analyze such biopsy slides to profile biomolecules found in CKD. The combination of “single cell and spatial multimodal data,” light microscopy, and AI would enable these researchers to predict RNA, protein, and metabolic profiles of various biomarkers found on renal light microscopy slides. 112 These two applications of AI would allow physicians to more easily analyze renal pathology and diagnose kidney disease.
Finally, Yin et al developed a successful CNN algorithm that can anatomically segment US kidney images. Kidney segmentation is the crucial first step in renal image analysis and kidney disease diagnosis. Although this model was only used to segment healthy kidneys, in the future, researchers may also be able to segment and diagnose kidneys exhibiting signs of kidney disease. 114
Neuropathy
Current approaches for diagnosing DN are labor-intensive and time-consuming, hindering frequent and large-scale screening for early detection.
Machine learning techniques leveraging diverse data modalities have shown promise in classifying DN (sub)-phenotypes in proof-of-concept studies.
Foundation models, such as LLMs, may enhance clinical diagnosis and complement existing ML techniques by integrating quantitative, qualitative, and human factors.
Current Status of AI to Diagnose Neuropathy
Diabetic neuropathy, a range of diverse clinical manifestations resulting from nerve damage due to diabetes, is the most prevalent complication of diabetes with high mortality risks. 116 Peripheral and autonomic neuropathies are the most common types, affecting nearly 50% and 30% of patients with diabetes, respectively.117,118 The American Diabetes Association recommends an annual neuropathy assessment. 119 However, timely diagnosis and large-scale screening for DN have been challenging, given the lack of specificity, labor-intensiveness, and the time-consuming nature of traditional diagnostic methods. Untreated DN can exacerbate nerve damages, leading to severe complications and impaired quality of life. Currently, treatments to reverse the underlying nerve damage are not available.
To address the need of efficient and early detection for DN, various innovative approaches leveraging the rapidly advancing AI and ML methods have been developed to classify (sub)-phenotypes of DN. Classical ML algorithms and recurrent neural network models using clinical measures, such as biochemistry laboratory tests, cardiovascular autonomic reflex testing, heart rate variability, or electrocardiograms, have shown promise in identifying phenotypes of cardiac autonomic neuropathy in proof-of-concept studies.120,121 Automated CNN-based systems demonstrated high efficiency and accuracy in differentiating patients with and without peripheral diabetic neuropathy (PDN) using corneal confocal images, which is demonstrated in Figure 6.122-124 This imaging method is used to quantify small nerve fiber degeneration in small fiber neuropathy. 125
