Abstract
Objectives:
Achieving optimal glycemic outcomes in young children with type 1 diabetes (T1D) is challenging. This study examined the durability of continuous glucose monitoring (CGM) coupled with a family behavioral intervention (FBI) to improve glycemia.
Study Design:
This one-year study included an initial 26-week randomized controlled trial of CGM with FBI (CGM+FBI) and CGM alone (Standard-CGM) compared with blood glucose monitoring (BGM), followed by a 26-week extension phase wherein the BGM Group received the CGM+FBI (BGM-Crossover) and both original CGM groups continued this technology.
Results:
Time in range (70-180 mg/dL) did not improve with CGM use (CGM+FBI: baseline 37%, 52 weeks 41%; Standard-CGM: baseline 41%, 52 weeks 44%; BGM-Crossover: 26 weeks 38%, 52 weeks 40%). All three groups sustained decreases in hypoglycemia (<70 mg/dL) with CGM use (CGM+FBI: baseline 3.4%, 52 weeks 2.0%; Standard-CGM: baseline 4.1%, 52 weeks 2.1%; BGM-Crossover: 26 weeks 4.5%, 52 weeks 1.7%, P-values <.001). Hemoglobin A1c was unchanged with CGM use (CGM+FBI: baseline 8.3%, 52 weeks 8.2%; Standard-CGM: baseline 8.2%, 52 weeks 8.0%; BGM-Crossover: 26 weeks 8.1%, 52 weeks 8.3%). Sensor use remained high (52-week study visit: CGM+FBI 91%, Standard-CGM 92%, BGM-Crossover 88%).
Conclusion:
Over 12 months young children with T1D using newer CGM technology sustained reductions in hypoglycemia and, in contrast to prior studies, persistently wore CGM. However, pervasive hyperglycemia remained unmitigated. This indicates an urgent need for further advances in diabetes technology, behavioral support, and diabetes management educational approaches to optimize glycemia in young children.
Keywords
Introduction
Achieving and maintaining target glycemic outcomes in very young children with type 1 diabetes (T1D) is challenging due to their unpredictable eating behaviors, erratic physical activity and moods, inability to articulate symptoms of hypo- and hyperglycemia,1,2 frequent intercurrent illnesses, and complete reliance on caregivers for diabetes management. Hence, young children with T1D spend little time in optimal glycemic range 3 and are at risk for both severe hypoglycemia and hyperglycemia.
Clinical trials of continuous glucose monitoring (CGM) devices have been shown to improve glycemic control in older children and adults4,5; however, data on CGM effectiveness in very young children are limited.6-8 CGMs provide real-time glucose data and alerts, as well as comprehensive retrospective glucose patterns and trends to inform diabetes management decisions for caregivers of young children with T1D. Recent improvements in CGM technology including smaller device size, enhanced accuracy, and ability to monitor data remotely offer great promise for leveraging CGM to optimize glycemic outcomes.
The
Subsequently, the current analysis was undertaken to examine the durability of CGM use and glycemic outcomes in a subsequent 26-week extension period. Specifically, the two initial CGM groups continued CGM use with less intensive follow-up, whereas the original BGM group crossed over to use CGM in combination with FBI training (BGM-Crossover Group) during the 26-week extension. Herein, we present results from the full 52-week study in all three treatment groups.
Methods
The study was conducted at 14 pediatric endocrinology practices in the United States with enrollment visits taking place between February 2017 and August 2018. The protocol and informed consent forms were approved by institutional review boards. Written informed consent was obtained from the parent or guardian of each participant and assent was obtained from each participant when applicable. Major eligibility criteria included clinical diagnosis of T1D for at least three months, age two to less than eight years, total daily insulin requirement ≥0.3 units/kg/d, no use of real-time CGM in 30 days prior to enrollment, and hemoglobin A1c (HbA1c) 7.0% to <10.0% (53-<86 mmol/mol). The full eligibility criteria have been published previously, 3 the protocol is available at https://public.jaeb.org/datasets, and details are provided on clinicaltrials.gov (NCT02912728).
26-Week Randomized Clinical Trial
All participants completed a two- to three-week prerandomization screening period using masked CGM (G4 Platinum® Professional; Dexcom, San Diego, CA) to collect baseline glucose data. Following successful completion of the screening period, participants were randomized to one of three groups: (1) CGM+FBI Group used the Dexcom G5 real-time CGM with standardized training and a family behavioral intervention, (2) Standard-CGM Group used the Dexcom G5 real-time CGM with standardized training without the family-based intervention, and (3) fingerstick BGM control group without CGM for a period of 26 weeks. 9 During the 26-week randomized phase, all study groups were matched for attention with a similar visit schedule. All groups were provided the same chart of glycemic targets (Supplemental Appendix). At four time points, BGM control group participants wore masked Dexcom G4 Platinum Professional CGM for one week in order to collect sensor glucose data.
26-Week Extension Phase
Following completion of the randomized trial, participants randomized to the BGM control group crossed over to initiate real-time CGM (BGM-Crossover Group) and received the standardized CGM training and FBI. All three groups were provided real-time Dexcom G5 CGM during this 26-week extension phase to wear through completion of the 52-week study (Figure 1). Of note, the Dexcom G5 CGM required calibrations every 12 hours, hence the study teams recommended participants perform fingerstick blood glucose checks at least three times a day to prevent overnight calibration alerts.
