Abstract
Diabetes Technology Society hosted its annual Diabetes Technology Meeting from November 1 to November 4, 2023. Meeting topics included digital health; metrics of glycemia; the integration of glucose and insulin data into the electronic health record; technologies for insulin pumps, blood glucose monitors, and continuous glucose monitors; diabetes drugs and analytes; skin physiology; regulation of diabetes devices and drugs; and data science, artificial intelligence, and machine learning. A live demonstration of a personalized carbohydrate dispenser for people with diabetes was presented.
Keywords
Introduction
From November 1 to November 4, 2023, Diabetes Technology Society (DTS) gathered health care professionals (HCPs), industry representatives, academicians, researchers, and US regulatory officials for the Diabetes Technology Meeting (DTM). This four-day meeting included two workshops, 12 sessions, a live demonstration, and a keynote presentation by the Associate Director of the Center for Biomedical Informatics and Data Science at Northwestern University Feinberg School of Medicine, all covering current research and emerging topics in diabetes technology. Table 1 presents the agenda for the meeting, including a list of workshop and session topics. This meeting report summarizes the key points of each presentation.
Agenda for the 2023 Diabetes Technology Meeting.

Abbreviations: AID, automated insulin delivery; CGM, continuous glucose monitor; iCoDE-1, Integration of Continuous Glucose Monitoring Data into the Electronic Health Record; iCoDE-2, Integration of Connected Diabetes Device Data into the Electronic Health Record; IFCC, International Federation of Clinical Chemistry and Laboratory Medicine.
Symposium: Standardization of Continuous Glucose Monitoring Performance Evaluations—Developments From the IFCC Working Group on CGM
Guido Freckmann, MD
Institut für Diabetes-Technologie, Forschungs und Entwicklungsgesellschaft mbH an der Universität Ulm, Ulm, Germany
Johan H. Jendle, MD, PhD
School of Medicine and Health, Institute of Medical Sciences, Örebro University, Örebro, Sweden
Continuous Glucose Monitoring Accuracy: Contrasting Conformité Européenne Marking With the Governmental Controls of the United States and Australia
John S. Pemberton, BSc
Department of Endocrinology and Diabetes, Birmingham Children’s Hospital, Birmingham Women’s, and Children’s NHS Foundation Trust, Birmingham, UK
The Conformité Européenne (CE) marking does not guarantee the accuracy of a continuous glucose monitor (CGM) device, even for its indicated populations.
There is a lack of CGM-specific standards to guide notified bodies during conformity assessments.
Relying solely on the CE marking system for CGM regulation puts those with type 1 diabetes (T1D) at risk.
A Scoping Review of CGM Performance Evaluations From the Last 20 Years
Stefan Pleus, PhD
Institut für Diabetes-Technologie, Forschungs und Entwicklungsgesellschaft mbH an der Universität Universität Ulm, Ulm, Germany
The use of different designs and results to present studies for the clinical performance evaluation of CGM systems is a major challenge in comparing their results.
The presentation summarized the findings from a recently published scoping review of CGM performance studies published between 2002 and 2022. 1
The review found large variability in nearly all aspects of study design as well as gaps in the reporting of important details. Therefore, a comprehensive list of recommendations for reporting is provided and the implications for the standardization of CGM performance studies were discussed. 1
Comparison Measurement Approaches for CGM Performance Studies
Rolf Hinzmann, MD, PhD
Roche Diabetes Care GmbH, Mannheim, Germany
The accuracy performance evaluation of a CGM system measures two factors simultaneously: the analytical performance of the CGM sensor and the performance of the algorithm to predict blood glucose from interstitial fluid (ISF) glucose. This is different from typical accuracy performance evaluations in clinical chemistry where the matrix of the analyte is the same for the test method and the comparison method. The accuracy of this prediction depends, among others, on the dynamics of the glucose flow between ISF and blood.
Glucose dynamics are influenced by the glucose excursions induced in the study and, together with the comparator method and the matrix chosen for comparison (capillary, venous, or “arterialized” venous blood), influence the measured analytical performance of a CGM system. This can explain different results of different performance studies for the same CGM system.
CGM system accuracy performance evaluations are currently not standardized. Standardization is necessary to achieve objective comparability of different CGM systems.
Standardized Testing Procedures for the Clinical Performance Evaluation of CGM Systems: A Proposal by the IFCC Working Group on CGM
Guido Freckmann, MD
Institut für Diabetes-Technologie, Forschungs und Entwicklungsgesellschaft mbH an der Universität Ulm, Ulm, Germany
The absence of clearly defined requirements for the characteristics of comparator data and testing procedures in CGM performance studies is a major obstacle in comparing the results of different studies.
The presentation introduced a proposal for such requirements and procedures from a group of experts in the field of CGM performance testing.
It was emphasized that the testing scenarios should include a wide range of glucose concentrations and dynamics to ensure that CGM systems are subjected to critical, real-world conditions during performance testing.
The first presentation from a UK point of view corroborated that increasing accuracy of CGM systems was a major contributor in their now widespread adoption. However, it was shown that market approval through CE marking may not guarantee sufficient accuracy, and there can be a mismatch between intended use and clinical evidence, particularly for use in populations like minors. This indicates a lack of CGM-specific standards to guide notified bodies during conformity assessments. Finally, the opportunity to implement results of the International Federation of Clinical Chemistry and Laboratory Medicine (IFCC) working group on CGM (WG-CGM) in the context of the UK’s goal to establish its own guidelines for market approval was discussed.
Transitioning to the work done by the WG-CGM, a recent review summarized 129 studies on the analytical performance of CGM systems published between 2002 and 2022. 1 Large variability in many study design aspects was identified, highlighting the need for standardization of CGM performance studies. This variability impairs the comparability between studies, leading to the situation where different performance results are observed in the same CGM system. An example of variability in mean absolute relative difference (MARD) results in the same CGM system is shown in Figure 1. An additional complication is given by the often incomplete reporting of study design aspects. The review article provides a list of topics recommended in reporting CGM performance studies. 1
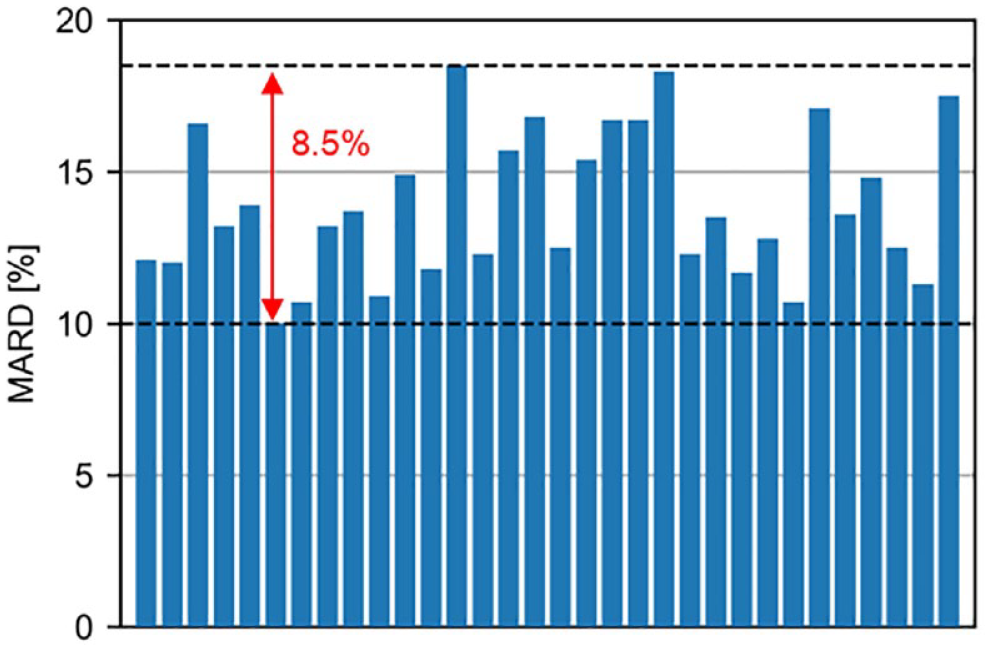
Mean absolute relative differences of the same CGM system observed in performance studies published between 2015 and 2022. Abbreviations: CGM, continuous glucose monitor; MARD, mean absolute relative difference. Source: Figure reproduced from the work of Freckmann et al. 1
Another presentation specifically discussed comparator measurement approaches in CGM performance studies. As CGM systems measure glucose concentrations in ISF but the results are compared with glucose concentrations in blood, it is impossible to establish a conventional metrological traceability chain. To nevertheless select an appropriate comparator measurement approach, analytical factors of the comparator method itself as well as the sample origin (matrix) have to be considered. In particular, the physiologic differences between glucose concentrations in blood samples of different origins (capillary, venous, “arterialized” venous) have been shown to influence the observed accuracy, and their advantages and disadvantages for use in CGM performance studies were discussed.
In addition to the comparator measurement approach, the absence of clearly defined requirements for the characteristics of comparator data, which can impact observed CGM performance, and the lack of clearly defined testing procedures also impair the comparability of study results. The last presentation emphasized the importance of considering combinations of glucose concentrations and rates of change (RoCs) when designing testing procedures, in particular for the manipulation of glucose levels during in-clinic sessions. To identify critical combinations of glucose levels and RoCs, real-world CGM data were analyzed. As a result, it was proposed that CGM performance studies should aim to produce sufficient comparator data with high and low glucose levels, as well as combinations of glucose levels and RoCs that indicate impending hypoglycemia and hyperglycemia. To produce these comparator data, a standardized study protocol was proposed.
Workshop A: Digital Health Tools—Panel 1: Interactive Apps for Behavioral Change
William Polonsky, PhD
Behavioral Diabetes Institute, San Diego, California, USA
Michelle L. Litchman, PhD, FNP-BC, FAANP, FADCES, FAAN
The University of Utah College of Nursing, Salt Lake City, Utah, USA
Andrew Farmer, DM, FRCGP
Nuffield Department of Primary Care Health Sciences, University of Oxford, Oxford, United Kingdom
The mechanisms through which interactive applications (apps) can achieve behavior change for people with diabetes include setting personal goals, strengthening motivation, developing a feeling of accomplishment, and providing opportunities for social support.
The untapped opportunities represented by interactive apps to produce a step change in health care for people with diabetes include the use of biometric sensors offering feedback and motivation for behavior change alongside the use of artificial intelligence (AI)-powered predictive analytics and anticipatory designs to provide timely, personalized interventions.
The key challenges in implementing the use of behavior change apps across entire health systems are the need to develop (1) a range of funding models that would enable them to be targeted at those patients who would most benefit, (2) a legal and data security framework to address liability concerns and personal data, and (3) more research to better understand the extent of clinical effectiveness of apps for different groups of people, with different types of illness, cared for in different health settings.
Felix C. Lee, MPharm, MSc, MBA
Sanofi, Bridgewater, New Jersey, USA
Apps have the potential to modify behavior—and we know that in managing chronic conditions, such as diabetes, drugs alone are not enough. 2 Sustained behavior change is needed in type 2 diabetes (T2D) to improve health outcomes.
Interactive apps are “bi-directional” in nature, unlike drugs, and have the ability to deliver digital interventions personalized to the individual, rather than a “one-size-fits-all” approach. Behavior change models, such as the COM-B framework 3 to improve a person’s capability, provide opportunity, and enhance motivation and the BJ Fogg MAP 4 model of Motivation, Prompts, and Ability can help inform intervention designs to elicit behavior change.
Part of the debate is whether we can show sufficient evidence that digital interventions delivered through smartphone apps can bring about behavior change and how we interpret the evidence.
David T. Ahn, MD
Mary & Dick Allen Diabetes Center, Hoag Memorial Hospital Presbyterian, Newport Beach, California, USA
To date, the most successful behavior change apps have been integrated with hardware (eg, CGMs, automated insulin delivery [AID] systems, etc).
The existing body of clinical evidence for diabetes apps is limited (eg, limited sample size, short study duration, etc).
Human interaction (or the perception of it) plays a key role in behavior change.
Digital health tools, such as interactive apps, can be important tools to improve the management of chronic conditions, such as diabetes. People with diabetes can use these patient-facing digital health solutions for various purposes, such as tracking glucose levels or calculating insulin doses. A large spectrum of diabetes apps currently exists in the marketplace. An example of some of the available apps is shown in Figure 2. They range from full suite solutions (including connected blood glucose monitors [BGMs], activity tracking, food blogging, data from wearables, coaching, and insulin dosing information) to apps that offer one or a subset of these options. However, simply having a wide array of features may not necessarily lead to behavior change.

Examples of the top iPhone diabetes applications in the Apple App Store as of January 11, 2024. Source: Apple App Store. 5
Interactive apps can take advantage of text messages, prompts, and/or notifications to engage the people using them. The ubiquity of smartphones in daily life therefore provides an opportunity for interactive apps to become incorporated into a person’s lifestyle and behavior. An essential feature of an interactive app is having a two-way interaction between the user and the app. Personalized messages, whether by the use of large language models or the addition of texting features, such as emojis and GIFs, can therefore enhance the app experience. It is important to consider the duration and frequency of the interactions, as they may not be well-defined and could be specific to the population and disease.
Among the most successful behavioral change apps are those tied to products or services. For example, users of apps that accompany CGMs will typically log in to the app multiple times a day and adjust their behavior in response to the information displayed by the app. The effectiveness of interactive apps depends on whether there is a sustained change in behavior that can complement the current standard of care and treatment with drugs, which is especially true for diseases like diabetes that are rarely managed through medications alone. By facilitating behavioral change in daily life, apps can promote healthier choices.
The concept of a “diabetes app” may need to change significantly. An app specific to diabetes management could become burdensome to the user, especially if multiple logins per day are required. Designing apps to generate more organic and meaningful interactions could lead to better retention over time. The best interactive apps would be those that can smoothly integrate into daily life through a variety of modalities, such as limiting notifications to only those that are timely and relevant or introducing a community component. Embedding social media into an interactive app, however, has both positive and negative consequences. Social support can be effective by providing emotional support, normalizing health technology, and making information accessible to others. At the same time, online communities risk exposing the user to negative social influences and bring up issues of privacy.
When designing interactive apps, it is important to consider whether specific populations are being reached. App usage and preferred modality will differ across generations, age ranges, and racial and ethnic groups. Fluency in English and access to health care can also act as barriers to digital health tool usage. However, apps also have an opportunity to help break down existing health disparities and improve accessibility, such as offering services in different languages and having settings for those who are visually impaired or hard of hearing.
In the existing space of digital health and diabetes apps, there are still challenges that remain to be addressed, including questions about cost, scale, liability, and clinician reimbursement. Further research is needed to evaluate the efficacy of digital health interventions in addition to adoption and implementation practices. However, it is unclear what type and level of evidence is sufficient to support the value of diabetes apps.
The versatility of technology allows for apps to be personalized to the individual user, rather than a one-size-fits-all approach. In addition, apps should be intuitive and convenient to encourage long-term use and sustained behavior change. Integrating data from sensors, wearables, and the electronic health record (EHR) can further personalize content and increase the frequency of timely, relevant reminders. As technology in the digital health space improves, interactive apps have the potential to meaningfully interact with users and reinforce behavioral changes that lead to improved outcomes for people with diabetes.
Workshop A: Digital Health Tools—Panel 2: Telehealth and Coaching
Warris Bokhari, MBBS
Amazon, Seattle, Washington, USA
David Kerr, MBChB, DM, FRCPE, FRCP
Sutter Health, Santa Barbara, California, USA
Wei-An (Andy) Lee, DO
Los Angeles General Medical Center, Los Angeles, California, USA
Shared virtual group visits show promise in providing cost-effective ways to improve medication adherence, self-monitoring, and medical consultation in the underserved population in Los Angeles; however, there are still significant barriers to adoption.
Telehealth navigators have been one of the most significant interventions in improving adoption of telehealth interventions for the safety-net population in Los Angeles County.
Although currently out of reach, AI-based digital twin (DT) modeling of patients in the Medicaid population can provide needed novel interventions to persons with diabetes in this population.
Anand K. Iyer, PhD, MBA
Welldoc, Inc., Columbia, Maryland, USA
The management of chronic conditions like diabetes, hypertension, and heart failure (HF) require persons to meticulously manage several parameters, including medications, lab measurements, diet, and exercise.
AI-powered digital health can help both people with chronic conditions as well as their HCPs to better manage these conditions in a personalized and efficacious manner.
The presentation will focus on the role of AI-powered digital health and its associated outcomes and benefits to the health care ecosystem.
Sacha Uelmen, RDN, CDCES
Association of Diabetes Care & Education Specialists, Chicago, Illinois, USA
National telehealth trends indicate that diabetes care and education specialists offering telehealth services as a mode of delivery improve patient outcomes and are more sustainable services.
Including the interprofessional team throughout the health care journey improves patient satisfaction and health outcomes for people with diabetes.
Reducing barriers to diabetes care through telehealth requires additional consideration of topic, timing, and place while allowing for micro-learning.
As the number and complexities of treatments and devices increase, there was universal agreement from the panel that the need for human coaches will increase. However, with the increasing use of digital health technologies to support diabetes care, the services offered by human coaches will change beyond navigating the complexities of the health care system to also include guidance for people with diabetes on how to access and use technologies. This will create a new type of community health worker—“tech-coaches and “tech-navigators”—which will require new approaches to employment, training, and reimbursement. Soon, AI will likely be used to deliver coaching, although currently this is at a very early stage. The advantages of AI potentially could include reducing routine, non–patient-facing tasks; providing evidence-based decision support for clinicians; and stratification of patients according to their needs, including identification of high-risk individuals. An AI-generated image of a chatbot and an HCP is shown in Figure 3.
An image generated by artificial intelligence of (a) a chatbot and (b) a health care professional with the assistance of DALL·E 2. Source: Figure courtesy of David Kerr.
For human coaching, the disproportionate burden of diabetes impacting individuals and families experiencing health disparities is creating opportunities for new approaches to care delivery (eg, shared telehealth appointments), but barriers remain in terms of reimbursement codes, training, and resource intensities, as well as improved knowledge of health beliefs and cultures (eg, there is a need for greater involvement of patients receiving Medicaid). Furthermore, feedback loops to continuously show the value of human coaching are required. For digital coaching, developing and maintaining trust is essential, but, at the same time, false empathy should be avoided.
