Abstract
Background:
Skin reactions due to technological devices pose a significant concern in the management of type 1 diabetes (T1D). This multicentric, comparative cross-sectional study aimed to assess the psychological impact of device-related skin issues on youths with T1D and their parents.
Methods:
Participants with skin reactions were matched in a 1:1 ratio with a control group. Diabetes-related emotional distress was evaluated using the Problem Areas in Diabetes–Teen version (PAID-T) for participants aged 11 to 19 years and the Problem Areas in Diabetes–Parent Revised version (PAID-PR) completed by parents. In addition, glucose control was assessed through glycated hemoglobin (HbA1c) values and continuous glucose monitoring (CGM) metrics.
Results:
A total of 102 children and adolescents were consecutively recruited. Adolescents with skin issues had higher PAID-T scores compared to those without (79.6 ± 21.1 vs 62 ± 16.8; P = .004). Parents of youths with skin reactions also reported higher PAID-PR scores than the control group (34.0 ± 11.0 vs 26.9 ± 12.3; P = .015). No differences were observed in HbA1c levels (6.9 ± 0.8% vs 6.8 ± 0.8%, P = .555) or CGM glucose metrics between the two groups. Remarkably, 25.5% were forced to discontinue insulin pumps and/or glucose sensors (21.5% and 5.9%, respectively).
Conclusions:
Our study highlighted the increased emotional burden experienced by youths with T1D and their parents due to device-related skin reactions, emphasizing the need for further research and interventions in this crucial aspect of diabetes management.
Introduction
Recent years have witnessed remarkable progress in the development of insulin pumps and continuous glucose monitoring (CGM) systems, introducing a new era in diabetes management.1,2 The integration of real-time glucose data with continuous subcutaneous insulin infusion (CSII) therapy through automated insulin delivery systems has emerged as a game-changing approach, leading to substantial improvements in metabolic and psychological outcomes for individuals with type 1 diabetes (T1D). 3 However, despite the continuous evolution of device features and functionalities, some barriers hinder their widespread adoption.4,5 One major concern related to the use of diabetes devices is the occurrence of skin reactions. Prolonged skin exposure to chemical agents present in the adhesives of sensors and insulin infusion sets (IISs) and catheters is the main cause of dermatological complications. 6 Skin issues associated with technological devices are frequent and clinically heterogeneous in both youths and adults with T1D.7-9 The most common skin lesions include eczema, lipoatrophy, wounds, lipohypertrophy, hyperpigmentation, and scars. 10 Of particular concern is contact dermatitis, a persistent inflammatory skin condition characterized by itching, erythema, and eczematous lesions occasionally complicated by impetiginization. 11 Despite increasing interest in the literature and heightened awareness among diabetes-care providers, skin reactions persist as unresolved challenges in diabetes management. 12 Studies investigating the effects of device-related skin reactions on glycemic outcomes and their consequent impact on the psychological well-being of affected people are currently lacking.
The objective of this study was to evaluate the emotional burden in youths with T1D and skin reactions and their parents. In addition, the study aimed to explore the clinical implications associated with these adverse events.
Methods
Participants and Study Procedures
This multicenter, comparative cross-sectional study was conducted across eight Pediatric Diabetes Centers affiliated with the Italian Society for Pediatric Endocrinology and Diabetes. The study followed the ethical regulations outlined in the Declaration of Helsinki and received approval from the Clinical Research Ethics Committee of Trento (n. A831). The recruitment of study participants took place from September 2022 to September 2023 during quarterly follow-up outpatient visits. Prior to the study procedures, written informed consent was obtained from each participant and at least one parent. Inclusion criteria were as follows: diagnosis of T1D based on the latest American Diabetes Association criteria, 13 age <20 years at the time of recruitment, documented history of skin reactions (ie, eczema, vesicles, or bullous skin lesions) due to diabetes devices in the year preceding the recruitment visit, and proficiency in the Italian language. Exclusion criteria included the presence of dermatological disorders unrelated to T1D, severe comorbidities or use of medications known to interfere with glucose control or psychological well-being. For the control group, youths with T1D without any history or evidence of skin reactions were consecutively recruited in a 1:1 ratio. Matching criteria included age, gender, body mass index (BMI), diabetes duration, and type of treatment regimen.