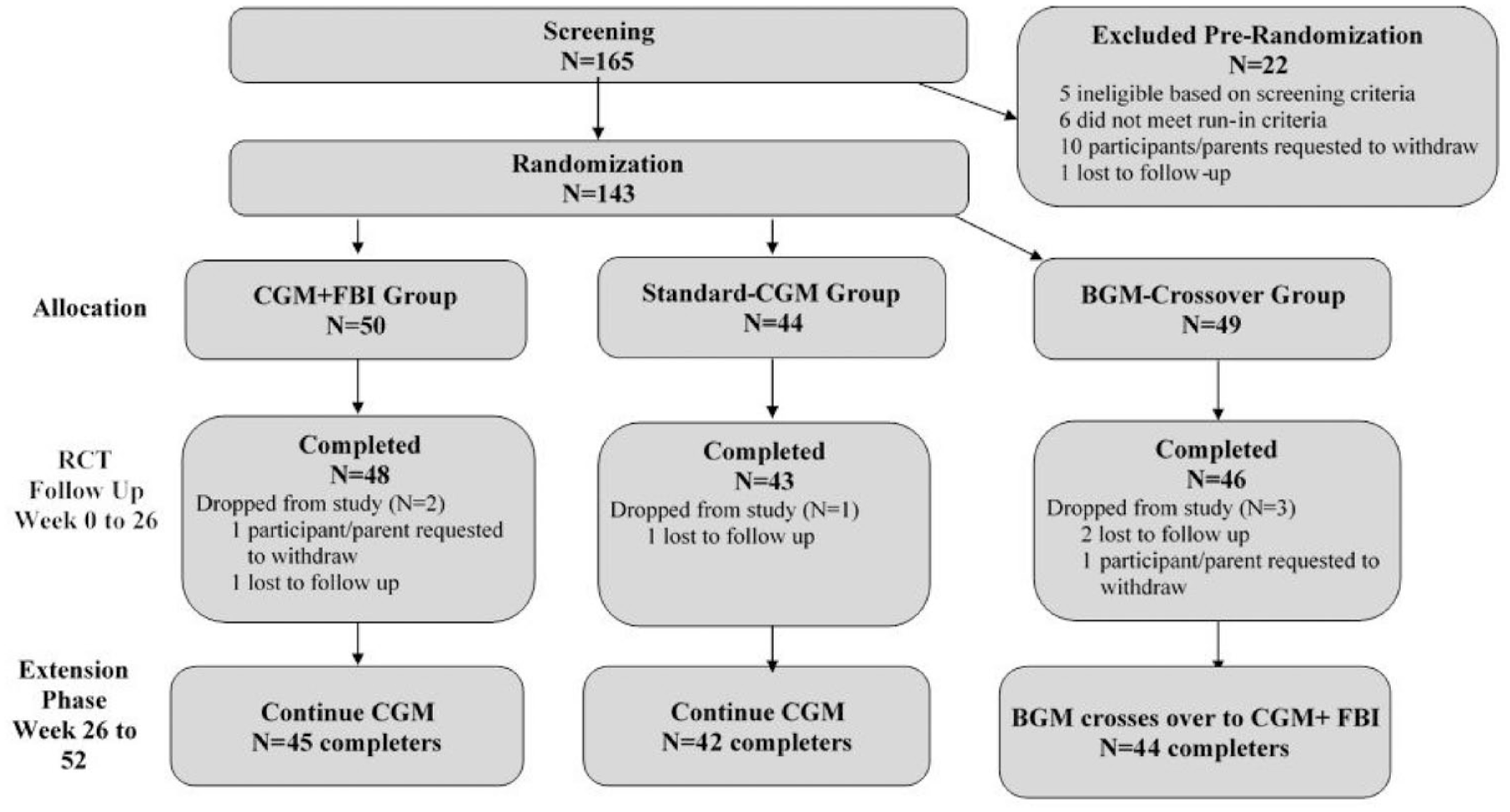
Flowchart of study.
In the 26-week extension phase, study contact was reduced in the CGM+FBI Group and Standard CGM Group to in-clinic visits at 39 and 52 weeks. The BGM-Crossover Group, who initiated real-time CGM at the 26-week visit, had a phone call at 27 weeks and in-clinic visits at 28, 39, and 52 weeks to receive standardized CGM education and FBI.
Training and Education
The standardized CGM education provided to the two CGM groups during the first 26 weeks and the BGM-Crossover Group in the extension phase involved five 30-minute standardized sessions of CGM training. These sessions were delivered by experienced diabetes educators and focused on how CGM works, CGM basics, troubleshooting and nonadjunctive use of CGM, using CGM to minimize highs and lows, and evaluating CGM data. Low glucose alerts were initially set to alarm when sensor glucose reached <80 mg/dL. The high alert was initially set to 400 mg/dL and was then lowered over the course of the CGM training sessions to 300 mg/dL at week 3 unless the family preferred not to make this change. The BGM-Crossover Group received the standardized CGM education sessions at the screening, 26-, 27-, 28-, and 39-week visits (Supplemental Table 1). Caregivers were also trained on use of remote CGM data monitoring through the Dexcom SHARE/FOLLOW applications.
The FBI provided to the CGM+FBI Group during the initial 26 weeks of the randomized trial and to the BGM-Crossover Group in the extension phase was developed based on survey and qualitative data from interviews with caregivers of young children with T1D.10-13 The BGM-Crossover Group received the FBI sessions at 26, 27, 28, and 39 weeks, with a review session at 52 weeks (Supplemental Table 1). Sessions were ~30 minutes, delivered via telephone or in person by a trained research assistant. The standardized intervention materials addressed obstacles to CGM use and caregiver feelings, attitudes, and behaviors related to effective and sustained CGM use, and interventionists taught caregivers relaxation, problem-solving, and communication strategies. Specifically, sessions included strategies to minimize CGM insertion pain and wearability concerns, discussion of parental expectations of CGM use, and education about glucose variability and links between glucose levels and mood. The FBI also covered discussions about parents’ emotional reactions to above and below range CGM data, review of how to treat above and below range glucose, and reframing attention to benefits of CGM alerts for optimizing diabetes management. Caregivers were counseled in seeking social support, communicating with children about their devices, and teaching other adults about CGM in planning for leaving the child with another caregiver.
Adverse Events
Reportable adverse events included severe hypoglycemia (defined as an event that required assistance from another person due to altered consciousness), hyperglycemia resulting in treatment at a health care facility or that involved diabetic ketoacidosis (as defined by the Diabetes Control and Complications Trial), 14 device-related events with potential effects on participant safety, and all serious adverse events regardless of causality.
Statistical Analysis
Glycemic outcomes included percent of time in target range (70-180 mg/dL), mean glucose, coefficient of variation, percent of time in hyperglycemia (>180, >250, and >300 mg/dL), percent of time in hypoglycemia (<70 and <54 mg/dL), rate of hypoglycemic events per week, and HbA1c. 15
CGM-measured outcomes were calculated at baseline using the masked data collected during the screening phase. For the CGM+FBI Group and Standard-CGM Group, CGM outcomes were calculated at each follow-up visit by pooling data from the four weeks prior. For the BGM control group, CGM outcomes were calculated using data from the masked CGM at each study visit in the initial 26 weeks of the randomized trial and then by pooling real-time CGM data from four weeks prior to study visits in the extension phase (BGM-Crossover Group).
HbA1c was measured by the central lab at randomization and 13, 26, 39, and 52 weeks at the University of Minnesota using the Tosoh A1c 2.2 Plus Glycohemoglobin Analyzer. If the central lab result was missing, values were imputed using local results (lab or point of care), if available.