Soon, a generation of young people who do not have experience prior to the digital revolution of the past two decades will be applying for medical, nursing, and other professional training in health care. Their understanding and expectation of the use of digital tools to support patient care, including health coaching, will require new approaches to education and health care delivery. The future of health coaching is likely to be a hybrid of human and machine delivered care, and digital health will become, simply, health.
Workshop B: Metrics for Assessing Glycemia and Complications—Panel 1: Analytical and Clinical CGM Metrics
Guillermo Umpierrez, MD
Emory University School of Medicine, Atlanta, Georgia, USA
Eda Cengiz, MD, MHS, FAAP
University of California, San Francisco, School of Medicine, San Francisco, California, USA
The Glycemia Risk Index
David C. Klonoff, MD, FACP, FRCP (Edin), Fellow AIMBE
Mills-Peninsula Medical Center, San Mateo, California, USA
The Glycemia Risk Index (GRI) is a single-number summary on a 0 to 100 scale of the quality of glycemia that can be used by clinicians and population health researchers to determine the glycemic effects of prescribed and investigational treatments. 6
The GRI corresponds closely to 330 clinicians’ rankings of the overall quality of glycemia of 225 CGM tracings from four types of insulin users and is a best-fit composite of the seven most important metrics displayed on an ambulatory glucose profile (AGP).
The GRI accounts for glycemic variability in its portrayal of the quality of glycemia and addresses the question of how a patient is doing by providing an actionable score.
Update on Time in Range
Richard M. Bergenstal, MD
International Diabetes Center, HealthPartners Institute, Minneapolis, Minnesota, USA
Measurement: Time in range (TIR)/time below range (TBR) is becoming both a personal and a population health quality measure and increasingly being considered a measure of risk of vascular complications.
Motivation: TIR/TBR is proving to motivate people with diabetes to keep working on improving glycemia. Since the data are so accessible on a mobile phone and the changes in glucose with meals are so easy to see, people with diabetes are motivated to change eating patterns to minimize glucose excursions with meals.
Management: Distinct combinations of TIR/TBR can guide the most effective and the safest selection of diabetes management medications and lifestyle changes, thus approaching a form of precision or personalized diabetes management.
Tight TIR
Viral N. Shah, MD
Indiana University School of Medicine, Indianapolis, IN, USA (current)
Barbara Davis Center for Diabetes, University of Colorado, Aurora, Colorado, USA
Healthy individuals without diabetes spent 96% of their time in a glucose range of 70 to 140 mg/dL.
In our study, hemoglobin A1C (HbA1c) and time in 70 to 140 mg/dL are the best predictors of diabetic retinopathy in T1D. 7
Therefore, time in tight glucose range (70-140 mg/dL) should be considered as one of the metrics for glycemic control in people with diabetes.
Continuous Glucose Deviation Interval and Variability Analysis
Manuel Eichenlaub, PhD
Institut für Diabetes-Technologie, Forschungs und Entwicklungsgesellschaft mbH an der Universität Ulm, Ulm, Germany
There are currently a large number of metrics, parameters, and methods to characterize the accuracy of CGM systems, which makes it difficult to compare and discuss CGM accuracy.
This presentation introduces the Continuous Glucose Deviation Interval and Variability Analysis (CG-DIVA), which is a new approach for the comprehensive characterization of CGM point accuracy based on US Food and Drug Administration (FDA) integrated continuous glucose monitoring (iCGM) system guidelines. It contains a visual demonstration of the expected deviations of a CGM system in different glucose levels as well as the differences in accuracy from sensor to sensor. 8
It is argued that the CG-DIVA can simplify the discussion of CGM accuracy and replace the high number of conventional approaches.
Glycemic Ratio for Critically Ill People
James S. Krinsley, MD
Division of Critical Care, Stamford Hospital, Columbia University College of Physicians and Surgeons, Stamford, Connecticut, USA
The glycemic ratio is the quotient of mean intensive care unit (ICU) blood glucose and estimated preadmission blood glucose, based on HbA1c.
The glycemic ratio is more predictive of mortality than is the Stress Hyperglycemia Ratio (the quotient of admission blood glucose and preadmission blood glucose, based on HbA1c). 9
The use of this new metric highlights the interaction of acute and chronic glycemia on the risk of mortality of critically ill patients.
Reaching glycemic targets is one of the fundamental pillars of diabetes treatment and is essential to prevent complications of diabetes. Continuous glucose monitoring use has become an essential component of standard of care for diabetes treatment and has introduced new metrics to ascertain glycemia and prevent diabetes complications.
The GRI is one of the newest additions to the group of glycemic index metrics. 6 It is a composite metric for the quality of glycemia and is developed using a model to predict the clinician ranking based on seven standard CGM data metrics in the AGP. It is a single-number summary on a 0 to 100 scale weighted according to the risk of hypoglycemia, hyperglycemia, and glycemic variability. Like HbA1c, a high score is unfavorable and indicates that more work needs to be done to improve glycemia. The GRI can be displayed graphically on a GRI Grid that reflects the quality of the glycemia according to percentage quintiles. The GRI is an innovative glycemic metric with actionable scores and a graphical display that can be used by clinicians and researchers to determine the glycemic effects of prescribed and investigational treatments. While more research is needed to ascertain its correlation with diabetes complications and its usability by clinicians, it crafts a broad and practical glycemic angle to complement conventional glycemic metrics. The GRI calculator on the DTS website is shown in Figure 4.
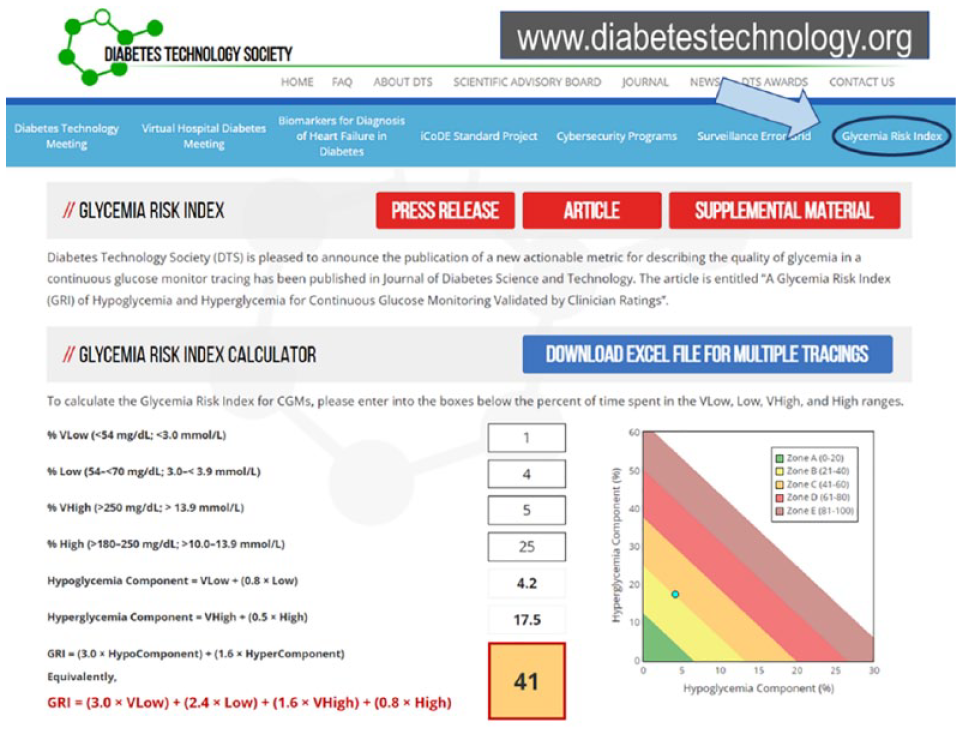
The Glycemia Risk Index Grid with the hypoglycemia component on the horizontal axis and the hyperglycemia component on the vertical axis. 10 Diagonal lines divide the graph into five zones (quintiles) corresponding to the best (zeroth-20th percentile) to the worst (81st-100th percentile) overall quality of glycemia. Source: Glycemia Risk Index. Diabetes Technology Society. https://www.diabetestechnology.org/gri/.
Time in range has become one of the most important standard of care metrics and a core outcome to assess glycemia both in clinical and research settings. 11 Time in range and TBR could be instrumental to motivate people with diabetes to improve their glycemia given that glycemic patterns are easily accessible to people with diabetes on their mobile phones and by easy-to-understand CGM data reports.
Time in tight range (TITR, 70-140 mg/dL [3.9-7.8 mmol/L]) introduces a secondary stricter measure of glycemic control that could be achieved by intensified diabetes treatment. Time in tight range and HbA1c have been shown to be the best predictors of retinopathy for people with T1D. 7 Time in tight range can become an additional metric for assessing glycemic control with a target that could be individualized based on age and risk of complications.
The CG-DIVA is a new approach for the comprehensive characterization of CGM point accuracy based on FDA iCGM guidelines. 8 It provides a simplified graphical visualization of CGM accuracy by demonstrating the expected deviations of a CGM system in different glucose levels and the differences in accuracy from sensor to sensor.
The glycemic ratio is the quotient of mean ICU blood glucose and estimated preadmission blood glucose based on HbA1c. It highlights the interaction of acute and chronic glycemia on the risk of mortality of critically ill patients and is more predictive of mortality than the Stress Hyperglycemia Ratio (the quotient of admission blood glucose and preadmission blood glucose, based on HbA1c). 9
The new glycemic metrics that have emerged in recent years give a more comprehensive view of glycemic profiles and provide more nuanced and personalized approaches to diabetes management. As the use of CGMs expands and evolves, it is essential to investigate and introduce novel glycemic metrics to diabetes management to improve the care and quality of life of people with diabetes.
Workshop B: Metrics for Assessing Glycemia and Complications—Panel 2: Sensors for Diagnosing Complications in Diabetes
Michael J. McShane, PhD
Department of Biomedical Engineering, Texas A&M University, College Station, Texas, USA
Andjela Drincic, MD
Division of Diabetes, Endocrinology, and Metabolism, University of Nebraska Medical Center, Omaha, Nebraska, USA
External Eye Screening as an Adjunct to Retinal Screening
Jorge A. Cuadros, OD, PhD
Herbert Wertheim School of Optometry and Vision Science, University of California, Berkeley, Berkeley, California, USA
Deep learning algorithms have been shown to provide accurate diagnosis of diabetic eye disease, but diagnostic quality imaging devices are costly and difficult to use.
Deep learning can detect biomarkers that humans may not be able to detect.
External images of the eye captured by smartphones (“eye-selfies”) can be used with deep learning algorithms to detect diabetes, diabetic eye disease, kidney disease, and several other conditions. This may be a useful and easily accessible tool for the future. 12
Monitoring Exercise for Precision Diabetes
Michael C. Riddell, PhD
Department of Kinesiology & Health Science, Muscle Health Research Center, York University, Toronto, ON, Canada
Glucose sensors have changed the way that exercise management is tackled in T1D.
Carbohydrate feeding should be initiated during exercise when sensor glucose level is < 140 mg/dL (< 7.8 mol/L) and a downward directional arrow is also observed.
Automated insulin delivery systems should be placed in “exercise mode” with a higher temporary target set 60 to 90 minutes before the onset of prolonged moderate-intensity exercise to allow circulating insulin levels to drop sufficiently. Pre-exercise carbohydrate feeding should be avoided to help reduce insulin-on-board.
A Multimodal Sensor Panel for Diabetes
Gerard L. Coté, PhD
Department of Biomedical Engineering, Texas A&M University, College Station, Texas, USA
The multimodal wearable arm band for predicting glycemic events has been developed. 13
The wearable includes six non-invasive sensors that measure physiologic responses to glycemic events, including single-sided electrocardiogram (ECG), bioimpedance, photoplethysmography (PPG), temperature, accelerometer, and gyro.
The sensors are combined with AI and deep learning algorithms to predict the glycemic events.
IEEE 2621: The Diabetes Device Cybersecurity Standard
Ted Osinski
Institute of Electrical and Electronics Engineers, Piscataway, New Jersey, USA
The Institute of Electrical and Electronics Engineers (IEEE) 2621 standard is a framework that can accommodate all types of medical devices and diabetes devices in particular. 14
The IEEE 2621 certification program provides an independent assessment of medical device cybersecurity.
The IEEE 2621 program and services will prepare medical devices for FDA submission.
Racial-Ethnic Disparities in Wearable Diabetes Devices
Devin W. Steenkamp, MBChB
Boston University Chobanian & Avedisian School of Medicine, Boston, Massachusetts, USA
We are witnessing ongoing challenges that underserved and minoritized patients face in adopting diabetes technologies.
There are practical considerations that clinicians may incorporate into care to support increased adoption of diabetes technology in populations who have traditionally not derived benefit.
Our local data from our safety-net institution show improved patient outcomes when technology is implemented into routine care.
Diabetic retinopathy screening is a mainstay of preventative care in diabetes, but many patients underutilize these services. Telemedicine-based retinopathy screening programs using AI and deep learning algorithms can improve access to care by bringing it to primary care and endocrinology offices. To date, there are three FDA-approved autonomous algorithms for diabetic retinopathy screening, all based on retinal images, offering screening and enhancing care through effective triage of patients with retinopathy. Artificial intelligence-based retinal image analysis has a potential to identify other disease processes, including risk of cardiovascular disease, kidney or liver disease, and cognitive disorders. In addition, AI offers a new potential for detecting signs of other diseases, including kidney disease and anemia disease, using external photographs of the eyes, especially the pupil and the iris. 12 Figure 5 depicts an example of a machine learning (ML) system being used to predict biomarker levels based on external eye photographs.
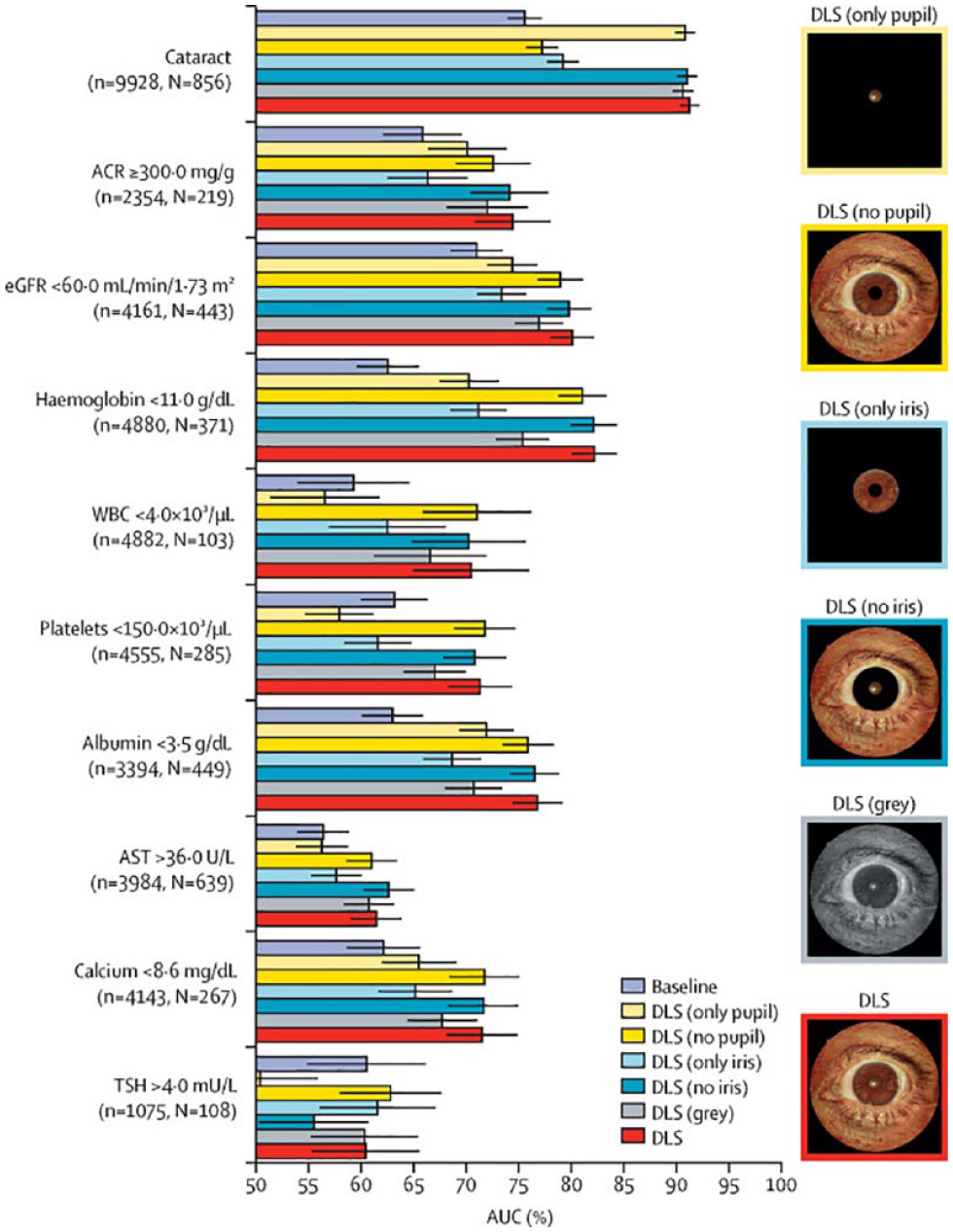
A deep learning system predicts parameters and biomarker levels based on external eye photographs. The results are shown for experiments where different regions of the images are masked or the image color is removed. Abbreviations: ACR, albumin-to-creatinine ratio; AST, aspartate aminotransferase; AUC, area under the receiver-operating characteristic curve; DLS, deep learning system; eGFR, estimated glomerular filtration rate; N, number of positive datapoints; n, number of datapoints; TSH, thyroid stimulating hormone; WBC, white blood cells. Source: Figure reproduced from Babenko et al 12 under the Creative Commons Attribution Non-Commercial No-Derivatives 4.0 International License (CC BY-NC-ND 4.0, https://creativecommons.org/licenses/by-nc-nd/4.0/).
