Abstract
Achieving optimal glycemic control in children with type 1 diabetes (T1D) is challenging even when wearing a continuous glucose monitor (CGM). We measured the impact of eddii, a gamified real-time app connected to a CGM, on glycemic control. An open label, eight-week randomized controlled trial (RCT) compared glycemic control utilizing the gamified CGM app connected to Dexcom G6 with only Dexcom G6 usage. Children with T1D using Dexcom G6 were enrolled (N=92, ages 5-12 years). Time-in-range (TIR) data were collected four weeks prior to and during the study period. The gamified CGM app utilization effect was measured by difference-in-difference (D-I-D) models. The TIR and time-above-range (TAR) improved among users of the gamified CGM app; 5.38% higher and 5.80% lower than controls (P = .001 and P = .019, respectively).
Keywords
Introduction
Wearable continuous glucose monitors (CGMs) have revolutionized diabetes management, becoming an increasingly adopted technology, particularly for individuals living with type 1 diabetes (T1D). 1 Today, more than five million people globally wear CGMs.2,3 While CGM devices improve diabetes therapy safety and effectiveness, 4 reduce the frequency and duration of hypoglycemia incidents, 5 and decrease glycemic variability, 4 interactive software that enable users to feel engaged with their CGM interface are limited. The CGM device users most often use manufacturer-supplied smartphone apps as interfaces for glucose monitoring. These apps, however, are not optimized to engage children and adolescents living with diabetes. These apps include the Dexcom G6 and G7, Medtronic Guardian, and Abbott Freestyle apps.
Achieving optimal glycemic control in children with T1D is challenging. 6 Today, emerging CGM real-time Application Programming Interface (rtAPI) integrations offer ample opportunity for improvement upon these preexisting apps. The mobile application, eddii (referred to hereafter as the gamified CGM app), is designed to display real-time CGM data in a format that engages children through games and rewards, helping them track and engage with their health. This gamified CGM app receives real-time glucose readings from the user’s Dexcom CGM every five minutes. The in-app character, “eddii,” in the shape of a leaf, reacts