A paired t-test, signed rank test, or McNemar’s test, as appropriate, was used to evaluate the change in each glycemic outcome between baseline and 52 weeks in the CGM+FBI Group and Standard-CGM Group and between 26 and 52 weeks in the BGM-Crossover Group. In addition, a linear or logistic regression model was used to compare each glycemic outcome between the CGM+FBI Group and Standard-CGM Group at 52 weeks. The model was adjusted for baseline value of the outcome, baseline HbA1c, and clinical site as a random effect.
P-values were two-sided and adjusted for multiple comparisons to control the false discovery rate using the adaptive Benjamini-Hochberg procedure. 16 All analyses included participants with available data who completed the 52-week visit. Baseline refers to prerandomization measurements. Analyses were conducted with SAS software version 9.4 (SAS Institute Inc., Cary, NC).
Results
The full 52-week trial (RCT + extension) was completed by 131 of the 143 participants (92%), including 45 of 50 in the CGM+FBI Group, 42 of 44 in the Standard-CGM Group, and 44 of 49 in the BGM-Crossover Group. Six participants dropped from the study during the initial 26 weeks (two in the CGM +FBI Group, one in the Standard-CGM Group, and three in the BGM control group) and six dropped during the extension phase (three in the CGM+FBI Group, one in the Standard-CGM Group, and two in the BGM-Crossover Group) (Figure 1).
At baseline, the 131 children were 5.7 ± 1.8 years of age, with 2.3 ± 1.9 years duration of diabetes. Participants were 50% female; 69% identified as non-Hispanic white, 13% as non-Hispanic Black, 12% as Hispanic or Latino, <1% as Asian, and 5% as another or more than one race/ethnicity. Sixty-two percent had private insurance. Thirty-five percent were using an insulin pump and 87% were CGM naïve at baseline. Baseline participant characteristics according to treatment randomization group are shown in Table 1.
Participant Characteristics at Baseline.
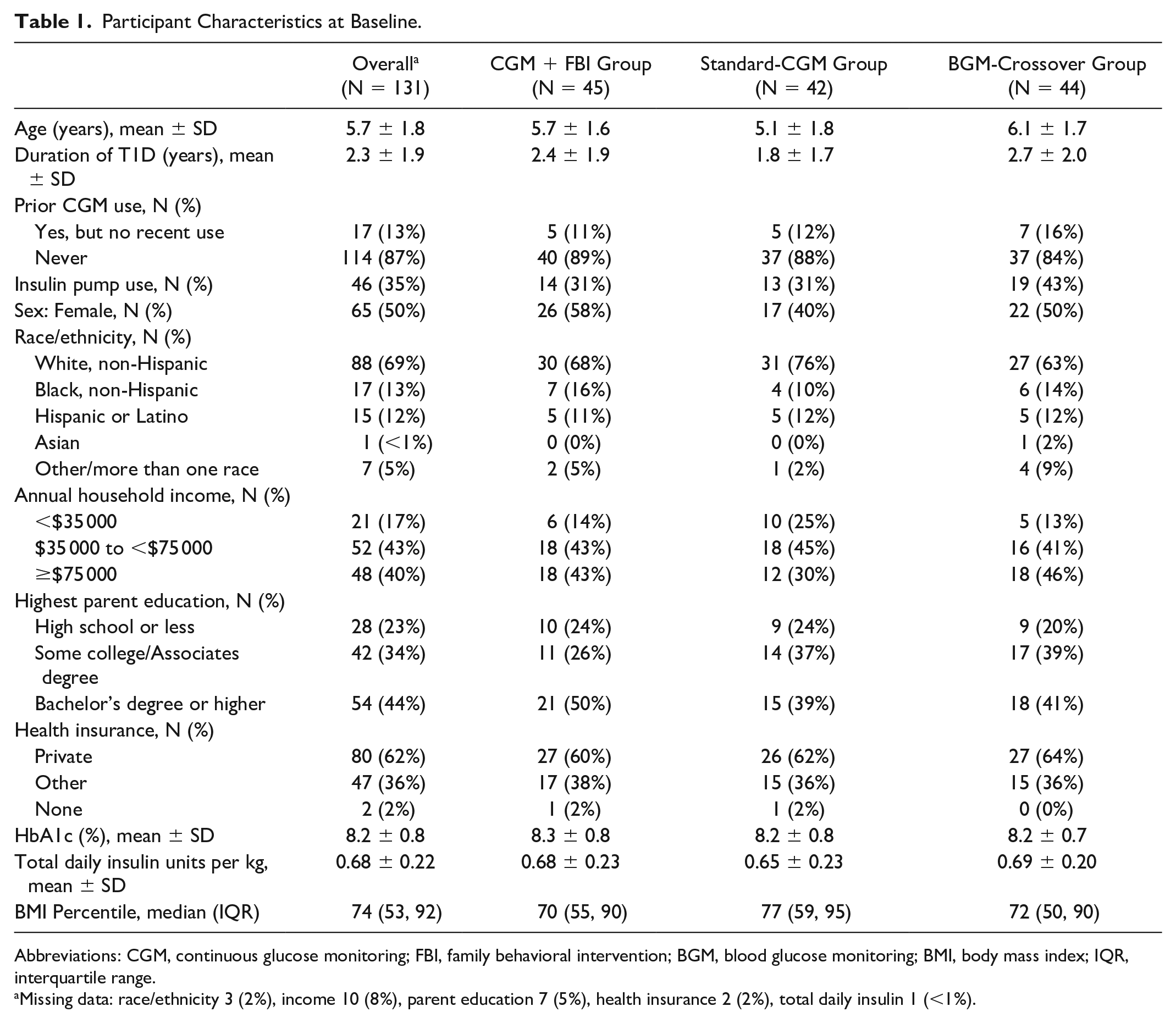
Abbreviations: CGM, continuous glucose monitoring; FBI, family behavioral intervention; BGM, blood glucose monitoring; BMI, body mass index; IQR, interquartile range.
Missing data: race/ethnicity 3 (2%), income 10 (8%), parent education 7 (5%), health insurance 2 (2%), total daily insulin 1 (<1%).
Glycemic Outcomes in the Standard-CGM Group and CGM+FBI Group
Time in target range (70-180 mg/dL) was low at baseline and did not significantly change over the study. The mean time in target range was 37% ± 12% at baseline and 41% ± 12% at 52 weeks for the CGM+FBI Group and 41% ± 10% at baseline and 44% ± 13% at 52 weeks for the Standard-CGM Group. Neither change was statistically significant within groups (P-value baseline vs 52 weeks >.05 for both groups) nor was there a difference between treatment groups at 52 weeks (P = 0.82) (Table 2, Figure 2). Similarly, there were no statistically significant changes in mean glucose or HbA1c from baseline to 52 weeks during the study period in either group.