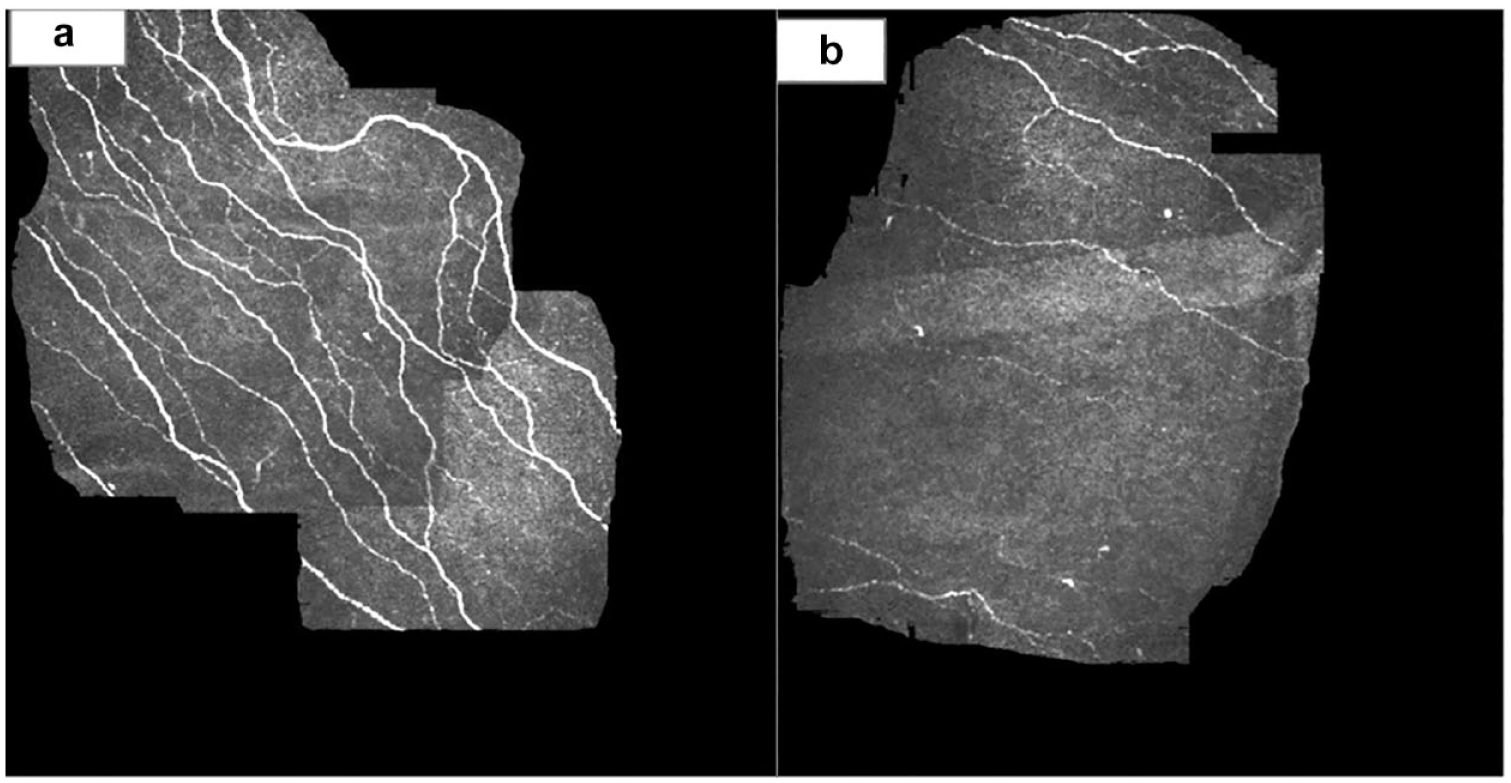
Corneal confocal microscopy showing the sub-basal nerve plexus. (a) Normal structure corneal nerve fibers in a healthy subject. (b) Loss of corneal nerve fibers in a recently diagnosed subject with type 2 diabetes. Figure and caption reformatted from Papanas and Ziegler 126 under the CC BY-NC license 4.0 (https://creativecommons.org/licenses/by-nc/4.0/).
Other data, such quality of life, lifestyle, personality, and psychology traits, as well as magnetic resonance images for functional connectivity of the brain have also been explored to identify sub-phenotypes of PDN, including treatment responses and distinctions between painful and painless PDN.127-130 Notably, a novel noninvasive, contactless diagnostic point-of-care-device powered by ensemble ML models can also identify PDN accurately by analyzing facial erythema of patients. 131
However, while many ML models and systems have achieved significant successes in identifying DN, they relied on limited sample sizes derived from institution-specific databases. External validation studies were rarely conducted, limiting the generalizability and potential utility of these models in larger populations.
Future Use of AI to Diagnose Neuropathy
The ML algorithms have the potential to improve early detection of DN through the integration of multimodal clinical data. Existing models have shown promising utility, yet external validations studies are still needed in larger and more diverse patient populations before these models can be adopted in clinical settings.
Future ML algorithms and AI to diagnose DN may further benefit from integrating human factors to minimize the risk of quantitative fallacy, which is a decision-making process based solely on quantitative metrics. 132 Prior studies have largely followed traditional model training paradigm where models were developed and trained from scratch to specialize in classifying phenotypes of DN. We anticipate that the emerging foundation models, including (multimodal) LLMs, that were pretrained with vast amount of data may play an increasing role in health care applications, including DN diagnosis. The LLMs have shown the capability to diagnose complex clinical cases, analyze extensive medical records and texts for those with delayed diagnoses, and generate empathetic responses.98,133-135 These findings underscore the potential advantages of integrating LLMs with ML diagnostic methods to take both quantitative data and human factors that may be captured in clinical texts and were previously unattainable.
