Abstract
Background:
Type 1 diabetes (T1D) is a complex condition requiring constant monitoring and self-management. The landscape of diabetes management is evolving with the development of new technologies. This survey aimed to gain insight into the perceptions and experiences of people with T1D (PWD) and their caregivers on the use of technology in diabetes care, and identify future needs for T1D management.
Methods:
PWD and caregivers (≥18 years) living in five European countries (France, Germany, Italy, Spain, and the United Kingdom) completed an online survey. Data were collected during July and August 2021.
Results:
Responders included 458 PWD and 54 caregivers. More than 60% of PWD perceived devices/digital tools for diabetes management as useful and 63% reported that access to monitoring device data made their life easier. Nearly half of participants hoped for new devices and/or digital tools. While approximately one-third of all PWD had used teleconsultation, perceptions and usage varied significantly between countries and by age (both P < .0001), with the lowest use in Germany (20%) and the highest in Spain (48%). The proportions of PWD contributing to diabetes care costs varied by device and were highest for smart insulin pen users at 83% compared with 44% for insulin pen users and 37% for insulin pump users. One-quarter (24%) of PWD and 15% of caregivers felt they lacked knowledge about devices/digital tools for T1D.
Conclusions:
Most PWD and caregivers had positive perceptions and experiences of new technologies/digital solutions for diabetes management, although improved support and structured education for devices/digital tools are still required.
Introduction
Diabetes affects 60 million people in Europe 1 and type 1 diabetes (T1D) accounts for 10% of all diabetes cases, with prevalence increasing worldwide. 2 Modeling projections indicate that by 2040 there will be a global increase in prevalent T1D cases of 13.5 to 17.4 million (60%-107% higher than in 2021). 3 T1D is a complex condition that requires 24/7 self-management and has a substantial impact on the quality of life. 4 Management of T1D also requires ongoing monitoring and support from health care professionals (HCPs). 4 The landscape of solutions available to people with T1D (PWD) has changed over the past decade due to the development of innovative treatments and the introduction of new technologies, such as continuous glucose monitoring (CGM), automated insulin delivery systems, smart pens, smart phone diabetes applications (apps), and teleconsultation.5-7
The degree of technologization depends on different health care systems (eg, regulatory restrictions, reimbursement by health insurers, and degree of co-payment), the financial resources of a country, availability of diabetes specialists, socioeconomic variables, and personal preferences or barriers regarding diabetes technologies.8-12 In a worldwide survey, approximately 45% of HCPs reported that they cannot rely on their national/regional health care system to cover diabetes technologies, and 56% cannot rely on insurance companies’ reimbursement to cover the cost of diabetes technologies. 11 In addition, HCPs require specific training and resources to be able to prescribe diabetes technology such as insulin pumps and CGM, and distance from the nearest diabetes specialist can be a barrier to people with diabetes being able to access these technologies. 9
Although information on new tools for digital diabetes management is available, more research is needed into PWD’s needs, perspectives, and user experiences of the technical and digital options on offer. 13 Furthermore, although it is known that caregivers play an important role in the treatment of T1D and are the most important people for providing support to PWD, relatively little is known about caregivers’ views and experiences with regard to modern technologies.14,15 From the results of this survey, the experiences and perspectives of PWD and their caregivers living in Western Europe were analyzed to assess the digital health solutions and other services to support diabetes management and identify future needs for improving diabetes management.
Methods
Survey Design
This survey was conducted on the digital social platform, Carenity (a social network for people living with chronic conditions and their caregivers), in the T1D community (PWD and caregivers) in France, Germany, Italy, Spain, and the United Kingdom. The questionnaire was developed by Carenity with experts from the United Kingdom, France, and Germany with different specialisms (clinician, psychologist, or nurse educator). Two PWD, from the United Kingdom and France, shared their experience and views to refine the questionnaire. Data were collected during July and August 2021. The survey contained a variety of questions on the experiences and perceptions of living with T1D. Some questions for caregivers concerned the PWD they cared for and some were about the caregivers themselves.