Glycemic Outcomes in the CGM Groups.
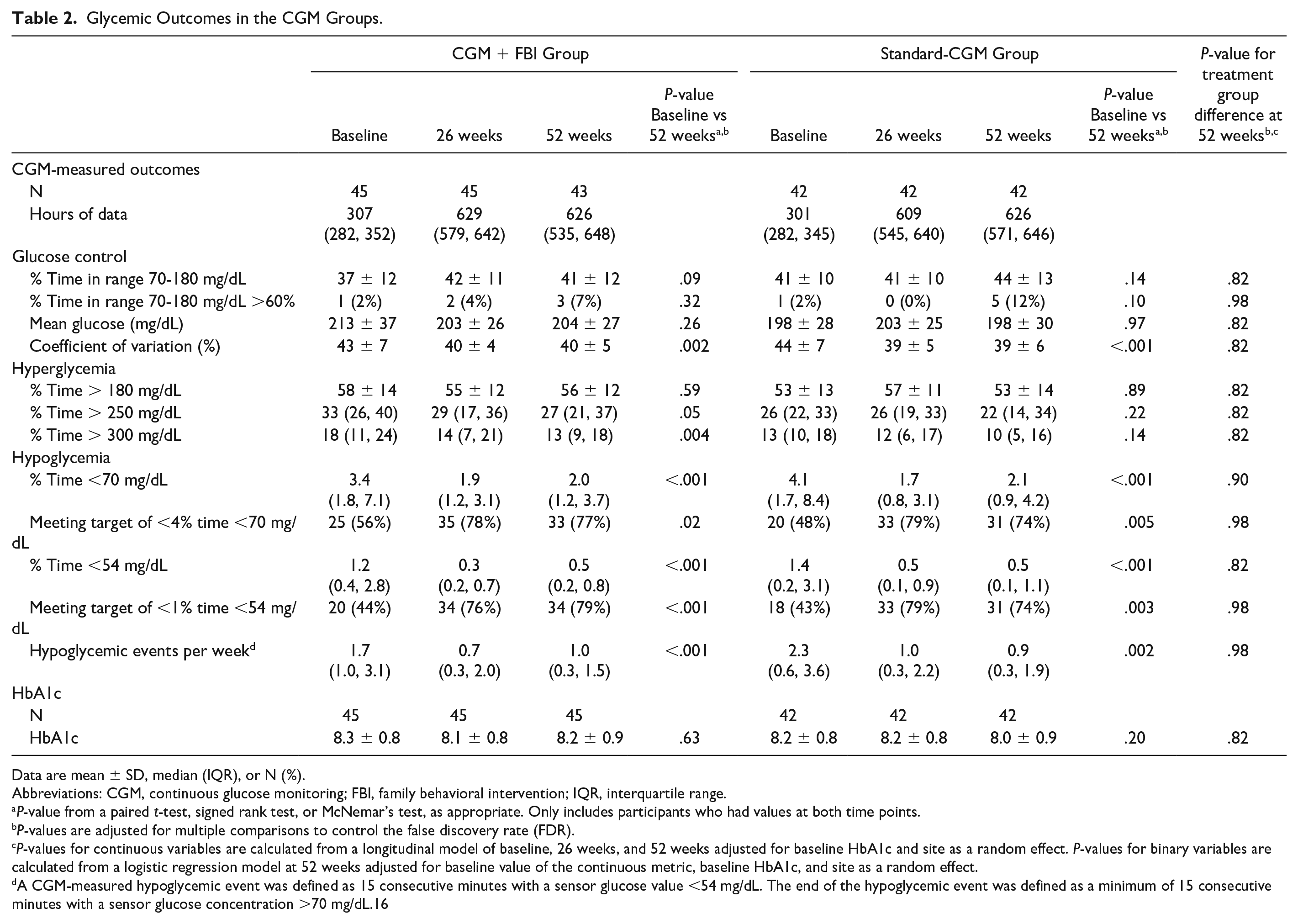
Data are mean ± SD, median (IQR), or N (%).
Abbreviations: CGM, continuous glucose monitoring; FBI, family behavioral intervention; IQR, interquartile range.
P-value from a paired t-test, signed rank test, or McNemar’s test, as appropriate. Only includes participants who had values at both time points.
P-values are adjusted for multiple comparisons to control the false discovery rate (FDR).
P-values for continuous variables are calculated from a longitudinal model of baseline, 26 weeks, and 52 weeks adjusted for baseline HbA1c and site as a random effect. P-values for binary variables are calculated from a logistic regression model at 52 weeks adjusted for baseline value of the continuous metric, baseline HbA1c, and site as a random effect.