For people living with diabetes, exercise conveys great challenges for glucose control: prolonged light to vigorous intensity aerobic activities promote a strong glucose-lowering effect, but resistance training may lead to post-exercise hyperglycemia. Continuous glucose monitors have revolutionized the way that diabetes is managed during exercise. Recently, TIR goals for training and competition have been published for athletes with T1D and consensus reports have been developed for glucose management during exercise 15 based on sensor glucose readings as well as trend arrows. Automated insulin delivery systems offer the advantage of having an exercise setting, but challenges related to physiology of exercise subtypes and pre-exercise blood glucose and carbohydrate ingestion mandate vigilance.
A multimodal non-invasive sensor panel for diabetes can complement or supplement CGM data to help with the hypoglycemia prediction. 13 Such non-invasive wearables monitor a number of psychophysiological features, such as skin temperature (decreases with hypoglycemia), skin conductivity (decreases with hypoglycemia due to elevated perspiration), ECG features (lengthened QT interval and reduction in heart rate variability with hypoglycemia), accelerometer/gyros (to monitor tremor), and electroencephalogram features (increase in delta/gamma and decrease in alpha frequency with hypoglycemia). When combined with CGM data, they offer excellent sensitivity and specificity for hypoglycemia prediction.
There are ongoing efforts to create a cybersecurity ecosystem for connected diabetes devices, including BGMs, CGMs, insulin pumps, insulin pens, and AID systems. The hope is to extend the scope to other medical devices in the future. IEEE adopted the IEEE 2621 standards as of March 2022, 16 outlining a framework for a connected electronic product security evaluation program. It offers various tiers of security and functional assurance levels. The IEEE certification should make the FDA submission process smoother. 14
There are significant racial disparities in the use of diabetes technology; non-white patients are 50% less likely to use technology even after adjusting for factors, such as insurance and annual income. 17 Health systems-based interventions that have shown to increase technology use include increased patient access to diabetes educators as well as provider education to raise awareness of technology benefits with the aim of increasing referrals. Institutional support for a technology coordinator is needed and the development of a clinic-based interdisciplinary education team is crucial for the success of such programs.
Keynote Presentation: iCoDE 1 and 2
Juan C. Espinoza, MD, FAAP
Ann & Robert H. Lurie Children’s Hospital of Chicago, Chicago, Illinois, USA
Electronic health record integration of diabetes technology is critical to improving patient care and lowering barriers to access.
Data and interoperability standards decrease barriers to integration adoption.
Cross-sector collaboration is critical to advance the state of EHR integration and interoperability.
iCoDE-1 (Integration of Continuous Glucose Monitoring Data into the Electronic Health Record) focuses on moving diabetes technology data from various devices into the EHR. By providing standards and recommendations, iCoDE-1 will (1) streamline workflows, (2) lower the barriers to integration, and (3) increase data access to enhance the overall experience for both patients and clinicians.
The current state of interoperability is defined across four levels: Level 1 (No Interoperability), Level 2 (Structural Interoperability), Level 3 (Syntactic Interoperability), and Level 4 (Semantic Interoperability). 18 Exploration of technical architecture, standards, and best practices led to an adapted interoperability framework, for CGM data specifically, with seven levels of integration: Level 1 (Transcription), Level 2 (Static Documents), Level 3 (Variable Documents), Level 4 (Discrete Structure Data), Level 5 (Continuous Structured Data), Level 6 (Device or App Metadata), and Level 7 (Advanced Analytics). 18 There are challenges in achieving Level 7 interoperability, and a practical framework is needed.
The final 2022 iCoDE Report, endorsed by organizations such as the American Association for Clinical Chemistry comprehensively covers technical standards and clinical workflow recommendations. It also contains a project implementation guide that considers the feasibility of the integration process, including a Diabetes Data and Technology Integration Framework that is shown in Figure 6. 18
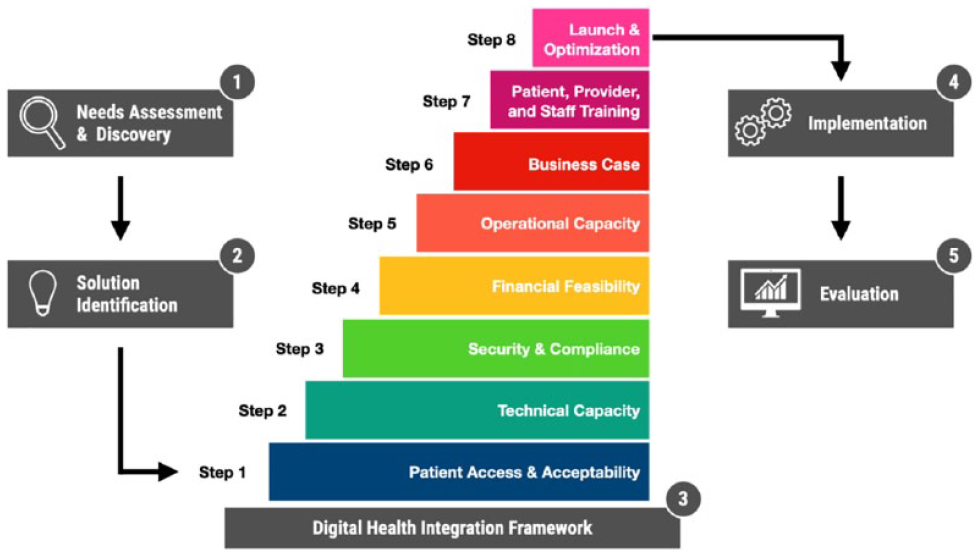
Diabetes Data and Technology Integration Frame. Source: Figure reproduced from Espinoza et al 19 (https://www.frontiersin.org/articles/10.3389/fcdhc.2022.867284/full) under the Creative Commons Attribution 4.0 International Public License (CC-BY 4.0, https://creativecommons.org/licenses/by/4.0/).
Differences in data access, metrics, and visualization in insulin devices compared with CGMs have led to the launch of the iCoDE-2 (Integration of Connected Diabetes Device Data into the Electronic Health Record) standard project. 20 iCoDE-2 will focus on the integration of insulin delivery devices into the EHR and will include recommendations for data access, standardization, visualization, and clinical workflows.
Session 1: Diabetes Data Science
William T. Cefalu, MD
Division of Diabetes, Endocrinology, & Metabolic Diseases, National Institute of Diabetes and Digestive and Kidney Diseases, National Institutes of Health, Bethesda, Maryland, USA
John C. Pickup, MA, DPhil, FRCPath
King’s College London School of Medicine, London, UK
Eirik Årsand, PhD
UiT Arctic University of Norway, Tromsø, Norway
Overview of Diabetes Data Science
Boris Kovatchev, PhD
University of Virginia, Charlottesville, Virginia, USA
Diabetes is one of the best quantified human conditions—vast amounts of glucose monitoring (eg, CGM) and other metabolically-relevant data (eg, physical activity), is readily available.
Data science methods (eg, prediction and classification techniques), ML, and AI are therefore rapidly entering the space of glycemic-control analytics.
A recent pinnacle in these developments is the first clinical testing of a Neural-net Artificial Pancreas—a new system developed at the University of Virginia, which exclusively uses data science methods to exert AID control. 21
Precision Monitoring in Diabetes
Norbert Hermanns, PhD
Research Institute of the Diabetes Academy Mergentheim (FIDAM), Bad Mergentheim, Germany
The availability of multiple sensors enables assessment of relevant parameters, behaviors, or cognitions in a daily routine.
The identification of clusters with specific patterns of correlation between these data sources is supported by the integration of glycemic, behavioral, and mental health data. In principle, this enables the development or selection of specific intervention and management approaches for such specific subgroups to advance precision medicine.
Just-in-time interventions are a fairly new and promising tool to address individual behavioral, cognitive, or glucose-related patterns in a very timely manner.
Digital Twinning Technologies in T1D
Andrea Facchinetti, PhD
Department of Information Engineering, University of Padova, Padova, Italy
Enhancing T1D therapy through DT technologies: DT technologies have emerged as indispensable tools for optimizing T1D therapy.
Personalized model generation from CGM, insulin, and meal data: By harnessing data from CGMs, insulin intake, and meal patterns, we can develop personalized models that offer deeper insights into each individual’s unique metabolic responses.
Therapeutic refinement via scenario exploration: Leveraging these personalized models, we engage in therapeutic fine-tuning using simulation scenarios, enabling us to explore and evaluate various “what if” scenarios for optimizing diabetes management strategies.
Diabetes is one of the best-quantified diseases. The emergence of diabetes technologies, such as CGMs, insulin pumps, and other wearable devices, such as physical activity trackers and heart rate monitors, has generated huge amounts of data, forming the basis of increasingly complex models of human metabolism. Data science can be used for detection, prediction, and classification. Examples include the detection of meals and physical activity using a recurrent neural network algorithm trained on CGM data; prediction of hypoglycemia using ML models; and classification of CGM profiles using ML, where it is possible to identify clusters corresponding to, among other archetypes, T1D and T2D, multiple daily injection (MDI) treatment, and insulin pump therapy. Another application is AI-powered decision support, such as the ADVICE4U system, being trialed for insulin dose optimization. 22 A Neural-net Artificial Pancreas is under development at the University of Virginia, which can approximate insulin dosing algorithms of AID systems. 21 Finally, the Virtual Diabetes Control and Complications Trial (DCCT) Project at the University of Virginia aims to replay the DCCT trial from computer-simulated CGM data. 23
New monitoring techniques have identified many contextual variables that affect diabetes control, including behavioral (eating, physical activity, medication), biological (heart rate, stress, illness, sleep), and psychological (mood, anxiety) factors. Precision monitoring aims to integrate these variables into management to provide personalized therapeutic interventions, for example, informing and improving AID systems and their algorithms. There is evidence for an association between poor glycemic control and self-care behaviors (low adherence), mental health issues (negative emotions, diabetes distress), and poor sleep quality (sleep apnea), but causality and directionality need further study. 24 However, identifying individuals so affected by monitoring techniques may provide valuable personalized management options. Challenges include monitoring measurement performance, stability of associations, interoperability of data sources, and the need for rigorous testing of personalized interventions versus standard care, both in randomized controlled trials and real-world care. Figure 7 presents a model of precision monitoring in diabetes.
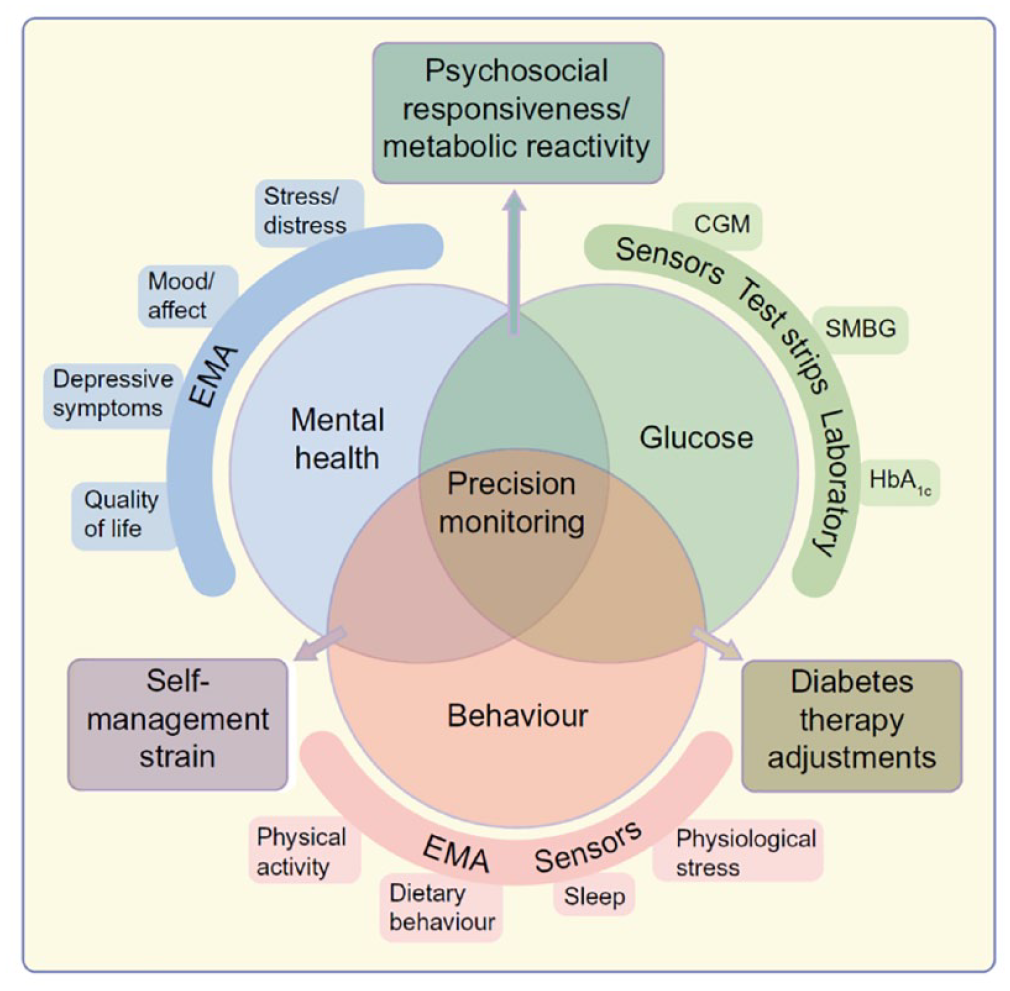
A conceptual model of precision monitoring in diabetes. Source: Figure reproduced from Hermanns et al 25 under the Creative Commons Attribution 4.0 International License (CC-BY 4.0, https://creativecommons.org/licenses/by/4.0/).
A DT is a relatively new concept in medicine, whereby a virtual, computer-generated, and intelligent representation of a real-world patient and their disease is created, using real-time and historical data to depict past and present states and simulate a predicted future. It is implemented by the internet of things (wearable sensors), tailored to individual cases, and powered by data integration. In diabetes care, it has the potential to enable precise and personalized treatment. Using data from several sources, a computer model represents a DT (updated in real time) which can be used to simulate various scenarios, such as predicting the effects of different drugs, doses, regimens, and lifestyle interventions. ReplayBG is an example of a DT tool developed at the University of Padova that uses insulin, carbohydrate, and CGM data to create a model on which the effect of test carbohydrate intake and insulin interventions can be evaluated as a simulated CGM output profile. 26 Challenges for future clinical implementation include data privacy, the complexity of human models, and medical liability.
Some of the main issues and knowledge gaps in data science are the validity and quality of data, understanding the heterogeneity of patient variables, the need for easy access to large data resources, the lack of focus on existing data, such as physical activity, ensuring patients understand terms of use when sharing their data, and improving clinician trust in “black box” models. Next steps include incorporating AI into control algorithms for AID systems, feasibility clinical trials of data science methods, such as DTs, and researching which instruments are best for monitoring contextual factors.
Session 2: Novel Indications for CGMs
Julia K. Mader, MD
Division of Endocrinology and Diabetology, Department of Internal Medicine, Medical University of Graz, Austria
Kong Y. Chen, PhD
National Institute of Diabetes and Digestive and Kidney Diseases, Bethesda, Maryland, USA
CGMs in People Without Diabetes
Tracey L. McLaughlin, MD, MS
Stanford University School of Medicine, Stanford, California, USA
Continuous glucose monitoring is currently accepted for use among individuals with a diagnosis of diabetes.
Among people without a diagnosis of diabetes, CGMs have potential to be used as a diagnostic tool for latent hyperglycemia or as a therapeutic tool for behavior modification and precision nutrition to reduce glycemic excursions and variability and potentially prevent diabetes and/or cardiovascular disease.
The optimal subgroup of the non-diabetic population who will benefit from this technology, the metrics used to define TIR, and the long-term clinical benefits have not yet been determined but will be a hot topic of the next decade.
CGMs in Pregnancy
Stephanie A. Fisher, MD, MPH
Northwestern University Feinberg School of Medicine, Chicago, Illinois, USA
Pregnancy physiology reflects a state of increased insulin resistance with advancing gestation, leading to higher blood glucose levels in pregnancies affected by diabetes; however, tighter glycemic control than in nonpregnant adults is required to reduce risk of maternal and neonatal complications related to diabetes in pregnancy, with the following CGM goals for pregnancy: TIR (target blood glucose = 63-140 mg/dL) > 70%, time above range < 25%, and TBR < 4%. 27
Although data on CGMs in pregnancy are limited, the Continuous Glucose Monitoring in Women with Type 1 Diabetes in Pregnancy (CONCEPTT) randomized trial 28 of CGM versus standard care for T1D in pregnancy demonstrated that use of CGMs promotes a reduction in HbA1c, a higher percentage of TIR, and reduced incidence of large-for-gestational age neonates, neonatal hypoglycemia, and neonatal ICU admissions.
Continuous glucose monitoring provides notable benefits for optimization of diabetes care during pregnancy for both patients and providers, but several areas surrounding CGM use in pregnancy require further study, especially for management of T2D and gestational diabetes.
CGMs in Kidney Disease
Connie M. Rhee, MD, MSc
VA Greater Los Angeles Healthcare System, Los Angeles, California, USA and David Geffen School of Medicine, University of California, Los Angeles, Los Angeles, California, USA
Review glycemic derangements in kidney disease.
Discuss the strengths and limitations of various glycemic metrics in kidney dysfunction.
Evaluate existing data on the use of CGMs in kidney disease.
The Use of CGMs in the Hospital
Kathleen M. Dungan, MD, MPH
The Ohio State University, Columbus, Ohio, USA
Use of CGMs in the hospital may be effective for identifying and treating hypoglycemia and hyperglycemia.
Adjunctive use of CGMs in the ICU may facilitate reductions in the need for point-of-care glucose values.
Implementation of CGMs in the hospital setting requires a coordinated team effort.
In using CGMs in people without diabetes, potential benefits include diagnostic and therapeutic applications. Continuous glucose monitor could help to identify individuals with glucose values outside a healthy range, high glycemic variability, and reactive hypoglycemia, especially in people with prediabetes. Therapeutic uses include tracking lifestyle modifications, personalized nutrition, and/or athletic performances. Continuous glucose monitoring could also be used in pharmacotherapies for weight loss in combination with other tools to provide feedback to change health behaviors. Continuous glucose monitor use in people without diabetes also poses certain risks; for example, frequent false hypoglycemia could lead to anxiety, impaired sleep, excess eating/weight gain, unneeded visits to health care facilities, and physiological (skin) and psychological reactions. Risks can be minimized by counseling, proper education, and intermittent rather than continuous use. People without diabetes who are at risk (eg, older age, higher body mass index, family history, and prediabetes) should be identified to optimize the use of CGMs in this population. The main goal is to minimize glucose spikes and prevent and/or delay diabetes development.