Participants
Invitations to complete the survey were sent to Carenity users registered as a PWD or as a caregiver of a PWD. A message about the survey with a link was also posted on the Carenity Facebook page and on the Facebook pages of other T1D patient groups. Invitations were also sent to PWD registered in Carenity partners’ panel. Eligibility criteria included adults (male or female aged ≥18 years at the time of inclusion into the survey) living in France, Germany, Italy, Spain, or the United Kingdom, and diagnosed with T1D (PWD) or living with an adult diagnosed with T1D (caregivers). Participants gave their informed consent.
Statistical Analysis
The sample size was 100 respondents in each country (France, Germany, Italy, Spain, and the United Kingdom; soft quotas), 90% PWD and 10% people living with PWD (caregivers). The total expected sample size for the survey was 450 PWD and 50 caregivers.
Data quality checks were performed on all completed questionnaires. If a respondent took <5 seconds per question (totaling ~3 minutes to answer all 37 questions), their questionnaire was excluded from analysis. If a respondent did not answer a question, or provided an irrelevant response in the field marked “Other,” their response on that particular question was also excluded, as were inconsistent responses (such as a response to one question that contradicted the response on another).
As a first step, a detailed descriptive analysis was performed for each question (univariate analysis). For closed-ended questions with qualitative variables, the number of respondents (n) and percentages (%) were calculated; for closed-ended questions with quantitative variables, mean, 95% confidence interval, median, first and third quartiles (p25 and p75), standard deviation, and grouping in classes (n, %) were calculated. Analysis of open-ended questions included grouping by theme, number of respondents and occurrences by themes, and verbatim extracts.
Bivariate descriptive analysis was performed depending on the sample sizes (n ≥30 for each category). To study the impact that variables have on one another and to determine links or dependencies between variables, different hypothesis tests were performed depending on the variable type. For qualitative variables, χ2 independency tests were performed (if the conditions were not met, the Kruskal-Wallis test was performed). For a quantitative and a qualitative variable, Student’s test (t test) to compare two groups (if the conditions were not met, the Wilcoxon-Mann-Whitney test was performed) or the ANOVA (analysis of variance) test to compare more than two groups was used (if the conditions were not met, the Kruskal-Wallis test could be performed). The link between quantitative variables was evaluated by the Pearson’s correlation. The Cochran-Armitage test was used to test whether two qualitative variables are independent, when at least one is an ordinal qualitative variable. Statistical tests were performed once all application conditions are met.
Objectives
Broadly, the aims of this European, real-world survey were to assess PWD and caregiver perceptions, usage, and experiences of devices/digital tools to aid diabetes management. More specifically, the survey was designed to ask by what means glucose was monitored and insulin administered, how useful survey participants found devices/digital tools, and what respondents thought about facilities such as teleconsultation and mobile consulting services. These data were gathered with a view to achieving a greater understanding of the attitudes and lived experiences of PWD and their caregivers, which may help shape future developments in diabetes management and technology.
Results
Responder Profile
Overall, 774 participants consented to taking part in the survey and 637 questionnaires were completed. In total, there were 512 evaluable responders included in the analysis, evenly distributed between the five countries: 458 PWD and 54 caregivers. Just more than half of PWD were female. Mean age at the time of data collection was 47.8 years, with a mean age at diagnosis of 24.4 years (Table 1). Over half of caregivers were female, with a mean age of 43.2 years. The main relationships between caregivers and PWD were couple (42%) or close family (PWD is parent/uncle/aunt: 31%). The level of education of responders ranged from General Certificate of Secondary Education (GCSE) to PhD (or equivalent qualifications outside the United Kingdom); 24% of PWD and 13% of caregivers were educated to GCSE level, 32% and 30% had completed A-levels, and 43% and 56% were educated to bachelor’s degree level or higher.
Responder Profile.

Abbreviations: PWD, people with diabetes; T1D, type 1 diabetes.
Use of Devices and Digital Tools for Diabetes Management
Most PWD responders (64%) used continuous and/or flash blood glucose monitoring (87% of PWD used only one monitoring device), and 3% answered they did not know what they used (Table 1). Caregivers’ answers were similar from their knowledge of the PWD’s care. Two-thirds (64%) of PWD used an insulin pen, 26% an insulin pump, 7% smart insulin pen, and 3% did not know what they used, with similar responses from caregivers (Table 1). The device used for insulin administration differed significantly according to age (P = .04), a difference that appears to be driven mainly by smart pen use, which is more common in younger PWD (Supplemental Figure S1A). Depending on the country, the insulin administration device differed significantly (P < .0001); for example, insulin pump usage ranged from 16% to 23% in Spain, Italy, the United Kingdom, and Germany, whereas 50% of PWD in France used insulin pumps (Supplemental Figure S1B).
