Abstract
Background:
Managing glucose levels during exercise is challenging for individuals with type 1 diabetes (T1D) since multiple factors including activity type, duration, intensity and other factors must be considered. Current decision support tools lack personalized recommendations and fail to distinguish between aerobic and resistance exercise. We propose an exercise-aware decision support system (exDSS) that uses digital twins to deliver personalized recommendations to help people with T1D maintain safe glucose levels (70-180 mg/dL) and avoid low glucose (<70 mg/dL) during and after exercise.
Methods:
We evaluated exDSS using various exercise and meal scenarios recorded from a large, free-living study of aerobic and resistance exercise. The model inputs were heart rate, insulin, and meal data. Glucose responses were simulated during and after 30-minute exercise sessions (676 aerobic, 631 resistance) from 247 participants. Glucose outcomes were compared when participants followed exDSS recommendations, clinical guidelines, or did not modify behavior (no intervention).
Results:
exDSS significantly improved mean time in range for aerobic (80.2% to 92.3%, P < .0001) and resistance (72.3% to 87.3%, P < .0001) exercises compared with no intervention, and versus clinical guidelines (aerobic: 82.2%, P < .0001; resistance: 80.3%, P < .0001). exDSS reduced time spent in low glucose for both exercise types compared with no intervention (aerobic: 15.1% to 5.1%, P < .0001; resistance: 18.2% to 6.6%, P < .0001) and was comparable with following clinical guidelines (aerobic: 4.5%, resistance: 8.1%, P = N.S.).
Conclusions:
The exDSS tool significantly improved glucose outcomes during and after exercise versus following clinical guidelines and no intervention providing motivation for clinical evaluation of the exDSS system.
Introduction
Type 1 diabetes (T1D) is characterized by the autoimmune destruction of pancreatic beta cells, resulting in a lack of endogenous insulin production. 1 People with T1D must dose exogenous insulin subcutaneously through multiple daily injections (MDI) or by insulin pump. 2 Continuous glucose monitors (CGMs) help people with T1D monitor their glucose levels in real time and alert users when they reach dangerous levels. 3 Physicians and diabetes educators oftentimes recommend that their patients adjust their insulin doses based on CGM responses to carbohydrate intake and physical activity. However, these recommendations are typically provided once every 3 months. Personalized recommendations are typically not provided to patients more often than this. Maintaining stable glucose levels can be challenging for people with T1D, especially during spontaneous forms of physical activity/exercise. 4 Regular exercise has been shown to improve outcomes in the long term, but it also frequently leads to low glucose (glucose < 70 mg/dL), which causes significant acute complications.5,6 It is crucial to develop effective strategies for managing glucose levels during exercise for people with T1D.
Managing insulin dosing and carbohydrate intake before, during, and after physical activity can present significant challenges. Prior studies have demonstrated that individuals with T1D often exhibit highly variable responses to different forms of exercise. 7 For instance, resistance-based exercise often elicits a significant increase in catecholamine secretion, leading to a surge in hepatic glucose production. 8 The increase in hepatic glucose production may overcompensate for the increased muscle glucose utilization rate during heavy resistance exercise and may cause a rise in blood glucose level, particularly if done fasted. 9 Conversely, during aerobic exercise, the sustained high demand for glucose by peripheral tissues can surpass counterregulatory mechanisms, resulting in a decrease in blood glucose levels. 7 Prolonged aerobic exercise oftentimes results in large drops in glucose levels and poses increased risk for hypoglycemia. 10 Many individuals with T1D may avoid exercise due to fear of low glucose. 11
A decision support tool is a software system that can provide personalized recommendations to help people with T1D manage their glucose levels. 12 It can integrate data from wearable sensors to provide insulin, carbohydrate, and exercise recommendations throughout the day. 13 Although several systems have been developed to help people adjust their basal insulin, calculate mealtime insulin boluses, or recommend carbohydrates to maintain safe glucose throughout the day12,14-17, few tools exist that can provide exercise-specific treatment recommendations. 18
Clinical guidelines are often used to provide recommendations around exercise in people with T1D.19,20 They comprise heuristics, established by experts in the field, that provide recommendations before, during, or after anticipated exercise by considering basic contextual data like CGM or insulin dosing data. A recent example, proposed in Moser et al, 21 presents recommendations such as consuming carbohydrates, adjusting insulin boluses, delaying exercise, or terminating exercise prematurely based on CGM levels. These guidelines are often difficult for patients to follow as they can be complicated. Some decision support systems have integrated these types of guidelines into smartphone apps to make it easier for patients to follow the recommendations. 22 However, clinical-based guidelines may not be suitable for all people as they do not account for individual differences in insulin sensitivity, diet and exercise responses that can have a major impact on glucose levels during and following exercise. 23
Digital twins are mathematical model representations of real-world phenomena. Digital twin-based tools can use personalized models to provide more individualized and effective treatment recommendations. Digital twins in diabetes can be designed using metabolic simulators which have been developed to model glucose dynamics in people with type 1 diabetes.24-27 These simulators are typically designed using ordinary differential equations (ODE) to model how carbohydrates, insulin, and other hormones impact glucose dynamics in metabolism. Some simulators also include impacts of exercise on glucose dynamics.27,28 Data-driven digital twins have been used in combination with coaching and medication interventions to help reduce HbA1c in people with type 2 diabetes. 29 In this study, we propose a new exercise decision support tool framework (exDSS) based on Ordinary Differential Equation (ODE) based metabolic twin models. 30 that may be used to provide personalized treatment recommendations prior to either aerobic or resistance exercise. By integrating data from CGMs, insulin pumps, meal logs, and wrist-mounted heart rate monitors, exDSS is designed to provide safe and effective insulin, carbohydrate, and behavioral recommendations prior to exercise to help people with T1D exercise more safely and avoid low glucose (<70 mg/dL). We evaluate whether exDSS can be used to improve glucose outcomes in an in slico population of people with T1D by reducing time in low glucose during and following exercise compared with (1) no intervention and (2) consensus clinical guidelines around exercise.
Methods
In slico study participants were represented by a previously validated metabolic model 27 and an extension to this model that includes the impact of exercise on this metabolic model shown in Supplemental Figure 1. In slico study participants were given meals and exercise scenarios acquired from a large, free-living exercise study from the T1Dexi study data set (Supplemental Material 1). Heart rate from the T1Dexi study participants was used to represent exercise in the in slico study participants using wrist-worn fitness sensors (Verily, Mountain View, CA). Each participant from the T1Dexi data set was matched to a digital twin from an exercise-enabled version of the OHSU metabolic simulator 30 based on insulin sensitivity and weight at the start of each exercise session 31 (Supplemental Material 2). It is important to match digital twins with real-world T1Dexi study participants based on insulin sensitivity and weight so that the virtual patients’ glucose dynamics behave as similarly as possible to the real-world person. An example of how well the digital twins matched the real-world glucose is shown in Figure 1. The interventions listed in Supplemental Table 1 and Figures 2 to 4 were evaluated on the digital twin in the time window of 90 minutes before the start of exercise until 1 hour postexercise (Figure 5). An optimal intervention for the digital twin was selected by exDSS based on which intervention minimized the sum of the low blood glucose index (LBGI) and the high blood glucose index (HBGI) during and following exercise (Supplemental Materials 3-4). The participant then followed the optimal recommendation provided by exDSS during the exercise session. Participants repeated all exercise scenarios under two other arms of the study: (1) when participants took no intervention and (2) when participants took an intervention based on clinical guidelines (Supplemental Materials 5). The primary outcome measure for this study was the percent time that glucose was below range (TBR; glucose <70 mg/dL) during and up to 1 hour from the end of aerobic and resistance exercise sessions. Secondary outcome measures included the percent time in target range (TIR; 70-180 mg/dL), and low blood glucose index (LBGI) plus HBGI. We hypothesized that participants following recommendations from exDSS would spend less TBR compared with when participants (1) did not follow an intervention prior to exercise and (2) followed clinical guidelines on exercise. The primary and secondary outcome measures were assessed from the start of exercise to 1 hour after the end of exercise. Statistical significance was calculated using linear mixed effects modeling, assuming a random effect of participant. P-values were adjusted for multiple comparisons using the Benjamini-Hochberg procedure. 32 In the mixed effects linear regression model, exDSS was a binary fixed effect parameter (0 = did not use exDSS, 1 = used exDSS) while the participant ID was the random effect parameter across multiple exercise sessions.