In pregnancy, CGMs can be clinically and cost-effective, allowing for closer monitoring of people with T1D throughout pregnancy for fetal exposure to maternal glycemic excursions, especially during periods of heightened insulin resistance in the second and third trimesters, and reducing the burden of self-monitoring blood glucose using glucometers. Use of CGMs has the potential to improve hypoglycemia awareness, patient and provider experience, and pregnancy outcomes. A glucose range of 63 to 140 mg/dL should be targeted during pregnancy. 27 Continuous glucose monitoring indications for use in pregnancy include all pregnant people with T1D and may be considered for pregnant people with T2D or gestational diabetes with labile glycemic control. The FDA endorses the use of CGMs for females with T1D and T2D. The CONCEPTT trial, the largest trial of CGMs in pregnancy, showed positive outcomes for pregnant people and their neonates. 28 More data on CGM use in pregnant people with gestational diabetes and T2D are needed. 28
With over 37 million US adults living with kidney disease, 29 patients with chronic kidney disease (CKD) and end-stage kidney disease (ESKD) are frequently at risk for dysglycemia, which could result in higher mortality from frequent hypoglycemia. Adequate glycemic monitoring through CGMs can lead to better glycemic outcomes and aid the management of diabetic kidney diseases. Continuous glucose monitoring has emerged as a more convenient and patient-centered method for frequent glycemic assessment, which provides a more comprehensive picture of their glycemic status. With several pilot and feasibility trials showing promising data against self-monitoring of blood glucose (SMBG), future research is needed to determine and improve the accuracy, efficacy, safety, and usability of CGMs in patients with advanced CKD, including those on renal replacement therapy, such as dialysis.
Because CGM devices are designed for personal or home use, adapting CGMs for use in a hospital setting faces major barriers, including issues with accuracy, implementation, communication/ documentation, regulatory concerns, and cost. A recent hybrid CGM protocol for the ICU has shown that it can reduce the frequency of point-of-care blood glucose measurements while safely maintaining glucose levels in very sick patients. 30 Previous small CGM studies in the ICU have also shown improvements in outcomes. CGMs used by patients with T2D and T1D in non-ICU hospital settings showed reasonable glycemic controls, as seen in Figure 8. However, CGM sensor versus point-of-care blood glucose meter agreement was not clearly associated with abnormal physiological parameters, and further accuracy studies are needed. Diabetes Technology Society recently published a consensus statement for CGM use in the hospital, which can be used as a guide. 32
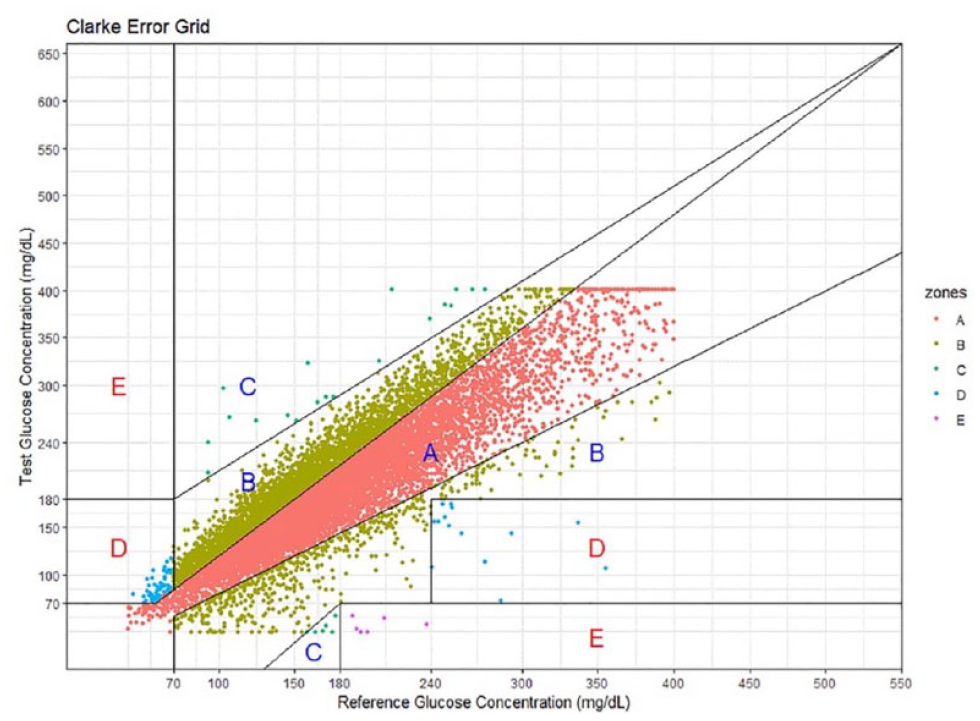
A Clarke error grid analysis of continuous glucose monitor (test) and point-of-care (reference) data pairs collected from patients with type 1 diabetes and type 2 diabetes in a non-intensive care unit hospital setting. Source: Figure reproduced from Spierling Bagsic et al. 31
Session 3: Regulation of Diabetes Devices and Drugs
Alberto Gutierrez, PhD
NDA Partners LLC, Bethesda, Maryland, USA
Naomi Schwartz, MS
Medcrypt, Inc., San Diego, California, USA
Update in Digital Health Regulation
Matthew C. Diamond, MD, PhD
Center for Devices and Radiological Health, United States Food and Drug Administration, Silver Spring, Maryland, USA
The FDA, Health Canada, and the UK’s Medicines and Healthcare products Regulatory Agency have developed ten guiding principles for Good Machine Learning Practice for Medical Devices. 33 These internationally harmonized practices promote stakeholder engagement and the development of technologies using AI and ML.
In addition, five guiding principles have been identified for Predetermined Change Control Plans for ML-enabled medical devices. 34
The Digital Health Policy Navigator is an FDA tool that can be used to determine whether the software function of a device or product may be under the FDA’s oversight, based on relevant policies and guidances. 35
Medical Device Cybersecurity
Jessica Wilkerson, JD
Center for Devices and Radiological Health, United States Food and Drug Administration, Silver Spring, Maryland, USA
Cybersecurity is patient safety. If you do not have a cybersecure device, you do not have a safe device.
Cybersecurity is an enabler of interoperability, by ensuring devices can interoperate securely, reliably, and effectively.
Center for Devices and Radiological Health (CDRH) looks forward to continuing to work with stakeholders to advance medical device cybersecurity and interoperability in diabetes-related devices.
Regulatory Considerations for Comparator Glucose Technology
Deanna Bousalis, PhD
Center for Devices and Radiological Health, United States Food and Drug Administration, Silver Spring, Maryland, USA
This presentation described what a glucose comparator method is and general recommendations for selecting one.
The talk included a discussion on some characteristics of glucose comparator methods to consider when deciding which is the best for a specific study or situation.
Finally, the presentation described regulatory considerations for transitioning to use of a different glucose comparator method in post-market activities for existing diabetes devices.
Regulation of Diabetes Drugs
Alexander Fleming, MD
Kinexum, Harpers Ferry, West Virginia, USA
The FDA’s Division of Diabetes, Lipid Disorders, and Obesity may be the busiest division in Center for Drug Evaluation and Research (CDER)—why, what, who, and how.
Center for Biologic Evaluation and Research (CBER) is busy with T1D therapies—what, how, when.
Combination products for diabetes and obesity are heating up—what and why.
While the term
Eight elements of a decentralized clinical trial.
Digital health technologies and CGMs will increasingly be used to support studies for detecting and intervening against prediabetes and undiagnosed T2D. Center for Biologic Evaluation and Research has recently approved processed human islets as the first biologic therapeutic product for people with T1D. Center for Biologic Evaluation and Research is also overseeing the investigation of manufactured insulin-secreting cells, such as that of Vertex. Center for Drug Evaluation and Research has been evaluating ways to delay the onset of T1D—the FDA approved the first drug with this indication in November of 2022, with a breakthrough designation. 37 Glucagon-like peptide-1 receptor agonists (GLP1-RAs) and related peptide drug products for diabetes and obesity comprise the hottest of all therapeutic areas. Tirzepatide and semaglutide are largely responsible for making Lilly and Novo the two largest market cap pharma companies, worldwide.
Session 4: Pump Technology
Robert A. Gabbay, MD, PhD
American Diabetes Association, Arlington, Virginia, USA
Douglas Muchmore, MD
La Jolla Endo, LLC, La Jolla, California, USA
Advances in Infusion Sets
Sarnath Chattaraj, PhD
Medtronic Diabetes, Northridge, California, USA
Insulin infusion using continuous subcutaneous insulin infusion (CSII): Insulin-induced chronic inflammation is an important target for lengthening the duration of infusion set wear: loss of preservative and insulin aggregation are major contributors in causing insulin-induced chronic inflammation.
Current state of infusion sets and SubQ insulin infusion: With the launch of Medtronic Extended Infusion Set, it appears that researchers have now solved a long-standing challenge of extending insulin infusion set wear duration, thereby reducing user burden of CSII.
Future research: Where do we go next with SubQ insulin infusion? The presentation was focused on the future prospects of SubQ infusion via infusion sets and comparable devices like infusion-CGM combo devices, patch pumps, and so on.
Advances in Infusion Set Needles and Cannulas
Jeffrey I Joseph, DO
Jefferson Artificial Pancreas Center, Sidney Kimmel Medical College of Thomas Jefferson University, Philadelphia, Pennsylvania, USA
Insertion and maintenance of an infusion set cannula damages epithelial cells, adipose cells, connective tissue, capillaries, and lymphatic vessels, leading to a surrounding layer of debris and acute inflammatory tissue.
A soft and flexible cannula inserted at an angle significantly decreases the thickness, density, and continuity of the surrounding layer of inflammatory tissue.
Controlled release of anti-inflammatory medications from the cannula surface into the adjacent adipose tissue may enhance the dose-to-dose precision of insulin absorption, extend the duration of wear time beyond seven days, and minimize site-loss due to avascular fibrous tissue formation.
Combining Glucose Monitoring and Insulin Infusion in an Integrated Device: Challenges and Solutions
Lutz Heinemann, PhD
Science Consulting in Diabetes, Düsseldorf, Germany
Measuring glucose levels in the subcutaneous tissue and applying insulin at the same site would reduce handling efforts for people with diabetes.
A number of questions are associated with such a single-site approach.
Different considerations for such an approach will be presented.
Determining Flow Rate From an Insulin Pump
Ralph Ziegler, MD
Diabetes Clinic for Children and Adolescents, Muenster, Germany
Accuracy of insulin delivery is different in different insulin pump models.
Especially at low flow rates in insulin pumps, accuracy is less, in basal rates as well as in boluses.
Differences in accuracy of flow rates in insulin pumps may be important in the variability of glucose levels.
Last Things First—The Importance of Design for Manufacturability
Michael Schoemaker, PhD
Pharmasens AG, Biel, Switzerland
Using real-life case studies, possible reasons for the difficulties away from the technical challenges in developing patch pumps are explored.
Often, manufacturability requirements for product design are not identified and implemented early in the product development cycle.
At the end of development, a medical device (possibly even approved by the authorities) is present, but extensive redesign and possibly a new development cycle are required to be able to manufacture the product economically in the desired quantities.
To lengthen infusion set wear duration as a means of reducing patient burden, Medtronic Diabetes has focused on three infusion set elements: (1) the H-Cap intended to improve insulin stability, (2) improved extended wear tubing, and (3) improved adhesive material. These advances have reduced unexplained hyperglycemia, ketonemia, and infusion site discomfort. The new Extended Infusion Set has shown ~50% survival to seven days of wear in real-world usage data; additional technical advancements may further improve upon this performance. 38
Introduction of an infusion device into the subcutaneous space results in local inflammation, and the degree of inflammation that occurs over time is related to several factors, including sharpness of the introducer needle and flexibility of the infusion device (eg, steel needles result in far greater inflammation as compared with Teflon cannulas). Local inflammation results in increased blood flow, potentially explaining the well-recognized acceleration of insulin absorption as a function of increased infusion set wear time. However, micro-computed tomography (CT) studies show that the volume of bolus delivered is reduced by nearly 50% by day 2 of set wear. It is unclear as of now how this finding may alter pharmacokinetics and pharmacodynamics of insulin as a function of infusion set wear time. Future work is targeted to reducing local tissue reaction by redesign of infusion cannula characteristics, such as making the cannula very soft and flexible.
Combining glucose monitoring and insulin infusion has obvious appeal, but several challenges must be addressed. There are two basic approaches: single-port design vs dual proximate ports, as illustrated in Figure 10. One issue to resolve is the question of whether local insulin action affects the accuracy of glucose assessment. However, studies have shown that this effect on local glucose levels is smaller than expected. Another issue is whether the local dilution of ISF due to insulin infusion will alter local glucose levels. This effect can be mitigated by several strategies, including using predictive algorithms or administering an extended bolus. A third challenge is whether insulin serves as an interfering substance in glucose sensor electrodes; however, various studies have shown that either no artifacts appear or that the artifacts can be eliminated. In conclusion, combining glucose monitoring and insulin infusion into a single device appears to be feasible.
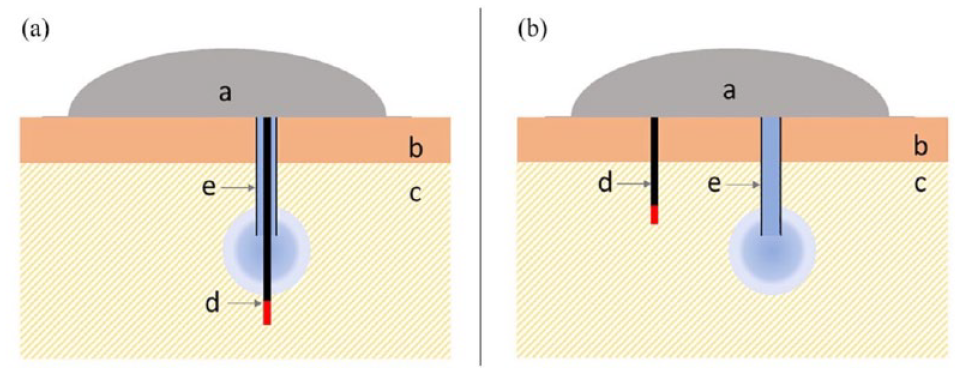
Two approaches to combining glucose monitoring and insulin infusion at the same body site through (a) a single-port and (b) a dual-port device. Labels: a, integrated body-worn device; b, dermis; c, subcutaneous tissue; d, glucose sensor with glucose sensitive tip (red); e, insulin catheter. Source: Figure reproduced from Schoemaker et al. 39
Accuracy of insulin basal rate and bolus delivery is clinically critical, especially in the pediatric setting but also in AID systems that rely on delivery of micro-boluses. Testing of a variety of pumps and infusion sets showed a high degree of accuracy for a ten-unit bolus. However, a one-unit bolus showed greater variability, with some systems exhibiting considerable variability and bias. Patch pumps appeared to have greater variability in delivery as compared with tubing-based pumps. Similar results were seen in basal rate variability. Automated insulin delivery systems may be able to account for some of the variability in pump performance, but if bolus accuracy is not adequate, then the AID system may have difficulty in “catching up” with dosing misadventures.
The FDA’s Design Control Guidance for Medical Device Manufacturers 40 provides a well-articulated and logical framework for device development. However, this guidance does not address concerns regarding manufacturability (ie, production costs) and profitability (which is heavily dependent on forecasted market size and pricing opportunities) that device developers face. Estimates of manufacturing costs and market size must be factored in at the earliest stages of product development (along with, of course, user requirements) as developers move through the design development, validation, and verification processes. A sad example of how this can go badly was given for the Calibra Medical Finesse bolus patch pump, which began development in 2004, achieved FDA clearance in 2010, and, after two different corporate acquisitions, was finally launched in 2021 (having undergone many additional design integrations) and achieved market penetration of only ~1000 users by 2022.
Session 5: Blood Glucose Monitoring Technology
Hubert W. Vesper, PhD
Centers for Disease Control and Prevention, Atlanta, Georgia, USA
Halis K. Aktürk, MD
Barbara Davis Center for Diabetes, University of Colorado, Aurora, Colorado, USA
Reference Comparators for BG Sensor Testing
Timothy S. Bailey, MD, FACE, CPI
AMCR Institute, Escondido, California, USA
The current standard for glucose comparator is the YSI 2300.
This device is no longer manufactured and support for it is being phased out, so that investigators and device companies need to find alternatives for glucose comparator measurement. 41
There are a number of alternatives in the marketplace; these will be discussed as to their benefits and drawbacks.
Interfering Substances
Andreas Pfützner, MD, PhD
Pfützner Science and Health Institute, Mainz, Germany
Little is known about the potential impact of interfering substances on CGM technologies.
We have developed an in vitro test system for standardized dynamic interference testing for CGM devices.
The interfering substances that we found in our in vitro evaluations with commercially available needle sensors should be tested for clinical relevance in clinical trials.
Platforms to Present Glucose Data
Mark A. Clements, MD, PhD, CPI, FAAP
Children’s Mercy Hospitals & Clinics, Kansas City, Missouri, USA
The learner will understand the efforts to create standardization in the display of glucose data.
The learner will understand the primary needs addressed by the visual display of glucose information for individuals and populations.
The learner will understand how glucose data can enable population health management and clinical decision support.
The Role of BGM in Today’s World
Yong Mong Bee, MBBS, MRCP(UK), FRCP (Edin)
Department of Endocrinology, Singapore General Hospital, Singapore
Self-monitoring of blood glucose continues to play an integral part in diabetes management in resource-constrained settings despite the introduction of CGMs.
Structured SMBG is the key to unlocking the benefits of BGMs.
To achieve optimal diabetes management, self-management education on SMBG and a closed-loop system with regular physician feedback should be implemented.
