Abstract
The adoption of diabetes technology for the management of type 1 and insulin-treated type 2 diabetes has greatly increased. The annual volume of discarded continuous glucose monitoring (CGM) devices, considering only Dexcom and Freestyle Libre brands, totals more than 153 million units and Omnipod® contributes an additional estimated 43.8 million units.
Although these technologies are clinically effective, their environmental impact is unknown. Batteries are a pivotal, yet often overlooked, component in diabetes technologies and can exert a detrimental impact on the environment.
In this commentary article, we explore the environmental impact of batteries used in diabetes devices. Furthermore, we highlight various strategies, including recycling of used batteries and alternative design approaches, that may reduce the environmental burden, as they become the ubiquitous standard of care for people with diabetes.
Keywords
Introduction: The Need and the Problems of Diabetes Devices
Climate change is likely to be the next biggest threat to human health. As a result of burning fossil fuels, humans have contributed significantly to the release of greenhouse gases, resulting in a rise in average global temperatures of around 1°C, with associated long-term changes. 1 Climate change has an impact on people with diabetes; however, it is equally critical that any impact that diabetes has on climate change is minimized. 2 Overall, there is increased adoption of diabetes devices by people with type 1 diabetes (T1D) and, increasingly, insulin-treated type 2 diabetes (T2D).
Continuous glucose monitoring (CGM) sensors are disposed after 7 to 14 days of wear, or earlier in the case of failure. Approximately 91 million Freestyle Libre (Abbott) sensors are used annually worldwide (approximately 3.5 million people across 44 countries using 26 sensors per year). 3 They are disposed as clinical waste as they contain a contaminated sharp and are, therefore, incinerated.
In terms of insulin pump therapy, there are two main types: those with tubing and patch or tubeless pumps, such as Omnipod® by Insulet. Each Omnipod® consists of a wearable “pod” with three batteries within it, and each pod is disposed every three days. With approximately 360,000 users worldwide, 4 this equates to 43·8 million pods disposed per year and more than 131 million batteries disposed per year. These numbers are likely to further increase. Although diabetes technologies are clinically important and cost-effective, their environmental impact is unknown.
There are several important considerations for sustainability. These include the environmental impact of (1) sensor production, (2) resource consumption, and (3) disposal of devices. 5 It is imperative to address these issues and strive for sustainable approaches to minimize the carbon footprint of optimal glucose management.
Manufacturers, acknowledging the concern around sustainability, have begun to innovate by downsizing devices. Slimmer and compact sensors not only improve user satisfaction but also effectively reduce the weight of used plastic. The Abbott FreeStyle Libre 3 has achieved a size reduction of over 70%, translating to a 41% reduction in plastic compared with its antecedents. 6 Similarly, the Dexcom G7 sensor is 60% smaller than its predecessor, 7 and the Simplera™ CGM is 50% smaller than its previous generation. 8 Compared with the sensor, the spring-loaded needle inserter contains more plastic, and future product designs should focus on re-using and further minimizing the applicator to reduce plastic usage, with preferential use of biodegradable plastics. Achieving this without increasing complexity, cost, and usability is a significant challenge.
However, beyond the product design, one important, but typically ignored component, is the system battery. Here, we focus on the environmental impact of batteries within diabetes technologies, the potential for recovery of minerals from disposed devices, and alternative design strategies that should be pursued to address the sustainability of devices as they become the ubiquitous standard of diabetes care.
Batteries: The Key Component for Powering Diabetes Devices
Within each single-use CGM device is a power source to drive the sensor potentiostat, instrumentation, and wireless communication. In the case of the Abbott FreeStyle Libre and Dexcom sensors, the most used devices, this is a battery, in the form of button cell form as shown in Figure 1. Similarly, the “pod” component of the Omnipod® system is disposed every 3 days and contains three button cell batteries to drive the infusion system, electronics, and wireless communication.