through animations to real-time glucose readings from the Dexcom CGM (Figure 1). This new CGM app encourages health and lifestyle data entry through gamification by awarding the child-user “hearts” that can be redeemed to play mini-games, customize the character home page (eg, purchase clothing and pets), or win gift cards (eg, Roblox). Parents can track their child’s glucose through the guardian interface of this gamified CGM app and set real-life rewards for their child. In this study, we measured the impact of using a gamified CGM app on glycemic control.
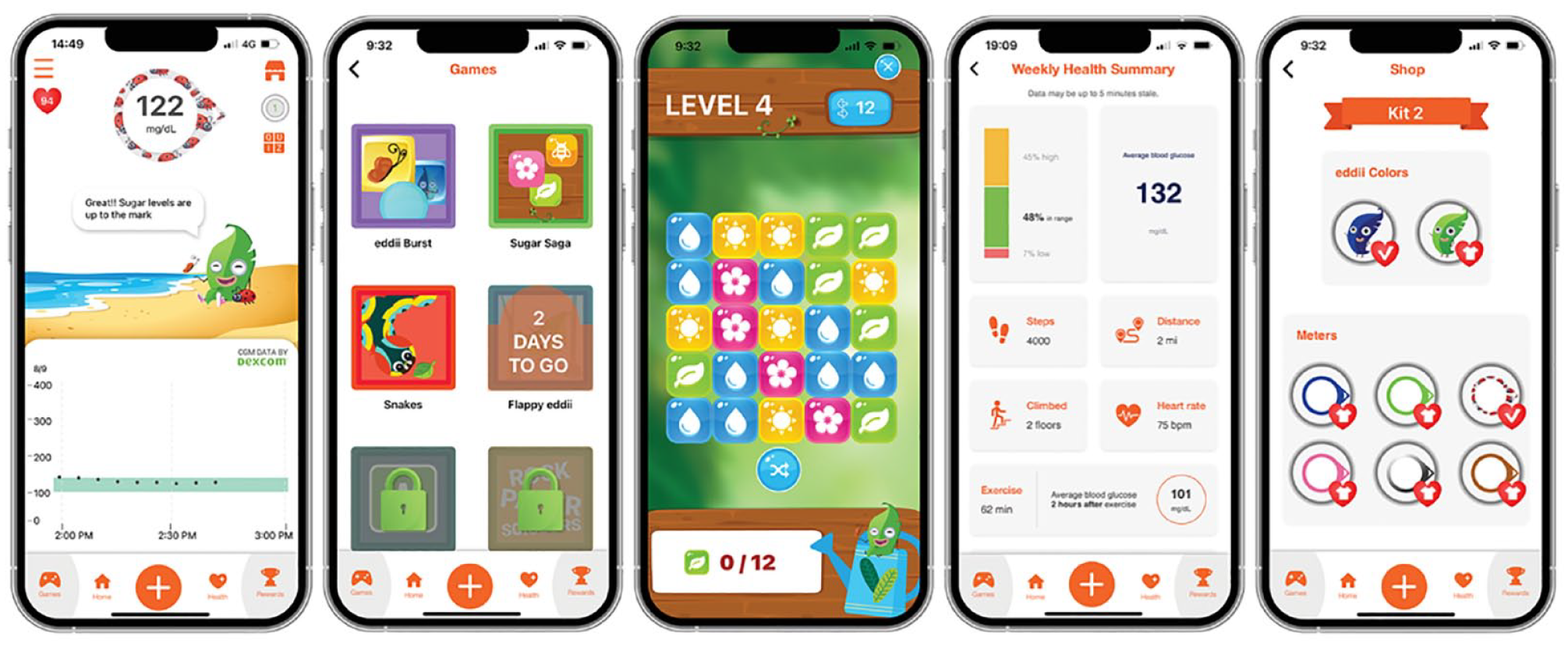
Gamified CGM (eddii) app interface.
Methods
Recruitment and Informed Consent
Candidates were recruited using social media ads and asked to participate in an eight-week study. Legal guardians were invited together with their child(ren) to schedule a virtual video call with the study team. Legal guardians indicated their consent to the study terms by signing the informed consent form provided to them subsequently through DocuSign. Children aged seven to 12 years provided their assent through the child’s electronic signature through DocuSign. All children gave their assent both verbally, during their video call, in the presence of their guardian, the principal investigator, a witness, and in writing by ticking a checkbox on the child assent form. For children five to six years of age, parents were asked to check the box and sign the child assent form on behalf of the child.
Study Timeline
The intervention group was asked to use the Dexcom G6 app during the two onboarding weeks and then switch to using the gamified CGM app on day 15 for the remaining six weeks. The intervention group participants continued using the gamified CGM app for an additional four-week follow-up period to document any delayed impact of the gamified CGM app. Participants randomized to the control group were asked to use the Dexcom G6 app and their usual diabetes management regimen for the two onboarding weeks, and then continue doing so for the remaining six weeks. Control group participants continued using the Dexcom G6 app for an additional four-week follow-up period, similar to the intervention group, for comparison, as shown in Figure 2.
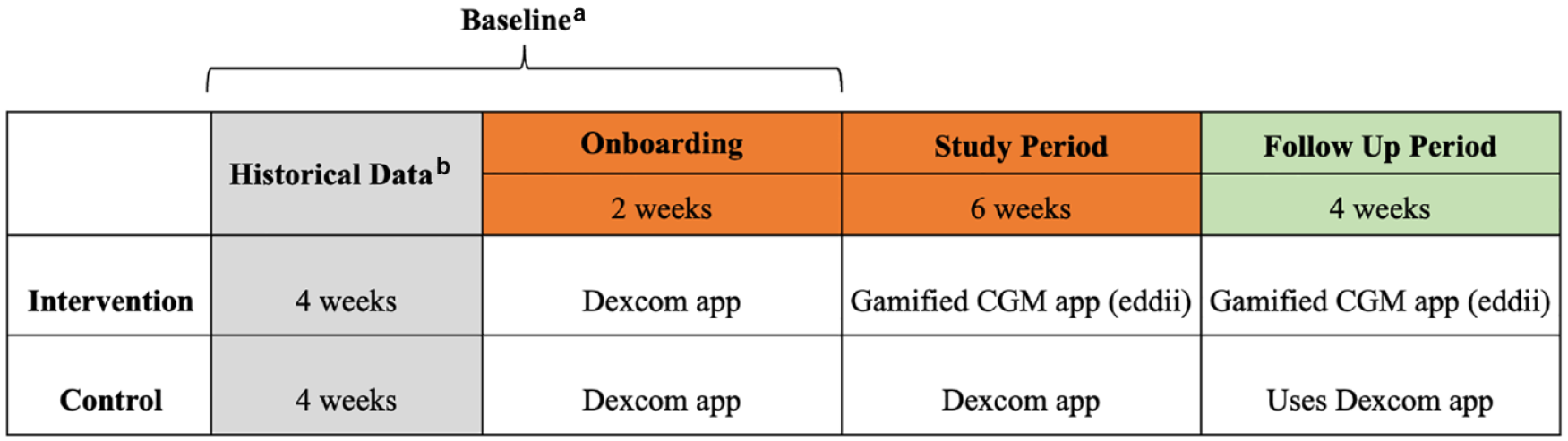
Study timeline.