The FDA requires accuracy testing for CGM devices. This accuracy test is done in children and adults with diabetes by inducing hyperglycemia and hypoglycemia and comparing CGM data with the gold standard YSI 2300. However, this device is no longer manufactured, and different alternatives to use as gold standards in CGM accuracy clinical trials are discussed. In a 2021 study, SUPER GL compact was compared with YSI 2300 STAT Plus, and SUPER GL’s performance was found to be comparable with YSI 2300. 42 YSI gives two values per sample with two independent electrodes, and it requires calibration. Users should be trained, as poor sampling technique and diluted samples may affect results. In addition, older YSI devices may also affect the accuracy of the results. 43
Substance interference testing is an important part of BGM and CGM testing before marketing, and there are guidelines published by regulatory agencies, such as the FDA and European Union European Medicines Agency. Substance interference may have major implications on the safety of diabetes technologies, such as CGMs and AID systems. In vitro test systems are developed using about 70 potentially interfering substances, such as carbohydrates, sugar alcohols, nutritional compounds, drugs, and endogenous substances. Acetaminophen, vitamin C, and hydroxyurea are known substances that interfere with some of the currently FDA-approved CGMs. 44 Further clinical studies are required to investigate whether in vitro substance interference has any clinical correlation.
There are several platforms to display glucose data, including manufacturer, aggregated, and EHR software. The flow of glucose data between different platforms is shown in Figure 11. These platforms supply different features for patients and clinicians for clinical decision-making. The AGP is suggested for standardizing the analysis and presentation of glucose monitoring data. 46 Tidepool and Glooko are commonly used aggregator software packages. Electronic health record integration is an essential part in health care, and several initiatives are established for guidance and standardization of this integration.18,47 There are technology integration barriers, such as data sourcing, account linkage, data fidelity, data exchange, data storage, workflows, and governance.
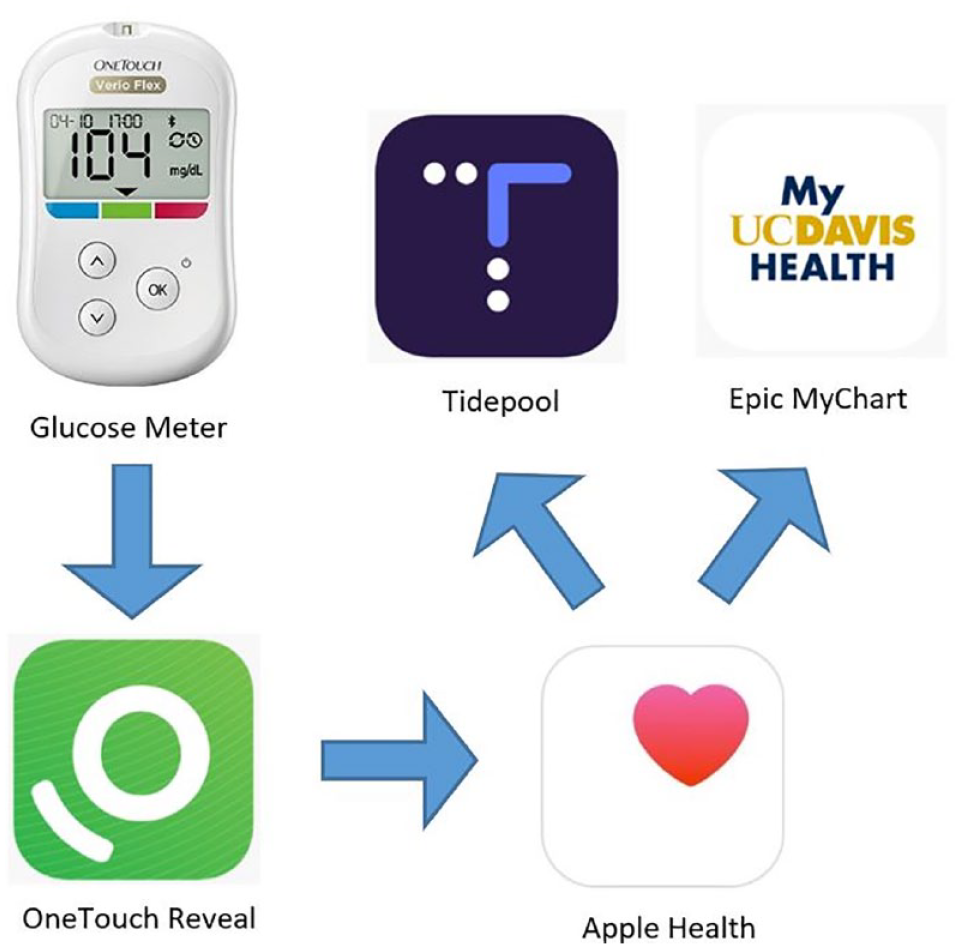
Blood glucose data can be transferred from the glucose meter via Bluetooth, mobile health apps, and the cloud and be viewed in aggregator software and the electronic health record. Source: Figure reproduced from Crossen et al 45 (https://diabetes.jmir.org/2022/1/e33639) under the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/).
Self-monitoring of blood glucose with glucose meters is still the most common form of diabetes management in the world despite the advances in CGMs. High cost, low insurance coverage, and inadequate diabetes health services are major reasons that limit CGM use in developing countries. Self-monitoring of blood glucose was used in the mainstay study, DCCT, 48 and frequent monitoring of blood glucose levels has been shown to improve glycemic outcomes. Structured SMBG was found to be superior to unstructured SMBG in patients with T2D. 49 Another study found that telemonitoring and titration of insulin using a connected glucose meter resulted in significant improvements in glycemia in patients with T2D. 50
Session 6: Emerging AID Technology
Jeffrey I Joseph, DO
Jefferson Artificial Pancreas Center, Sidney Kimmel Medical College of Thomas Jefferson University, Philadelphia, Pennsylvania, USA
Carol J. Levy, MD, CDCES
Division of Endocrinology, Diabetes, and Metabolism, Icahn School of Medicine at Mount Sinai, New York City, New York, USA
Bihormonal Systems: Automating Both Insulin and Glucagon Delivery
Steven J. Russell, MD, PhD
Massachusetts General Hospital Diabetes Research Center, Boston, Massachusetts, USA
Coordinating automated delivery of insulin and glucagon allows more aggressive insulin delivery without increasing hypoglycemia.
Bihormonal systems are able to achieve better glycemic control than insulin-only systems while also reducing the need for oral carbohydrates to prevent or treat hypoglycemia.
Stable glucagon formulations and/or analogs make bihormonal systems practical and commercially scalable.
The Loop Algorithm
Rayhan A. Lal, MD
Stanford University, Stanford, California, USA
Current open-source AID algorithms offer transparency and are important for autonomy.
Open-source AID systems allow personalization through direct control of system aggressiveness.
Open-source AID systems provide interoperability and equity.
AID Systems. Are We There Yet? A Clinician’s Perspective
David N. O’Neal, MD, FRACP, FRCP (Edin)
University of Melbourne, Melbourne, VIC, Australia
Of the current commercially available AID systems, pertinent clinical gaps include meals, exercise, and user acceptance.
Potential avenues for refining AID systems to address these gaps may include optimizing the form factor to minimize intrusiveness and burden on the user; minimizing the need for any user input to reach glucose targets; and increasing cost-effectiveness to maximize access.
The ideal system from the perspective of those at the coal face may be a patch pump incorporating a single insertion for multianalyte sensing combined with a cannula delivering insulin to facilitate full closed-loop functionality.
Improving the Performance of an AID System With Sodium-Glucose Cotransporter 2 Inhibitor
Ahmad Haidar, PhD
McGill University, Quebec, Canada
Sodium-glucose cotransporter 2 inhibitor (SGLT2i) studies in T1D have been conducted.
Two studies assessing empagliflozin with AID systems are appropriate to discuss at this meeting.
Highlights of a study assessing carbohydrate counting and empagliflozin in an AID system will be presented.
Personal Patient Experience With Loop—Patient 1
Subjective user experience—including design and great platform integrations—is a critical motivator for users of Loop. 51
Loop offers tools for temporary conditions (illness, menstrual cycles, etc) absent from commercial systems.
For a data curious individual, the algorithm transparency of open-source systems builds refreshing trust.
Personal Patient Experience With Loop—Patient 2
I believe that any closed-loop insulin delivery system can be adjusted to help someone control insulin dosing and glucose levels with less mental burden and better sleep than manual-only control.
I have direct personal experience with Do-It-Yourself (DIY) Loop 51 and the Open Artificial Pancreas System (OpenAPS) project, 52 although I have mentored friends who use commercial alternatives.
I prefer DIY because the underlying algorithm is open for anyone to study and understand; I can make contributions to improvements in the algorithm, user interface, and documentation; I can control the app using my phone or my watch; and I can select a target range and safety level that keeps me within my preferred glycemic range most of the time.
While multiple commercial AID algorithms are available for patient use within the United States and internationally, opportunities still exist to further improve glycemic control and user experiences. Four speakers reported updates on system features and research findings. This was followed by presentations by two individuals with T1D discussing their decision to use a DIY system.
Dr Steven J. Russell presented data on the use of bihormonal systems for people with T1D. He reviewed the challenges with dysfunctional alpha cells associated with insulin excess increasing the risk of hypoglycemia. He discussed the impact of micro-bolus glucagon delivery with the bionic pancreas and other systems. He noted that a bihormonal system enables more aggressive insulin delivery due to glucagon, calling it a “powerful lever” to improve TIR. System use considerations due to more stable glucagon were reviewed.
Dr Rayan Lal discussed open-source AID systems. Open APS algorithms make four predictions about the future based on insulin delivery and entry of meals. Specific system features exist, including glucose momentum and retrospective correction of meal doses based on the nature of foods. Data from a patient who used an adjunctive off-label GLP1-RA with the loop algorithm to reduce the need for meal bolusing was shared. He stressed that access challenges exist for some groups and that open-source systems may reduce those disparities. In one study, shown in Figure 12, HbA1c and TIR improved in people with diabetes after an open-source AID system was implemented. 53
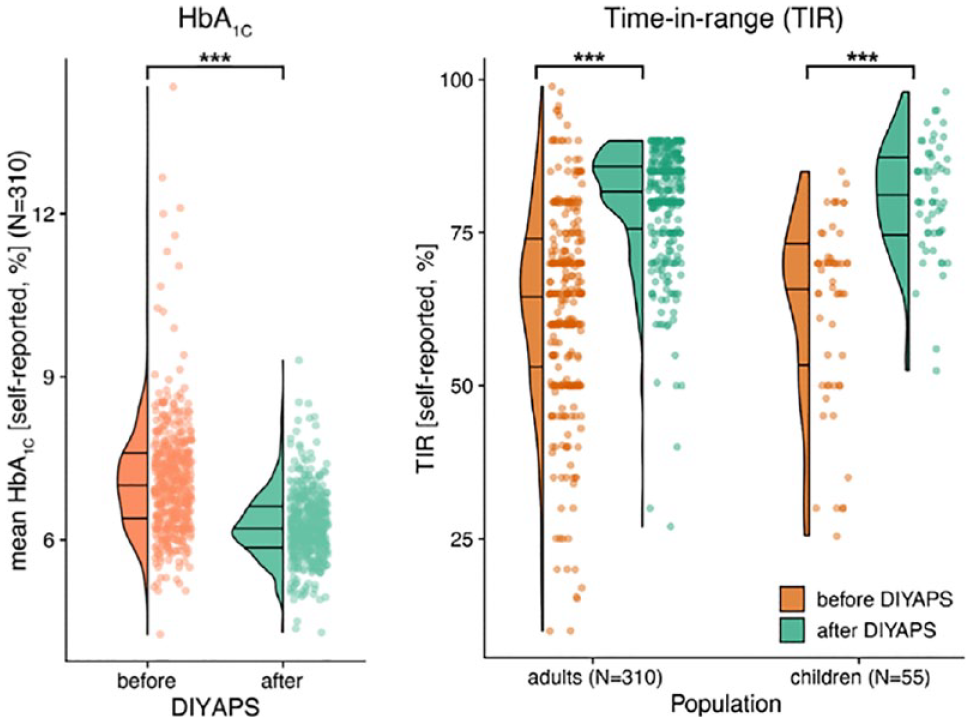
Self-reported hemoglobin A1C and time in range of adults and children with diabetes, before and after an open-source automated insulin delivery system was implemented. Abbreviations: DIYAPS, do-it-yourself artificial pancreas system; HbA1C, hemoglobin A1c. Source: Figure reproduced from Braune et al 53 (https://www.jmir.org/2021/6/e25409) under the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/).
Dr David N. O’Neil followed with a review of AID systems: a clinician’s perspective. While significant improvements over time have occurred, there are continued limitations, including whether a goal of TIR of 70% for most people with diabetes is adequate, challenges around exercise planning, and pre-meal bolusing. Dr O’Neil shared his “wish list.” Suggestions included the need for fully closed-loop systems, higher and tighter TIR, form factor considerations, ease of device wear, robustness across all clinical environments, and accessibility for all, as well as other features.
Dr Ahmad Haidar then presented research evaluating the off-label use of SGLT2i’s to improve AID system performance, while balancing this with the risk of euglycemic diabetic ketoacidosis. Preliminary data from a small two-week crossover trial of empagliflozin 2.5 mg showed increased TIR without increase in ketosis; a longer duration multisite parallel design trial is now underway. Prior to closing, Dr Haidar briefly touched on two new projects underway using semaglutide in commercially available AID systems. One of these will evaluate whether semaglutide can help simplify meal dosing.
The last two speakers were individuals with T1D who shared their experiences using the DIY Loop system. Both eloquently presented the reasons they use non-commercial systems and their hopes for the future.
Session 7: Drugs for Diabetes
Mohammed E. Al-Sofiani, MBBS, MSc
King Saud University, Riyadh, Saudi Arabia / Johns Hopkins University, Baltimore, Maryland, USA
Signe Schmidt, MD, PhD
Steno Diabetes Center Copenhagen, Herlev, Denmark
Gold Student Award Winner: DR-CIB: An Algorithm for the Preventive Administration of Corrective Insulin Boluses in T1D Based on Dynamic Risk Concept and Patient-Specific Timing
Elisa Pellizzari, MSc
University of Padova, Padova, Italy
Tirzepatide/Semaglutide for Obesity and T2D
Eric Zijlstra, PhD
Profil, Neuss, Germany
The latest incretin-based medicines provide improvements in glucose control and weight loss unmatched by any other pharmaceutical therapy for diabetes and obesity. Weight loss of 15% or more as seen with these medicines can have a disease-modifying effect in people with T2D.
Greater physiological responses (improvements in beta-cell function, insulin sensitivity, or glucagon suppression) with tirzepatide probably explain observed greater improvements in glucose control versus semaglutide in phase 2 and 3 clinical trials.
Phase 2 data from retatrutide, a triple agonist in clinical development, show even greater and more robust body weight-lowering effects.
Weekly Insulin for T1D and T2D
Ronald M. Goldenberg, MD, FRCPC, FACE
LMC Diabetes & Endocrinology, Concord, Ontario, Canada
The two weekly basal insulins in development, insulin icodec and insulin efsitora alfa, should help address insulin therapeutic inertia in T2D and improve adherence to basal insulin.
Clinical trials in T2D with weekly basal insulin demonstrate good glycemic efficacy compared with daily basal insulin comparators with relatively low rates of hypoglycemia.
Once-weekly insulins will likely be available by 2024 to 2025 and will probably be used clinically in T2D as basal insulin initiation or as a switch from daily basal insulin.
Inhaled Insulin
Alfonso Galderisi, MD, PhD
Yale University, New Haven, Connecticut, USA
Subcutaneous insulin delivery still exposes people living with T1D to inadequate post-meal glucose control and peripheral non-physiologic hyperinsulinemia. Alternative insulin delivery routes, such as the inhalation route, could provide a faster insulin delivery and potentially reduce the total daily insulin dose. Human regular insulin adsorbed onto Technosphere microparticles can be safely delivered to the lungs with a breath-powered inhaler and demonstrates faster absorption with respect to subcutaneous rapid-acting insulin analogues with a time of maximum plasma concentration ~8 minutes compared with ~50 minutes for insulin lispro.
In adults with T1D using MDIs, inhaled insulin has been proven to be non-inferior to rapid-acting analogues with respect to glycemic control after six months of real-life use.
Evidence from a meal study during hybrid closed-loop treatment supports the non-inferiority of inhaled insulin for meal glucose control and its ability to reduce early glucose peaks. Inhaled insulin holds the potential to ease diabetes management in people using MDIs or hybrid closed-loop systems.
Tzield/Teplizumab—Why and for Whom?
Stephen E. Gitelman, MD
Division of Endocrinology, Department of Pediatrics, University of California, San Francisco, San Francisco, California, USA
Teplizumab is an anti-CD3 monoclonal antibody that targets T cells, delivering a partial agonist signal and thereby inhibiting autoimmune attack on beta cells.
Teplizumab has received FDA approval for treatment of individuals at stage 2 risk for T1D (two or more autoantibodies, dysglycemic response on oral glucose tolerance test, HbA1C = 5.7%-6.4% or 10% or greater increase in HbA1C even in the normal range) to delay progression to clinical diabetes (stage 3).
A recently completed phase 3 study for children with new onset T1D with teplizumab was successful. Next steps with teplizumab in T1D include: determine those who are most likely to respond to therapy; determine means to extend the durability of the drug response, including repeated courses of teplizumab or combination with other agents; and evaluate responses in younger children.
Only half of the people with T2D and as little as one fifth of people with T1D achieve the recommended glycemic goals.2,54 These numbers document an urgent need for more treatment options to close this therapeutic gap and meet individual needs and preferences.
Incretin-based therapies offer glycemic, weight-related, and many other benefits for people with T2D. Semaglutide, a GLP1-RA for daily oral or once-weekly subcutaneous administration, has been available for treatment of T2D since 2017. 56 More recently, the gastric inhibitory polypeptide/GLP1-RA tirzepatide for once-weekly subcutaneous administration was approved, and the benefits of the dual agonist therapy are summarized in Figure 13. 57 The list of incretin-based therapies seems to continue growing, with other dual agonists and even triple agonists currently under development.
A summary of the benefits of dual glucagon-like peptide-1 receptor agonist and glucose-dependent insulinotropic polypeptide receptor agonist therapy in different organ systems in type 2 diabetes. Source: Figure reproduced from Samms et al 55 (https://www.sciencedirect.com/science/article/pii/S1043276020300485) under the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/).