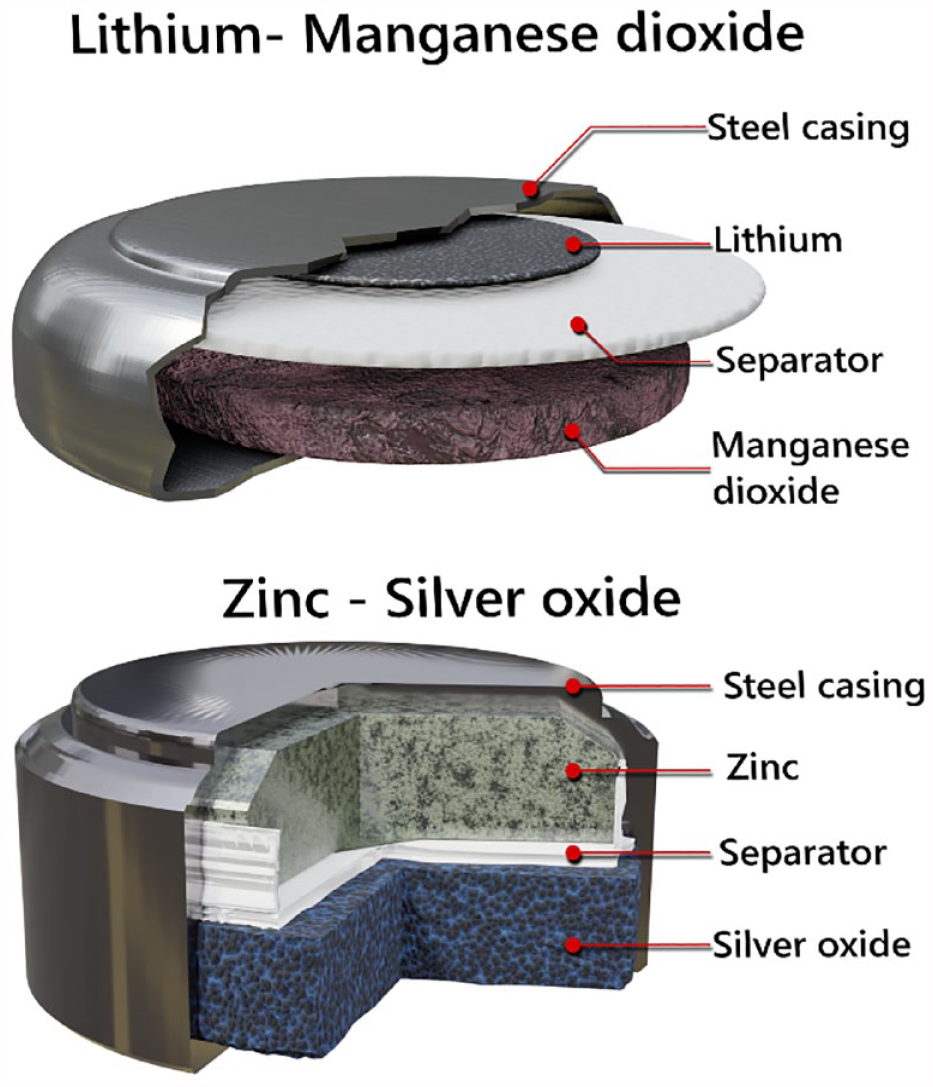
Schematic diagram for two different types of coin cells used in glucose sensors and the insulin pump, Omnipod®.
There are two popular battery chemistries (Table 1) used by manufacturers in diabetes technology: (1) zinc-silver oxide system (e.g., used within the Freestyle Libre sensor and Omnipod® pump) and (2) lithium metal-manganese dioxide system (e.g., used within Dexcom sensors).
Properties of current diabetes devices including glucose monitoring sensors and insulin pump and battery type with the estimated valuable materials used in corresponding batteries.

Calculation is based on cell disassembly in the lab with an estimated mass.
With three batteries in each pod, equating to 131.4 million batteries disposed.
The battery systems exhibit overall similarity, with some key distinctions. The negative electrode (anode) is either metallic zinc or metallic lithium (Figure 1), while the positive electrode (cathode) is silver oxide or manganese oxide, respectively.16,17 A polymer or glass fiber separator is positioned between these electrodes to prevent direct electrical contact. Additionally, the separator is saturated with an electrolyte, a salt in a solvent, which helps in ion transfer for battery operation. In the case of the zinc-silver oxide system, hydroxide (OH) ions are utilized as the transport medium, employing sodium hydroxide (NaOH) or potassium hydroxide (KOH) aqueous solution as the electrolyte. 18 In the lithium system, lithium ions serve as the transport medium, requiring lithium salts dissolved in organic solvents.