Randomization Procedure
Participants were enrolled on a rolling basis and assigned a study ID upon randomization. Randomization was done in blocks of four through parallel assignment, as follows: four participants were grouped together and half of the group (two out of four) were randomly assigned to each study arm in a 1:1 ratio.
Inclusion and Exclusion Criteria
Child participants were eligible if willing and able to provide informed consent. Inclusion criteria included T1D diagnosis and history of Dexcom CGM use for at least four weeks. Exclusion criteria included individuals unwilling or unable to consent, unavailable iPhone operating system (iOS) device, or candidate being enrolled in another study. Parent or legal guardian eligibility criteria included (1) ability and willingness to provide written informed consent and authorize health information release, (2) child met study inclusion criteria and child assent has been obtained, (3) willingness to be responsible for the gamified CGM app use, and (4) iOS smartphone access. The study Consolidated Standards of Reporting Trials (CONSORT) 7 flow chart is shown in Figure 3.
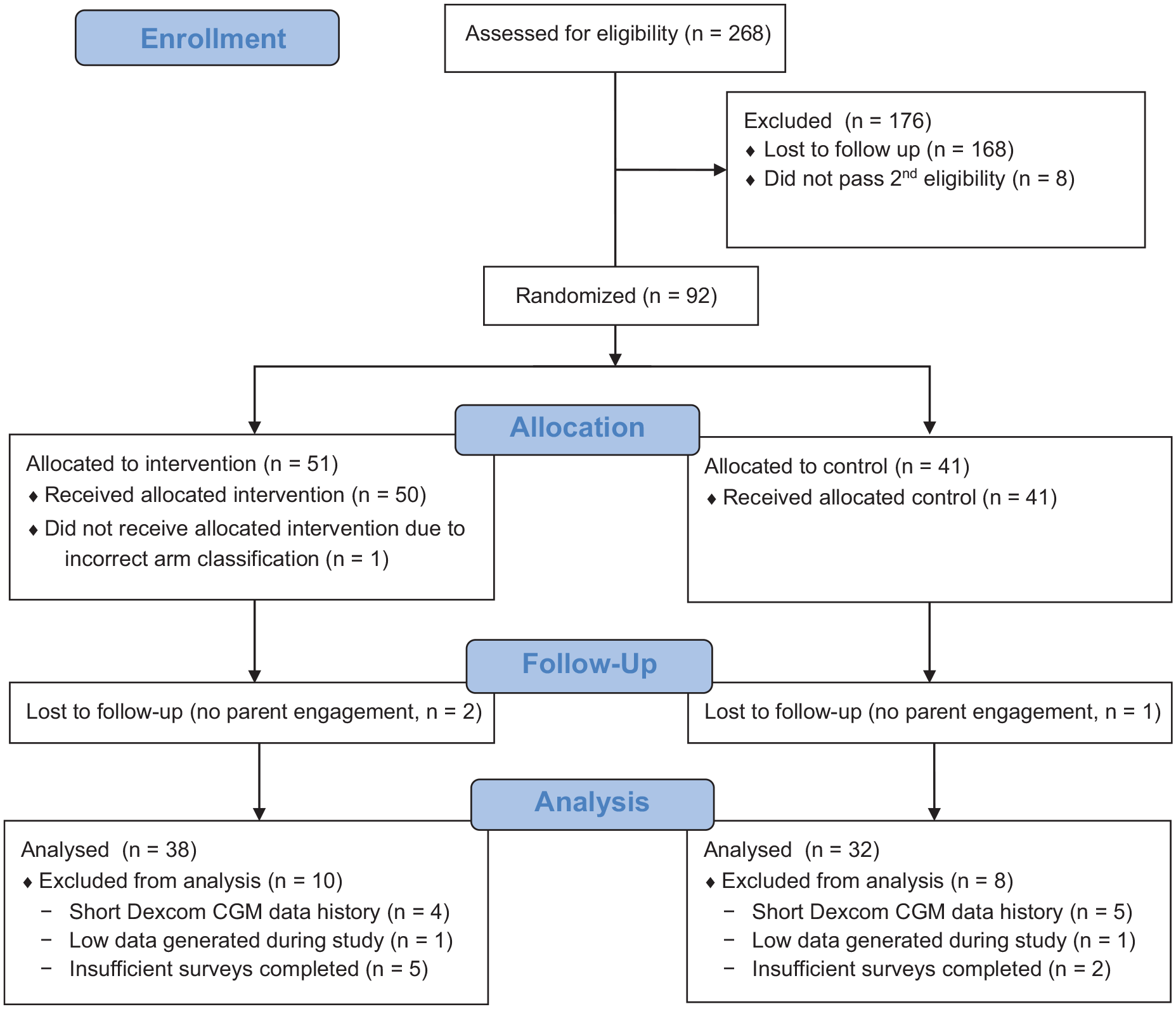
Study CONSORT flow chart.
Power Analysis
Power analyses were conducted to estimate the required sample size by two methods comparing baseline and end-of-study time-in-range (TIR) mean values targeting two main research goals: (1) intervention within group TIR changes, and (2) between-groups average TIR difference.
Post hoc power analysis and sample size calculation were conducted and supported estimates of an acceptable sample size for a future, larger study. No measures of spread were evaluated for these estimates. The power of this study was 49% for α=.05. To adequately compare the baseline and final TIR in the study arm with a 70% power (α=.05), a sample size of 115 is needed.
Measures
TIR changes