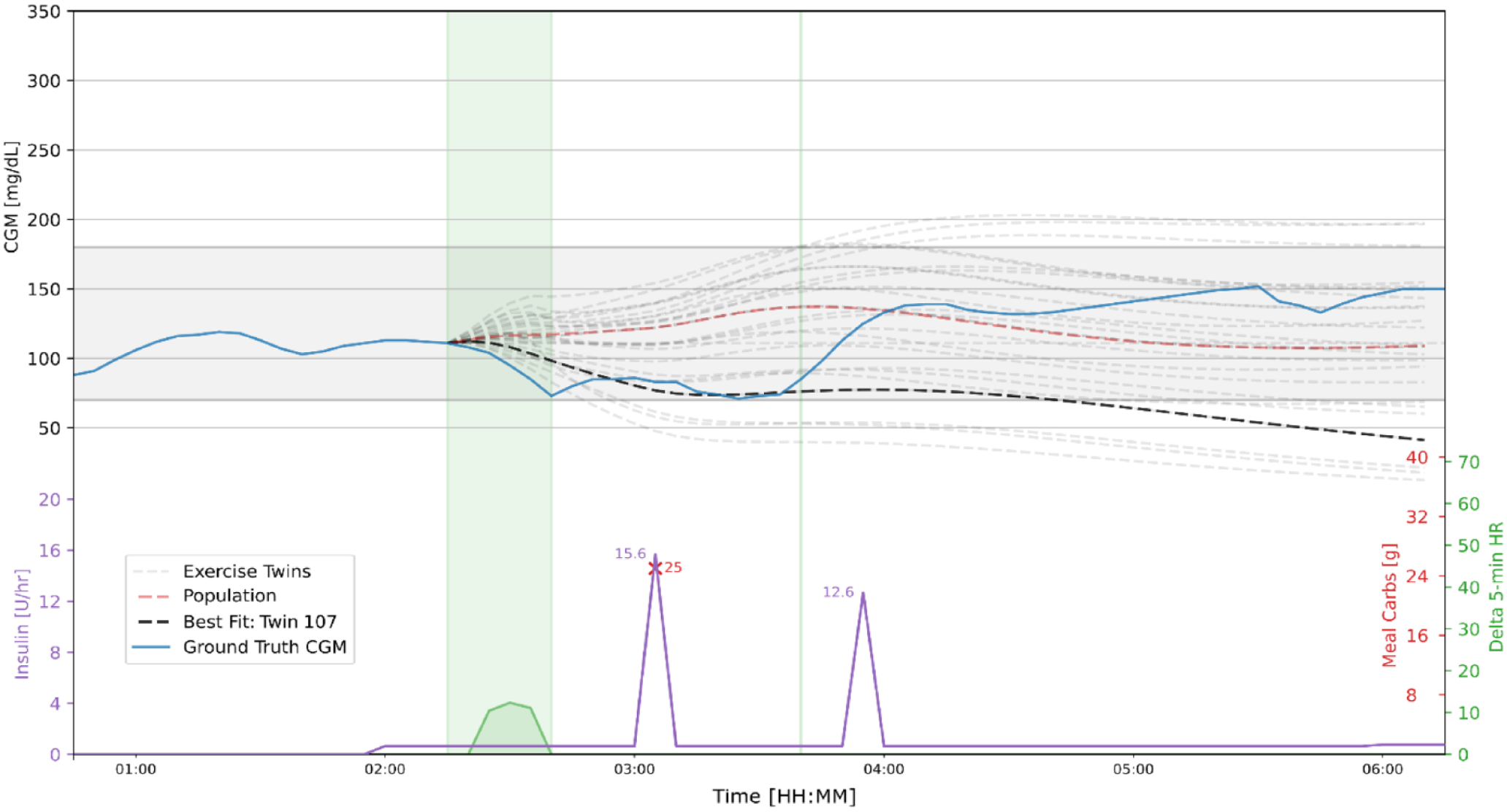
Example of digital twin fit during aerobic exercise (shaded green area on the right). Top panel is CGM during exercise. Blue trace is real-world data from a T1Dexi participant; note that hypoglycemia occurred postexercise. Digital twin candidates are light gray dashed. Population model is shown by dashed red line. Best twin (black dashed) is the twin most closely matching real-world CGM during the 90 minutes following the start of exercise. Bottom green trace shows how heart rate increases during exercise. Carbohydrate intake is shown as red X’s with amount of carbohydrates in grams. Insulin infusion rates are shown in purple.
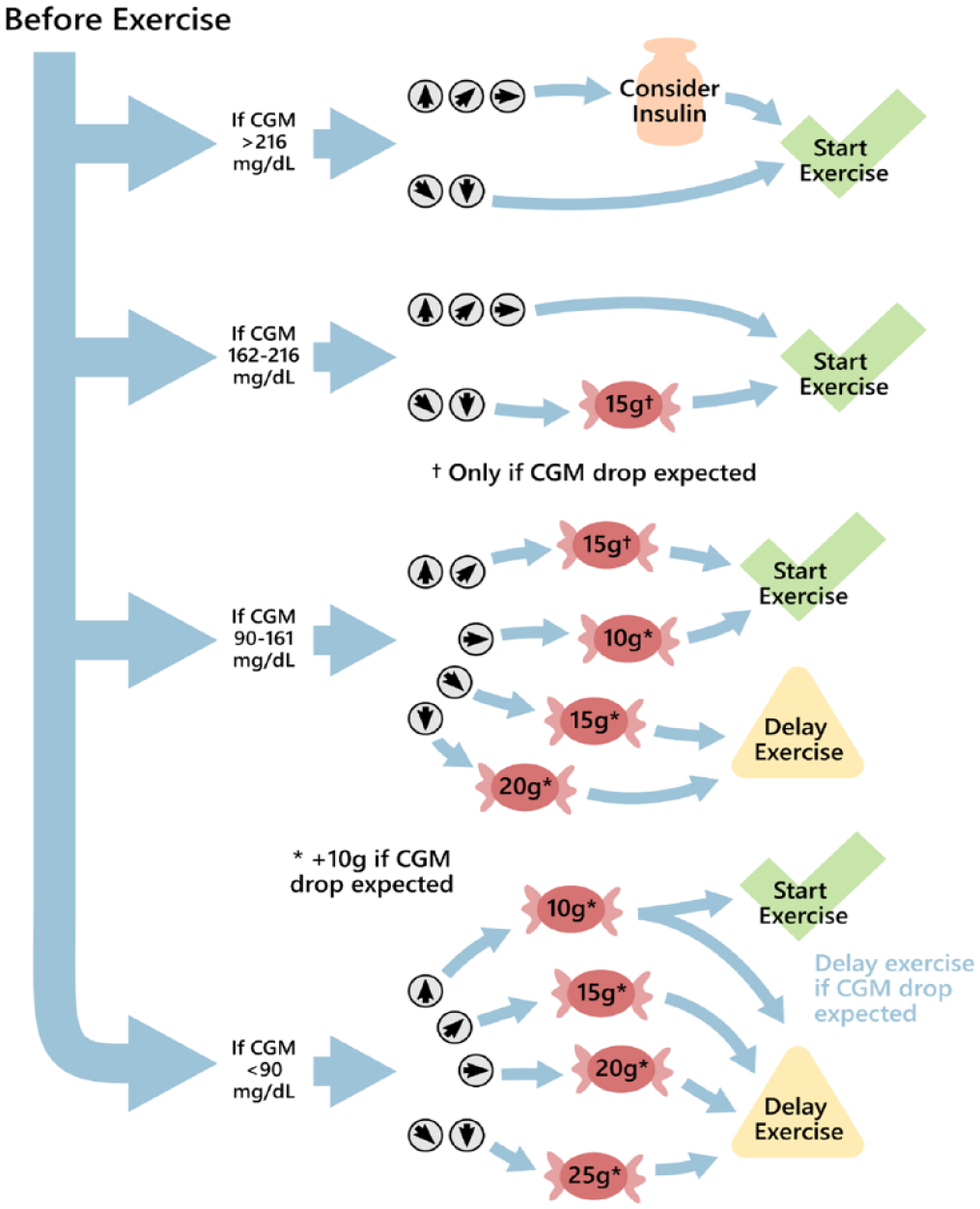
Moser et al clinical guidelines provide recommendations prior to exercise. Clinical guideline recommendations are based on CGM values and CGM trend arrows. 21 CGM trend arrows are represented by small gray circles with arrows, while red candy symbols indicate recommended carbohydrate intake. The carbohydrate amount is specified within each red circle. In the case of an expected CGM drop, such as during aerobic exercise, 10 g of carbohydrates may be added to the recommendations indicated by “*,” while 15 g may be added to the recommendation indicated by “†.” Simulated participants may be prompted to either start exercise, or delay exercise by 20 minutes. A single recommendation was provided via this flowchart 45 minutes before exercise.