Battery chemistries for small CGM sensors and insulin pumps are chosen for multiple reasons. First, due to size constraints, high energy density batteries are preferred in terms of both weight and volume. Zinc-silver oxide systems offer approximately 350 Wh/kg and 750 Wh/L, while lithium metal-manganese dioxide systems provide around 300 Wh/kg and 600 Wh/L.18,19 Second, in order to maintain stable and reliable performance over time, a flat discharge plateau is a prerequisite for a stable power delivery. 16 Zinc-silver oxide batteries exhibit a near constant flat voltage output of approximately 1.5 V throughout its operational lifespan, 18 whereas lithium metal-manganese dioxide batteries can sustain a stable voltage output of ~3 V for most of its operational life, 19 as illustrated in Figure 2.
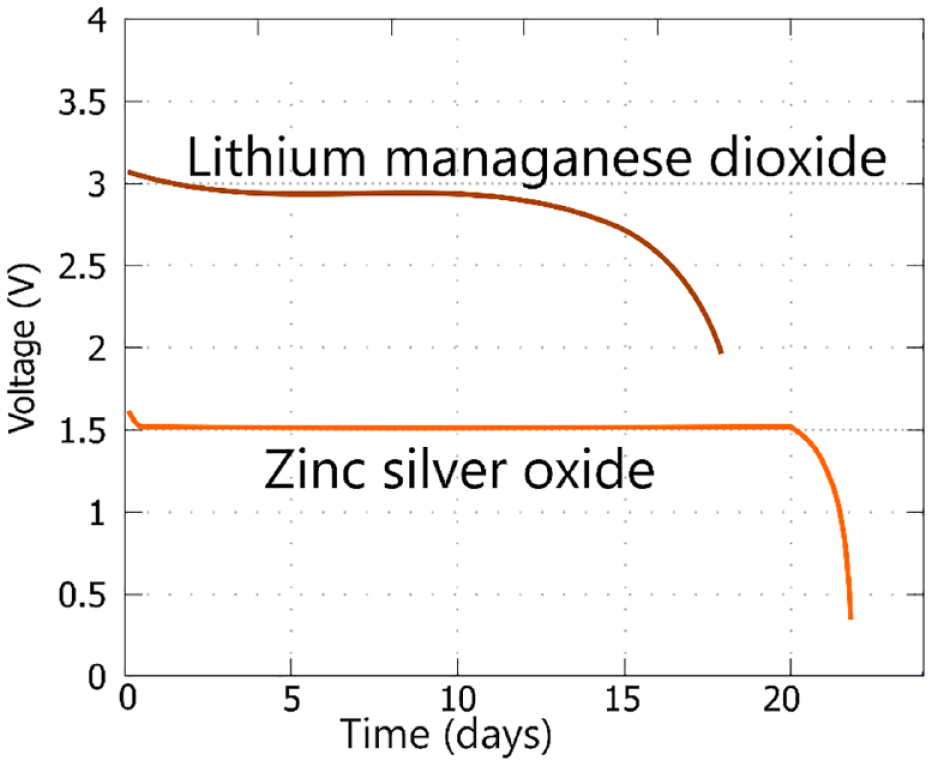
Schematic for typical discharge profiles for Lithium manganese dioxide battery and zinc-silver oxide batteries at room temperature and a load of 30 kΩ.