Foot Ulcers
Artificial intelligence can be used to classify foot wound images to detect ischemia and infections.
Artificial intelligence can be used to predict complications due to DFUs.
An AI-derived phenotype of frailty can predict a need for a major limb amputation.
Enhancing Diagnosis and Management of DFUs With AI
The DFUs represent a critical challenge in diabetes management, necessitating an integrated approach that incorporates emerging technologies. Artificial intelligence has the potential to significantly enhance the personalization of DFU management by analyzing comprehensive data sets, including patient history, lifestyle, and genetic information. 136 While current evidence supporting the benefits of AI in managing DFUs is still emerging, recent studies offer promising results in several areas, which is illustrated in Figure 7. These advancements include the automation of wound classification, comprehensive insights into potential risk factors, support in decision-making, prediction of major adverse events such as the risk of major amputation, and the facilitation of remote triaging for hard-to-heal wounds through digital biomarker surrogates like frailty. Such advancements may assist in tailoring treatment plans based on the wound healing process, amputation risk, and other individual factors. Each of these developments is summarized in this section.
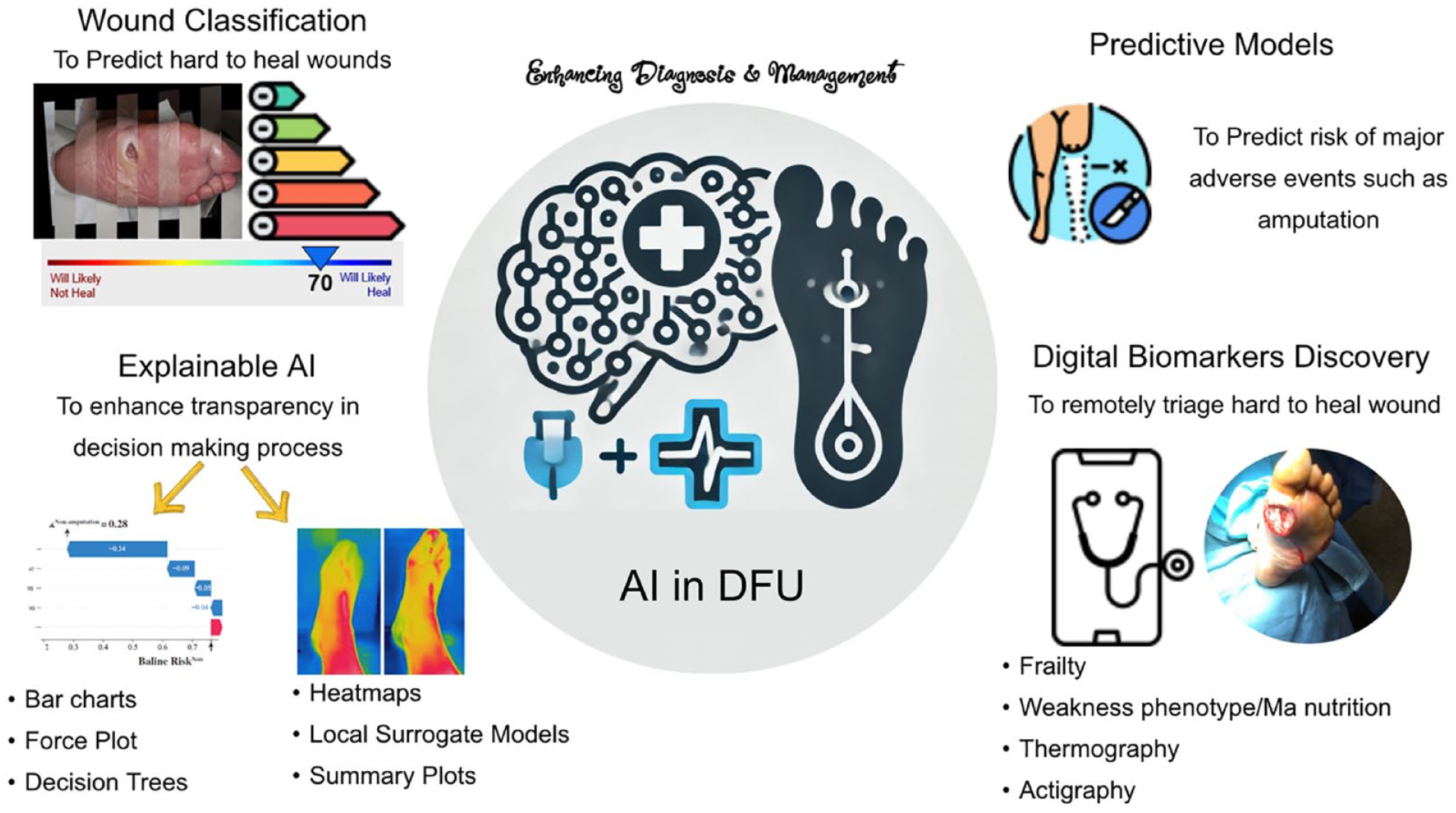
AI in DFU. The evidence supporting the benefits of AI in enhancing the management of diabetic foot ulcers remains limited. However, recent studies offer promising results in several areas. These include the use of AI to automate wound classification, provide comprehensive insights into potential risk factors, aid in decision-making,137,138 predict major adverse events such as the risk of major amputation, and potentially facilitate remote triaging of hard-to-heal wounds. This is achieved through the use of digital biomarker surrogates, such as frailty, to assess wound complexity. Component of figure adapted from Xie et al under the CC-BY-NC license (http://creativecommons.org/licenses/by-nc/4.0/). Component of figure adapted from Crepaldi et al under the CC-BY license (https://creativecommons.org/licenses/by/4.0/).
Revolutionizing Wound Classification With AI