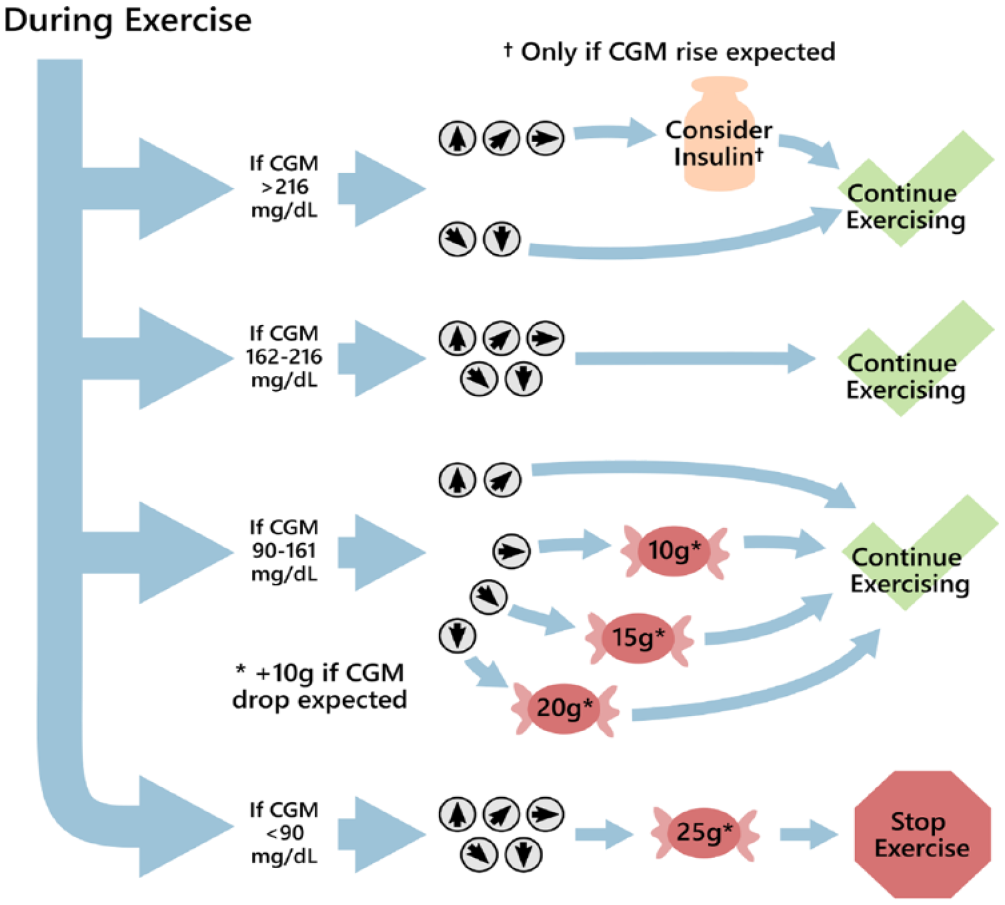
Moser et al clinical guidelines provide recommendations during exercise. Recommendations are based on CGM values and trend arrows, as per Moser et al. 21 CGM trend arrows are depicted by small gray circles with arrows, while red candy symbols indicate recommended carbohydrate intake. The amount of carbohydrates is specified in grams within each red circle. If an expected CGM drop occurs, such as during aerobic exercise, 10 g of carbohydrates may be added to the recommendations indicated by “*.” Simulated participants were prompted to continue exercising for the intended duration or halt exercise immediately based on their glucose levels. A single recommendation was provided via a flowchart 15 minutes after the start of exercise. † Indicates that insulin is recommended only if a drop in glucose is expected during exercise.
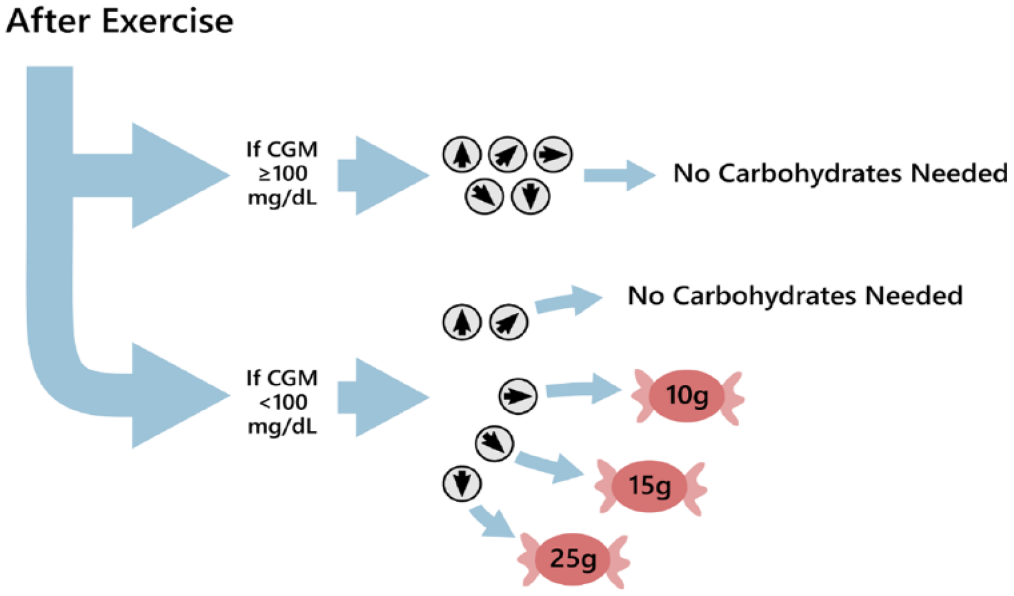
Moser et al. clinical guidelines provide recommendations after exercise. Recommendations are based on CGM values and trend arrows, as per Moser et al. 21 CGM trend arrows are depicted by small gray circles with arrows, while red candy symbols indicate recommended carbohydrate intake. The amount of carbohydrates is specified in grams within each red circle. A single recommendation was provided via this flowchart 15 minutes after stopping exercise.
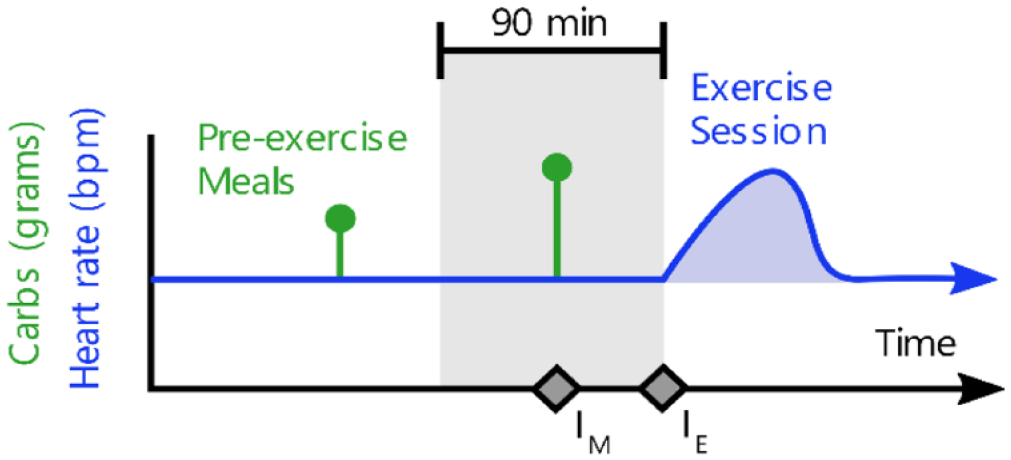
exDSS provides treatment recommendations before an anticipated exercise session. A schematic trace of average heart rate is shown in blue, while reported meals are indicated by the green impulses. Treatment recommendations are provided for any meals consumed within 90 minutes prior to the start of exercise (gray region), indicated by the point . At the start of exercise, indicated by the point, simulated participants receive another treatment recommendation. No recommendations are provided during exercise or after, and no recommendations are provided for meals outside of the 90-minute pre-exercise window.
Results
Primary and secondary outcome measures are provided for aerobic (Table 1) and resistance exercise (Table 2). Across T1Dexi study exercise sessions, exDSS significantly reduced TBR compared with the no intervention arm for both aerobic (mean 5.1% vs 15.1%, no intervention, P < .0001) and resistance exercise (mean 6.6% vs 18.2% no intervention, P < .0001). Following the clinical guidelines resulted in a comparable reduction in percent TBR relative to the no intervention arm (4.5%, P < .001), and resistance exercise (8.1%, P < .001) and these reductions were not significantly different than the exDSS arm.
Aerobic Exercise Glucose Outcomes.