Environmental Challenges With the Currently Used Batteries
The environmental impact of battery usage encompasses the entire supply chain, from the acquisition of raw materials to battery waste disposal. Among the many metals utilized, lithium and silver are crucial components. Our analysis based on the estimated number of sensors and pumps used worldwide per year (Table 1) reveals, on a global scale, that more than 600 kg of lithium and 14,000 kg of silver oxide are consumed and disposed of. The financial implications of this metal consumption cost approximately roughly $7080 for lithium and $49,441,000 for silver oxide. To gain a broader perspective on the global supply-demand dynamics of these key metals, Figure 3 demonstrates the supply-demand data. The market for silver suggests supply and demand competition ran at parity between 2016 and 2020 but, notably, demand exceeded supply by approximately 5% to 7% in the years 2021 to 2022. Based on the IEA 2023 report, a similar trend emerges for lithium metal, where demand outstrips supply by approximately 1.5% despite rapid increases in supply in 2022. 14 Furthermore, multiple forecasts predict a shortage of lithium metal within the next 5 to 10 years, driven by the huge demand stemming from electric vehicles and portable electronics.20 -22
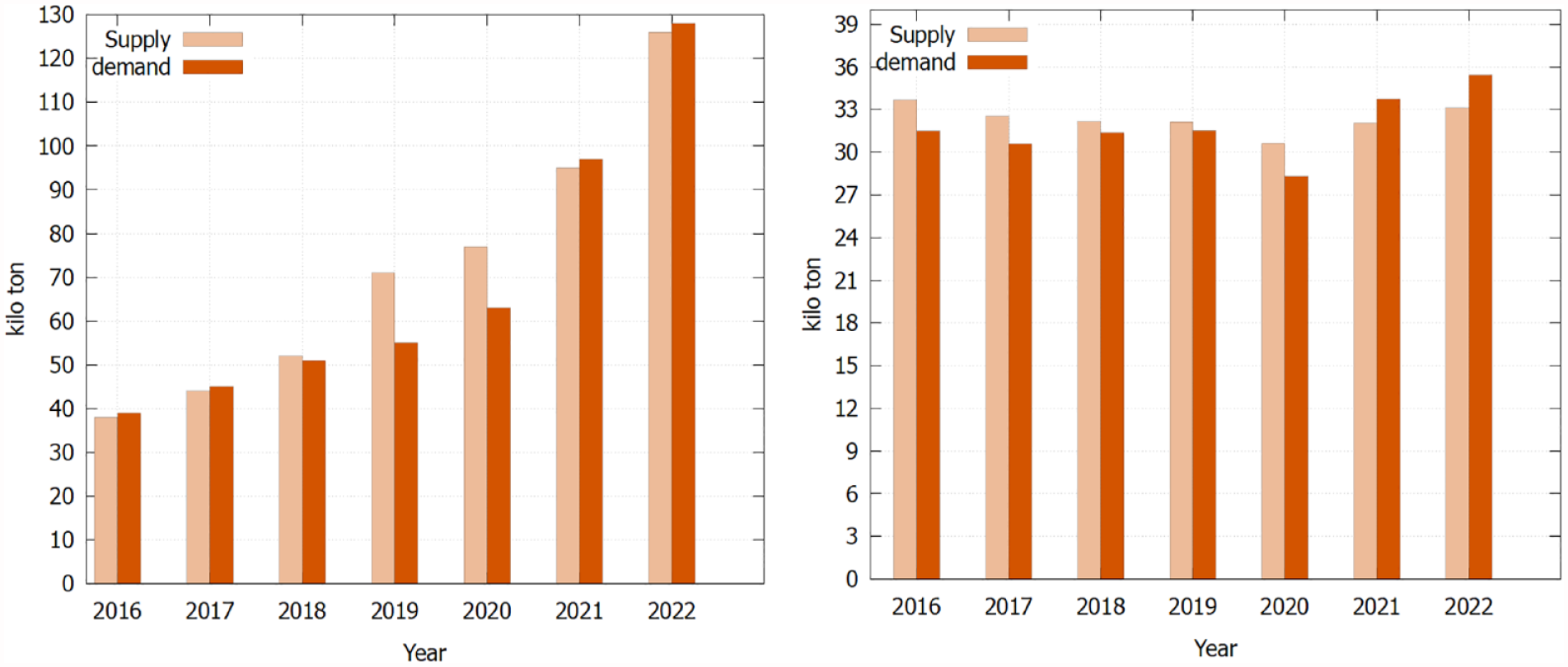
The biggest supply sources for lithium and silver metals primarily originate from mines, but their global distribution is uneven. Approximately 50% of the world’s lithium reserves are concentrated in the South American “Lithium Triangle,” spanning Chile (Salar de Atacama), Bolivia (Salar de Uyuni) and Argentina (Salar de Olaroz). 22 In 2021, China processed 58% of the world’s lithium, 23 while Central and South America account for approximately 50% of global silver production. 13 This regional dependency can risk supply chain disruption. For example, in Peru, social unrest caused a 7.3% decline in silver production in 2022. 13
The current mining processes for these metals have several environmental drawbacks. Extracting 1 ton of lithium requires 250 tons of rock mineral Spodumene ore, or 750 tons of brine, 24 and approximately 1900 tons of water. 25 In Chile’s Salar de Atacama, 65% of water reserves are depleted for mining activities, further straining already low groundwater levels in this high-altitude arid region. 22 Silver mining also has significant environmental repercussions, as metals like silver or gold are extracted using corrosive chemicals such as cyanides and ferricyanides. 26 One-fifth (20%) of the world’s cyanide is used in gold and silver mining, risking contamination of water and soil from accidents and leaks.26,27 In 2000, a cyanide spill from a dam breach at Romania’s Baia Mare gold-silver 28 contaminated drinking water and rivers in Romania, Hungary, and Yugoslavia, killing over 150 tons of fish and drastically reducing plankton29,30 It was deemed the worst environmental disaster since Chernobyl 1986 incident.