Perceptions of Devices and Digital Tools for Diabetes Management
Most PWD (63%) felt that having access to data generated by T1D monitoring devices made their life easier. More than 60% of PWD perceived various devices and digital tools for diabetes management as useful (Figure 1a); for each device/tool, below 10% of PWD reported that they had used it and not found it useful. Continuous and/or flash glucose monitoring was found to be useful by 51% of users and perceived as potentially useful by 28% of nonusers. Smartphone applications for tracking blood glucose were found to be useful in 45% of current/past users and considered as potentially useful in 31% of nonusers. Only one-quarter of PWD had used a smartphone application that recommended insulin dose but, overall, more than two-thirds of PWD thought that they were or could be useful. One-third (35%) of PWD had accessed data sharing software and found it useful, whereas 30% had not but believed it could be useful. One-fifth of PWD had consulted mobile coaching services and found them useful; 42% had not used mobile coaching services but believed they could be useful. Overall, caregivers’ views were comparable to those of PWD; for each device/digital tool, more than half of caregivers perceived them as useful and below 13% of caregivers reported that they had used them but not found them useful; for each device/digital tool, around 20% of caregivers did not know whether they were used by the PWD. Apps for tracking blood glucose, apps that recommend an appropriate insulin dose, and mobile coaching services were less used by older PWD (Figure 1b). PWD who had used these tools but found they were not useful were younger on average.
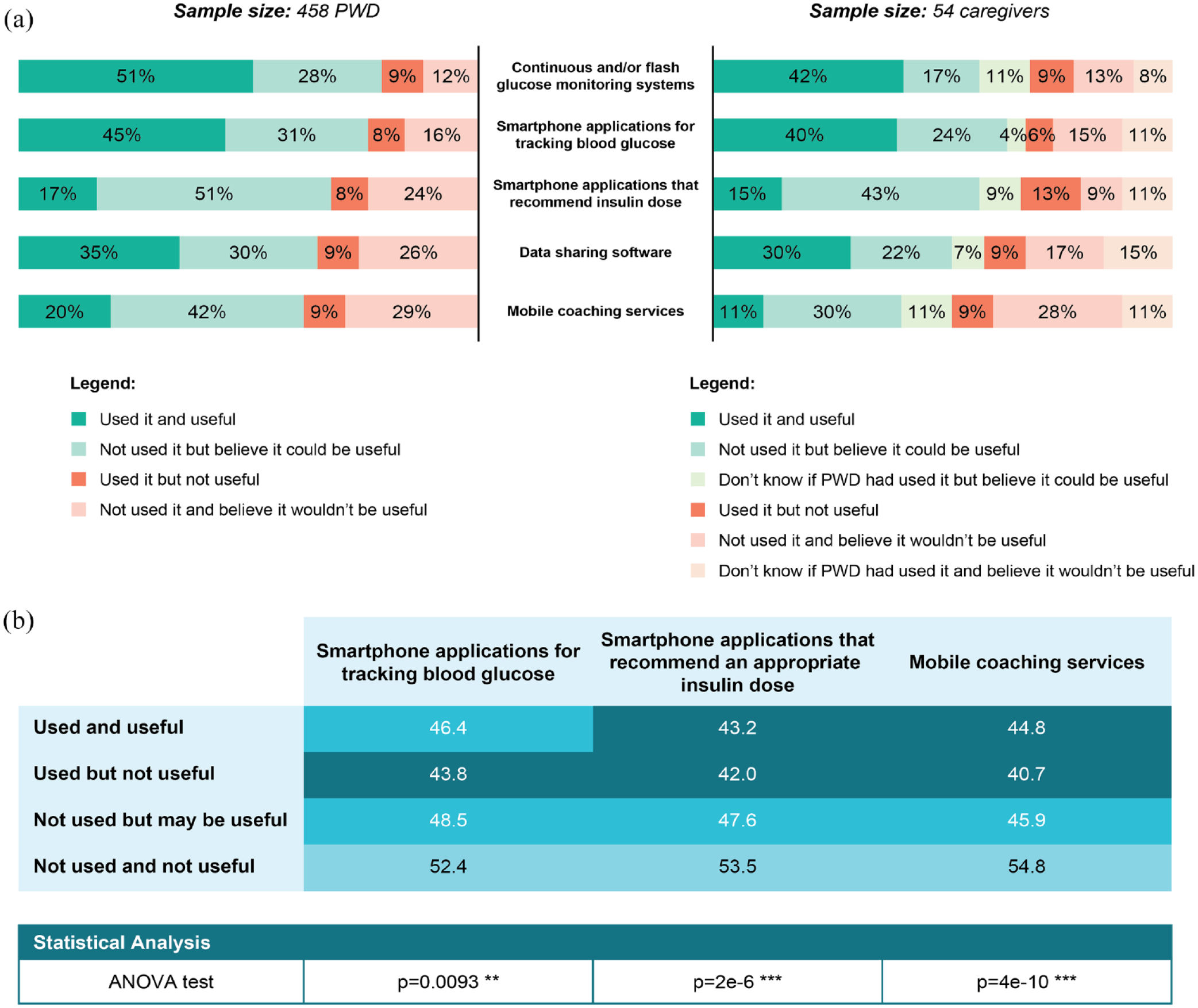