The TIR, hyperglycemia, and hypoglycemia changes were gathered for four weeks before and recorded after using the gamified CGM app in the study arm and over the same period for the controls. Reported values follow American Diabetes Association’s (ADA) standards and the Standards of Medical Care in Diabetes–2022 8 for CGM data in Ambulatory Glucose Profiles (AGPs).9-11 Glycemic outcomes data were collected through the Dexcom Clarity.
Safety and feasibility
Mixed format questionnaires assessed the safety and feasibility of the gamified CGM app. Participants in both arms were asked to complete questionnaires at enrollment and every two weeks during the study period. At the time of enrollment, participant guardians were asked to complete a baseline medical questionnaire.
The intervention group and their guardians were asked to rate their level of satisfaction with the overall app effectiveness (1-10 scale, with 10, highly effective). In addition, comments on what were the best and worst things about their experience were invited. The number and nature of interactions with the gamified CGM app and app crashes were recorded in the backend infrastructure of the app. Safety was evaluated as freedom from serious adverse events directly attributed to the gamified CGM app.
Self-care inventory
The self-care inventory (SCI) 12 was used with author permission (La Greca 2004) to assess the participant quality of self-care during the study. The SCI is a 14-item self-report measure developed to assess a patient’s perception of adherence to treatment recommendations. A higher SCI score indicates greater self-care. The lowest and highest scores that an individual could record are 1 and 5. As CGM enables both “glucose testing” and “glucose recording,” these two procedures were collapsed into one “glucose tracking” item.
Statistical methods
Participant data were analyzed according to their randomized assignment. Exclusion from data analysis was warranted in 19 study cases due to (1) incorrect study arm classification, N=1; (2) loss to follow up, N=1; (3) insufficient Dexcom CGM historical data availability, N=10; (4) insufficient data generated during the study period from their Dexcom CGM, N=2; or (5) insufficient responses to surveys, N=5.