Disposal of batteries contained within glucose monitoring sensors and insulin pumps have further adverse environmental consequences. The incineration process can release harmful gases, particularly from zinc-silver oxide batteries, posing health risks when zinc-containing fumes are inhaled.31 -33 Additionally, the solid residue in landfill contains heavy metals, like silver, which pose a high risk of contaminating the groundwater.34,35 The toxicity of battery material is a direct threat to organisms and animals, as well as to human health. 35
Disposing lithium-manganese dioxide batteries can pose even greater environmental risks than rechargeable lithium-ion batteries. Unlike Lithium-ion batteries with graphite anodes, lithium-manganese dioxide batteries use lithium metal, which can react violently with air or water. For example, dissolving 1 kg of lithium metal in water releases approximately 32 megajoule (MJ) of energy, capable of vaporizing 12.4 kg of water. 36 Furthermore, this dissolution releases flammable hydrogen gas, and the organic solvents used to make the Lithium battery electrolyte are flammable themselves, making the disposal of untreated batteries a significant fire hazard. Unlike silver metal, incineration or pyrometallurgical techniques to treat batteries before landfilling are ineffective for lithium recovery due to its high oxygen affinity. 37 As a result, lithium oxide ends up in slag with impurities, and lithium recovery from slag with impurities is not yet economically viable. 38
With the rapid deterioration of the supply-demand dynamics, environmentally harmful mining processes for metal production, and the risks associated with battery disposal, there is an urgent need for improved measures to mitigate the environmental footprint from the use of these batteries within diabetes devices.
Potential Strategies to Reduce Environmental Impact of These Batteries
To reduce the environmental impact of diabetes devices, some noteworthy initiatives have emerged. Abbott has launched a “Sensor-Kit-Take-Back-pilot” program in the United States to encourage users to return used sensors. 39 Collected sensors, batteries included, will be sent to specialized waste-to-energy facilities. Through controlled combustion, the batteries as the sensor waste will be transformed into renewable energy. Similarly, Insulet has launched a “Pod Take-Back Program”; however, this pilot is only available to residents in Massachusetts, United States. 40 This approach not only reduces the volume of solid waste destined for landfills but also has the potential for energy recovery during the waste-burning process. This generates a renewable energy source and reduces carbon emissions by offsetting the demand for fossil energy and reducing methane production in landfills.
Instead of single-use primary batteries, rechargeable batteries in these devices can offer some benefits to minimize their disposal. For example, some commercially available diabetes devices include the Medtronic Guardian transmitters, which are rechargeable (require charging once weekly) and last up to one full year, minimizing the amount of battery waste. In terms of insulin pumps, the conventional tubed insulin pump Tandem t:slim X2 insulin pump has a rechargeable lithium battery (advised to charge 10-15 minutes daily), while the MiniMed™ 780G insulin uses one primary AA (1.5 V) lithium battery.41,42 The MiniMed™ 780G pump can also accept a rechargeable AA nickel-metal hydride (NiMH) rechargeable battery (HR6). 42 Thus, choosing a conventional tubed pump can be a more sustainable choice from a battery waste perspective.
For people on multiple daily injections, reusable smart insulin pen devices represent the new generation of technologies. The InPen™ (Medtronic) is a reusable smart insulin pen that uses Bluetooth technology to send dose information to a mobile app, consisting of a lithium battery lasting approximately 1 year. The NovoPen® 6 and NovoPen Echo® Plus (Novo Nordisk) lithium battery lasts approximately 4 to 5 years. Each of these is currently non-replaceable but provides a design opportunity to extend the lifespan of these pens.
