Abstract

New continuous glucose monitors (CGMs) are released periodically and supported by method comparison studies. Four recently released CGMs trials (three of which were clinical trials) reported on the safety and accuracy of the devices.1-4
This letter proposes that three criteria be part of method comparison trials:
An error grid plot of the differences between the CGM and reference.
A tally of the occurrences when the CGM failed to provide a result.
A statement of whether any data were deleted.
The four evaluations are analyzed for these criteria in Table 1.
Properties of Four CGM Trials.
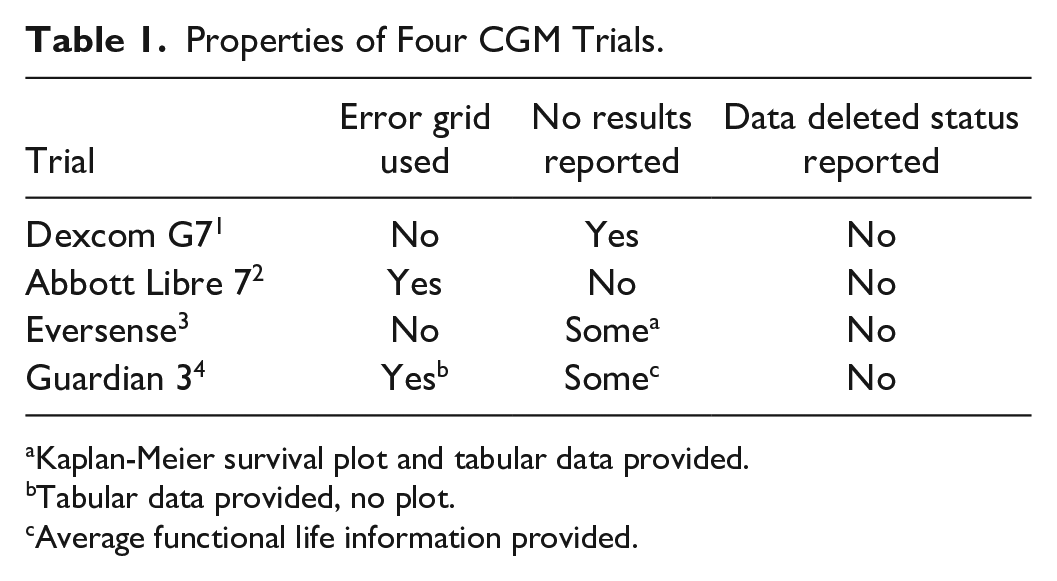
Kaplan-Meier survival plot and tabular data provided.
Tabular data provided, no plot.
Average functional life information provided.
An error grid provides the most important information about clinical accuracy. A Bland-Altman plot should also be provided. A previous study showed that no result is the most common adverse event for CGMs and often requires medical intervention. 5 The most common glucose metric is MARD (mean average relative deviation). As a parametric parameter, its value is highly influenced by outlier data. And, the 2013 glucose meter standard ISO 15197 recommends deleting data when an error was suspected.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