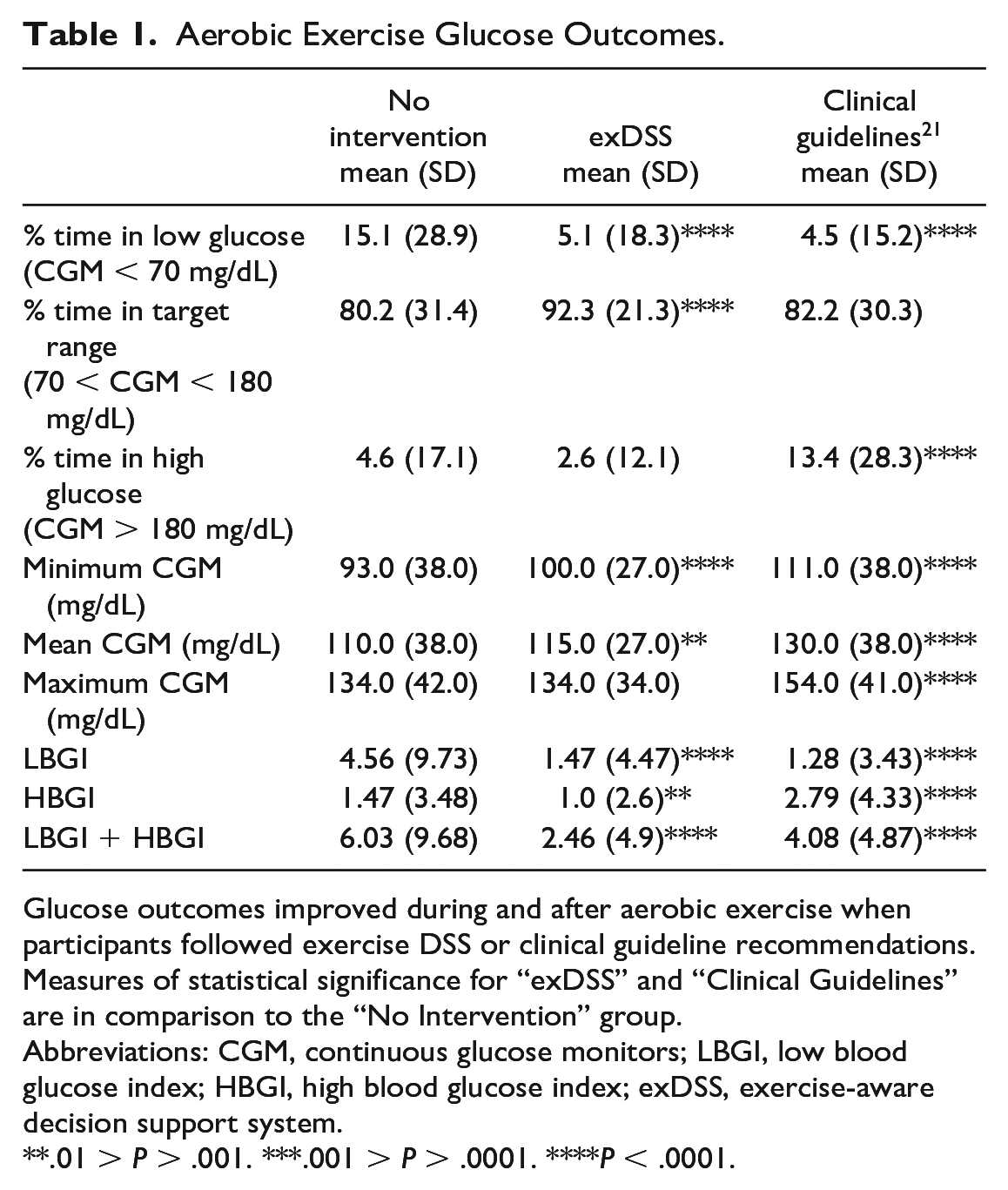
Glucose outcomes improved during and after aerobic exercise when participants followed exercise DSS or clinical guideline recommendations. Measures of statistical significance for “exDSS” and “Clinical Guidelines” are in comparison to the “No Intervention” group.
Abbreviations: CGM, continuous glucose monitors; LBGI, low blood glucose index; HBGI, high blood glucose index; exDSS, exercise-aware decision support system.
.01 > P > .001. ***.001 > P > .0001. ****P < .0001.
Resistance Exercise Glucose Outcomes.
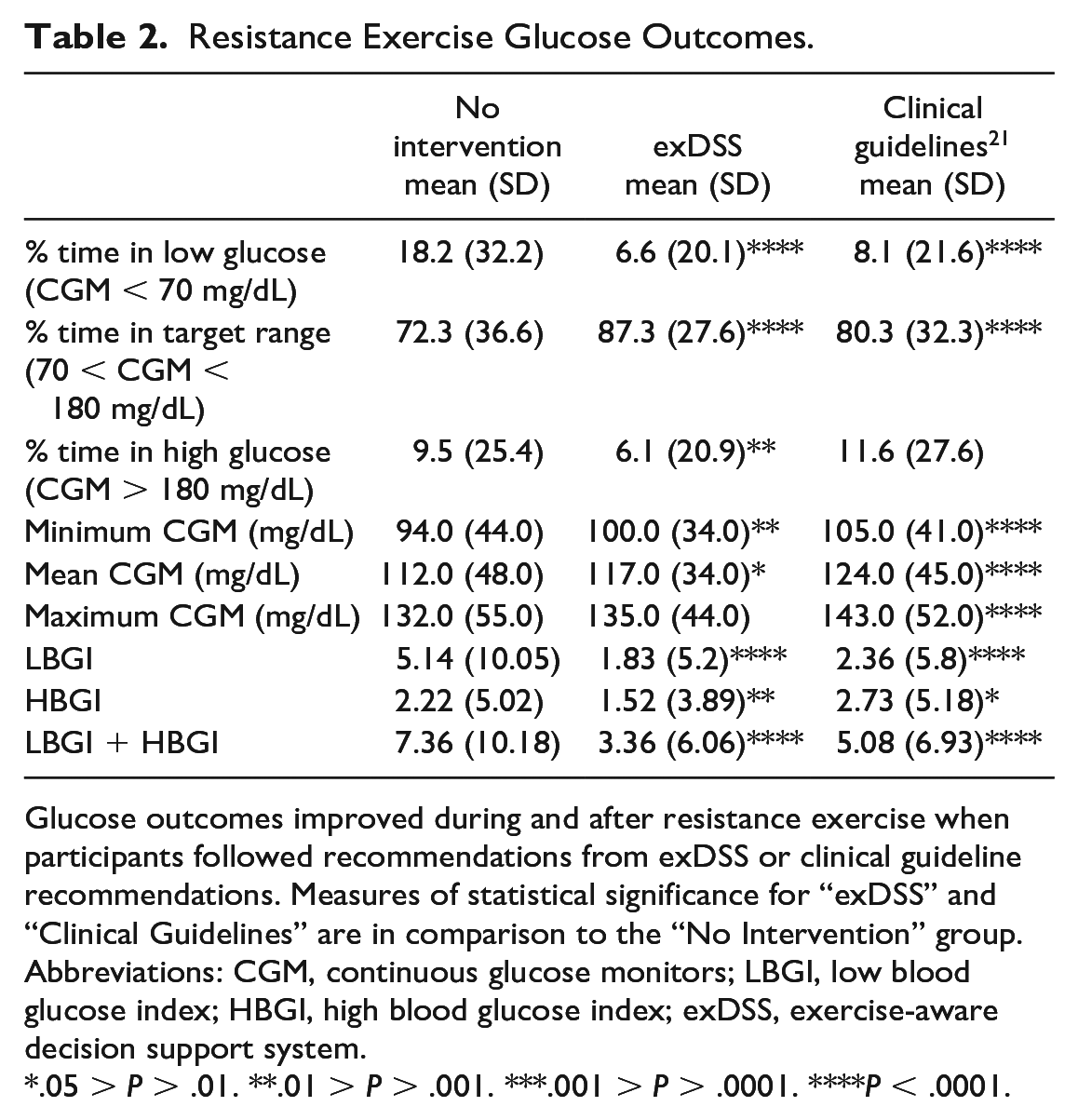
Glucose outcomes improved during and after resistance exercise when participants followed recommendations from exDSS or clinical guideline recommendations. Measures of statistical significance for “exDSS” and “Clinical Guidelines” are in comparison to the “No Intervention” group.
Abbreviations: CGM, continuous glucose monitors; LBGI, low blood glucose index; HBGI, high blood glucose index; exDSS, exercise-aware decision support system.
.05 > P > .01. **.01 > P > .001. ***.001 > P > .0001. ****P < .0001.