A CGM-measured hypoglycemic event was defined as 15 consecutive minutes with a sensor glucose value <54 mg/dL. The end of the hypoglycemic event was defined as a minimum of 15 consecutive minutes with a sensor glucose concentration >70 mg/dL.16
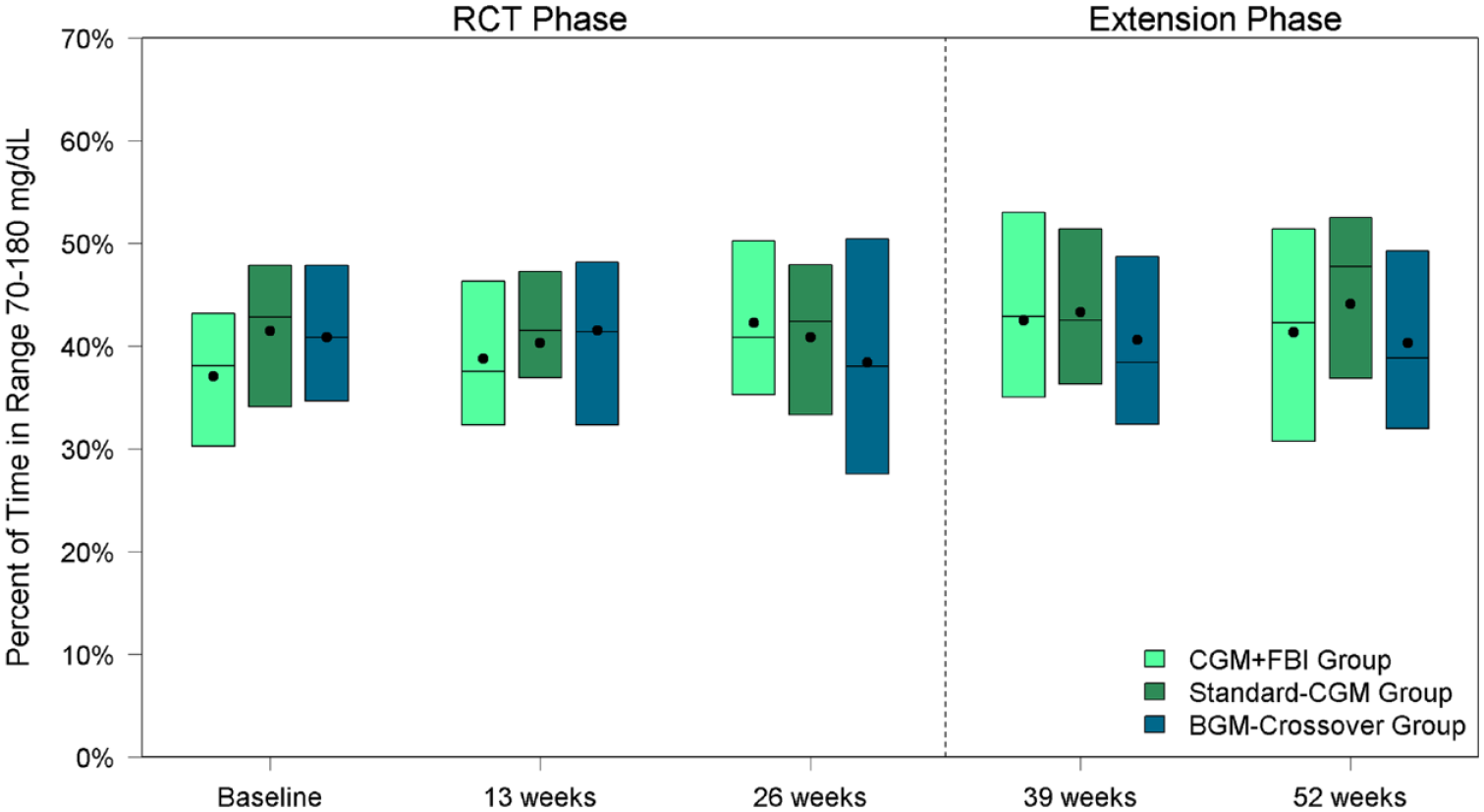
Percent of time in range 70 to 180 mg/dL by treatment group and visit.
In both the Standard-CGM Group and CGM+FBI Group, median percent time <70 mg/dL decreased within six weeks of starting real-time CGM and remained significantly lower than baseline throughout the entire one-year of the study. The reduction in hypoglycemia was from 3.4% (50 min/d) at baseline to 2.0% (29 min/d) at 52 weeks in the CGM+FBI Group (P-value baseline vs 52 weeks <.001) and from 4.1% (59 min/d) at baseline to 2.1% (31 min/d) at 52 weeks in the Standard-CGM Group (P-value baseline vs 52 weeks <.001) (Table 2, Figure 3). There was no treatment group difference in median percent time <70 mg/dL between CGM+FBI Group and Standard-CGM Group at 52 weeks (P = .90). Median percent time <54 mg/dL also decreased within six weeks of starting real-time CGM and remained significantly lower than baseline in both initial CGM groups (P-value baseline vs 52 weeks <.001 for both groups) but was not different between groups at 52 weeks (P = .82). Moreover, median percent time <54 mg/dL was reduced for both daytime (6 am-<10 pm) and nighttime (10 pm-6 am) hours in both groups (Supplemental Table 2).
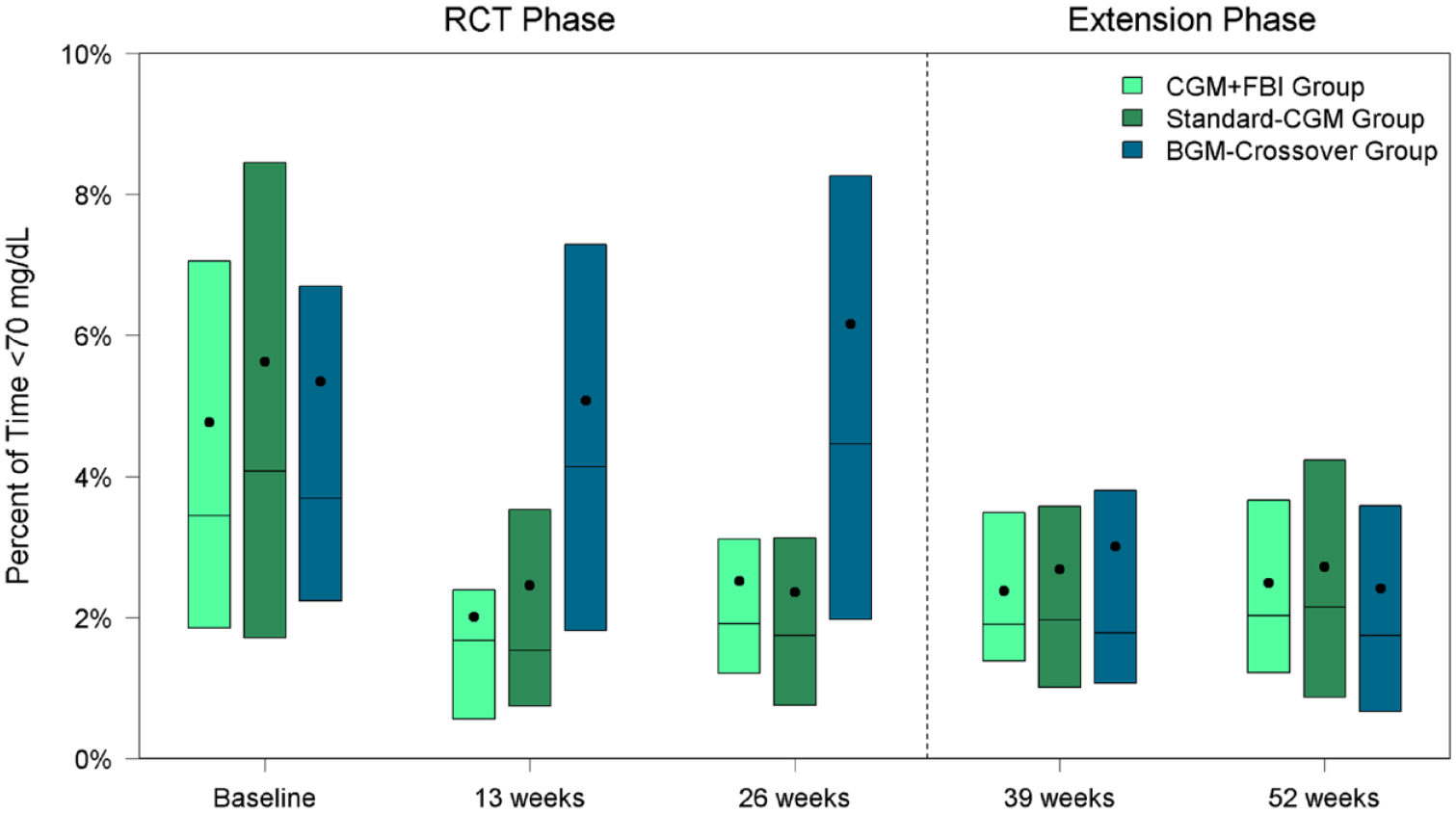
Percent of time <70 mg/dL by treatment group and visit.