(a) Use and perceptions of technological devices and digital tools for T1D management in PWD and caregivers, and (b) mean age per use and perception of devices in PWD. In (b), the lighter the shade of blue, the higher the mean age of PWD.
Approximately one-third (35%) of PWD had used teleconsultation and 9% had used it regularly. Among the 65% who had never used teleconsultation, two-thirds would have liked to do so. Use of teleconsultation differed significantly by age group and country (Figure 2). Face-to-face appointments from time to time were thought to be important by 40% of PWD. Additional experiences and perceptions of teleconsultation are presented in Supplemental Figure S2.
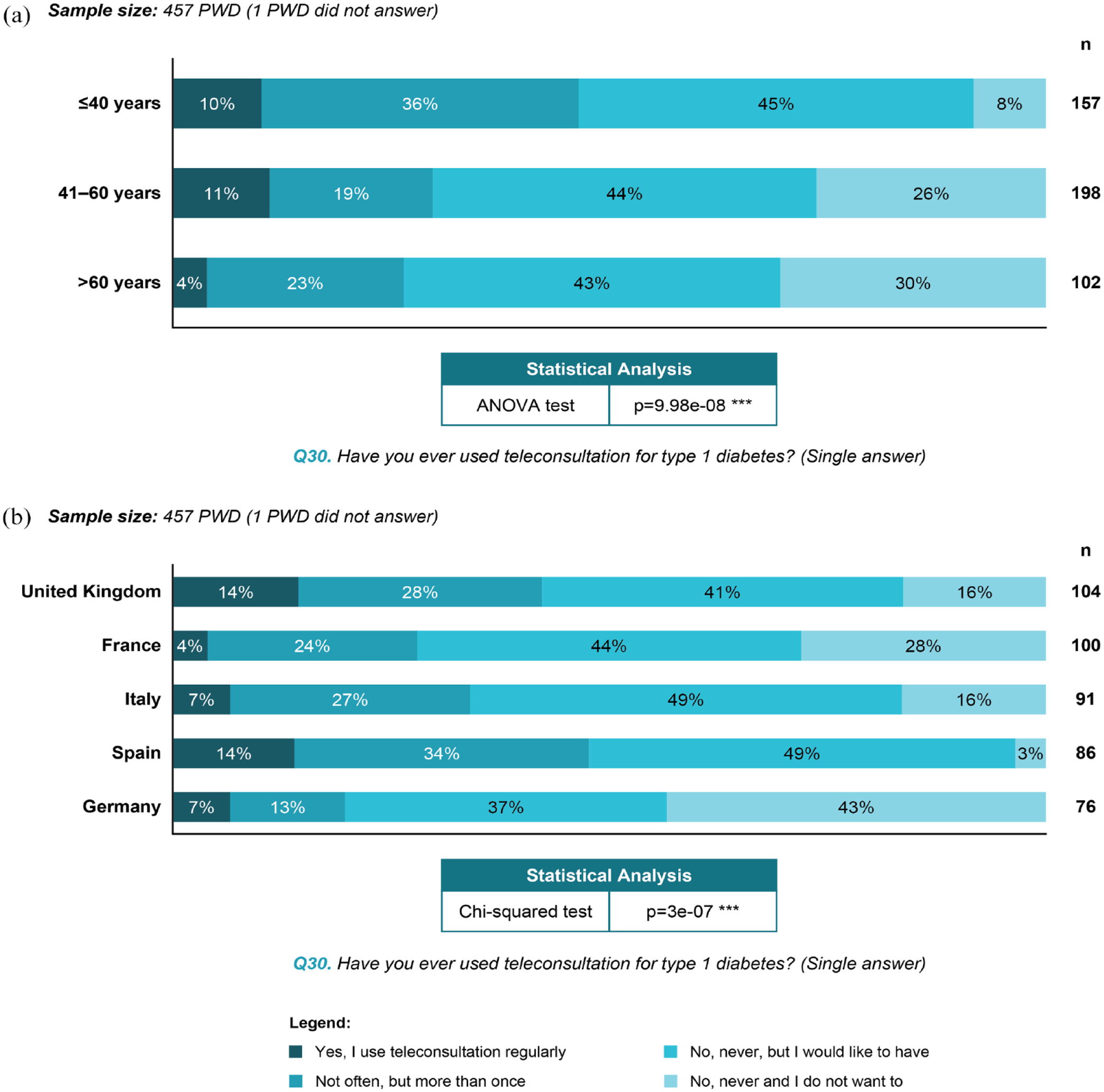
Use of teleconsultation in PWD by (a) age group, and (b) country.Abbreviations: PWD, people with diabetes; ANOVA, analysis of variance.
Needs for Support With Diabetes Technology
When PWD were asked about the costs of T1D care generally, it was not considered a financial issue for 99% of the responders. However, 46% of PWD contributed to the costs of their T1D care. This differed significantly, depending on the insulin device used (P = .0003): 83% of smart insulin pen users paid/contributed versus 44% insulin pen and 37% insulin pump users (Figure 3). Approximately half of the responders (52% PWD and 48% caregivers) reported that they would find it useful if devices and digital tools were fully reimbursed and widely available.
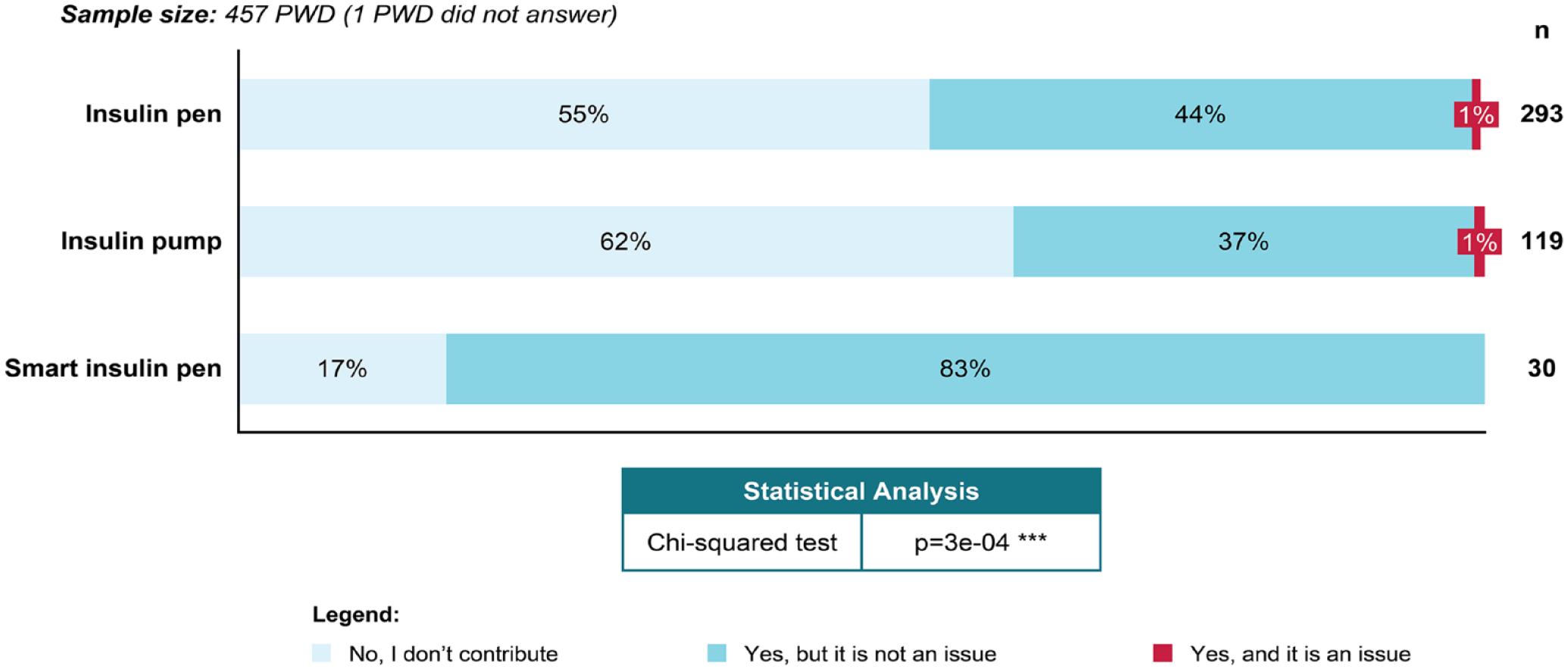
Contribution to costs of T1D care by insulin administration device in PWD.