Data Collection
At the time of recruitment, demographic, anamnestic,and clinical data were gathered, including age, sex, time of diabetes onset, concomitant disorders, type of treatment regimen (multiple daily injections or CSII), type of glucose monitoring (self-blood glucose monitoring or CGM), details of technological devices used, and anthropometric measures (height, weight, BMI). For patients with evidence of skin reactions, information regarding implicated device brands, timing of dermatological complications, and any discontinuation of insulin pump and/or glucose sensor use was also collected.
Assessment of Psychological Burden
The assessment of diabetes-related emotional distress involved the use of the Problem Areas in Diabetes–Teen version (PAID-T) for participants aged 11 to 19 years and the Problem Areas in Diabetes–Parent Revised version (PAID-PR) completed by parents of each participant. The PAID-T and PAID-PR scales are validated tools to measure perceived disease-related burden in adolescents living with diabetes and in parents caring for a child with diabetes, respectively. The PAID-T comprised 26 items, 14 whereas the PAID-PR consisted of 18 items categorized into two factors (“concrete burden” and “unpredictable burden”). 15
The translation and validation process of both questionnaires consisted of the following phases: forward translation, backward translation, and patient testing.
Assessment of Glucose Control
Glucose control of study participants was evaluated by analyzing the following parameters: glycated hemoglobin (HbA1c) measurement, mean HbA1c value from the year preceding recruitment, CGM metrics from the 14-day period prior to the recruitment visit, including mean and standard deviation (SD) of sensor glucose levels, percentage of time below 70 mg/dL (3.0 mmol/L), percentage of time between 70 and 180 mg/dL (3.9 and 10 mmol/L) (%TIR), percentage of time above 180 mg/dL (10 mmol/L) (%TAR), glucose management indicator (GMI), percentage of coefficient of variation (%CV) expressed in percentage, when available. These metrics were obtained from dedicated web-cloud platforms. In addition, events of severe hypoglycemia and diabetic ketoacidosis were recorded.
Statistical Analysis
Statistical analysis was performed using SPSS 22.0 (IBM, Armonk, New York) for Windows package. Descriptive statistics were used to summarize demographic and clinical characteristics. Numerical data were expressed as mean ± SD, whereas categorical variables were presented as absolute frequencies and percentages. The Mann-Whitney test was performed to compare continuous variables between the two groups (group with skin reactions and controls). The chi-square test or Fisher test, when appropriate, was used for categorical variables. The significance threshold was set to .05.
Results
A total of 102 participants were consecutively recruited. The study group consisted of 51 children and adolescents with skin reactions, matched in a 1:1 ratio with the control group without skin reactions. Detailed anamnestic and clinical data for both the study and control groups are provided in Table 1. Among children and adolescents with skin reactions, 62.8% were males, with a mean age of 12.4 ± 4.5 years. The mean age at T1D onset was 5.7 ± 3.2 years. Most participants (64.7%) met the criteria to complete the PAID-T questionnaire. All the participants in our study population used a CGM system, and 80.4% were concomitantly on CSII therapy. Skin reactions were primarily caused by glucose sensors in 72.5% of individuals, by ISS in 11.8%, and by both components in 15.6%. Among CGM users, reactions from two different brands were recorded in 5.9% of cases.
Comparative Analysis of Clinical Characteristics, Glycemic Control Parameters, and Acute Complications Between the Study and Control Groups.