Participants who followed the exDSS recommendations had increased mean percent TIR from 80.2% to 92.3% (P < .0001) for aerobic exercise, and from 72.3% to 87.3% (P < .0001) for resistance exercise. Participants who followed the clinical guidelines also had increased TIR compared with the no intervention group for resistance exercise (80.3%, P < .0001), but not for aerobic exercise (82.2%, P = N.S.). Compared with the no intervention case, participants following the clinical guidelines had higher minimum CGM values (aerobic: 111 vs 93 mg/dL, P < .0001; resistance 105 vs 94 mg/dL, P < .0001), maximum CGM (aerobic: 154 vs 134 mg/dL, P < .0001; resistance 143 vs 132 mg/dL, P < .0001), and mean CGM (aerobic: 130 vs 110 mg/dL, P < .0001; resistance 124 vs 112 mg/dL, P < .0001). Participants who used exDSS had similar maximum CGM values for both types of exercise compared with the no intervention group (aerobic: 134 mg/dL, P = N.S.; resistance: 135 mg/dL, P = N.S). Participants using exDSS also showed a statistically similar percent time above range (TAR) compared with the no intervention group for aerobic exercise (2.6% vs 4.6%, P = N.S.) and a decrease for resistance exercise (6.1% vs 9.5%, P = 0.007).
Figure 6 shows that use of exDSS helped to keep virtual participants’ mean glucose during and following exercise within the safe glucose region (70-180 mg/dL). Participants who followed the exDSS recommendations experienced less variation in mean glucose during and after exercise such that the 95% confidence interval for all exercise sessions were closer to the safe glucose range (aerobic: 71.8-155.5 mg/dL, resistance: 67.5-181.9 mg/dL), as compared with following no treatment recommendations (aerobic: 53.8-168.9 mg/dL, resistance: 49.6-203.9 mg/dL). Participants who followed clinical guideline recommendations also had an increase in mean glucose for both types of exercise, such that the 95% confidence interval for all exercise session were closer to the safe glucose range (aerobic: 79.0-189.9 mg/dL, resistance: 69.7-209.8 mg/dL), compared with following no treatment recommendations.
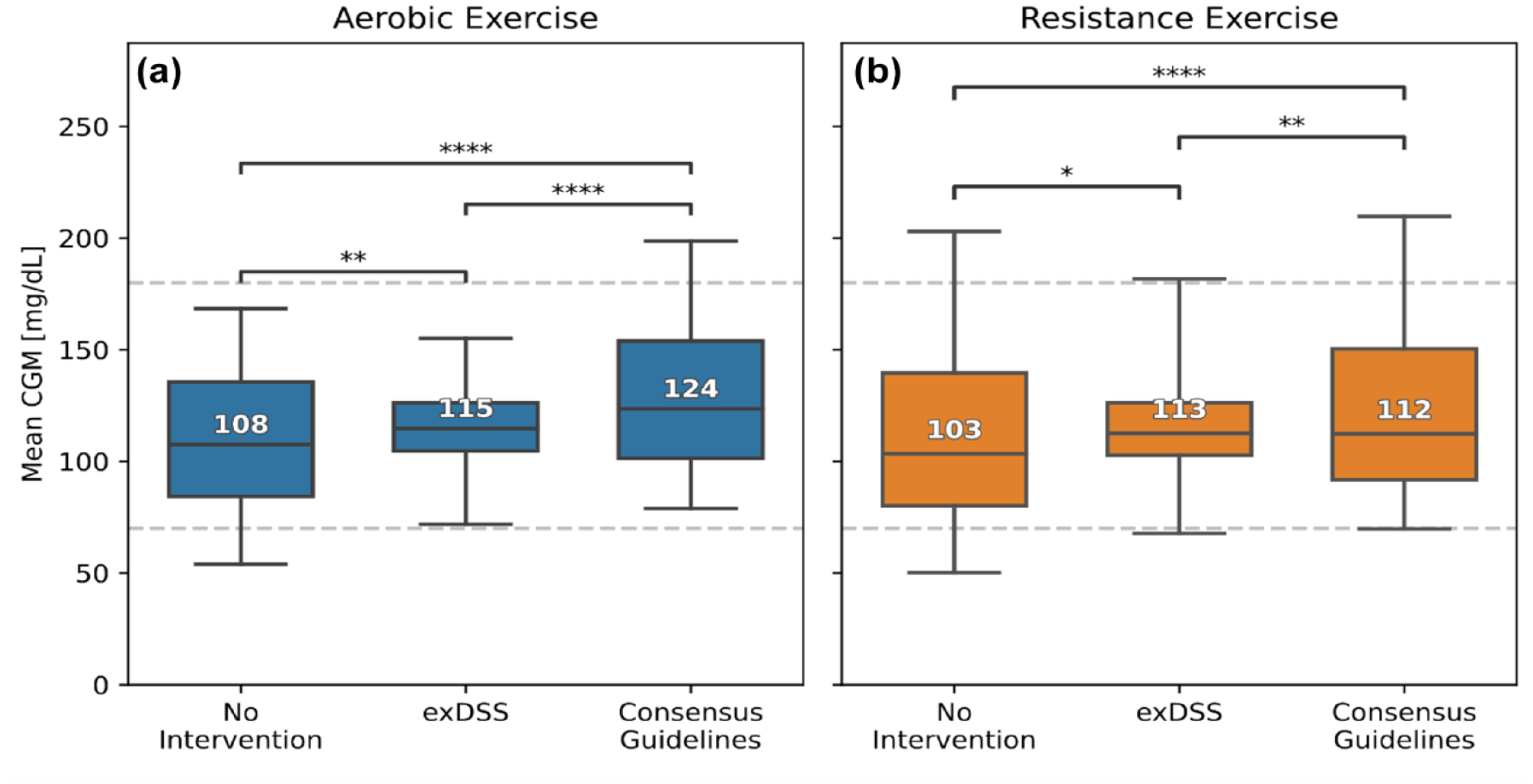
Participants who followed exDSS recommendations had higher mean glucose during exercise, with the 95% confidence intervals falling within the target glucose range of 70–180 mg/dL (dashed line). The mean CGM during exercise plus 1 hour of the postexercise recovery period is shown for the T1Dexi study’s aerobic exercise (a) and resistance exercise sessions (b). The “Clinical Guidelines” group followed recommendations from Moser et al. 21 The median and interquartile ranges of all recorded video sessions with complete data in the T1Dexi data set are indicated by boxes and the 95% confidence interval is indicated by whiskers. The median for each group is quantified by the numbered values. Brackets indicate statistical significance between intervention groups by mixed effects modeling (*.05 > P > .01. **.01 > P > .001. ***.001 > P > .0001. ****P < .0001).
Figure 7 shows that LBGI + HBGI was significantly reduced for participants who used exDSS compared with the no intervention arm (aerobic: 0.8 vs 2.6, P < .0001; resistance: 1.0 vs 3.6, P < .0001) and the clinical guidelines intervention (aerobic: 2.5, P < .0001; resistance: 2.7, P < .0001). Participants who adhered to the clinical guidelines also had a decreased LBGI + HBGI compared with the no intervention arm (aerobic: 2.5 vs 2.6, P < .0001; resistance: 2.7 vs 3.6, P < .0001), but the reduction was less when participants followed the clinical guidelines compared with the exDSS recommendations.