There were no statistically significant changes from baseline in mean percent time >180 mg/dL in either group, but there was a reduction in the CGM+FBI Group in median percent time >250 mg/dL (baseline 33% vs 52 weeks 27%, P-value .05) and median percent time >300 mg/dL (baseline 18% vs 52 weeks 13%, P-value .004) (Table 2). The Standard-CGM Group also observed a reduction in these hyperglycemia metrics, but it did not reach the threshold for statistical significance (median percent time >250 mg/dL: baseline 26% vs 52 weeks 22%, P-value .22; median percent time >300 mg/dL: baseline 13% vs 52 weeks 10%, P-value .14). There were no treatment group differences in hyperglycemia metrics between the CGM+FBI Group and Standard CGM Group at 52 weeks.
Glycemic Outcomes in the BGM-Crossover Group
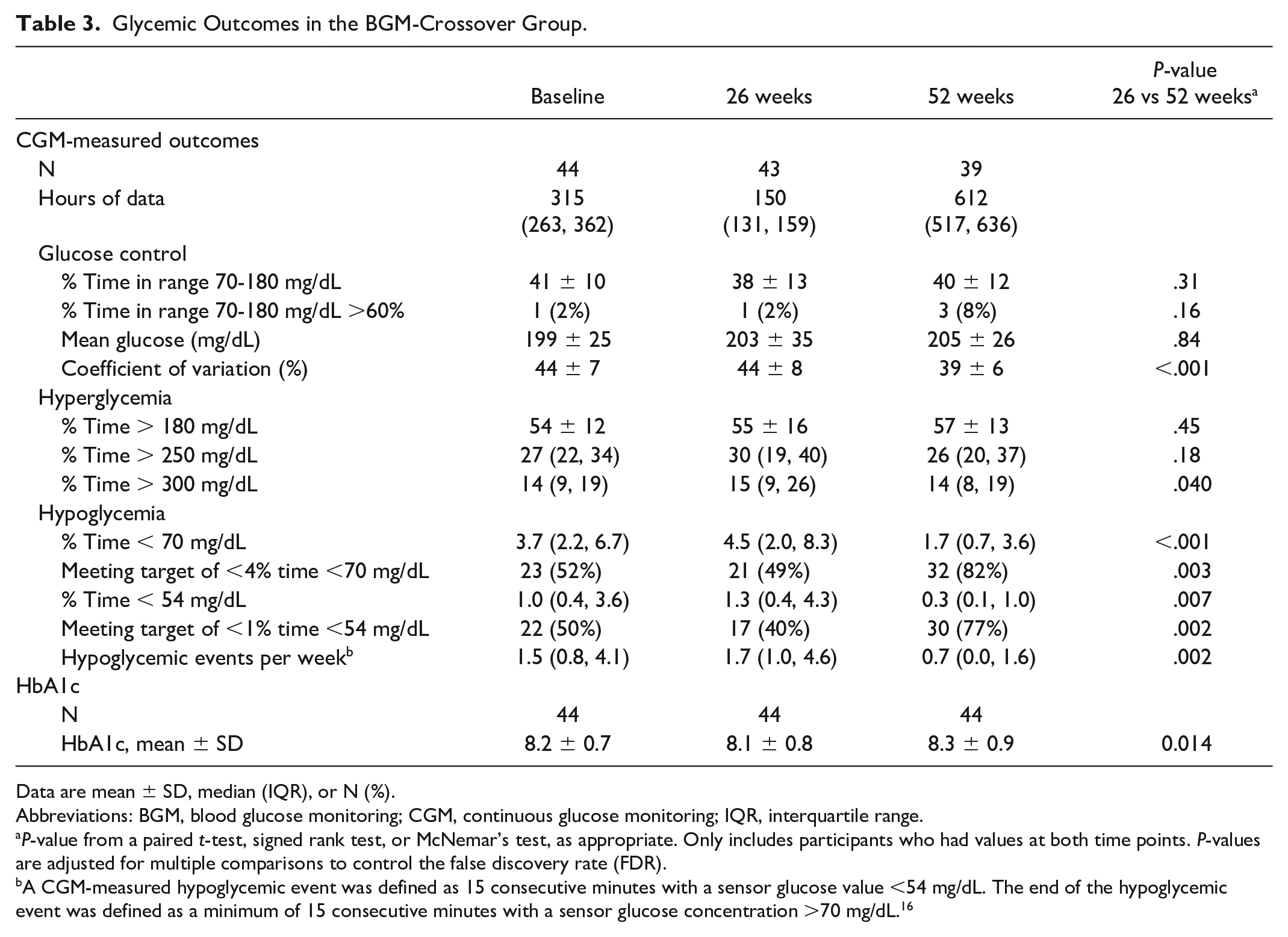
As described above, upon completion of the initial 26-week randomized trial, the BGM-Crossover Group initiated real-time CGM along with the FBI during the 26-week extension phase. CGM use in the BGM-Crossover Group did not result in a significant change in mean time in range from 26 to 52 weeks (38% at 26 weeks vs 40% at 52 weeks, P-value .31) (Table 3, Figure 2). There was also no significant change in mean glucose. Mean HbA1c increased from 8.1% at 26 weeks to 8.3% at 52 weeks (P-value .01).
Glycemic Outcomes in the BGM-Crossover Group.
Data are mean ± SD, median (IQR), or N (%).
Abbreviations: BGM, blood glucose monitoring; CGM, continuous glucose monitoring; IQR, interquartile range.