Artificial intelligence is making significant strides in improving wound classification methods, which are crucial for accurate diagnosis and optimal treatment planning. Traditionally, wound classification was performed manually by specialists, often resulting in inconsistencies due to the absence of standardized guidelines. The introduction of AI has revolutionized this process by providing more objective and precise grading of wounds and their complexity. Several studies have highlighted the potential of AI-based image processing models to enhance wound classification and assist in the triage of hard-to-heal wounds.
For instance, Ahsan et al 139 used DLAs to automatically classify DFUs, particularly those complicated by infection and ischemia. Using the Diabetic Foot Ulcer 2020 (DFU2020) database, 140 which contains 1459 classified wound images with resolutions ranging from 1600 × 1200 to 3648 × 2736, their model achieved an impressive accuracy of 99.49% for detecting ischemia and 84.76% for detecting infections. Similarly, Patel et al 141 employed a deep CNN model to categorize wounds into four categories: diabetic, pressure, surgical, and venous ulcers. Using standard 2D images of wounds and incorporating information about wound location, their model reported an accuracy range of 78.10% to 100% for whole image classification.
Enhancing Decision-Making Assessment With Explainable AI (XAI)
One of the significant barriers to the use of AI in determining wound complexity is the lack of transparency and the inherent black-box nature of these models. These AI models process and interpret data instantaneously from an existing data set, without storing the learned data as clear digital memory, making their decision-making processes opaque. The use of XAI 142 offers a solution by enhancing transparency and providing guided inference to understand the AI system’s decision-making processes.
Several studies have employed XAI, which is a set of methods and processes to assist people to understand and trust ML algorithms to classify wound images, including DFUs. For instance, Sarp et al 143 used the Local Interpretable Model-Agnostic Explanations (LIME) model 143 on 8692 chronic wound images labeled by wound care specialists. They reported an average precision, defined as the ratio of true-positive predictions to the total number of positive predictions made, of 83% for classifying the images into four categories: DFU, lymphovascular wounds, pressure injury wounds, and surgical wounds. The predictions made by the model are explained by an “explainer,” which highlights the visual features most important to the model using a heatmap-based explanation method. In these heatmaps, warmer colors indicate the more critical areas of the wound in the importance map. This information allows clinicians to understand the model’s rationale and decide whether to trust its output.