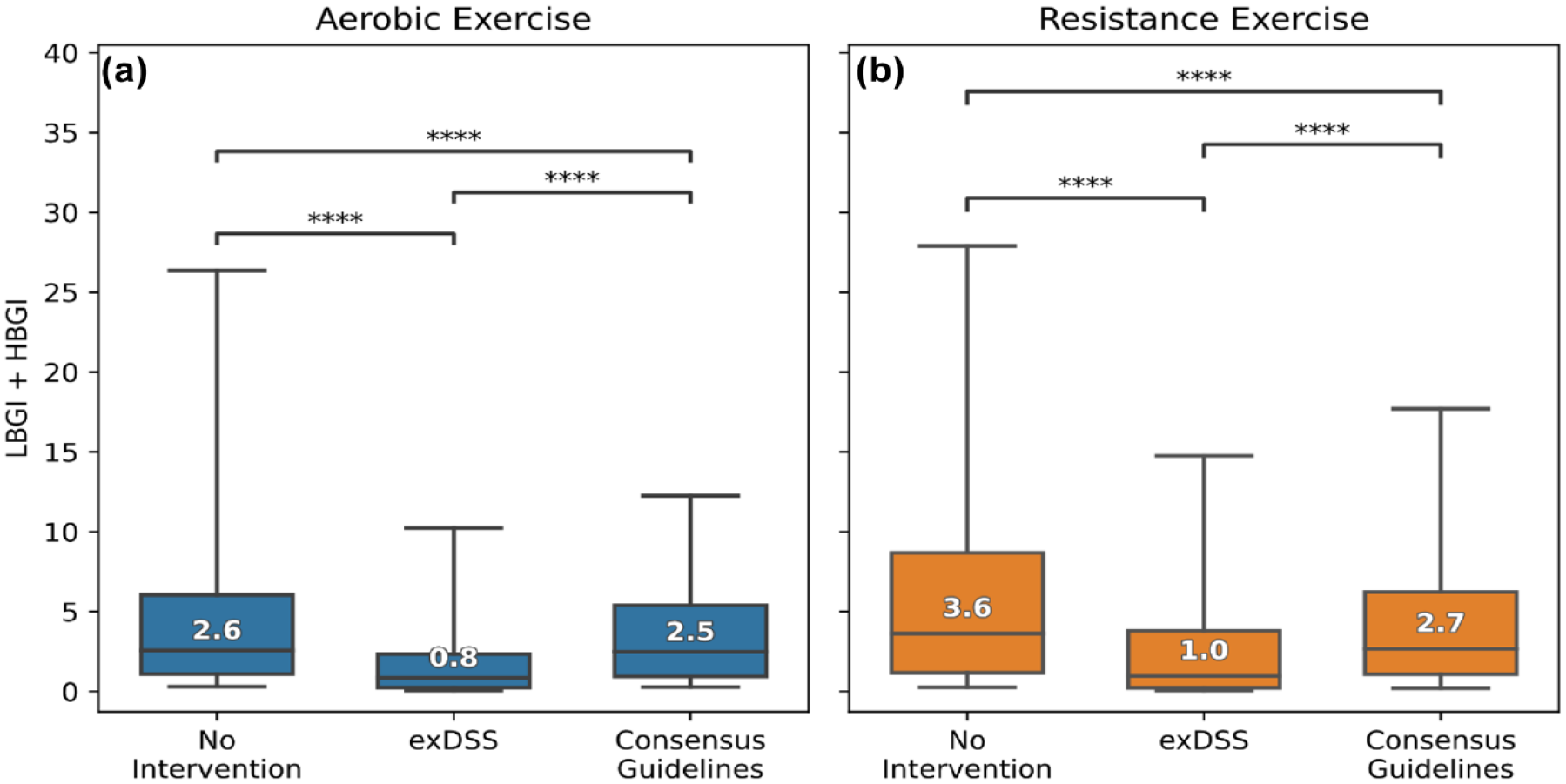
Participants who followed exDSS recommendations had the lowest LBGI + HBGI during and after exercise, and therefore the best glucose outcomes, compared with the no intervention arm and the clinical guidelines arm. Low blood glucose index plus high blood glucose index (LBGI + HBGI) during and 1 hour after the end of exercise are shown for aerobic (a) and resistance exercise (b). The “Clinical Guidelines” group followed recommendations from Moser et al. 21 The median and interquartile ranges of all recorded exercise sessions with complete data in the T1Dexi data set are indicated by boxes, while the 95% confidence interval is indicated by whiskers. Brackets indicate statistical significance between intervention groups by mixed effects modeling (****P < .0001).
Recommendations From exDSS
We examined the frequency that each possible recommendation in Supplemental Table 1 was selected as the optimal recommendation (ropt). Figure 8 shows the number of times each exDSS recommendation was chosen as the optimal recommendation by exDSS during aerobic and resistance exercise. The most frequent recommendations provided by exDSS were similar for both types of exercise. The most selected recommendations were to eat 30 g of carbohydrates immediately prior to exercise (34.4% of aerobic sessions, 34.7% of resistance sessions). This intervention likely explains why glucose levels were higher in the postexercise period compared with the no intervention arm and also likely explains the reduction in TIR for the exDSS arm.
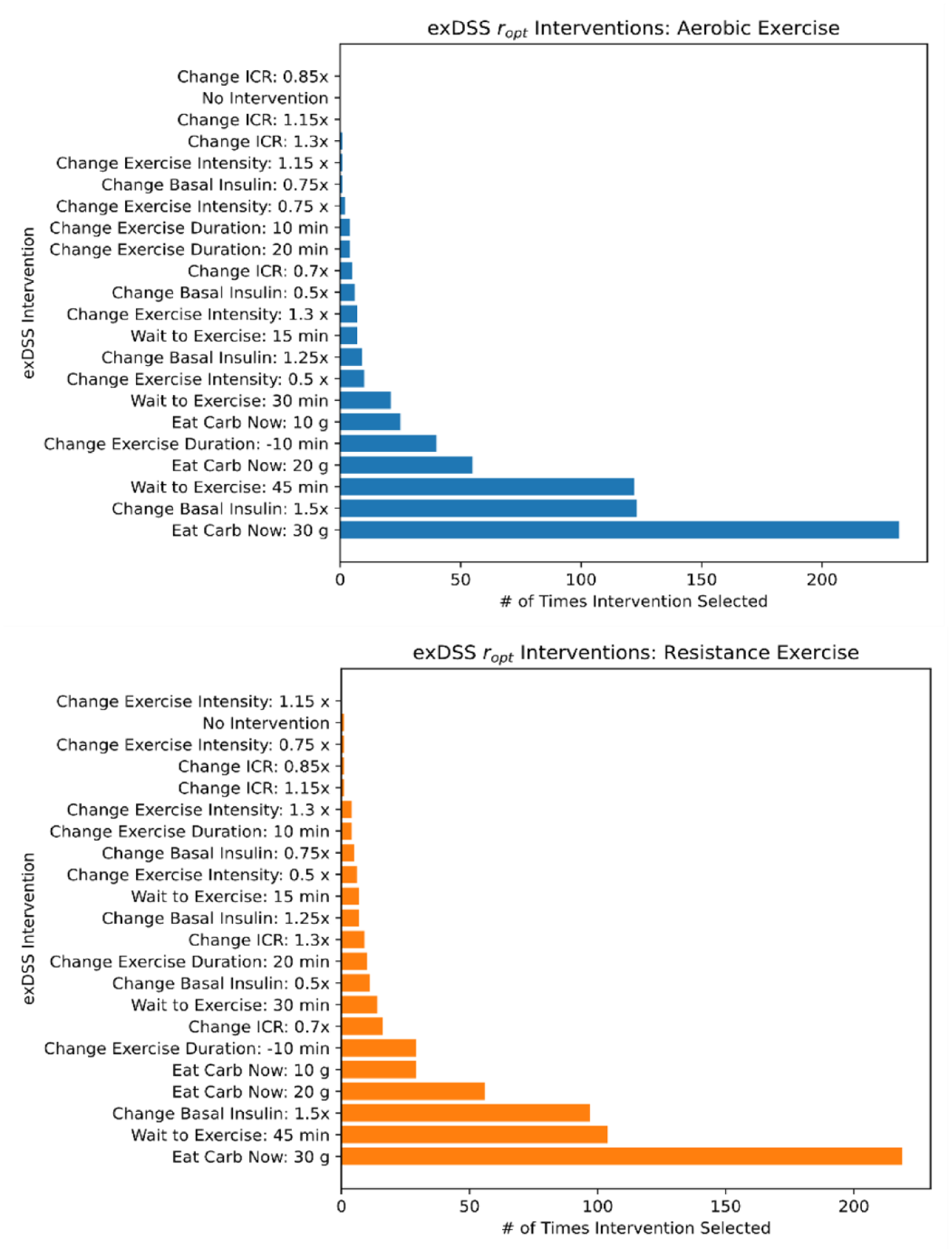
exDSS recommends a variety of glucose-lowering and glucose-raising interventions during and after aerobic (top) and resistance (bottom) exercise. The exDSS recommendations for all aerobic and resistance exercise sessions with complete data in the T1Dexi study data set are summarized. The Y axis represents all available DSS recommendations, while the X axis displays the number of times each intervention was selected by the lowest (LBGI + HBGI) metric in simulation.
The second and third most commonly selected recommendations for both exercise types were increasing the basal insulin infusion rate by 50% (18.2% of aerobic sessions, 15.4% of resistance sessions) and waiting 45 minutes before starting exercise (18.1% of aerobic sessions, 16.5% of resistance sessions). Increasing basal insulin prior to exercise is not typically recommended in clinical guidelines,19,21 and so it was surprising to see this as a common recommendation. However, if a participant were experiencing higher glucose levels prior to exercise, increasing the basal insulin would have the impact of improving the person’s TIR during and following exercise. We used mixed effects analysis to explore whether higher glucose levels and rapidly increasing glucose levels were associated with recommendations to increase insulin prior to start of exercise. Using mixed effects modeling, we observed that recommendations to increase basal insulin were associated with increasing CGM prior to exercise for both aerobic (+0.60 mg/dL*min, P = 0.006) and resistance exercise (+0.49 mg/dL*min, P = 0.041). Recommendations to increase basal insulin were also associated with higher CGM values at the start of exercise for aerobic exercise (+14.5 mg/dL, P = 0.002), but not resistance exercise (+7.0 mg/dL, P = 0.173). This suggests exDSS tends to recommend increased insulin prior to exercise for sessions where participants had increasing CGM trends prior to exercise (aerobic and resistance) and also when glucose was higher at the start of aerobic exercise.