The difference-in-difference (D-I-D) models were conducted to estimate the effect of the intervention by comparing the changes in outcomes over time (baseline vs final) between intervention group and control group, adjusted by patient`s age, race/ethnicity, sex, devices, perceived control, automated insulin, acceptance of diagnosis, and time wearing CGM. Specific adjusted between-group D-I-Ds were calculated by least squares means (LSMEANS). All statistical analyses were completed using STATA 17 and all hypothesis tests were conducted at a significance level of α=.05.
Results
Our study compared a new gamified CGM app with the Dexcom G6 app in children with T1D. Prescreening questionnaires were sent to 356 candidates. Eligible candidates (N=197) were invited to provide informed consent. Participants (N=92) were randomized to either the intervention or the control group. As five study arm participants became ineligible per protocol, we assigned the following three consecutive enrollees to intervention instead of control, as replacements. Some participants withdrew or became ineligible for inclusion in data analysis, which led to an imbalance of six participants between the intervention and control groups. Although 89 participants completed the total six weeks of study, only data from 70 study participants (intervention group, 38; control group, 32) were eligible for inclusion in the final glycemic control data analysis (Figure 3).
Participant Demographics
Participant baseline characteristics are outlined in Table 1. Most enrollees were girls (57%), non-Hispanic (97.1%), and white (84.3%). The average age was 8.6 years (SD 2.1). In the intervention group, 81.6% of the children used an insulin pump. The study arm participants