Predictive AI Models for Complications
In another study, Xie et al 137 used an explainable prediction model to estimate the risk of in-hospital amputation in patients with DFUs. They employed the Light Gradient Boosting Machine (LightGBM) model, originally developed by Microsoft, 144 for classification, using data from 618 hospitalized patients with DFUs. Their model included data from demographic features; medical and medication history; clinical and laboratory data; Wagner Ulcer Classification; Wound, Ischemia, and foot Infection (WIfI) Classification; and clinical outcomes (amputation and death) from the EHR during hospitalization. The model predicts whether a patient will not have any amputation, or undergo a minor or major amputation during hospitalization, with reported areas under the curve (AUC) of 0.90, 0.85, and 0.86, respectively.
To interpret the model output, they used the SHapley Additive exPlanations algorithm. 145 This algorithm provided insights based on baseline risk and patient characteristics. For example, their model’s calculated predicted probability for nonamputation showed that a high Wagner score, high foot infection score, long diabetes duration, and the presence of gangrene reduced the probability of nonamputation by −0.34, −0.05, −0.04, and −0.09, respectively. Conversely, the remaining features included in their model contributed +0.04 to the probability. A similar explanatory approach was applied to the probabilities of minor and major amputations. 137
Determining Digital Biomarkers to Triage Hard-to-Heal Wounds
Recent studies also suggest that AI may help determine digital biomarkers that indirectly assist in assessing wound healing complexity. For example, Mishra et al
146
used a validated AI model to determine frailty using a 20-second repetitive elbow flexion extension
147
and demonstrated that this model could distinguish those with hard-to-heal wounds based on stages 3 and 4 of the Society for Vascular Surgery (SVS) WIfI risk stratification system for major limb amputation. They found that AI-derived frailty phenotypes, including exhaustion phenotypes measurable remotely, enabled the distinction of stage 3 and 4 wounds from stage 1 and stage 2 wounds with large to very large effect sizes (Cohen effect size
Heart Failure
Machine learning models have shown promise in predicting incident HF risk among patients with diabetes, outperforming traditional scores.
Novel AI approaches using energy waveform electrocardiogram (ewECG) features demonstrate high accuracy in detecting subclinical left ventricle dysfunction in asymptomatic patients with T2D, potentially enabling early intervention.
Future research should focus on validating AI models in larger, diverse cohorts and exploring the integration of multimodal data to enhance predictive performance and clinical utility.
Current Status of AI to Diagnose HF
Heart failure is one of the most prevalent and severe cardiovascular complications in people with diabetes. 148 Despite its high prevalence, HF often remains underdiagnosed because of its insidious onset. 149 Early detection of HF could enable timely treatment to prevent disease progression. Artificial intelligence has emerged as a potential tool to predict and detect HF in high-risk populations, such as those with diabetes. 148
Four studies investigating the application of AI in HF risk prediction and detection among people with diabetes are summarized in Table 1. Pandey and colleagues developed the WATCH-DM score, an integer-based clinical risk score derived from a random survival forest model, which improved the prediction of five-year HF incidence compared with traditional Cox models (C-index 0.77 vs 0.73) in 8756 patients with T2D from the Action to Control Cardiovascular Risk in Diabetes (ACCORD) trial. 150 The authors further validated the ML-based WATCH-DM score in other cohorts, including 3554 patients with T2D and 5384 patients with prediabetes from the Atherosclerosis Risk in Communities (ARIC) Study, Cardiovascular Health Study, Framingham Heart Study, and Multi-Ethnic Study of Atherosclerosis, demonstrating improved performance (C-index 0.80) compared with the integer-based WATCH-DM score (C-index 0.71) and showing further improvement when integrating natriuretic peptides. 151 Finally, high WATCH DM score also identifies individuals who may be more likely to benefit from preventive therapies such as SGLT2i and can inform treatment approaches in the EHR.
Summary of AI-Based Models for Heart Failure Risk Prediction and Detection in Patients With Diabetes.