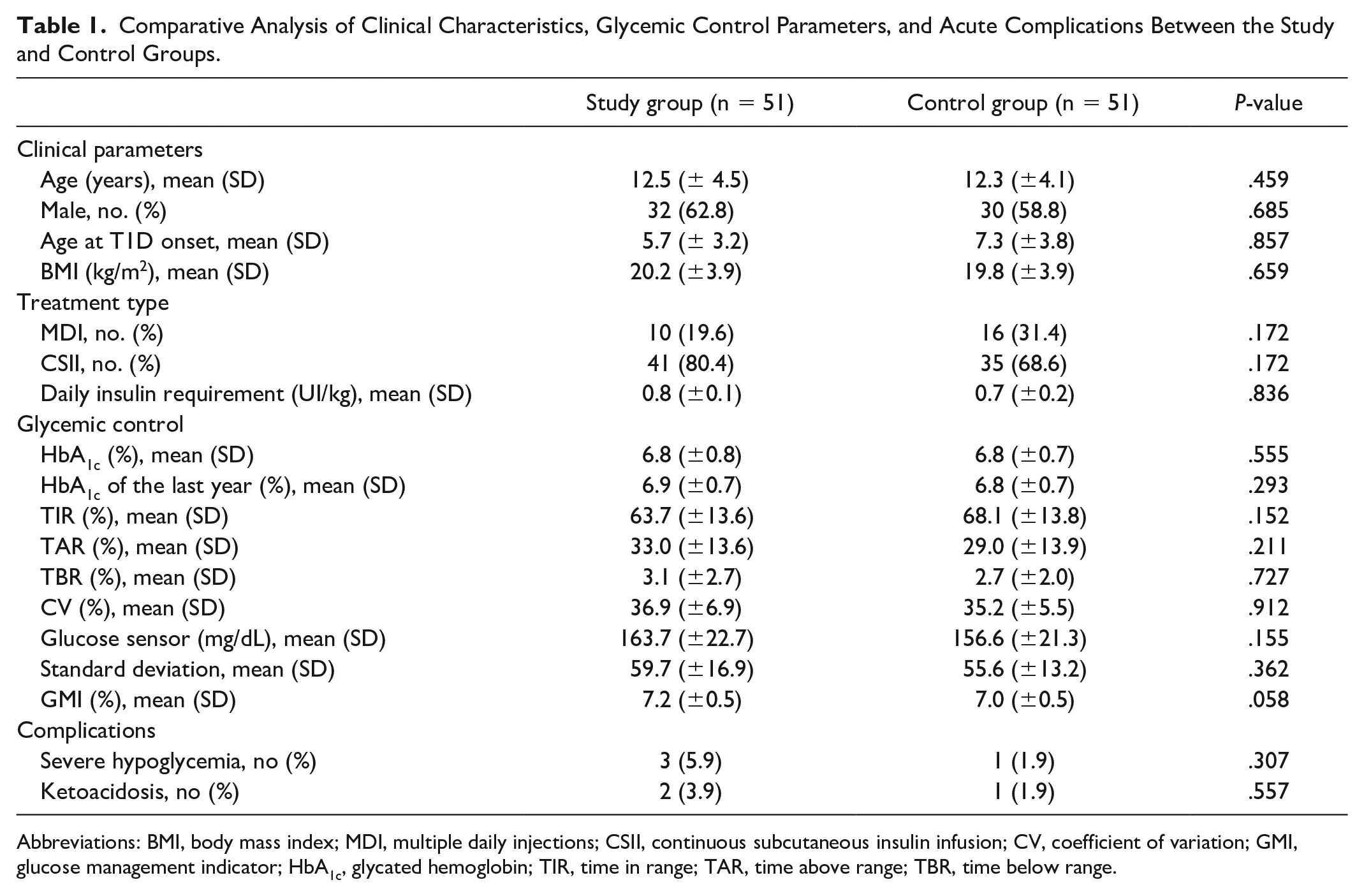
Abbreviations: BMI, body mass index; MDI, multiple daily injections; CSII, continuous subcutaneous insulin infusion; CV, coefficient of variation; GMI, glucose management indicator; HbA1c, glycated hemoglobin; TIR, time in range; TAR, time above range; TBR, time below range.
Problem Areas in Diabetes (PAID) scores in both teen and parent versions were higher in participants experiencing skin reactions than the control group (Figure 1). Adolescents with skin issues had significantly higher PAID-T scores compared to those without (79.6 ± 21.1 vs 62 ± 16.8; P = .004). Parents of youths with skin reactions reported significantly higher overall PAID-PR scores than the control group (34.0 ± 11.0 vs 26.9 ± 12.3; P = .015). Analyzing PAID-PR factors, the “unpredictable burden” score was higher in the study group (22.5 ± 6.9 vs 18.1 ± 8.8; P = .040), whereas no significant differences were found for the “concrete burden” score (11.6 ± 5.8 vs 10.8 ± 9.1; P = .109).