People with T2D and T1D may soon be offered basal insulins for once-weekly subcutaneous administration. Two drugs are expected to reach market in 2024 to 2025, that is, insulin icodec and insulin efsitora alfa.58,59 The reduction from seven to just a single basal insulin administration per week is thought to increase therapy adherence and reduce therapeutic inertia.
For people with diabetes on basal-bolus therapy, inhaled rapid-acting insulin is available. 60 Compared with subcutaneous administration, the inhalation of insulin provides a faster onset and a shorter duration of insulin action. This pharmacodynamic profile is a better match for the rise in blood glucose after most carbohydrate-rich meals.
A new algorithm for insulin bolus advice for people with T1D is being proposed for inclusion in a decision support system. Based on dynamic risk measure and personalized timing of bolus administration, the algorithm suggests post-prandial corrective insulin doses. In simulation studies, the new dosing approach is shown to be effective and safe.
The above-mentioned therapies target hyperglycemia in people already diagnosed with diabetes. Teplizumab, on the other hand, is indicated to delay the onset of stage 3 T1D. 61 The anti-CD3 monoclonal antibody targets the T cell receptor and inhibits the immune attack on beta cells without causing unacceptable side effects. Recently, it has been shown that teplizumab may also preserve beta-cell function in children and adolescents with newly diagnosed T1D.
Session 8: AI and ML
Boris Kovatchev, PhD
University of Virginia, Charlottesville, Virginia, USA
Riccardo Bellazzi, PhD
Università di Pavia, Pavia, Italy
Bridge to AI
Xujing Wang, PhD
Division of Diabetes, Endocrinology, & Metabolic Diseases, National Institute of Diabetes and Digestive and Kidney Diseases, National Institutes of Health, Bethesda, Maryland, USA
Diabetes research and care are increasingly data centric.
Great opportunities exist to accelerate discovery, innovation, and translation in diabetes research leveraging the latest advancements in data science, including those in AI.
There are also significant gaps and challenges when leveraging AI, such as in integrating large multimodal data types, in accessing and integrating data from different diabetes technology platforms, and in AI ethics.
Consensus Guidelines for Using AI and ML to Improve Glucose Management in Diabetes: Best Practices, Pitfalls, and Opportunities
Peter G. Jacobs, PhD
Oregon Health & Science University, Portland, Oregon, USA
In this presentation, I will review best practices and pitfalls that can arise when using ML within applications in diabetes as determined through consensus by experts in the field.
Data sets now available in ML will be reviewed along with best practices on feature representation from these data sources and use of data sheets to fully describe data used to train and validate algorithms.
I will cover approaches at how ML can be used within AID and decision support for glucose forecasting, meal estimation, exercise detection, and hypoglycemia forecasting along with best-practice methods for presenting results.
Uncertainty-Aware Nocturnal Hypoglycemia Prediction in People With T1D on MDIs
Clara Mosquera-Lopez, PhD
Oregon Health & Science University, Portland, Oregon, USA
The uncertainty-aware overnight low glucose prediction model developed using CGMs, physical activity, and demographic data achieved an area under the receiver-operating characteristic curve of 0.8 and 0.71 when predicting nocturnal hypoglycemia during 0 to 4 and 4 to 8 hours after bedtime, respectively.
Predicted probability of nocturnal hypoglycemia was higher following physically active days.
A prediction-based bedtime snack intervention reduced probability and duration of nocturnal hypoglycemia in silico.
AI for Treating Diabetes
Moshe Phillip, MD
Schneider Children’s Medical Center of Israel, Petah Tikva, and Faculty of Medicine, Tel-Aviv University, Tel-Aviv, Israel
Artificial intelligence for treating people with diabetes is here to stay.
Artificial intelligence improves clinical outcomes and reduces the burden of patients and HCPs.
Artificial intelligence standardizes the workflow across providers and clinics.
Artificial intelligence and ML are key technologies of our times. Medicine is one of the most important fields for their application, and the near future will probably see a burst of AI-enabled medical devices that are likely to have a strong impact on biomedical research and clinical practice. Diabetes is one of the clinical domains where AI has been applied for decades, well before the rise of “big-data-driven” approaches and generative models.
The AI session of the 2023 DTM thus focused on results achieved so far and on future perspectives of AI in diabetes. Improvements in diabetes therapies from advancements in connected devices, AI/ML, and computing power are shown in Figure 14.
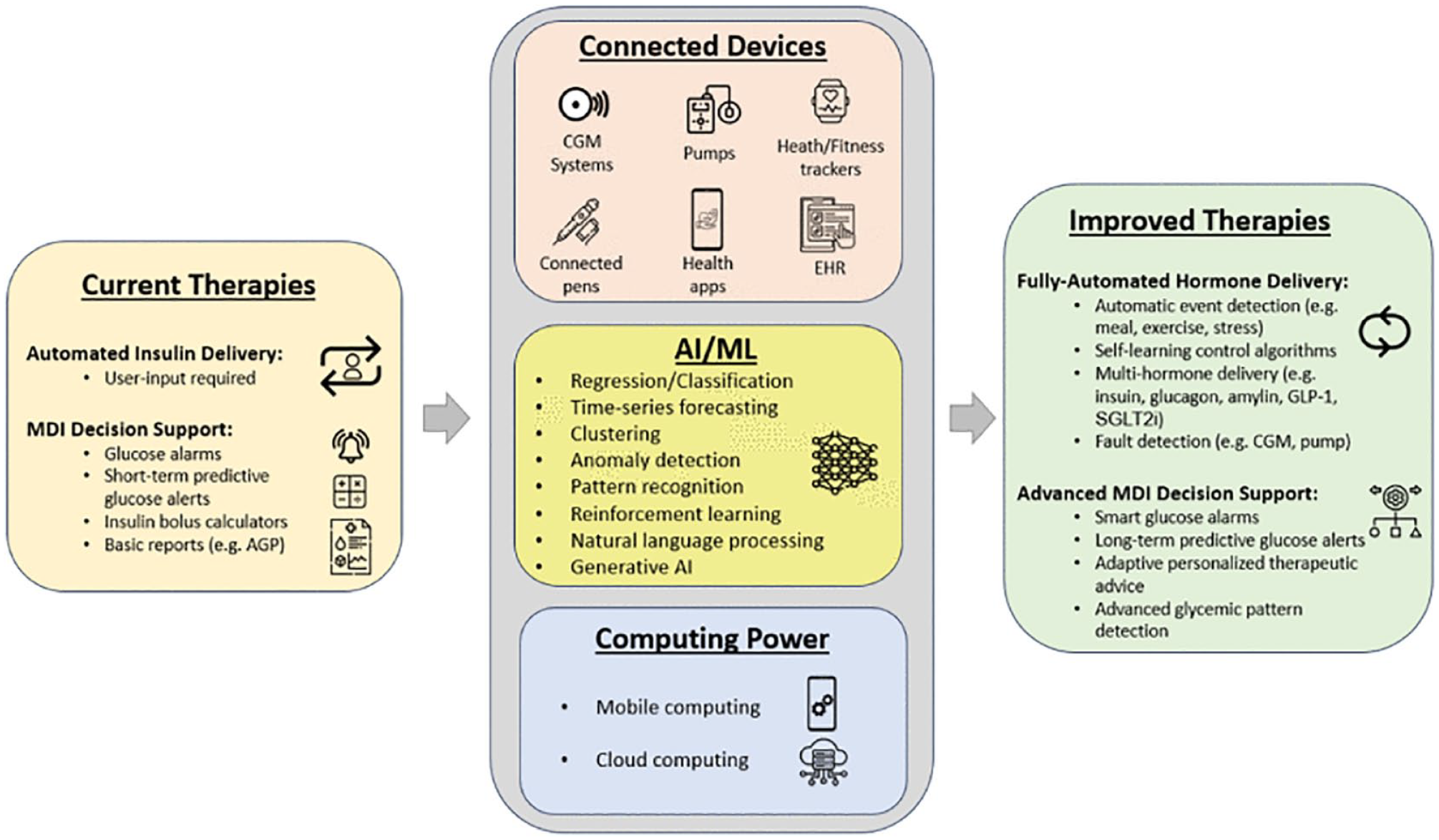
Advancements in connected devices, artificial intelligence, and computing power promote the development of improved therapies for diabetes treatments. Source: Figure reproduced from Jacobs et al 62 under the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). Abbreviations: AGP, ambulatory glucose profile; AI, artificial intelligence; CGM, continuous glucose monitor; GLP-1, glucagon-like peptide 1; EHR, electronic health record; MDI, multiple daily injection; ML, machine learning; SGLT2i, sodium-glucose cotransporter 2 inhibitor.
Dr Xujing Wang brought the vision of the National Institutes of Health (NIH) and the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK). As described by the October FDA report on AI-enabled and ML-enabled medical devices, 34 four types of applications appear to be most frequently implemented in diabetes: automated retinal screening, clinical decision support, predictive population risk stratification, and patient self-management tools. Dr Wang has highlighted several limitations of current AI technologies in medicine, including human factors, technological factors, and data quantity and quality, which the NIH and the NIDDK would like to overcome by financing specific programs and projects. For example, the NIH has recently started the BRIDGE2AI program, which has funded several AI-ready data generation projects, including the T2D Salutogenesis Data Generation Project called Artificial Intelligence Ready and Equitable Atlas (AI-READI). 63 Moreover, the NIDDK is renovating its information networks and knowledge bases to better leverage AI.
Dr Peter Jacobs talked about a recently published paper 62 that reported a consensus guideline for designing and implementing AI and ML systems to improve glucose management in diabetes. This guideline is based on 38 best practices and 23 pitfalls identified in the literature when applying AI and ML in diabetes. In his talk, he described all steps that led to the guidelines and their content, including expert consensus, data sets (both simulated and real cases), preprocessing, features and outcomes definition, ML goals and related assessment measures.
Dr Clara Mosquera-Lopez focused on addressing the risk of unaware nocturnal hypoglycemia in individuals with T1D on MDIs. Recognizing MDI patients’ heightened vulnerability, the talk showed how to combine long-term prediction and prevention through smart-snack interventions, considering prediction uncertainties. The study applied a novel AI algorithm, called Evidential Neural Network, to forecast the probability of hypoglycemia within an interval of 8 hours overnight. 64
Dr Moshe Phillip described the successful trajectory of his research in implementing an AI-based decision support system for diabetes management, comprising support to both clinicians and patients in optimizing insulin therapy. 22 In particular, a non-inferiority trial demonstrated that the frequent insulin dose adjustments suggested by a decision support system had the same quality and safety as those provided by clinical experts. 65 Dr Phillip has also highlighted that, in real-world settings, AI-based systems are likely to be adopted if more than just glucose outcomes are considered, including reducing providers’ burden and standardizing workflow among providers.
Session 9: Novel Analytes for Diabetes
Guillermo Arreaza-Rubín, MD
Division of Diabetes, Endocrinology, and Metabolic Diseases, National Institute of Diabetes and Digestive and Kidney Diseases, National Institutes of Health, Bethesda, Maryland, USA
Alan H.B. Wu, PhD
University of California, San Francisco, San Francisco, CA, USA
Non-invasive Glucose Monitoring—When Will It Arrive?
Mark A. Arnold, PhD
The University of Iowa, Iowa City, Iowa, USA
Although it is impossible to predict when non-invasive glucose monitoring will be clinically or commercially successful, it is both possible and valuable to describe how a clinically viable non-invasive glucose sensor can be realized.
A set of fundamental principles is presented with the objective of guiding the research and investment communities toward meaningful advancements along the developmental path for successful non-invasive glucose-sensing technologies.
Two examples from the recent literature are used to highlight selected features of these guiding principles where the first corresponds to a pilot-study designed to assess non-invasive glucose measurements from PPG signals 66 and the second involves non-invasive glucose measurements using Raman scattering spectroscopy with a much larger cohort of people with diabetes. 67
Continuous Ketone Monitoring
Shridhara Alva, PhD
Abbott Diabetes Care, Alameda, California, USA
An integrated sensor that can monitor glucose and ketone continuously has demonstrated good correlation between ISF and blood concentrations.
The sensor leverages FreeStyle Libre 3 form factor and can provide alerts, warning of impending ketoacidosis.
Factory calibrated sensor shows good correlation with blood ketone in people with and without diabetes.
Biomarkers Screening for Asymptomatic Heart Failure
Ambarish Pandey, MD
UT Southwestern Medical Center, Dallas, Texas, USA
The risk of HF is increased among people with diabetes.
Cardiovascular biomarkers, such as natriuretic peptides can help identify people with diabetes who are at increased risk of HF.
Targeting high-risk patients with novel therapies, such as SGLT2i’s can help lower the risk of HF.
Novel technologies and testing for biomarkers continue to develop for diagnosis and monitoring of people with diabetes. With regards to glucose testing, the process has evolved from analysis via the central laboratory using automated instruments by qualified lab technologists, to BGMs, and now, to CGMs by caregivers and patients themselves. Each of these approaches requires the collection of blood, either through traditional venipuncture, capillary collections, or ISF analysis, respectively. The ultimate solution is a non-invasive sensor that can make glucose measurements without taking a blood sample or implanting a subcutaneous sensor. The popular approach toward a non-invasive sensor involves taking optical measurements across the skin and using an ML program to interpret results. In general, these methods involve illuminating the skin with a type of electromagnetic radiation (eg, infrared, radio waves, etc) and recording the absorption or reflection (eg, PPG or Raman light scattering) of the incident radiation to a detector. ML algorithms are commonly used to relate variations in the collected optical signals with the concentration of glucose in the skin matrix. As an example, glucose-dependent variations in non-invasive PPG measurements are presented in Figure 15.
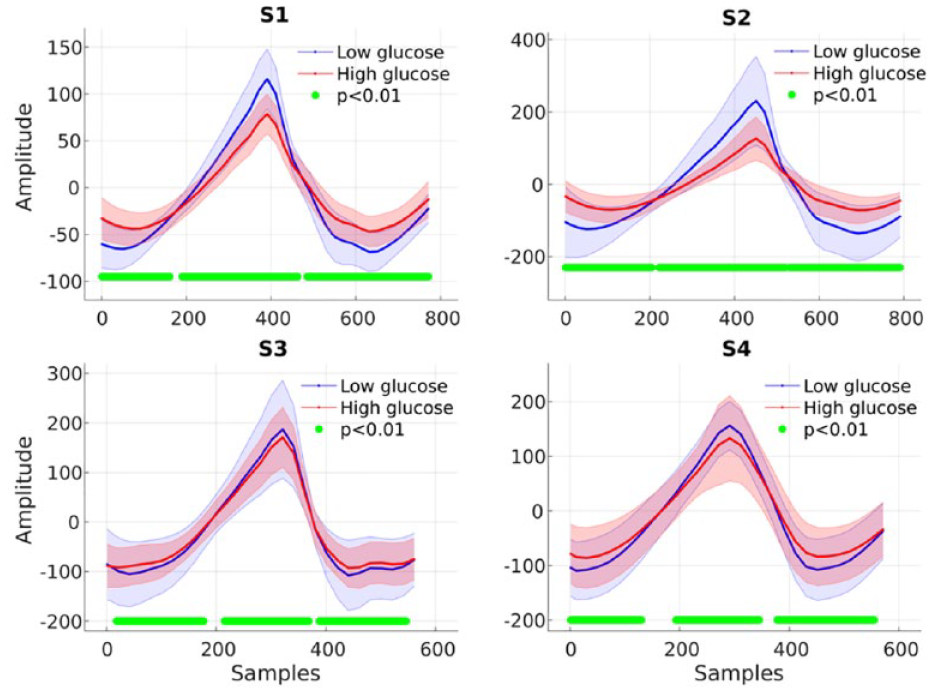
Differences in average photoplethysmography cycles during low glucose (blue) and high glucose (red) periods for four subjects (S1-S4). The photoplethysmography signal was recorded from a non-invasive in-ear sensor. Shaded areas correspond to confidence intervals, and the green bar denotes statistical significance (
In addition to glucose, sensors may be useful for other analytes relevant to diabetes monitoring, such as lactate and ketones. These tests are useful for the early detection of serious diabetic complications, such as metabolic acidosis and diabetic ketoacidosis. Individuals at high risk for ketosis include those with T1D and T2D, on insulin pumps, taking certain anti-diabetic medications, and who are on low carbohydrate ketogenic diets. Current laboratory ketone tests include urinalysis, central lab testing for measuring acetoacetic acid, and point-of-care testing for β-hydroxybutyric acid. Of the ketones that are released after acidosis, targeting the β-hydroxybutyrate provides the earliest indication of ketosis. In a study conducted by Abbott Laboratories, the non-diabetic subject was monitored using a prototype ketone sensor and monitored with standard blood testing with concordance of results between ISF and blood ketones.
Given that the presence of diabetes is a risk factor for the development of HF, there is interest in screening people with diabetes using blood-based biomarkers. B-type natriuretic peptide (BNP) and N-terminal pro B-type natriuretic peptide (NT-ProBNP) are released from the myocardium at the same time and are established tests for diagnosis and risk stratification for HF. In the STOP-HF trial, screening for HF with BNP coupled with an appropriate therapeutic intervention has produced reductions in major adverse cardiovascular events. 68 As with the initiation of any screening program, an economic analysis must be conducted to determine whether the costs to achieve improvements of clinical outcomes with appropriate intervention is less than what federal agencies and insurance carriers are willing to pay to achieve such success.
Session 10: New Technology Developments in Commercial CGMs
Nestoras Mathioudakis, MD, MHS
Johns Hopkins University School of Medicine, Baltimore, Maryland, USA
Elias K. Spanakis, MD
Baltimore VA Medical Center and School of Medicine, University of Maryland, Baltimore, Maryland, USA
Abbott Technology
Anila Bindal, MD
Abbott Diabetes Care, Alameda, California, USA
Introducing advancements in the FreeStyle Libre portfolio that integrate with currently available insulin delivery systems will provide personalized, connected solutions to improve the lives of those living with diabetes.
Our expanding partnerships with insulin pens and AID systems create integrated diabetes management solutions to give those living with diabetes options.
Continuous innovation within our portfolio includes a next-generation system designed for iCGM and ketone monitoring that is the first of its kind for those living with diabetes.
Dexcom Technology
Daniel R. Cherñavvsky, MD
Dexcom, Inc., San Diego, California, USA
At present time, CGMs are leading the disruptive technology for improving the treatment and management of diabetes, not only in T1D but also in T2D and gestational diabetes.