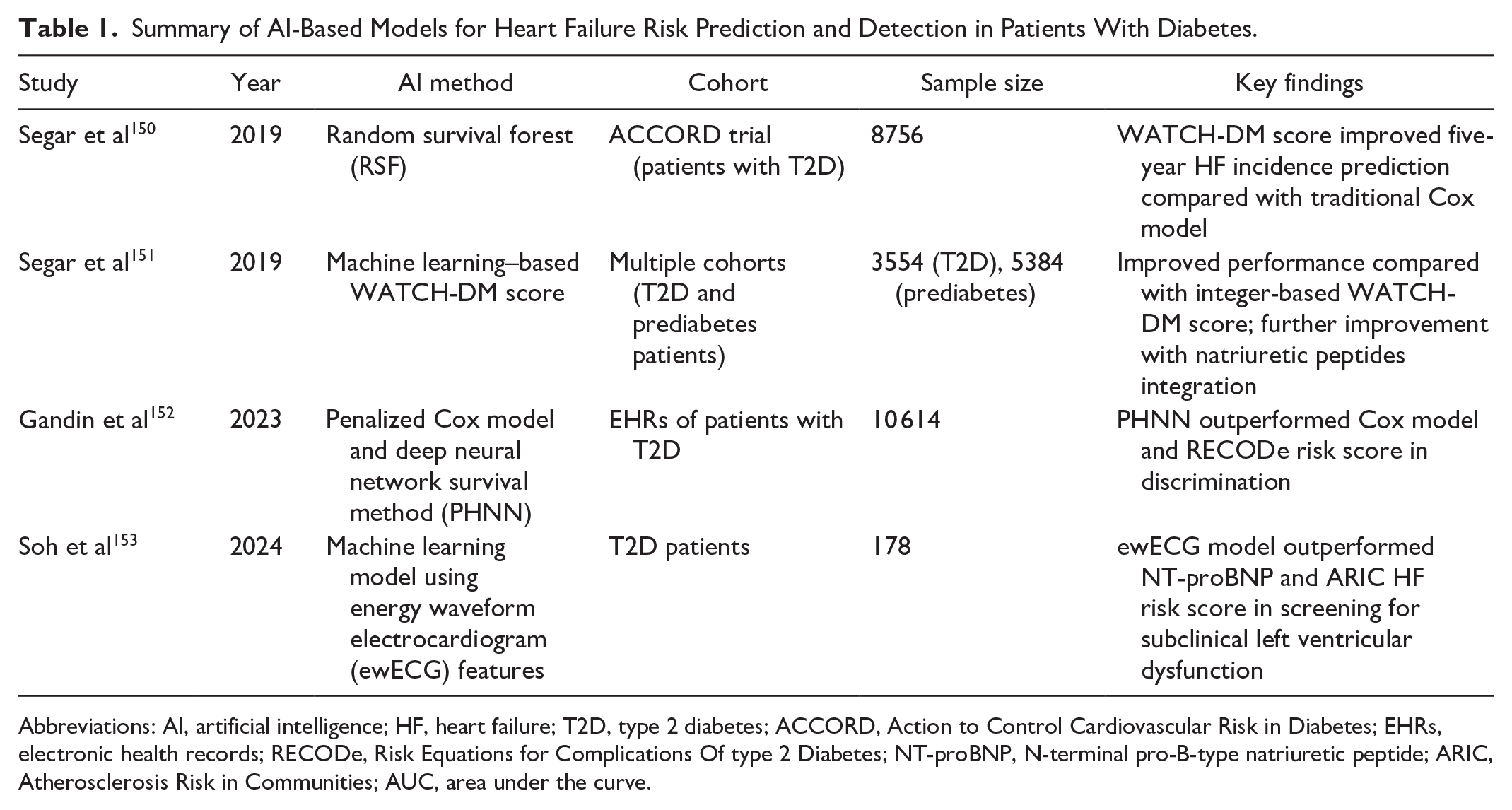
Abbreviations: AI, artificial intelligence; HF, heart failure; T2D, type 2 diabetes; ACCORD, Action to Control Cardiovascular Risk in Diabetes; EHRs, electronic health records; RECODe, Risk Equations for Complications Of type 2 Diabetes; NT-proBNP, N-terminal pro-B-type natriuretic peptide; ARIC, Atherosclerosis Risk in Communities; AUC, area under the curve.