Half of the PWD (48%) and 41% of caregivers hoped for new devices and/or digital tools. There was no statistical difference in the mean age of those hoping for new devices and those who were not (P = .19). The PWD who were hoping for new devices and/or digital tools paid more attention to controlling their T1D (Supplemental Figure S3). Approximately half of responders (53% PWD; 56% caregivers) would like to have more knowledge about various aspects of T1D; 24% of PWD and 15% of caregivers reported that they lack knowledge about devices and digital tools for T1D.
Discussion
Almost all PWD with T1D in this European survey reported using either a pen or pump to administer insulin. However, the proportion of PWD using insulin pumps varied significantly between countries. A total of 63% of all respondents used a CGM device. Other studies of CGM in Europe have also observed a relatively high uptake, with one transatlantic comparison reporting the adoption of CGM in Europeans to be greater than that seen in PWD in the United States. 16
Previous retrospective real-world studies suggest that apps for T1D management can improve glycemic control,17,18 and most PWD in this survey believed that the data produced by digital tools made their life easier. Most PWD believed that existing devices and digital tools were or could be useful in aiding T1D management.
Almost half of PWD contributed to the costs of their diabetes care. The costs associated with more advanced technology may represent a barrier to improving diabetes management; however, very few responders in this survey felt that contributing to the costs of their T1D treatment was an issue, perhaps because the cost was still limited or affordable to this population. While the level of reimbursement is significantly different between the different countries, PWD and caregivers felt that more must be done to improve the accessibility of technology for T1D