P-value from a paired t-test, signed rank test, or McNemar’s test, as appropriate. Only includes participants who had values at both time points. P-values are adjusted for multiple comparisons to control the false discovery rate (FDR).
A CGM-measured hypoglycemic event was defined as 15 consecutive minutes with a sensor glucose value <54 mg/dL. The end of the hypoglycemic event was defined as a minimum of 15 consecutive minutes with a sensor glucose concentration >70 mg/dL. 16
On the contrary, median percent time <70 mg/dL decreased from 4.5% (64 min/d) at 26 weeks to 1.7% (25 min/d) at 52 weeks (P-value <.001) and median percent time <54 mg/dL decreased from 1.3% (18 min/d) at 26 weeks to 0.3% (5 min/d) at 52 weeks (P-value .007) (Table 3, Figure 3). Again, the reductions in median percent time <54 mg/dL were observed during both daytime and nighttime hours (Supplemental Table 3).
There were no significant changes in mean percent time >180 mg/dL or median percent time >250 mg/dL, but there was a small reduction in median percent time >300 mg/dL from 15% at 26 weeks to 14% at 52 weeks (P-value .04) (Table 3).
Sensor Use
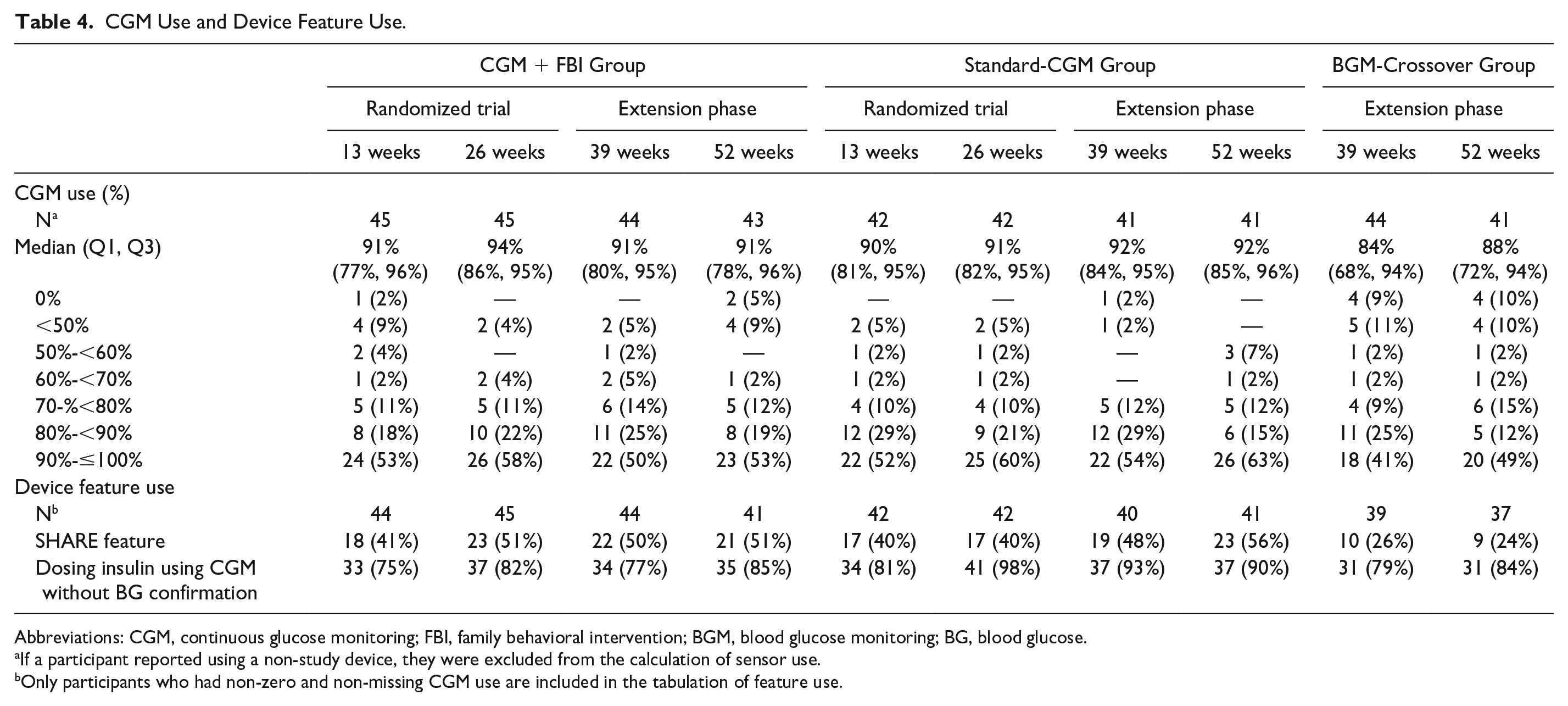
Sensor use was high and sustained throughout the extension phase. During month 12, median sensor use was 91% in the CGM+FBI Group, 92% in the Standard-CGM Group, and 88% in the BGM-Crossover Group (Table 4).
CGM Use and Device Feature Use.
Abbreviations: CGM, continuous glucose monitoring; FBI, family behavioral intervention; BGM, blood glucose monitoring; BG, blood glucose.
If a participant reported using a non-study device, they were excluded from the calculation of sensor use.
Only participants who had non-zero and non-missing CGM use are included in the tabulation of feature use.
Of those who were using the CGM at 52 weeks, 51% of the CGM+FBI Group, 56% of the Standard-CGM Group, and 24% of the BGM-Crossover Group reported using the SHARE feature of the mobile app to allow remote monitoring of real-time CGM data with another person. In addition, 85% of the CGM+FBI Group, 90% of the Standard-CGM Group, and 84% of the BGM-Crossover Group reported using CGM glucose values to dose insulin without fingerstick blood glucose confirmation. In all three groups, the median number of fingerstick blood glucose checks was ~6 per day prior to initiation of real-time CGM and decreased to ~4 per day by the end of the study.
Severe Hypoglycemia and Diabetic Ketoacidosis
During the extension phase, severe hypoglycemic events involving impaired cognition requiring the assistance of another person were reported for two participants in the CGM+FBI Group and four participants in the BGM-Crossover Group. Of these events, two in the CGM+FBI Group and one in the BGM-Crossover Group resulted in seizure or loss of consciousness. No severe hypoglycemic events during the extension phase were reported in the Standard-CGM Group. One participant in each group reported an episode of diabetic ketoacidosis during the extension phase.