Gandin et al 152 investigated the use of EHRs and AI techniques for HF risk prediction in 10 614 patients with T2D. They compared a penalized Cox model with a deep neural network survival method (PHNN), which outperformed the Cox model and traditional Risk Equations for Complications Of type 2 Diabetes (RECODe) risk score in terms of discrimination (C-index 0.768 vs 0.734 vs 0.670). The AI approach used 20 predictors, identified through feature selection, from different domains, such as age, body mass index, echocardiographic and electrocardiographic features, laboratory measurements, comorbidities, and therapies, corresponding to known trends in clinical practice.
Soh et al 153 developed an ML model using ewECG features to detect subclinical left ventricular dysfunction (LVD) in 178 patients with T2D. The ewECG model outperformed NT-proBNP and the ARIC HF risk score in screening for subclinical LVD, with an AUC of 0.81 compared with 0.56 and 0.67, respectively. The authors demonstrated that the ewECG model could be a valuable tool for identifying asymptomatic patients with T2DM who require echocardiography for subclinical LVD diagnosis, potentially preventing or delaying the progression to overt HF.
More recently, there has also been growing interest in developing AI-based clustering models to identify diabetic cardiomyopathy, a subclinical form of stage B HF among individuals with T2D. Such clustering approaches have identified subgroup of patients with high burden of abnormalities in cardiac structure and function and increased downstream risk of HF. Artificial intelligence–based echocardiographic algorithms have also been recently approved by FDA to diagnose HF with preserved ejection fraction and future studies are needed to determine how such algorithms may play a role in identifying individuals with T2D who may have subclinical HFpEF.
Future Status of AI to Diagnose HF
Future research should focus on validating AI models for HF risk prediction and detection in larger, diverse cohorts of patients with diabetes. Prospective studies are needed to evaluate the clinical utility and cost-effectiveness of implementing these AI algorithms in real-world settings. In addition, the integration of multimodal data, such as EHRs, echocardiography, and novel biomarkers, may further enhance the predictive performance of AI models. As the field of AI continues to evolve, emerging techniques such as explainable AI and federated learning should be explored to improve the interpretability and generalizability of these models, ultimately facilitating their adoption in clinical practice for personalized HF risk assessment and management in patients with diabetes.
Discussion
Globally, the burden, impact, and cost of diabetes show no sign of abating. Complications such as DR, nephropathy, neuropathy, and cardiovascular disease all contribute substantially to morbidity, mortality, and health care costs associated with diabetes.2,3 Beyond these well-established complications, emerging challenges confront health care providers and individuals living with diabetes. Hypoglycemia, particularly in the hospital setting, poses a critical risk, necessitating vigilant monitoring and management to prevent adverse outcomes.4,5 Mental health disorders, including depression and anxiety, frequently coexist with diabetes, compounding the complexity of disease management and compromising overall well-being. 6 Moreover, the lack of adherence to therapy, encompassing medication noncompliance, suboptimal dietary choices, and inadequate physical activity, undermines treatment efficacy and exacerbates disease progression. 7 Traditional diagnostic approaches for diabetes complications often rely on clinical assessments, laboratory tests, and imaging studies, which can be time-consuming, invasive, and subjective. Furthermore, these methods may not always detect complications at their early stages, when interventions are most effective in preventing progression and improving outcomes. 1
Artificial intelligence has already become a ubiquitous feature of modern living and more recently is impacting the structure and delivery of modern health care. The potential benefits from AI include improvements in pharmacoadherence, earlier prediction of hypoglycemia to prevent progression to severe events, more effective screening of individuals who may be at risk of microvascular complications, earlier detection of HF, and prevention and treatment of foot problems associated with diabetes. Outside of the complications associated with diabetes, there is also interest in applying predictive AI to better understand the heterogeneity of diabetes as we move toward precision medicine. 154
The main advantages from applying AI appear to be the potential identification of individuals at risk of diabetes complications, earlier detection at stage where interventions are more likely to be effective, a reduction in the personal burden associated with diabetes, and improved efficiencies of equitable care delivery. A yet underexplored area for AI is in enhancing professional and patient education related to the complications of diabetes.