interacted with gamified CGM app, on average, 12.5 times/day (SD 8.3). Interactions with the gamified CGM app ranged between 0.9 and 37.7, with a median of 10. On average, there were 3.0 daily lifestyle entries (SD 3.5, median 2.1, and range 0.0-18.6). Participants earned hearts for data entry and completing daily missions. The study arm participants spent a median of 246 “hearts” and a total of 95 gift cards were purchased by 37 participants in the intervention group.
Baseline Characteristics.
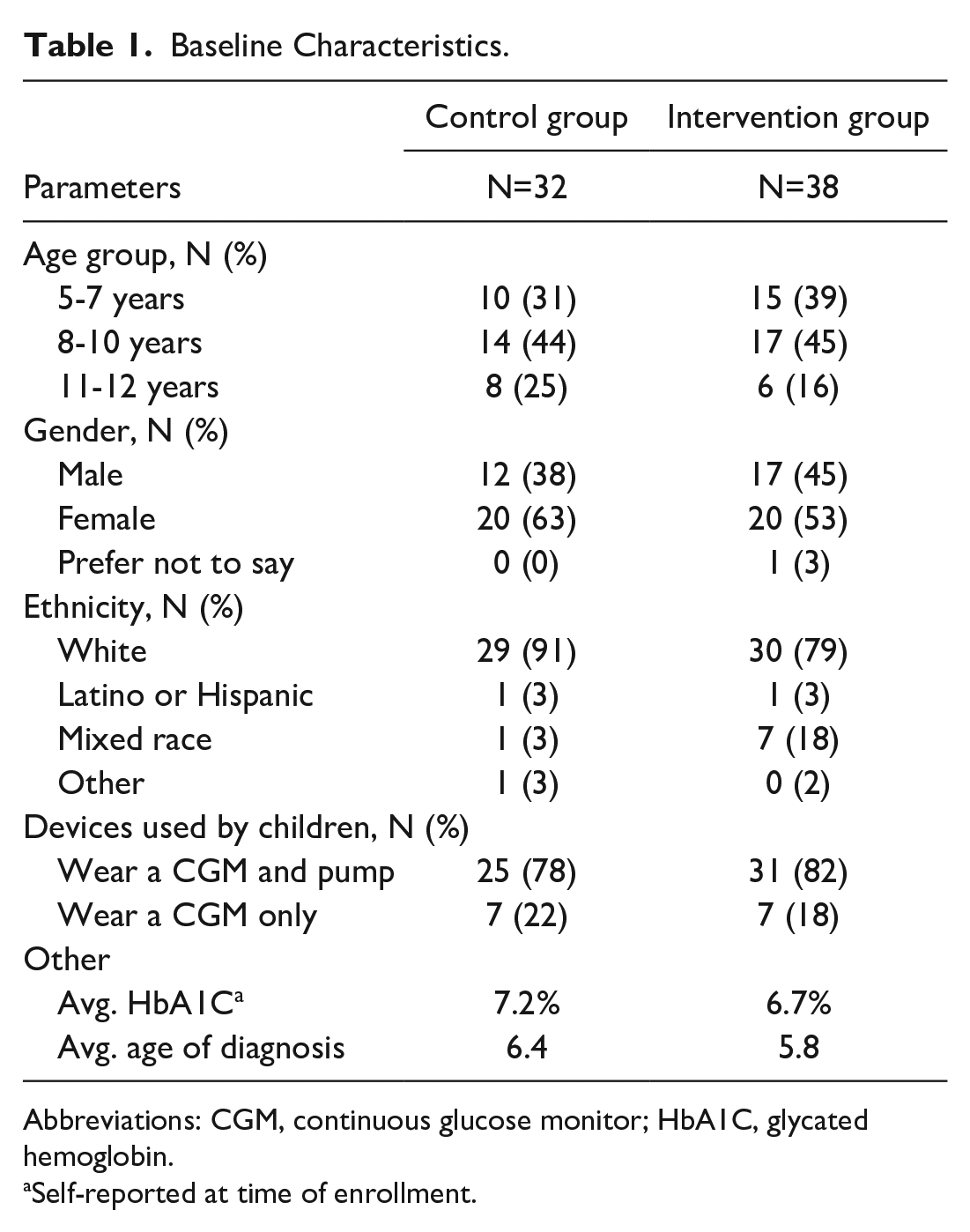
Abbreviations: CGM, continuous glucose monitor; HbA1C, glycated hemoglobin.
Self-reported at time of enrollment.
Questionnaires
The majority of participant parents were able to see and track their child`s glucose on the gamified CGM app (92.5%). Their experience using the gamified CGM app as very good or good (94.6%, P < .0001). Most respondents (72%) preferred the gamified CGM app compared with the Dexcom G6 app, whereas 22.6% indicated no preference, P < .0001 (see supplemental material). The child participants’ preference questionnaires indicated that 84.5% of them favored the gamified CGM app and 93.2% found the gamified CGM app to be either good, brilliant, or really good (see supplemental material).
D-I-D Model
To evaluate the gamified CGM app’s impact on glycemic control, the utilization effect was estimated by D-I-D models comparing outcomes’ changes within and between study groups. The outcomes evaluated were TIR, hyperglycemia, hypoglycemia, SCI, and SCI insulin. The LSMEANS-generated multivariable D-I-D model output for the control versus study arms comparison is presented in Table 2.
Main Results With LSMEANS Outputs From Multivariable D-I-D Models Adjusted by Baseline Characteristics.