Another common recommendation selected by exDSS was to delay the exercise event by 45 minutes. Delaying exercise may be helpful when meals are consumed shortly before exercise since both glycemia and insulin-on-board (IOB) are typically elevated. Meals with at least 5 g of carbohydrate were common in the 90 minutes preceding exercise in the T1Dexi data set (38.3% of aerobic sessions, 40.6% of resistance sessions). Delaying exercise may have helped to mitigate the impact of higher IOB from these meals. exDSS rarely recommended “No Intervention,” and frequently recommended decreasing the duration of exercise by 10 minutes during both types of exercise (5.9% of aerobic sessions, 4.6% of resistance sessions). Increasing the duration of anticipated exercise was rarely recommended for either aerobic (+10 minutes: 0.6%, +20 minutes: 0.6%) or resistance exercise (+10 minutes: 0.6%, +20 minutes: 1.6%). exDSS more frequently recommended increasing/decreasing the insulin-to carbohydrate ratio for meals before resistance exercise (combined 4.3%) compared with less often for aerobic exercise (combined 0.9%). exDSS rarely recommended increasing or decreasing the exercise intensity by any factor (combined 3.0% of aerobic sessions, 1.7% of resistance sessions).
Exercise Recommendations From Clinical Guidelines
We further investigated which treatments were commonly recommended by the clinical guidelines described in Moser et al 21 before, during, and after bouts of structured exercise. We quantified the number of times each scenario occurred during simulations for aerobic and resistance exercise sessions (Figure 9). Similarly to the exDSS, the clinical guidelines most often recommended that people consume carbohydrates to avoid drops in glucose. Figure 9 shows the typical amount of carbohydrates that were recommended by the clinical guidelines.
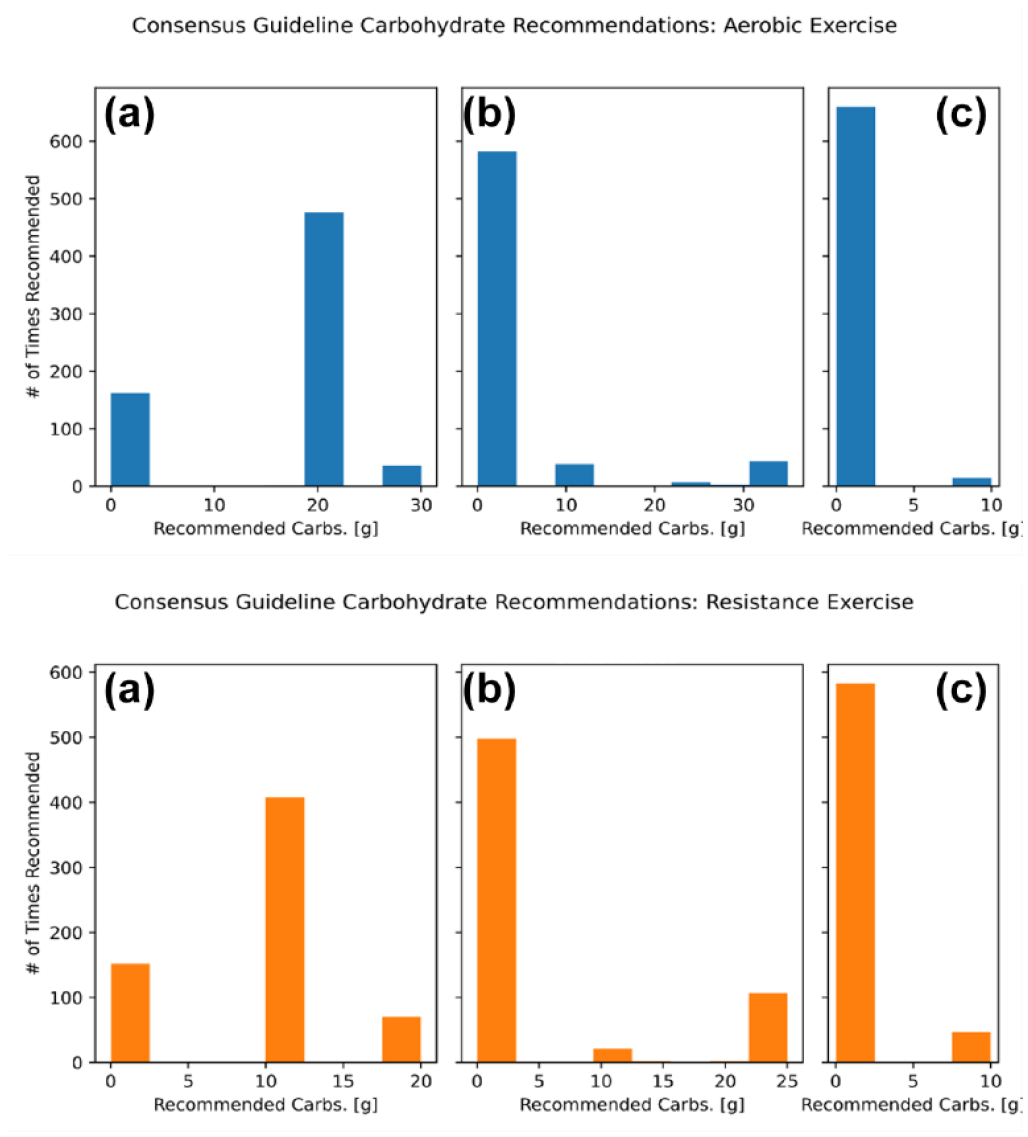
Clinical guidelines most frequently recommended eating carbohydrates before, but not during aerobic (top) and resistance (bottom) exercise. Carbohydrate recommendations provided by Moser et al 21 clinical guidelines are summarized for all aerobic exercise sessions with complete data in the type 1 diabetes exercise initiative (T1Dexi) study data set. Simulated participants were provided with a single recommendation at three separate time points, before exercise (a), during exercise (b), and after exercise (c).
For both exercise types, most in slico participants started exercise with a CGM value between 90 and 161 mg/dL in the 90 minutes prior to the start of exercise (Figure 10). Under these conditions, the clinical guidelines recommend consuming a carbohydrate immediately: typically 20 g before aerobic exercise (70.5% of sessions), and 10 g before resistance exercise (64.8% of sessions). 21 Participants were advised to delay exercise by 20 minutes in only 5.3% of aerobic sessions and 11.1% of resistance sessions.
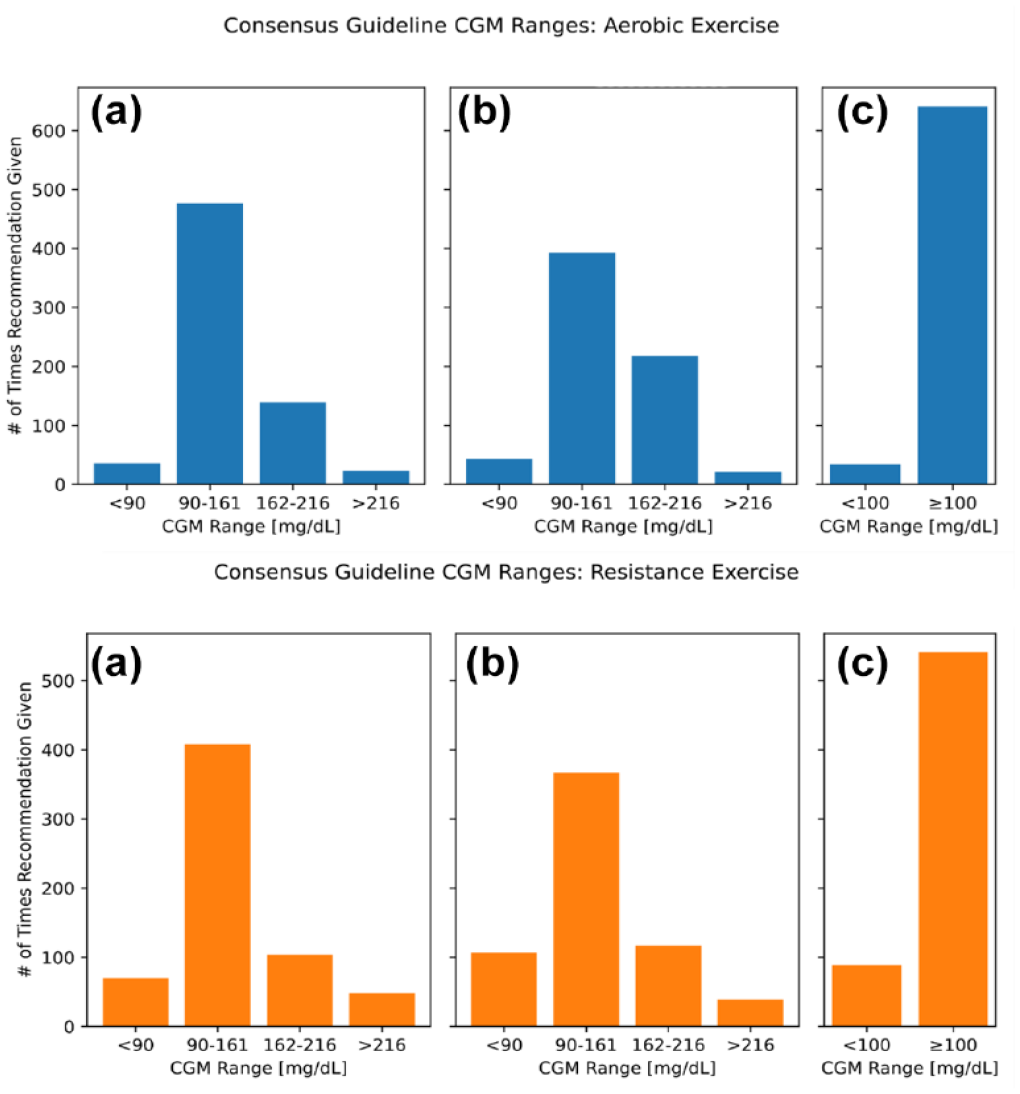
Clinical guidelines most frequently provided recommendations when participants were in the 90 to 161 mg/dL range for aerobic exercise. The CGM values observed at the time of each recommendation are displayed for aerobic exercise sessions. Participants received a single recommendation at three possible time points: before (a), during (b), and after (c) exercise. The CGM ranges are represented along the X-axis for each panel and correspond to the thresholds specified in the clinical guidelines. The Y-axis represents the number of times a CGM was observed at the time of a recommendation.