Conclusion
The application of AI in diabetes care is in its infancy but the adoption of AI appears to be irresistible. Future research will be required to validate AI as a tool for the earlier detection of diabetes-related complications and the clinical value and cost-effectiveness of using AI in real-world settings. A major initiative will also be required in parallel to ensure engagement of clinicians and people with diabetes. In turn, this will require considerations on how to reduce cognitive and automation bias within AI systems, ensure data privacy, reduce the risk of hallucinations and misinformation, and clarify accountability and responsibility. 155 With increased application of AI, diabetes care in the near future is likely to be effective and personalized.
Footnotes
Abbreviations
ACCORD, Action to Control Cardiovascular Risk in Diabetes; Adv., Advanced; Anthr., anthropomorthic; AUC, area under the curve; AI, artificial intelligence; ARIC, Atherosclerosis Risk in Communities; Carb. Int., carbohydrate intake; CKD, chronic kidney disease; Clin. Notes, clinic notes; CGM, continuous glucose monitoring; CNNs, convolutional neural networks; DLA, deep learning algorithm; Demogr., demography; DFUs, diabetic foot ulcers; DKD, diabetic kidney disease; DN, diabetic neuropathy; DR, diabetic retinopathy; Dx, diagnoses; EHR, electronic health record; ewECG, energy waveform electrocardiogram; eGFR, estimated glomerular filtration rate; XAI, explainable artificial intelligence; FDA, Food and Drug Administration; HF, heart failure; HbA1c, hemoglobin A1c; Hx, History; Hypo, hypoglycemia; JITAIs, just-in-time adaptive interventions; labs, laboratories; LLM, large language models; LVD, left ventricular dysfunction; LightGBM, Light Gradient Boosting Machine; LIME, Local Interpretable Model-Algorithm; ML, machine learning; MRI, magnetic resonance imaging; Meds, Medications; mtmDR, more-than-mild diabetic retinopathy; NT-proBNP, N-terminal pro-B-type natriuretic peptide; NLP, natural language processing; NDKD, nondiabetic kidney disease; OCT, optical coherence tomography; PDN, peripheral diabetic neuropathy; Phys. Act., physical activity; Proc., procedures; RSF, random survival forest; RECODe, Risk Equations for Complications Of type 2 Diabetes; SMBG, self-monitored blood glucose; SHAP, Shapley Additive exPlanations; SVS, Society for Vascular Surgery; SVM, support vector machines; T1D, type 1 diabetes; T2D, type 2 diabetes; US, ultrasound; Utiliz./Ins., utilization/insurance; VSC, visually significant cataracts; WIfI, Wound, Ischemia, and foot Infection.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ATA is a consultant for Liom. CNH is a consultant for Liom. DK has received research support from Abbott Diabetes Care and consultancy fees from Perfood. SLC has received research support from i-SENS, Inc. NM has nothing to disclose. MW has nothing to disclose. BN is serving as a consultant for BioSensics LLC and Mölnlycke Health on projects unrelated to the scope of this paper. S-JM is a consultant for Abbott, Curestream, Daweoong, EOFlow, G2e, iSense, Medtronic, Novo Nordisk, and Sanofi. AP has received grant funding (to the institution) from Applied Therapeutics, Gilead Sciences, Ultromics, Myovista, and Roche; has served as a consultant for and/or received honoraria outside of the present study as an advisor/consultant for Tricog Health Inc, Lilly USA, Rivus; Cytokinetics, Roche Diagnostics, Sarfez Therapeutics, Edwards Lifesciences, Merck, Bayer, Novo Nordisk, Alleviant, Axon Therapies, and has received nonfinancial support from Pfizer and Merck. AP is also a consultant for Palomarin Inc with stocks compensation and has received research support from the National Institute on Aging GEMSSTAR Grant (1R03AG067960-01), the National Institute on Minority Health and Disparities (R01MD017529), and the National Institute of Heart, Lung, and Blood Institute (R21HL169708). DCK is a consultant for Afon, Embecta, Glucotrack, Lifecare, Novo, Samsung, Synchneuro and Thirdwayv.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