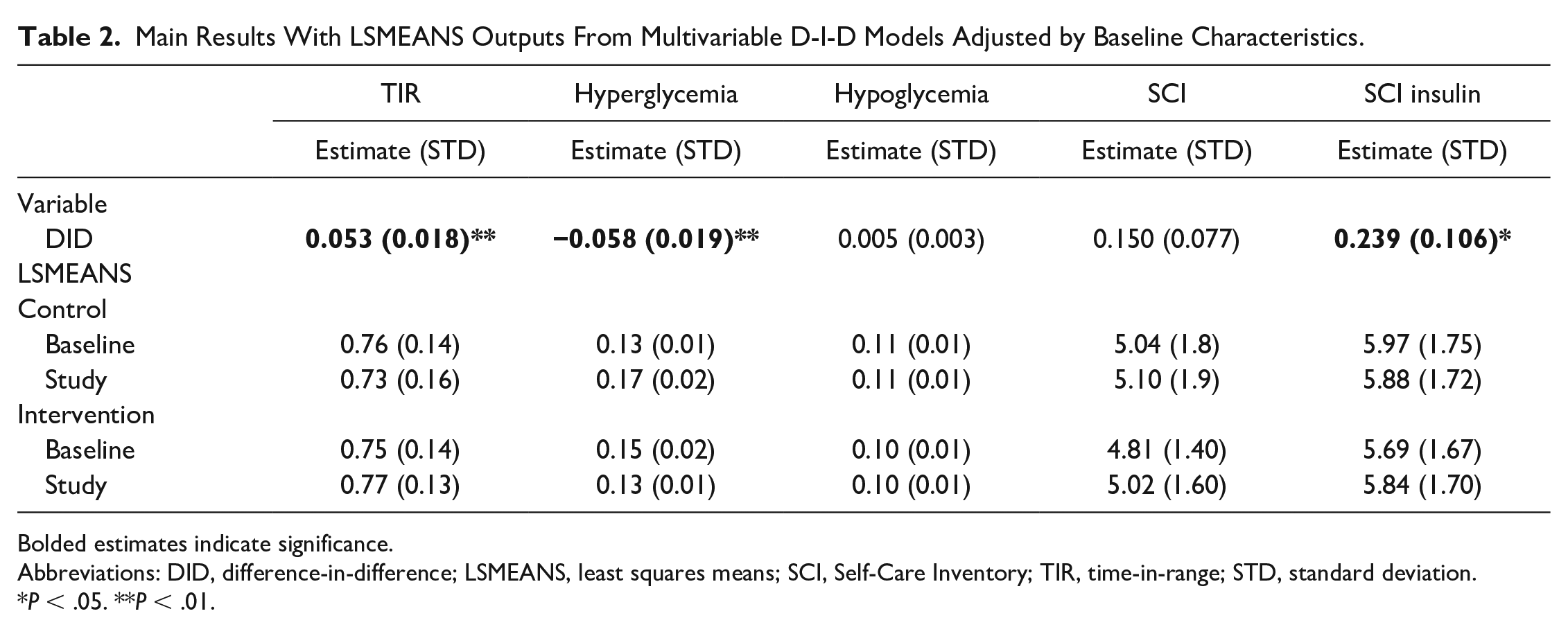
Bolded estimates indicate significance.
Abbreviations: DID, difference-in-difference; LSMEANS, least squares means; SCI, Self-Care Inventory; TIR, time-in-range; STD, standard deviation.
P < .05. **P < .01.
Among the control arm participants, TIR decreased from 76.4% at baseline to 72.7% at week 8, whereas for those in the study arm, the TIR improved from 75.5% to 77.3%, indicating a positive impact of the gamified CGM app on glucose control. The adjusted D-I-D for TIR was 5.3% (P < .01). Hyperglycemia also improved in the study arm, decreasing from 15% to 13% while increasing from 13% to 17% in the control arm, a difference favoring the gamified CGM app users. The adjusted D-I-D for hyperglycemia values was −5.8% (P < .01), indicating a positive impact of the gamified CGM app. Regarding hypoglycemia, no within-group differences were observed, but the hypoglycemia in the intervention group was slightly lower. The SCI increased slightly in the control arm, whereas the change from baseline was larger in the study arm, from 4.81 to 5.02. The adjusted D-I-D for the SCI was 0.15 score units. Finally, the SCI insulin values worsened slightly in the control arm, from 5.97 to 5.88, while improving in the study arm, from 5.69 to 5.85. The adjusted D-I-D for SCI insulin was 0.24 score units (P < .05). No adverse events were reported.
Discussion
To our knowledge, this is the first pediatric randomized controlled trial (RCT) evaluating the efficacy of a real-time gamified software interface for diabetes management, as measured by TIR. The children with T1D using the gamified CGM app demonstrated an improved TIR compared with the standard Dexcom G6 app users (5.3%, P = .001). Hyperglycemia also improved among gamified CGM app users, with an adjusted treatment effect of −5.8% (P < .01). Both control and intervention groups showed high baseline TIR (76% and 75%, respectively). This can be explained by the high number of participants using both a CGM and a pump in both arms of the study. Despite this, a significant improvement in TIR was shown in the intervention arm. Given this improvement, we hypothesize that gamified CGM interface can have an even larger impact on children living with T1D whose glycemia is not so well controlled. Whereas the gamified CGM app effect on SCI was modest, the adjusted SCI insulin scores in-between group comparison revealed a significant effect that favored the study arm.