Most virtual patients received exDSS recommendations when their glucose was in the range of 90 to 161 mg/dL for both aerobic or resistance exercise sessions (Figure 10). Only a small percentage of sessions had virtual patients receive clinical guideline recommendations for carbohydrate intake during aerobic exercise (13.6% of sessions, Figure 9 top panel B) or resistance exercise (21.0% of sessions, Figure 9 bottom panel B). Most participants had CGM levels above 100 mg/dL 15 minutes after stopping exercise when following clinical guidelines (95.0% of aerobic sessions, Figure 10 top panel C versus 85.9% of resistance sessions, Figure 10 bottom panel C). Participants were mostly not advised to eat any extra carbohydrates after exercise (97.8% of aerobic sessions, Figure 10 top panel C vs 92.5% of resistance sessions, Figure 10 bottom panel C).
Discussion
We presented in slico results of a decision support system, exDSS, for managing glucose levels in individuals with T1D during and after aerobic and resistance exercise and compared glucose outcomes for participants who followed exDSS versus those who did nothing versus those who followed clinical guidelines around exercise.
Both exDSS and clinical guidelines from Moser et al 21 significantly reduced the percentage time participants spent in low glucose events during and following exercise compared with the no intervention arm. However, participants who followed the exDSS were also able to significantly increase time in range for both aerobic and resistance exercise, whereas participants who followed the clinical guideline recommendations saw more modest improvements in time in range compared with the no intervention arm, and these improvements were not significant for aerobic exercise. The exDSS has the advantage over other published clinical guidelines in that they are personalized and specific to a given individual’s physiologic needs at the time that the recommendation is given. In this way, the exDSS has the ability to recommend increases basal rate insulin delivery for those participants who have higher glucose levels prior to exercise and/or who may have had higher resistance to insulin. On the contrary, clinical guidelines do not recommend increases in basal insulin prior to the start of exercise.19,21 These types of non-intuitive recommendations may be why participants who followed recommendations from the exDSS had a more substantial increase in time in range compared with following clinical guideline recommendations.
The most common recommendations selected by exDSS included pre-exercise carbohydrates, increased basal insulin infusion levels, or delaying exercise. Most of these recommendations are intuitive with the exception of increasing basal insulin delivery, which would not typically be recommended based on current clinical guidelines. The recommendation to increase insulin prior to exercise was associated with a higher glucose at the start of exercise and also a more rapid rise in glucose at the start of exercise. Nonetheless, the reason for this recommendation is multifaceted and could be due to other factors besides starting CGM and CGM trend including the type of exercise, insulin on board, meal carbs consumed prior to exercise, and the person’s prior response to this type of exercise. Unlike consensus guidelines which have specific CGM and CGM trend cut-off thresholds for different recommendations, the model takes many factors into account thereby leading to conditions when the recommendation is to increase insulin based on what the model predicts will happen. In this way, the exDSS is capable of providing recommendations that are similar to the already established clinical guidelines,19,21 but also can extend beyond these guidelines to be specific for individual physiologic needs.
Virtual participants following exDSS recommendations also had significantly reduced LBGI + HBGI across exercise and postexercise periods compared with no intervention, indicating an improvement in glucose management by avoiding large deviations from a target glucose range. Following clinical guidelines also resulted in a significantly smaller LBGI + HBGI, but the reduction was smaller and P-values were larger compared with exDSS.
Importantly, the recommendations provided by exDSS differed based on whether the participant was doing aerobic versus resistance exercise as shown in Figure 8. While the “eat carbohydrates now 30 g” was the most popular recommendation for both types of exercise, some recommendations were more common for aerobic than resistance exercise and vice versa. For example, increasing the aggressiveness of the carbohydrate ration by 30% of their typical carbohydrate ratio (i.e. deliver 30% more meal insulin) was the seventh most popular recommendation for resistance exercise, but it was much less common (13th most popular recommendation) for aerobic exercise. This makes intuitive sense, since aerobic exercise tends to cause more significant drops in glucose compared with resistance exercise. 33 The different frequency of recommendations for resistance versus aerobic exercise emphasizes the need to have models for the two types of exercise within digital twin models.
There were several limitations of this study. The results are based on in slico simulations and may overestimate expected improvements. A real-world study will be important to demonstrate the benefit of exDSS on improving glucose outcome measures. A second limitation is that the simulation presumed that participants always followed clinical guideline recommendations. This is unlikely given the complexity of the recommendation that the required preparation time to perform some of the recommendations, such as reducing the insulin delivery well before exercise start time. In this way, the results presented here may be considered as an upper bound on expected improvement in glucose outcomes. A third limitation is that it used only data from the T1Dexi study that were using insulin pumps. It did not include people using MDI therapy. The insulin data collected from people using MDI in the T1Dexi data set was not reliable as the insulin usage (i.e., insulin dose and timing) was all self-reported by participants. Furthermore, virtual participants were assumed to follow optimal insulin therapy during simulation, and always adhered to their prescribed insulin-to-carb ratio, correction factor, and basal rate. Further investigation is needed to assess the safety of changing insulin doses in participants who follow suboptimal insulin regimens. A final limitation is that the exercise sessions evaluated in this study were all evaluated on participants exercising during 30-minute exercise videos that were constructed to represent aerobic, resistance or mixed forms of exercise. Future work will need to evaluate exDSS on unstructured exercise sessions of different types and durations.
Conclusions
In conclusion, our study demonstrated that exDSS can provide safe and effective exercise recommendations for people with T1D in slico. Following exDSS recommendations yielded improved glucose outcomes, and in particular less TBR and more TIR during exercise as compared with no intervention and current clinical guidelines. A larger real-world clinical study is necessary to confirm results.
Supplemental Material
sj-docx-1-dst-10.1177_19322968231223217 – Supplemental material for Design and In Silico Evaluation of an Exercise Decision Support System Using Digital Twin Models
Supplemental material, sj-docx-1-dst-10.1177_19322968231223217 for Design and In Silico Evaluation of an Exercise Decision Support System Using Digital Twin Models by Gavin Young, Robert Dodier, Joseph El Youssef, Jessica R. Castle, Leah Wilson, Michael C. Riddell and Peter G. Jacobs in Journal of Diabetes Science and Technology
Footnotes
Abbreviations
AID, automated insulin delivery; CGM, continuous glucose monitoring; exDSS, exercise decision support system; HBGI, high blood glucose index; LBGI, low blood glucose index; MDI, multiple daily injection; T1D, type 1 diabetes; T1Dexi, type 1 diabetes exercise initiative; TAR, time above range; TBR, time below range; TIR, time in range.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PGJ reports grants from the National Institutes of Health, the Leona M. and Harry B. Charitable Trust, the Juvenile Diabetes Research Foundation, Dexcom, and the Oregon Health & Science University Foundation; consultancy fees from the Clinical Data Interchange Standards Consortium; participation as a member of an advisory board for Eli Lilly; and PGJ and JRC report stock options from Pacific Diabetes Technologies, outside the submitted work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Leona M. and Harry B. Helmsley Charitable Trust, 2018PG-T1D001. National Institutes of Health (NIH), through grant from the National Institute of Diabetes and Digestive and Kidney Diseases Grant 1F30DK128914-01.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