management, which may be in prevision of the potential extra cost from upcoming technologies. This perception was not limited to PWD and caregivers; the Diabetes Digitalization and Technology (D.U.T) 2020 report has shown that 80% of diabetologists felt that the lack of reimbursement was a great disadvantage of digitalization and new technologies. 19 In this survey, half of PWD and caregivers said that it would be useful if devices and digital tools were fully reimbursed and widely available, which may be linked to the variation observed in T1D management devices used between different countries. Significantly more smart pen users contributed to the cost of their T1D care compared with users of other insulin delivery devices, which was expected, given that costs associated with smart pens were not reimbursed in any of the countries at the time of the survey. Smart insulin pens may improve treatment adherence and glycemic control20,21; however, there is limited evidence published so far on the impact of smart insulin pens on diabetes management and only a small number of PWD in this survey (30, 7%) were smart pen users. It is possible that the proportion of smart pen users reported is inaccurately high as the availability of smart pens was limited in the countries included at the time of the survey.
The results of this survey indicate that current devices and digital tools are used by many PWD and are seen as an important innovation in diabetes treatment, with nearly half of participants reporting that they hoped for new devices and/or digital tools. Furthermore, many PWD and caregivers would like to have more knowledge about diabetes technology, which indicates a need for more support and education relating to diabetes technology.