Discussion
This one-year study of CGM use in early childhood demonstrated durability of real-time CGM use and a sustained reduction in hypoglycemia throughout the study, an important clinical benefit of CGM use in this age range given the frequency of hypoglycemia, risk of severe hypoglycemia events, and potential for long-term impact on cognitive development.3,10,17 However, CGM use was not associated with improved time in target glucose range or HbA1c.
The inability of real-time CGM to improve time in range parallels results from former trials of families with young children with T1D carried out by the DirecNet research group, using earlier and less accurate CGM devices.1,18 Even in those early studies, parents appeared to primarily use CGM to avoid hypoglycemia even at the expense of spending time in the hyperglycemic range. Despite improvements in CGM technology, taken together these data indicate that different or additional interventions are needed to help families not only reduce hypoglycemia, but to more effectively reduce time in hyperglycemic ranges.
Furthermore, the findings that children in all groups maintained >50% time in hyperglycemia (>180 mg/dL) throughout the entire 52-week study period are consistent with data from the T1D Exchange Clinic Registry,19,20 which reveal high mean HbA1c and a low percentage of young children achieving glycemic targets; only 24% of those two to less than six years achieve HbA1c <7.5% and only 7% achieve HbA1c <7.0%. Notably, the American Diabetes Association and International Society of Pediatric and Adolescent Diabetes have lowered the HbA1c target to <7.0% 21 based on emerging evidence that exposure to hyperglycemia negatively impacts central nervous system structure and function.21-27 Therapeutic advances, such as automated insulin delivery systems, can improve time in range in younger and older children with T1D by diminishing both hyper- and hypoglycemia through algorithms working 24 hours a day and only requiring intermittent intervention by caregivers.28-31 Additional research is needed to inform content focused on simultaneously addressing fear of hypoglycemia in caregivers and proactively treating or preventing hyperglycemia in their young children.
In the setting of standardized education, real-time CGM wear remained high throughout the extension phase for all groups in this population of late adopters to diabetes device technology who had either previously discontinued or were naïve to CGM. In fact, CGM use in this study was far above what has been previously reported using earlier CGM systems in young children with T1D.8,18 The DirecNet group performed the prior large study in this age group and found that only 41% of children were wearing CGM at least six days a week at six months, 1 declining further to 33% at 12 months. 18 Improvements in CGM (eg, fewer required fingersticks, remote monitoring capabilities, smaller device size) that reduce the burden and enhance the benefits of use for parents (eg, less worry, improved sleep) 11 likely contributed to 80% to 95% of young children wearing CGM at least six days a week during the extension phase and are mirrored in reports of real-world use.6,7 The high CGM wear rates over 52 weeks in this study are important, as consistent CGM wear is a critical component of advancing diabetes technologies including automated insulin delivery systems. Moreover, the durability of CGM wear underscores the importance of trained personnel to provide effective and long-lasting education, a valuable tool for future generalizability of device use in young children with T1D.
This study was strengthened by the involvement of 14 centers throughout the United States who recruited a large, racially, ethnically, and socioeconomically diverse cohort of young children with T1D and maintained a high retention rate over one full year. Notably, our cohort uniquely included a large portion of youth not using insulin pumps and without private health insurance. The results suggest that with standardized education, even late adopters to CGM who previously declined use of or lacked access to CGM can durably use diabetes technology. Hence, this intervention may be generalizable to the greater population of young children with T1D. This is of key importance to ensure that young children from structurally disadvantaged families have access to the glycemic benefits of both CGM and automated insulin delivery systems.
Due to the rapid pace of CGM advances, factory-calibrated systems with less complicated insertion requirements became commercially available in the United States during the study; however, these were not available to participants. Hence, the study was limited by use of older technology requiring twice daily fingerstick calibration, and clinical outcomes may have been more favorable with the use of newer systems. CGM devices were supplied by the study, removing barriers of insurance coverage and paperwork often experienced by families and diabetes clinical teams. This, in addition to the run-in eligibility criteria of at least 200 hours of masked CGM wear time over a two- to three-week period and screening HbA1c of 7.0% to 9.9%, may limit the generalizability of findings.
In summary, reduction in hypoglycemia and high rates of CGM use in young children with T1D were sustained over 12 months of CGM wear; however, improvements in time in target range and HbA1c were not achieved. Real-time CGM is a vital component of diabetes care in young children with T1D. Further advances in diabetes technology, behavioral supports, and education in diabetes management in young children are needed to shift the paradigm away from avoiding hypoglycemia at the expense of hyperglycemia to optimizing time in range.
Supplemental Material
sj-docx-1-dst-10.1177_19322968221084667 – Supplemental material for Long-term Continuous Glucose Monitor Use in Very Young Children With Type 1 Diabetes: One-Year Results From the SENCE Study
Supplemental material, sj-docx-1-dst-10.1177_19322968221084667 for Long-term Continuous Glucose Monitor Use in Very Young Children With Type 1 Diabetes: One-Year Results From the SENCE Study by Michelle A. Van Name, Lauren G. Kanapka, Linda A. DiMeglio, Kellee M. Miller, Anastasia Albanese-O’Neill, Persis Commissariat, Sarah D. Corathers, Kara R. Harrington, Marisa E. Hilliard, Barbara J. Anderson, Jennifer C. Kelley, Lori M. Laffel, Sarah A. MacLeish, Brandon M. Nathan, William V. Tamborlane, R. Paul Wadwa, Steven M. Willi, Kristen M. Williams, Kupper A. Wintergerst, Stephanie Woerner, Jenise C. Wong and Daniel J. DeSalvo in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
Abbreviations
BGM, blood glucose monitoring; CGM, continuous glucose monitoring; FBI, family behavioral intervention; HbA1c, hemoglobin A1c; SENCE, Strategies to Enhance New CGM use in Early childhood; T1D, type 1 diabetes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M.A.V.N., L.G.K., L.A.D., K.M.M., A.A.-O., P.C., S.D.C., K.R.H., M.E.H., B.J.A., J.C.K., S.A.M., B.M.N., W.V.T., K.M.W., K.A.W., S.W., and J.C.W. have no disclosures to report. L.M.L. reports grants and personal fees from Dexcom, outside the submitted work. R.P.W. reports grants from Dexcom, Eli Lilly, and Tandem Diabetes Care and personal fees from Dompe and Tandem Diabetes Care outside the submitted work. S.M.W. reports personal fees from Roche Diagnostics and Boehringer Ingelheim, outside the submitted work. D.J.D. reports research support from Insulet and personal fees from Dexcom outside the submitted work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by the Leona M. and Harry B. Helmsley Trust, Dexcom Inc provided nonfinancial support.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