More recently, CGMs have been considered for monitoring metabolic health in the general population, such as weight loss. Other potential uses are being investigated in clinical research for diseases and conditions that have the potential to produce severe dysglycemia with catastrophic outcomes.
The use of CGMs should be considered in conditions where the risk of severe hypoglycemia is a threat. However, more research must be done in these areas. Diabetes is the present, and the success of CGMs and general population metabolic health is the future, without a doubt.
Medtronic Technology
Robert A. Vigersky, MD
Medtronic, Washington, D.C., USA; Uniformed Services University of the Health Sciences, Bethesda, Maryland, USA
There are multiple inputs that allow the G4S sensor to be calibration-free and non-adjunctive when in automation (SmartGuardTM) compared with the previous generation of calibration-requiring, adjunctive sensor (GS3).
The result of this change increases time in SmartGuardTM, TIR, and sensor utilization and reduces sensor alerts and sensor-mandated exits from SmartGuardTM.
The next-generation sensor is disposable and is anticipated to further enhance the performance of the MiniMedTM 780G system.
Senseonics Technology
Hari Sree, PhD
Senseonics Inc., Germantown, Maryland, USA
Senseonics’ implantable technology for diabetes management offers the only long-term high accuracy CGM system with high user acceptability.
The implantable technology roadmap enables our product vision for a one-year fully implantable system with no required on-body system components.
The technology platform provides opportunities for sensing of other relevant analytes toward diabetes management.
Pacific Diabetes Technologies
Thomas Seidl, PhD
Pacific Diabetes Technologies, Portland, Oregon, USA
SynerG is a dual function glucose sensing and insulin delivery cannula that combines glucose level measurement with safe insulin delivery.
SynerG has been shown in clinical studies to accurately measure glucose while delivering insulin with a 30-minute warm-up and a single calibration.
SynerG eliminates waste and the need for multiple device changes and will promote treatment adherence through its convenience for the user.
Important progress has been made in commercial CGM technology; CGMs discussed at the 2023 DTM are shown in Figure 16. Abbott has developed several CGM systems. 69 The FreeStyle Libre 3 is a very small, extremely accurate, and reliable CGM, providing readings every minute that are sent directly to the patients’ smartphone. FreeStyle Libre 2 has the ability, through a software application, to continuously send glucose readings to the user’s smartphone. Recently, a modified FreeStyle Libre 2 sensor (FreeStyle Libre 2 Plus) has been FDA-cleared, which has a 15-day sensor wear time. The MARD of the FreeStyle Libre 2 Plus was reported at 8.2% in both adult and pediatric patients, with 99.9% of the Consensus Error Grid values in zones A + B (94.3% in zone A). The FreeStyle Libre 3 has been authorized to work with the My Life CAM APS FX app, Tandem, and the My Life Ipso AID systems. An upcoming partnership with Omnipod is currently under development. Abbott CGMs are also integrated with the Novo Nordisk and Bigfoot Smart Insulin Pens. Overall, the FreeStyle Libre portfolio has been shown to lead to higher treatment satisfaction and high persistence. Unpublished data have shown that patients using both Abbott and GLP1-RA medications are more likely to use both products. Finally, Abbott has received an FDA breakthrough designation for integrated glucose and ketone measurement designed to continuously monitor glucose and ketone levels.

Dexcom CGMs have been shown to lower HbA1c levels, increase TIR, reduce hypoglycemia and hyperglycemia, save costs of $5000 per year, and have strong satisfaction and retention rates among CGM users. 70 Dexcom CGMs are connected with several AID systems, such as Tandem, Omnipod, Ypsomed, diabeloop, CamAPS, Tidepool, and Beta Bionics, with studies showing statistically significant improvements in TIR. Dexcom CGMs are also connected with smart insulin pens, such as Lilly’s tempo smart button, Medtronic’s Inpen, and Novo Novordisk’s Novo Pen 6 and Novo Pen echo, as well as more than 30 digital health apps. Beyond diabetes, Dexcom is exploring whether CGMs can provide nutrition guidance to patients that lead to weight loss or even to T2D reversal and whether CGMs can be used to achieve better metabolic health. Another area is utilizing CGM devices to reduce hypoglycemia in diseases other than diabetes, such as in patients with post-bariatric surgery, congenital hyperinsulinemia, and glycogen storage disease T1, as well in the management of hyperglycemia in patients with cystic fibrosis and in patients receiving steroids for leukemias and COVID-19 infection. Other areas where the use of Dexcom CGMs can be expanded are the management of hospitalized patients with diabetes or hyperglycemia as well as for patients with CKD on hemodialysis.
Medtronic has developed several CGM systems, among them the GS3, which required calibration and blood glucose for bolus insulin. Newer sensors that include the FDA-approved GS4 and the CE-marked DS5 (SimpleraTM for stand-alone use and Simplera SyncTM for use with the 780G system) do not require any calibration or blood glucose value for bolusing insulin. 71 DS5 is fully disposable, smaller, and can be easily inserted. Combining Medtronic insulin pumps with CGMs has shown that the 780G with G4S has resulted in fewer alerts (either systems or optional alerts) compared with the 780G with GS3 or the 770G. The most significant contributor to the reduction of the system sensor alerts is the substantial reduction in the calibration alerts. Optional sensor alerts, as well as system pump and optional pump alerts, are similar in frequency between 770G, 780G with GS3, and 780G with G4S. Important reductions in system SmartGuardTM alerts have also been found with the newer 780G with G4S, mainly due to the reduction in the number of glucose entries required to enter. Using blood glucose values as a comparator, unpublished data show that the MARD is in the low 9% range for the G4S system.
Senseonics has developed Eversense, which is the only long-term (up to 180 days) FDA-approved implantable CGM. 72 Eversense has many benefits, such as discreet vibration alarms, great accuracy over a six-month period, and flexibility of wear. Eversense has been shown to have very high user acceptability, among them a high reported improved confidence over diabetes control, higher motivation with diabetes management, and less day-to-day burden. The new Senseonics sensors under development are based on the ROME (Redundant Optical Measurement Electronics) platform, which, compared with the current E3 Sensor, has two distinct sensing areas with four independent optical measurement system components. Beyond the differences in the electronics, the new sensor platform has, in addition to a glucose sensing molecule, a reactive oxygen species sensing molecule. The new sensor will have an integrated battery with an updated algorithm to enable use as an intermittently scanned CGM without the need for on-body system components. Future sensors can have, instead or in addition to a glucose-sensing molecule, other substrates that can detect lactate, ketones, or other clinically relevant analytes.
Pacific Diabetes Technologies’ goal is to produce a device capable of measuring glucose and simultaneously delivering insulin through a single device, eliminating asynchronous device changes and reducing needle insertions and adhesives as waste. 73 The SynerG core technology has on the top a stainless-steel needle, which runs through the entire length of the device through a polyethylene core. On the top of the core, there is a conduced layer on which there is a glucose-sensing chemistry. The subcutaneous part of the device is 6 mm underneath the skin. To develop the above technology, several issues had to be addressed, such as the insulin formulation interference and the transient dilution artifact. A prototype has been developed, which has been evaluated using YSI studies with an overall MARD of 9%, when the Dexcom CGM achieved a MARD of 10%. It requires less than 30 minutes of warm-up and a single calibration at 15 minutes of warm-up. Consensus Error Grid values in zones A + B were 98.0% (88.6% in zone A). The next steps include seven-day clinical studies in Australia and the US, industrial design, and production lines.
Session 11: Skin Physiology
Gerald R. Sydorak, MD, FACS
Seton Medical Center, Daly City, California, USA
Bijan Najafi, PhD, MSc
Baylor College of Medicine, Houston, Texas, USA
Device-Induced Inflammation
Ulrike Klueh, PhD
Department of Biomedical Engineering, Integrative Biosciences Center, Wayne State University, Detroit, Michigan, USA
Skin mast cells initiate the inflammatory response that culminates in tissue fibrosis.
Extracellular traps form in response to insulin preservatives and other pro-inflammatory stimuli.
Device performance optimization must address the root causes of the inflammatory response.
Identifying and Preventing Lipohypertrophy
Jane Jeffrie Seley, DNP, MPH, MSN, GNP, BC-ADM, CDCES
Weill Cornell Medicine, Division of Endocrinology, Diabetes & Metabolism, New York City, New York, USA
Since incorrect insulin injection technique is common among people taking insulin, ongoing assessments of injection sites and simulated observations of injection technique is crucial to assess for lipohypertrophy (LH) and gaps in knowledge.
The consequences of errors in insulin injection technique include glucose variability, site pain and discomfort, LH, and increased risk of infection.
Education on site selection, pen needle size, avoidance of pen needle reuse, skin lift (if needed), angle and force of injection, avoidance of areas with existing LH, and formulating a personalized insulin injection site rotation plan has improved outcomes.
Effects of Infusion Pumps
Jannet Svensson, MD, PhD
Steno Diabetes Center Copenhagen, Herlev, Denmark
Lipohypertrophy is a well-known complication to subcutaneous infusion of insulin with possible implications for the function of the algorithms in systems with AID.
There seems to be a lack of awareness concerning the importance of LH among people with diabetes and HCPs.
Lipohypertrophy is a common but preventable complication to subcutaneous infusion of insulin where rotation and multiple positions for infusions are the key.
Glycemic and Geriatric Challenges: Understanding Wound Healing Dynamics in Frail Diabetic Patients
Bijan Najafi, PhD, MSc
Baylor College of Medicine, Houston, Texas, USA
Understanding the frailty-diabetic foot ulcer (DFU) relationship: The presentation emphasizes the importance of understanding the connection between frailty in older adults and non-healing DFUs to tailor treatment plans and reduce lower extremity amputation risks.
Telemedicine for early frailty identification: The talk promotes the use of telemedicine to enable HCPs to efficiently evaluate and monitor frailty, facilitating early identification of patients at risk of non-healing DFUs.
Exploring frailty and vascular health connection: The presentation highlights the critical need to examine the association between frailty and vascular health to inform patient care pathways, distinguishing between those treatable in primary clinics and those needing specialized, multidisciplinary care.
Diabetes significantly impacts skin physiology, including its protective, regulatory, and sensory functions. Comprising the epidermis, dermis, and subcutaneous layers, the skin’s key roles like cell regeneration and moisture retention are adversely affected in people with diabetes. Elevated glucose levels in people with diabetes can weaken the skin’s barrier function, heightening infection risks and slowing wound healing. This condition also leads to altered blood flow and neuropathy, causing skin dryness and reduced sensation, and increasing the risk of diabetic ulcers, particularly in the lower extremities. In addition, diabetes disrupts collagen production, impacting skin elasticity and strength, and a compromised immune system further elevates infection risks.
Session 11’s four speakers addressed various aspects of skin health in diabetes. Topics included device-induced inflammation from CGMs and insulin administration tools, overlooked problems associated with LH resulting from incorrect insulin injection techniques, the adverse effects of insulin pumps and glucose monitoring on children’s skin health, and strategies for skin care and monitoring to mitigate these issues. They also discussed how frailty in people with diabetes can lead to increased skin breakdown, resulting in DFUs and delayed wound healing.
In the first talk, Dr Ulrike Klueh addressed device-induced inflammation caused by medical devices like CGMs and insulin administration tools. She highlighted that such inflammation could result from factors like needle-induced tissue damage, materials in device sensors or catheters, and tissue distortion from liquid infusion or insulin formulations. Utilizing animal models, including a diabetic mouse air pouch model, Dr Klueh’s research showed that insulin preservatives are inflammatory both in vitro and in vivo, but removing them via Zeolite filtration reduces their adverse effects. Her findings also demonstrated that preservative-free insulin formulations decrease inflammation at the infusion site. Furthermore, Dr Klueh presented a swine model to showcase the detrimental effects of these preservatives on inflammation, emphasizing their role in inducing extracellular trap and NETosis. She underscored the importance of further research to understand the mechanisms behind tissue reactions to medical devices.
In the second presentation, Dr Jane Jeffrie Seley addressed LH, a condition characterized by increased adipose tissue due to insulin injections, often overlooked by both people with diabetes and HCPs. She highlighted the discrepancy between self-reported and clinically diagnosed LH cases and the reduced effectiveness of insulin in LH-affected areas. Dr Seley emphasized the importance of proper injection techniques, such as using short needles and correct injection sites, to prevent LH. She advocated for continued patient education through visual aids and apps, particularly focusing on older adults, to enhance awareness and management of LH. Examples of the types of errors that could lead to LH, along with the proportion of errors made by patients in one study, is shown in Figure 17. 74
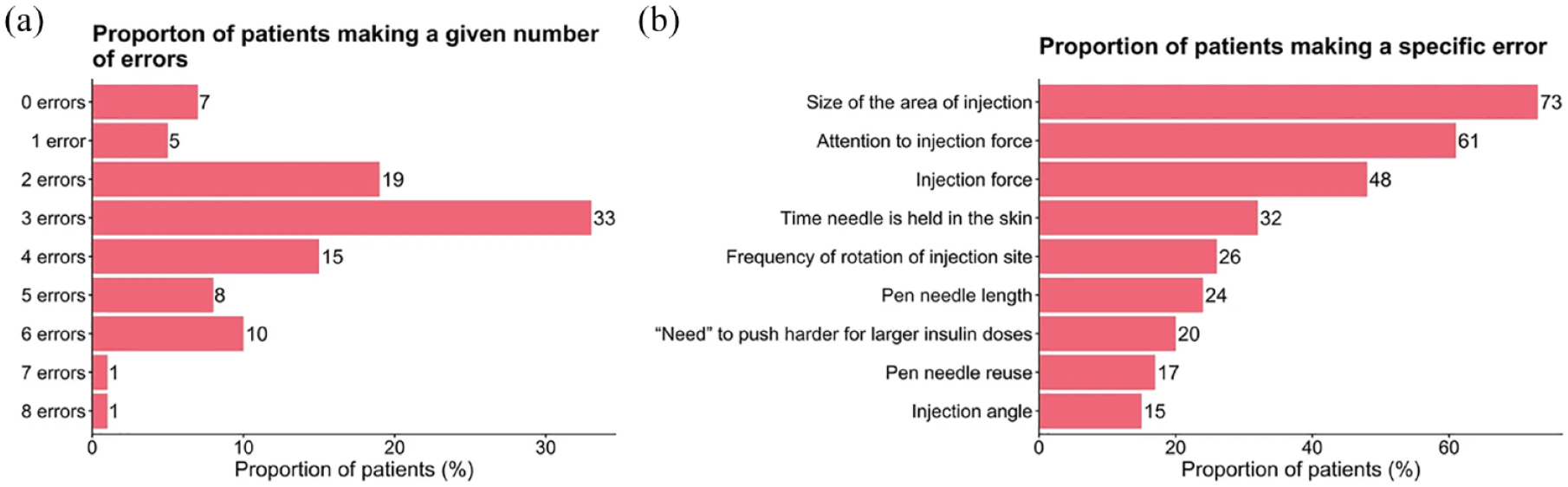
A summary of the (a) number and (b) type of errors made by 147 people across Canada with type 1 diabetes or type 2 diabetes while injecting insulin. Source: Figure reproduced from Bari et al 74 under the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/).
In the third talk, Dr Jannet Svensson discussed the impact of insulin pumps and CGMs on skin health in children with diabetes. Her study utilized ultrasound imaging to identify subcutaneous changes in a pediatric cohort using these devices, revealing that 40% developed hyperechogenicity at pump sites initially, increasing to 62% after six months, with some sites showing new vascularization. Dr Svensson also addressed skin reactions, including allergic and irritative contact dermatitis, and the importance of proper skin care, such as lipid lotion use, to mitigate risks. Echoing Dr Seley’s recommendations, she emphasized the risk of long needles causing intramuscular injections in children and the need to select injection sites with sufficient dermal depth. She concluded by stressing the ongoing issue of skin damage from infusion pumps and the value of ultrasound in both monitoring LH and guiding proper injection site selection and patient education.
In the fourth talk, Dr Bijan Najafi discussed the critical impact of frailty on skin health and wound healing in people with diabetes, particularly emphasizing its contribution to increased risks of DFUs and delayed healing. He pointed out that frailty, often associated with organ failures like cardiac or renal issues, could also lead to “skin failure,” making the skin more susceptible to breakdown and slower recovery. Dr Najafi highlighted the importance of understanding this link to devise personalized treatment strategies aimed at reducing the risk of lower extremity amputation in older adults with diabetes. He also spoke about leveraging telemedicine and AI for remote frailty assessment, as demonstrated in their study published in the Journal of Diabetes Science and Technology, where a simple elbow flexion-extension test analyzed by AI can reveal key frailty metrics, such as slowness, weakness, and exhaustion. 75 Notably, exhaustion emerged as a potential digital biomarker for hard-to-heal wounds, often related to malnutrition and poor perfusion. Dr Najafi stressed the preliminary nature of these findings and the need for further research in larger samples. He advocated for the integration of telemedicine in patient triage, differentiating between those manageable in primary care and those requiring urgent, specialized multidisciplinary care, a crucial approach for enhancing skin health and wound healing outcomes in the aging diabetic population.
Session 12: Hot Topics
Umesh Masharani, MB, BS
University of California, San Francisco, San Francisco, California, USA
James Geoffrey Chase, PhD
University of Canterbury, Christchurch, New Zealand
How Can Technology Improve Patient Adherence to Offloading?
Bijan Najafi, PhD, MSc
Baylor College of Medicine, Houston, Texas, USA
Effective treatment of DFUs heavily relies on adherence to offloading footwear and devices. However, there are notable challenges: non-removable devices face issues of complexity and patient resistance, while removable devices often suffer from low patient compliance.
The innovative “SmartBoot,” a collaborative creation by Dr Najafi and Dr David Armstrong’s team, integrates an inertial sensor for remote adherence monitoring. 76 This technology transmits data to a smartphone or smartwatch, utilizing gamification techniques to boost patient engagement, reinforce adherence to offloading, and facilitate tailored care via a specialized clinician dashboard.
Early trial outcomes suggest that the SmartBoot could significantly enhance wound healing in DFU patients, as evidenced by reduced healing times. This highlights the potential of remote patient monitoring as a transformative tool in the treatment of DFUs and as a critical component in strategies aimed at limb preservation among individuals with diabetes.