A strength of this work is that the survey was developed with a group of T1D experts from different European countries (the United Kingdom, France, and Germany) with different specialty backgrounds (clinician, psychologist, or nurse educator), and with the input of two PWD from the United Kingdom and France. Another strength of this survey is the sociodemographic profile of the responders. This survey provides insight into the experiences and perceptions of people living in five Western European countries with different health care systems providing different diabetes education and levels of integration of technology into standard care. The responders also had a large age range and varied educational background. In addition, the survey was completed online, preventing social desirability bias.
One limitation of the survey is that it lacks the perceptions of HCPs. However, the findings of two surveys conducted in Germany suggest that HCP attitudes toward digitization are aligned with those of PWD and caregivers. In the D.U.T 2020 report on diabetologists’ perspectives, 76% had a positive attitude toward digitalization 19 ; such positivity was also seen in 83% of PWD and caregivers in a 2022 report. 13
Another limitation of the online survey is that computer literacy is required; furthermore, invited participants were mainly registered users of the Carenity platform or members of Facebook groups related to T1D, and therefore likely to already be motivated and paying attention to the management of their T1D. The lack of standardized measurements makes it difficult to directly compare perceptions of the care pathway with other surveys. The timing of the survey may be considered a limitation as data were collected during one of the waves of the COVID-19 pandemic and, therefore, during a period of sudden changes in health care and diabetes care. Other limitations are the potential for recall bias and the small sample size for caregivers.
Conclusions
This survey highlights the positive experiences and perceptions of technology for diabetes management in many PWDs and their caregivers. To build on this, survey respondents would like to see new devices/digital tools become more widely available, preferably with full reimbursement. This could enhance the existing level of support for PWD and their caregivers, and would allow the benefits of disease technology to reach an even broader T1D community. In the process of technologization and digitalization of diabetes therapy, it is very useful to repeatedly capture the user perspective and to ask PWD about their attitudes, wishes, and expectations with regard to new digital solutions. This survey serves as a useful benchmark against which to assess future attitudes toward technology as it continues to evolve and becomes more integrated into T1D care.
Supplemental Material
sj-docx-1-dst-10.1177_19322968231208690 – Supplemental material for European Survey on Adult People With Type 1 Diabetes and Their Caregivers: Insights Into Perceptions of Technology
Supplemental material, sj-docx-1-dst-10.1177_19322968231208690 for European Survey on Adult People With Type 1 Diabetes and Their Caregivers: Insights Into Perceptions of Technology by Alfred Penfornis, Su Down, Antoine Seignez, Alizé Vives, Mireille Bonnemaire and Bernhard Kulzer in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors thank the survey participants for their participation. The authors thank Pierre Evenou, PhD (Sanofi), for coordinating the development, facilitating author discussions, and critical review of the manuscript. Medical writing support and editorial assistance were provided by Arthur Holland, PhD, of Fishawack Communications Ltd, part of Avalere Health, and were funded by Sanofi.
Abbreviations
ANOVA, analysis of variance; CGM, continuous glucose monitoring; D.U.T, Diabetes Digitalization and Technology; GCSE, General Certificate of Secondary Education; HCP, health care professional; PWD, people with type 1 diabetes; T1D, type 1 diabetes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Alfred Penfornis: Advisory panel member for AstraZeneca, MSD, and Novo Nordisk, and speaker for Abbott, Amgen, Bayer MSD, Sanofi, Novo Nordisk, Eli Lilly, and Medtronic. Su Down: Funding from the following companies for providing educational sessions and documents, and advisory boards: Abbott, AstraZeneca, Boehringer Ingelheim, Eli Lilly, GlaxoSmithKline, MSD, Mylan, Napp, Novo Nordisk, Roche, Sanofi, and Viatris. Antoine Seignez: Employee of Carenity, providing consultancy to Sanofi. Alizé Vives: Employee of Carenity, providing consultancy to Sanofi. Mireille Bonnemaire: Employee of Sanofi, holds stocks/shares in Sanofi. Bernhard Kulzer: Speaker’s honoraria and/or advisory boards and/or research grants from Abbott, Ascensia, Bayer, Becton Dickinson, Berlin Chemie, Dexcom, Insulet, Novo Nordisk, Roche, and Sanofi.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funded by Sanofi.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