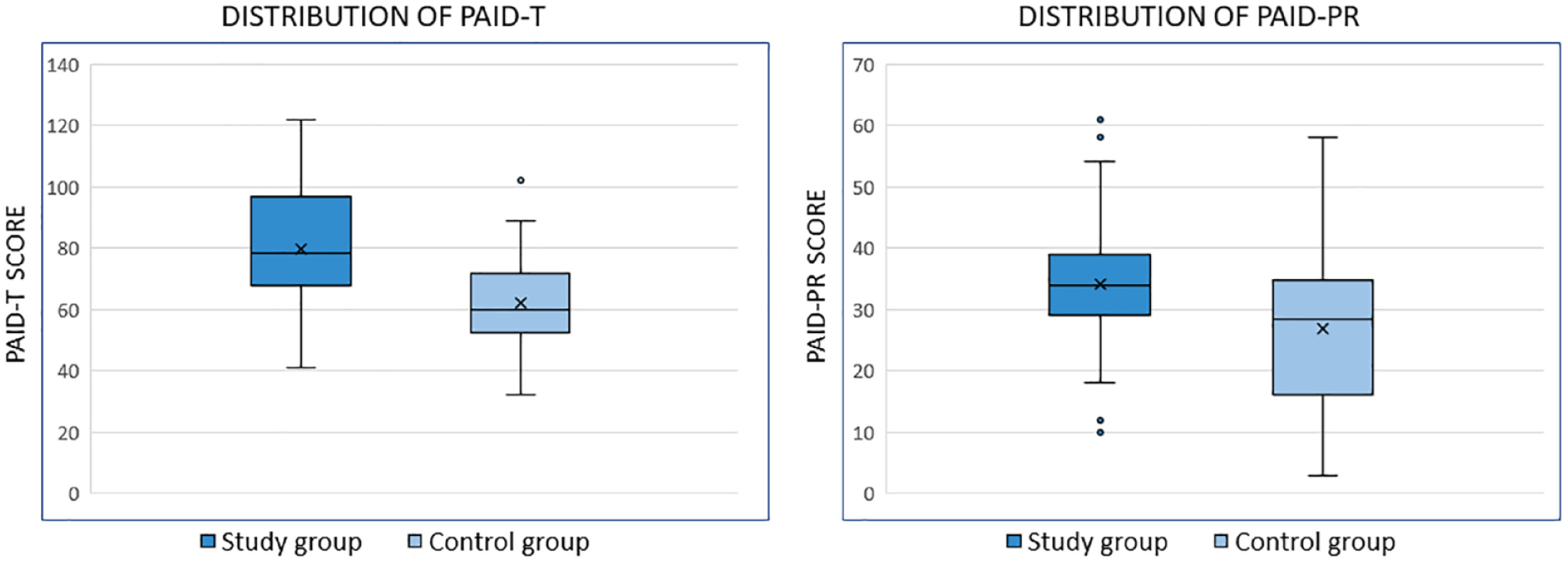
Box plots illustrating the distribution of PAID-T and PAID-PR scores in the study population and the control group. Notably, within the study population, PAID scores in both versions are significantly higher, indicating an increased emotional burden.
In terms of glucose control, comparative analysis revealed no differences for HbA1c (6.9 ± 0.8% vs 6.8 ± 0.8%, P = .555), mean HbA1c value from the year preceding recruitment (7.0 ± 0.7% vs 6.9 ± 0.7%, P = .293), and any of the CGM glucose metrics (P > .05) (Table 1). In addition, no significant differences were observed in the two groups in episodes of severe hypoglycemia and diabetic ketoacidosis in the last year (P = .307 and P = .557, respectively).
Table 2 illustrates the distribution of device brands associated with skin reactions. Notably, 41% of the study group required a brand change to ensure the continuation of CSII therapy and/or CGM, whereas 25.5% were compelled to discontinue the use of insulin pumps and/or glucose sensors (21.5% and 5.9%, respectively).
Presence of Skin Reactions Related to Different Devices Expressed as Absolute Frequencies and Percentages Out of the Total of Device Users Followed in the Pediatric Diabetes Centers Involved in the Study.