Peritoneal Insulin Delivery
Diane J. Burgess, PhD
University of Connecticut, Storrs, Connecticut, USA
Investigation of the root cause of the foreign body response (FBR) to insulin catheters.
Effect of size and shape on FBR.
Effect of anti-inflammatory drugs on FBR.
ML for Multiple Sensor Datastreams and Explainable AI
Ali Cinar, PhD
Illinois Institute of Technology, Chicago, Illinois, USA
Meal, physical activity, psychological stress, and sleep events can be detected and discriminated by applying ML to CGM and wearable device data.
The results of ML should be explainable and interpretable to HCPs and people with diabetes.
The outcomes of ML algorithms can be integrated with adaptive and personalized automatic control algorithms to provide AID that does not necessitate manual entries by the user.
Green Diabetes
Ed Krisiunas, MLS(ASCP), MPH
WNWN International, Burlington, Connecticut, USA
Post-COVID-19 has seen an incredible amount of interest in all facets of recycling with plastics being at the forefront in many parts of the world.
Some diabetes care companies have increased their effort in managing the materials/products used by their clients/patients by taking them back and recycling them into a new product.
The next goal on the horizon that is being actively pursued is the reusing of materials from patients and remanufacturing them back into the same device to be used again.
This session presented a series of exciting new advances and developments in “hot topic” areas relevant to diabetes care. Some key highlights and outcomes include: growing information delivery and care guidance via a range of software and smartphone apps; increased sensing capability, including the addition of a continuous ketone-sensing technology; increased device longevity with up to 180-day sensor lifetimes; increased co-design and interaction of glucose-sensing and insulin delivery systems; increased growth toward T2D use; and increased consideration of equity and economic costs.
For people with DFUs, novel sensors and wearable devices help to reduce the load on the ulcer. One hot new sensor is a SmartBoot, with a smart sensor, to replace irremovable and removable walkers. 76 The smart offloading system is shown in Figure 18. The SmartBoot uses Bluetooth to connect to the Internet and employs gamification, notifications, and feedback to encourage adherence. The SmartBoot also enables remote patient monitoring and data collection, allowing the HCP to provide individualized education at visits. As smart technologies continue to be developed, it is important to consider innovative ways to encourage compliance among users.

A smart offloading system for people with diabetic foot ulcers, consisting of a smart removable cast walker, a sensor, and a watch that provides the user with adherence information, notifications, and feedback. Source: Figure reproduced from Finco et al 76 under the Creative Commons Attribution license (https://creativecommons.org/licenses/by/4.0/).
Continuous intraperitoneal insulin delivery is limited by high manufacturing cost and FBR issues, with catheter obstructions occurring at a rate of > 10 per 100 patient-years. Catheter obstructions include tip blockage, tip encapsulation, and catheter encapsulation, and require surgery to address. A rodent animal model was developed to investigate the root cause of FBR-led catheter obstructions, and animals were given either diluent or insulin through a catheter. There was no difference in encapsulation between normal rats given diluent or insulin, and diabetic rats given insulin showed a slower rate of encapsulation as expected due to being immune compromised. The worst-case scenario for catheter obstructions was found to be when the catheter tip was buried under the liver. When a long-acting dexamethasone-eluting coating was added to the catheter, however, the tip blockage was eliminated and no FBR was found. Therefore, dexamethasone is promising in the development of intraperitoneal catheters for insulin delivery in people with diabetes.
Given that the effects of various events and stimuli appear in the glucose concentration with a certain time delay, basing treatment decisions and actions solely on glucose data may not be optimal. Multi-analyte sensors are those that provide information about the concentration of various analytes, including lactate, ketones, and cortisol, that could provide additional metabolic information before the change is reflected in the glucose level. Physiological/biometric variables (such as heart rate, skin temperature, and respiration rate) from wearable devices that have sufficient accuracy and that tend to be cheaper and more accessible to measure can also be used to make insulin-dosing decisions in real time. By detecting meals, exercise, sleep, and stress without these events needing to be manually entered by the user, glycemic excursions can be mitigated or prevented in the near future. Both historical data and current data can be analyzed by ML to make decisions about insulin doses proactively. However, challenges in this area include missing data, outliers, and artifacts, as well as data sovereignty and privacy. These types of ML algorithms may perform reliably in personalized medicine, where treatment decisions for the user are made with the user’s own data.
Methods for the collection and disposal of diabetes technologies exist, but recycling, reprocessing, and reusing remains a challenge. Some diabetes companies have programs that allow users to mail back used sensors for recycling and reuse. The different types of plastics used, often in the same device, however, makes recycling more complex. Ongoing research is being conducted to change product development, so that a device can be completely manufactured from the same type of plastic.
Session 13: Live Demonstration
Dorian Liepmann, PhD
University of California, Berkeley, Berkeley, California, USA
Jane Jeffrie Seley, DNP, MPH, MSN, GNP, BC-ADM, CDCES
Weill Cornell Medicine, Division of Endocrinology, Diabetes & Metabolism, New York City, New York, USA
House of Carbs—Personalized Carbohydrate Dispenser for People With Diabetes
Eirik Årsand, PhD
UiT Arctic University of Norway, Tromsø, Norway
Pietro Randine
UiT Arctic University of Norway, Tromsø, Norway
The House of Carbs is an automatic “juice machine” that delivers the user carbohydrates dependent on their current blood glucose status.
It exemplifies useful innovations that could be made given the availability of diabetes data, such as blood glucose values, insulin injections, and carbohydrate intake.
A mobile version of the House of Carbs will also be presented, demonstrating how people with T1D can manage physical activity more safely than today.
Eirik Årsand, Professor in Cyber Physical Systems and eHealth, and Pietro Randine, PhD candidate at UiT The Arctic University of Norway, Department of Computer Science & Norwegian Center for E-health Research in Tromsø, Norway, discussed their innovation, known as House of Carbs. 77 Using a CGM device with sharing possibilities via the Nightscout platform, they created an automatic “juice machine” that delivers carbohydrates in the form of juice, depending upon the current blood glucose status of the person with diabetes. A demo video showcasing the juice machine along with all its components was shown, and scenarios were discussed that reflect potential real-world applications of the innovation. To date, the juice machine has been tested on real patients for a total of 50 days. One of the main challenges for the researchers has been obtaining real-time data to inform further development of the device. Future plans include refining a portable version of the House of Carbs machine to support people with T1D during physical activity. The mobile House of Carbs is illustrated in Figure 19.
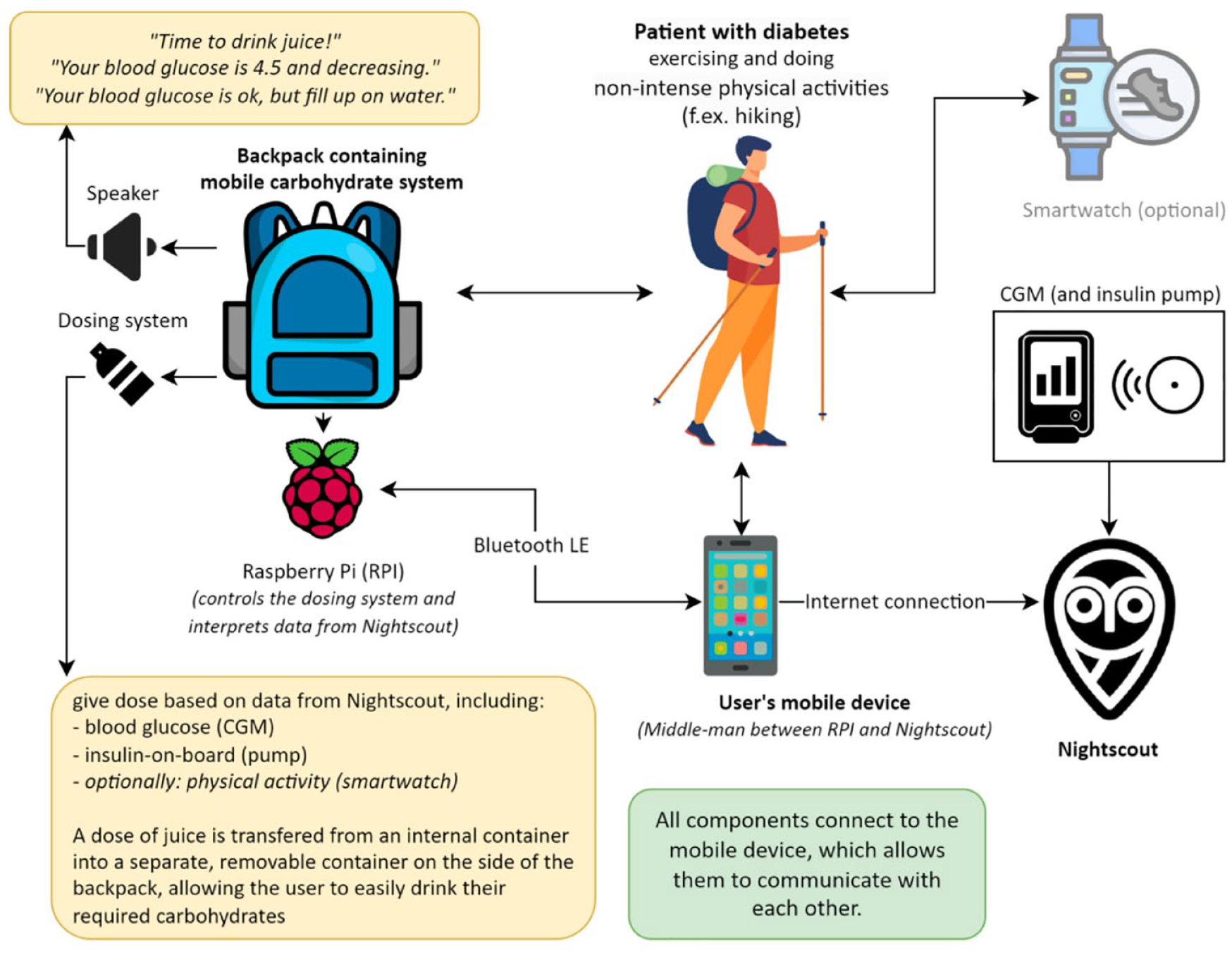
Schematic diagram of the mobile version of the House of Carbs Personalized Carbohydrate Dispenser for People with Diabetes. Source: Figure courtesy of Eirik Årsand, PhD, UiT Arctic University of Norway, Tromsø, Norway. 78
Conclusions
The DTM presented various perspectives on the current state of diabetes technology. The meeting’s presentations examined the development of diabetes technology through medical, scientific, regulatory, and engineering lenses.
Footnotes
Acknowledgements
For contributing key points to this report and reviewing the manuscript, we would like to thank David T. Ahn, MD, Mohammed E. Al-Sofiani, MBBS, MSc, Shridhara Alva, PhD, Mark A. Arnold, PhD, Guillermo Arreaza-Rubín, MD, Eirik Årsand, PhD, Timothy S. Bailey, MD, FACE, CPI, Yong Mong Bee, MBBS, MRCP (UK), FRCP (Edin), Richard M. Bergenstal, MD, Anila Bindal, MD, Warris Bokhari, MBBS, Deanna Bousalis, PhD, Diane J. Burgess, PhD, William T. Cefalu, MD, Sarnath Chattaraj, PhD, Danny Cherñavvsky, MD, Ali Cinar, PhD, Mark A. Clements, MD, PhD, CPI, FAAP, Gerard L. Coté, PhD, Jorge A. Cuadros, OD, PhD, Matthew C. Diamond, MD, PhD, Kathleen M. Dungan, MD, MPH, Manuel Eichenlaub, PhD, Juan Espinoza, MD, FAAP, Andrea Facchinetti, PhD, Andrew Farmer, DM, FRCGP, Stephanie A. Fisher, MD, MPH, Alexander Fleming, MD, Guido Freckmann, MD, Robert A. Gabbay, MD, PhD, Alfonso Galderisi, MD, PhD, Stephen E. Gitelman, MD, Ronald Goldenberg, MD, FRCPC, FACE, Alberto Gutierrez, PhD, Ahmad Haidar, PhD, Lutz Heinemann, PhD, Norbert Hermanns, PhD, Rolf Hinzmann, MD, PhD, Anand K. Iyer, PhD, MBA, Peter G. Jacobs, PhD, Jeffrey I Joseph, DO, Ulrike Klueh, PhD, Boris Kovatchev, PhD, James S. Krinsley, MD, Ed Krisiunas, MLS(ASCP), MPH, Rayhan A. Lal, MD, Felix C. Lee, MPharm, MSc, MBA, Wei-An (Andy) Lee, DO, Dorian Liepmann, PhD, Michelle L. Litchman, PhD, FNP-BC, FAANP, FADCES, FAAN, Julia K. Mader, MD, Umesh Masharani, MB, BS, Nestoras Mathioudakis, MD, MHS, Tracey L. McLaughlin, MD, MS, Michael J. McShane, PhD, Clara Mosquera-Lopez, PhD, David N. O’Neal, MD, FRACP, FRCP (Edin), Ted Osinski, Ambarish Pandey, MD, Elisa Pellizzari, MSc, John S. Pemberton, BSc, Andreas Pfützner, MD, PhD, Moshe Phillip, MD, Stefan Pleus, PhD, William Polonsky, PhD, Pietro Randine, Connie M. Rhee, MD, MSc, Michael C. Riddell, PhD, Steven J. Russell, MD, PhD, Michael Schoemaker, PhD, Thomas Seidl, PhD, Viral N. Shah, MD, Hari Sree, PhD, Devin W. Steenkamp, MBChB, Jannet Svensson, MD, PhD, Gerald R. Sydorak, MD, FACS, Sacha Uelmen, RDN, CDCES, Guillermo Umpierrez, MD, Hubert W. Vesper, PhD, Robert A. Vigersky, MD, Joseph Wang, PhD, Xujing Wang, PhD, Jessica Wilkerson, JD, Ralph Ziegler, MD, and Eric Zijlstra, PhD.
Abbreviations
AGP, ambulatory glucose profile; AI, artificial intelligence; AID, automated insulin delivery; app, application; BGM, blood glucose monitor; BNP, B-type natriuretic peptide; CBER, Center for Biologic Evaluation and Research; CDER, Center for Drug Evaluation and Research; CDRH, Center for Devices and Radiological Health; CE, Conformité Européenne; CG-DIVA, Continuous Glucose Deviation Interval and Variability Analysis; CGM, continuous glucose monitor; CKD, chronic kidney disease; CONCEPTT, continuous glucose monitoring in pregnant women with T1D; CSII, continuous subcutaneous insulin infusion; DCCT, Diabetes Control and Complications Trial; DFU, diabetic foot ulcer; DIY, Do-It-Yourself; DT, digital twin; DTM, Diabetes Technology Meeting; DTS, Diabetes Technology Society; ECG, electrocardiogram; EHR, electronic health record; ESKD, end-stage kidney diseases; FBR, foreign body response; FDA, U.S. Food and Drug Administration; GLP1-RA, glucagon-like peptide-1 receptor agonist; GRI, Glycemia Risk Index; HbA1c, hemoglobin A1C; HCP, health care professional; HF, heart failure; iCGM, integrated continuous glucose monitoring; iCoDE-1, Integration of Continuous Glucose Monitoring Data into the Electronic Health Record; iCoDE-2, Integration of Connected Diabetes Device Data into the Electronic Health Record; ICU, intensive care unit; IEEE, Institute of Electrical and Electronics Engineers; IFCC, International Federation of Clinical Chemistry and Laboratory Medicine; ISF, interstitial fluid; LH, lipohypertrophy; MARD, mean absolute relative difference; MDI, multiple daily injection; ML, machine learning; NIDDK, National Institute of Diabetes and Digestive and Kidney Diseases; NIH, National Institutes of Health; NT-ProBNP, N terminal pro B-type natriuretic peptide; OpenAPS, Open Artificial Pancreas System; PPG, photoplethysmography; RoC, rate of change; SGLT2i, sodium-glucose cotransporter 2 inhibitor; SMBG, self-monitoring of blood glucose; T1D, type 1 diabetes; T2D, type 2 diabetes; TBR, time below range; TIR, time in range; TITR, time in tight range; UK, United Kingdom; US, United States; WG-CGM, IFCC working group on CGM.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JHJ was also a speaker for Eli Lilly, Boehringer Ingelheim, Nordic Infucare, Novo Nordisk, Sanofi, and an advisory board member for Abbott, Eli Lilly, and Medtronic. DK has received research support from Abbott Diabetes Care and has been a Consultant to Sanofi, Better Therapeutics and Abbott Diagnostics. EC is an advisory board member and consultant for Novo Nordisk, Eli Lilly, Adocia, MannKind, Lexicon, Arecor. EC was also a speaker for Novo Nordisk. JP reports consultancy fees from Medtronic. NS is an employee of Medcrypt. DM is a consultant for Capillary Biomedical and a shareholder in Halozyme Therapeutics. HKA received research grant support through University of Colorado from Medtronic, Tandem Diabetes, Eli Lilly, Mannkind, Dexcom and consultation fees through University of Colorado from Tandem Diabetes, Medtronic and Dexcom. CJL has received research support by the NIDDK and Helmsley Foundation and industry support paid to the Icahn School of Medicine at Mount Sinai from Abbott Diabetes, Dexcom, Insulet, Novo Nordisk, Mannkind, Senseonics, and Tandem. CJL has received consulting fees from Eli Lilly, and Dexcom outside of this work. RB is a shareholder of Biomeris s.r.l. and Engenome s.r.l. EKS was partially supported by the VA MERIT award (#1I01CX001825) and CSP #2002 from the US Department of Veterans Affairs. EKS has received unrestricted research support from Dexcom and Tandem (to Baltimore VA Medical Center and to University of Maryland) for the conduction of clinical trials. EKS has received fees from the Medscape and the Endocrine Society (ESAP). BN is serving as a consultant for BioSensics LLC. JJS is a consultant for Lifescan Diabetes Institute. DCK is a consultant for Afon, Better Therapeutics, Integrity, Lifecare, Nevro, Novo, and Thirdwayv. TT, REA, AYD, AD, KYC, SS, AHBW, JGC have nothing to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The 2023 Diabetes Technology Meeting was supported by educational grants from Abbott, Dexcom, Embecta, Glytec, LifeScan, Medtronic, Novo Nordisk, Roche Diabetes Care, Roche Diagnostics, and Tandem.