Optimal glycemic control in children is challenging, particularly given children’s reliance on caregivers, unpredictable activity, and difficulty in effectively recognizing their own emotions and/or communicating their high or low glucose levels. 6 A lack of significant improvement in diabetes management with CGM use in children has been repeatedly reported.6-9 For instance, in 2022, a yearlong study (Strategies to Enhance New CGM Use in Early Childhood [SENCE]) investigating CGM effects and Family Behavioral Intervention (FBI) on adults’ and children’s glycemic control, showed a significant TIR improvement in adults, but failed to find a glycemic control improvement in children aged two to eight years or in young adults. 6 Other studies focusing on children using CGMs reported mixed results.13,14 Furthermore, a study by Juvenile Diabetes Research Foundation (JDRF) reported no significant improvement in glycemic metrics in children aged eight to 14 years using CGMs. 4 More recently, a study by the T1D Exchange Registry showed lower glycated hemoglobin (HbA1c) levels in CGM users than nonusers, 15 while a study by Straton et al 16 showed that adolescents using CGM showed a significantly lower HbA1C level than nonusers. These varied results, collectively, indicate the need for more advanced behavioral approaches to optimize glycemia in both children and adolescents.
Our gamified CGM app offered children a delightful interface, one that allowed them to customize their app and associate their diabetes with rewarding experiences, such as winning their favorite gift card, playing games, and interacting with the leaf character. We hypothesize that this engagement ultimately led to an improvement in glycemic control. Our findings demonstrate that a child-friendly and fun interface augments the already existing benefits of wearing a CGM and other devices in a low-risk manner that makes diabetes management less burdensome and a happier experience for children.
In the future, we believe that an even more interactive app, one that uses a multipronged approach to user engagement, can further improve glycemic control. These methods can include AI-driven emotional support, educational elements to help children take more ownership of their diabetes, and community features, in addition to more enhanced games, to name a few.
There are several methodological weaknesses that limit the generalizability of our findings, however. Our sample size was rather small, and the study duration was relatively short. Yet this study shows that gamified engagement can improve glycemic control in children with T1D while posing minimal risk to the user.
Supplemental Material
sj-docx-1-dst-10.1177_19322968231223759 – Supplemental material for Gaming the System: A Fun Continuous Glucose Monitor Interface Improves Glycemic Outcomes for Children
Supplemental material, sj-docx-1-dst-10.1177_19322968231223759 for Gaming the System: A Fun Continuous Glucose Monitor Interface Improves Glycemic Outcomes for Children by Farhaneh Ahmadi and Alonso Lucero in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors would like to acknowledge Dr G. Gutierrez and Dr Y. Deng for their guidance on statistical analyses’ selection. The authors would like to thank Ms M. Orosco on her assistance with conducting the clinical trial. The authors are grateful to Dr K. Mengwasser for guidance on research design, helpful insight on results’ discussion, and review of the manuscript. The authors would like to thank Dr R. Jafri for review of the manuscript. This clinical trial was conducted in collaboration with Dexcom and the authors thank Dr T. Walker for the fruitful collaboration.
Correction (May 2024):
Abbreviations
ADA, American Diabetes Association; AGP, ambulatory glucose profile; BG, blood glucose; CGM, continuous glucose monitor; CONSORT, Consolidated Standards of Reporting Trials; D-I-D, difference-in-difference; HbA1C, glycated hemoglobin; LSMEANS, least squares means; RCT, randomized controlled trial; SCI, self-care inventory; TAR, time-above-range; TIR, time-in-range; T1D, type 1 diabetes.
Author Contributions
F.A. served as the study sponsor, contributed to the study design and implementation, performed the random allocation sequence, enrolled participants (in the presence of a witness), assigned participants to the intervention and control groups, performed data analysis, and edited and reviewed the manuscript. A.L. contributed to the study implementation, data analysis, and writing of the manuscript. All authors approved the final version of the manuscript. F.A. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: F.A. and A.L. are full-time employees of Eddii, Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was sponsored by Eddii, Inc, New York, NY.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