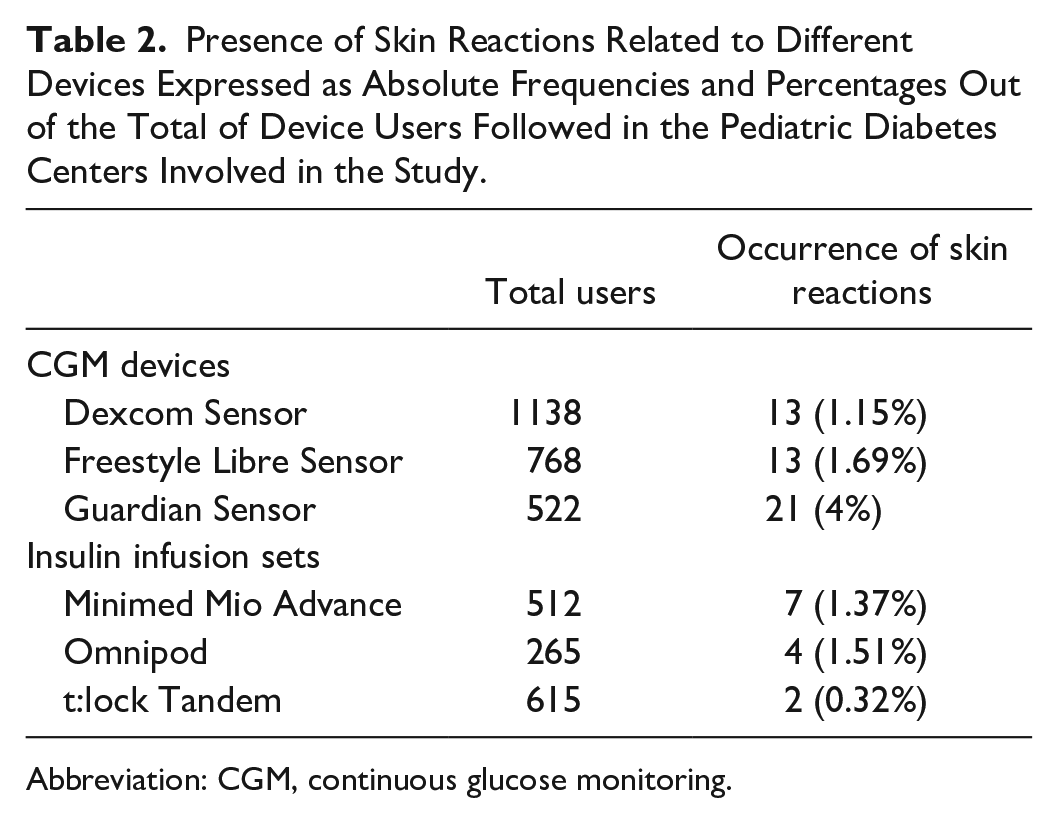
Abbreviation: CGM, continuous glucose monitoring.
Discussion
Our study demonstrated the increased disease-related burden experienced by youths with T1D and their parents caused by the occurrence of skin reactions. To date, the literature on the relationship between skin manifestations and quality of life in individuals with T1D is limited and contradictory. A previous mix-aged study demonstrated a significant impact of skin manifestations on the psychological well-being of individuals with T1D. 16 In contrast, a pediatric study by Burgmann et al 17 reported low or absent skin-related quality of life impairment in the majority of their study cohort. However, it is crucial to note that both studies did not differentiate device-related complications from other skin diseases, and disparate tools were used to assess the psychological impact. A cross-sectional study focusing on the adult population found that subjects with CSII-related skin issues experienced increased diabetes burden assessed by the PAID scale. These findings are consistent with our data, which revealed significantly higher PAID-T and PAID-PR scores in young people with these dermatological complications and their parents, respectively.
Results obtained from PAID-T for the 11 to 18 years group are noteworthy, considering the undeniable challenges in managing T1D during adolescence, marked by impaired diabetes self-management and subsequent suboptimal glucose control.18,19 Approximately one third of adolescents experience elevated diabetes distress. 20 This is manifested as a lack of comprehension from, or unconstructive interactions with, family and friends along with a sense of being overwhelmed by the demands of managing the disease. 21 In addition, this tricky transitional phase could be compounded by the emergence of other psychiatric symptoms, including anxiety, depression and disorder eating behaviors, adding complexity, and potentially worsening glucose control.22-24 Our study highlights that the eventual occurrence of device-related skin reactions can serve as an additional risk factor, contributing to the impairment of the psychosocial impact of diabetes on the lives of adolescents.