While recycling and rechargeable initiatives signal a positive step toward sustainability, the challenge posed by the increasing need for battery components necessitates further review. Specifically, addressing issues related to battery materials (i.e., silver and lithium metals), supply-demand imbalance, environmental repercussions from metal mining, and inefficient disposal, is imperative. Recycling of these batteries to reclaim valuable metals like lithium and silver presents a promising strategy to tackle this multifaceted problem. However, the current state of recycling for lithium and silver metals falls short of making it sustainable. In 2022, recycled silver met only ~18% of global demand. 13
Lithium recycling is even more challenging, with some recent reports suggesting less than 1% of global lithium metal markets undergo recycling.43,44 High-temperature methods, such as pyrometallurgy, are ineffective. An alternative approach, known as hydrometallurgy, has demonstrated feasibility on a small laboratory scale. Hydrometallurgical methods are more environmentally friendly than traditional pyrometallurgy. In this method, an acid or aqueous medium is used to dissolve the desired ions, creating an aqueous solution. Metal salts can be precipitated from this solution by adding suitable reagents. 45 To mitigate lithium’s reactivity, various methods can be employed, including cooling batteries, using inert gas atmospheres, or converting lithium metal into less reactive compounds like lithium oxide.46 -51 Subsequently, lithium is dissolved into the solution and can be precipitated as lithium salts, such as hydroxide or carbonate salts. 52
Compared with lithium, silver is less reactive, allowing traditional high-temperature pyrometallurgical methods to recover it with up to 95% purity11,22 However, the emission of toxic gases and zinc vapors necessitates more environmentally friendly approaches. Hydrometallurgical methods offer a sustainable alternative, enabling zinc and silver recycling with efficiencies as high as 99% 31 using non-toxic leaching agents. 33 This approach significantly improves the recycling economy, generating a profit of €296 per kilogram for used zinc-silver oxide batteries. Innovative technologies, such as electrowinning, a process used in metallurgy to extract metals from their ores using an electrolytic procedure, could be utilized to further increase recovery efficiency. 31
Beyond recycling batteries, another notable battery strategy is integrated battery design. Glucose sensors and insulin pumps often rely on commercial button cells, which can constrain the size and form factor of the overall device. The development of small-area high-energy-density batteries will facilitate production and reduce manufacturing costs, potentially achieving a positive environmental impact. Printed batteries are an emerging solution. Briefly, the components in print batteries are mostly thin layers which are deposited on flexible substrates using printing processes. To date, novel form factors, such as tattoo paper and fabric wearable cells, have been embedded, and an area capacity of up to 54 mAh/cm2 has been achieved at the lab scale. 53 Despite these promising results, further work is needed to incorporate printed batteries with circuit board, and microprocessor, evaluating the scalability and performance in a more realistic environment.
Other novel alternate battery designs include self-powered sensors, such as supercapacitors, which could be the next generation of wearable sensors that form the future of CGM devices. 53 However, there are several considerations to be addressed. First, ensuring the safety of alternative battery chemistries is crucial, particularly around issues such as toxicity. Second, manufacturing processes need to demonstrate scalability and cost-effectiveness to be adopted on a broader scale. Finally, compliance on safety, accuracy, and performance standards is essential to gain regulatory approval.
Conclusion
While lithium metal-manganese dioxide and zinc-silver oxide batteries have been widely used in diabetes technology devices, their ability to reliably power devices is not without limitations. To sustainably support people living with diabetes and optimize self-management, the challenges around finite resources and environmental concerns associated with their production and disposal must be addressed. Researchers and manufacturers must explore alternative strategies, such as recycling and battery designs, that work to reduce these limitations. Furthermore, authorities have the potential to hold manufacturers accountable for the entire recycling process of batteries and plastic waste associated with devices. For example, through implementing “extended producer responsibility” regulations, manufacturers can be incentivized to design more sustainable products and manage end-of-life disposal, significantly reducing the environmental impact of diabetes devices.
Footnotes
Acknowledgements
The views expressed are those of the author(s) and not necessarily those of the NHS, the NIHR, or the Department of Health and Social Care. This research/project was supported by the National Research Foundation, Singapore, and the National Environment Agency, Singapore, under its Closing the Waste Loop Funding Initiative (SCARCE Phase 2 Award No. CTRL-2023-1D-01).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MR has received honoraria for advisory board participation from Dexcom and Medtronic and has received support for investigator-initiated studies by Dexcom and Medtronic. PA has received support for investigator-initiated studies by Dexcom. NO has received research support from Dexcom, Medtronic, and Roche Diabetes, has participated in advisory groups for Dexcom, Medtronic, and Roche Diabetes, and has received fees for speaking from Sanofi, Dexcom, Tandem, and Roche Diabetes.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: PA was funded by NIHR Grant No. 132960. Infrastructure support was provided by the NIHR Imperial Biomedical Research Center and the NIHR Imperial Clinical Research Facility.