Our investigation revealed higher levels of diabetes distress among parents of youths with skin reactions compared to the control group. Emotional burden is common among families of children living with T1D, often manifested through symptoms of anxiety, depression, exhaustion, and a pronounced need for respite. 25 Furthermore, parents’ diabetes distress levels have been found to be closely related to children’s glycemic levels. 26 Interestingly, our analysis indicated that unpredictable burden was more salient than concrete burden in parents of youth with a history of skin issues. This form of burden is characterized by negative emotions and worry about the future. The PAID-PR scale evaluates this type of parent distress by exploring feelings of anger and fear when contemplating their child’s life with diabetes, as well as concerns about the potential occurrence of serious complications. 15 The emphasis on unpredictable burden highlights the emotional challenges experienced by parents in navigating the complexities associated with their child’s diabetes management, particularly when device-related skin reactions occur.
The presence of dermatological complications did not appear to significantly impact glucose control, as evidenced by the lack of differences in both HbA1 levels and CGM metrics between the study group and control group. These findings are consistent with other previous studies that investigated HbA1c variations in individuals facing skin reactions.9,27 Analysis regarding the influence of skin issues on glucose metrics cannot be compared directly with existing literature as no other study has still investigated this aspect. However, caution is needed in interpreting the lack of correlation between worsening CGM parameters and skin reactions observed in our study. The rate of glucose sensor failure in the study group resulted in CGM metric data not available for all participants, potentially representing a bias for the comparative analysis. Indeed, the prevalence of drop-out from CSII therapy and/or CGM emerged as one of the most alarming findings of our investigation. Particularly, more than 20% of children and adolescents with skin issues were forced to discontinue insulin pump and/or glucose sensors use. This finding is supported by a previous study that remarked that the reported hassle of constantly wearing devices is a major barrier to device use among adolescents with T1D. 28
The change of device brands and, if necessary, discontinuation of the culprit device, represent the ultimate therapeutic options in severe cases characterized by the failure of all available preventive measures. Consistent with the literature evidence, youths experiencing dermatological complications, along with their families, are advised to engage with a multifaceted approach to preserve skin integrity. 29 Considerations about proper device placement, meticulous skincare practices, and gentle removal of patches are routinely discussed. In addition, protective physical skin barriers, such as films, tapes, or adhesives are recommended in cases of occurrence of eczema or wounds.
The diversity of device brands implicated in the occurrence of skin reactions is closely related to the range of potentially harmful substances present in the adhesives of sensors and ISS. These substances have been identified in all diabetes devices routinely used in clinical practice.30-32 In addition, in the manufacturing process, various materials may be mixed, posing a challenge in precisely identifying the components of CGM and ISS.
Our study has some weaknesses. First, the cross-sectional design limits our ability to establish causality and evaluate the dynamics of skin reactions over time. The study group comprised individuals with a recent history of skin reactions, which may not have been present during the 14-day period of collecting CGM metrics. This may have resulted in a potential bias in the assessment of glycemic outcomes. Furthermore, the absence of additional tools aimed at investigating the quality of life of study participants represents a limitation in providing a clearer understanding of the psychosocial impact of skin issues due to diabetes devices. In addition, the relatively modest sample size of the study cohort constrained our ability for meaningful subgroup analyses to explore potential differences among individuals experiencing skin reactions. Nonetheless, the strengths of our investigation lie in its originality, offering a perspective on the complex interplay between device-related dermatological complications and well-being of youth with T1D and their parents. Furthermore, it is noteworthy the comprehensive inclusion of various glucose control parameters, including CGM metrics.
Conclusions
In conclusion, our study highlights that device-related skin issues contribute to increase emotional distress in youths with T1D and their parents. This finding emphasizes the significance of addressing this insidious complication related to the daily use of technology for diabetes management. Recognizing the psychological impact of skin reactions, further longitudinal studies are needed to fully elucidate the long-term consequences and explore potential intervention strategies to mitigate the emotional burden experienced by individuals and their families.
Footnotes
Acknowledgements
The authors thank all the patients and their parents for their study participation.
Abbreviations
CGM, continuous glucose monitoring; CSII, continuous subcutaneous insulin infusion; HbA1c, glycated hemoglobin; IIS, insulin infusion set; PAID-PR, Problem Areas in Diabetes–Parent Revised version; PAID-T, Problem Areas in Diabetes–Teen version; T1D, type 1 diabetes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SP received speaking honoraria from Roche and Movi SpA. NM received speaking honoraria from Movi SpA, Theras, and Novonordisk. MB received speaking honoraria from Movi SpA. All other authors declare no conflicts of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
