Abstract
Ultra-rapid-acting insulin analogs (URAA) are a further development and refinement of rapid-acting insulin analogs. Because of their adapted formulation, URAA provide an even faster pharmacokinetics and thus an accelerated onset of insulin action than conventional rapid-acting insulin analogs, allowing for a more physiologic delivery of exogenously applied insulin. Clinical trials have confirmed the superiority of URAA in controlling postprandial glucose excursions, with a safety profile that is comparable to the rapid-acting insulins. Consequently, many individuals with diabetes mellitus may benefit from URAA in terms of prandial glycemic control. Unfortunately, there are only few available recommendations from authoritative sources for use of URAA in clinical practice. Therefore, this expert consensus report aims to define populations of people with diabetes mellitus for whom URAA may be beneficial and to provide health care professionals with concrete, practical recommendations on how best to use URAA in this context.
Introduction
Insulin is indispensable in the treatment of people with type 1 diabetes mellitus (T1D) and continues to be an important pillar of advanced therapy of people with type 2 diabetes (T2D). Besides recombinant human insulin, which is identical to the native hormone and either short-acting or intermediate-acting depending on the formulation, insulin analogs play an important role in insulin therapy today. 1 Based on adjustments in the amino acid sequence, insulin analogs have a significantly altered pharmacokinetic and pharmacodynamic profile compared with native human insulin, resulting in either faster or prolonged insulin action. Consequently, rapid-acting insulin analogs (RAA)—including insulin lispro, aspart, and glulisine—are particularly used to counteract postprandial glucose (PPG) excursions, whereas long-acting or basal insulins, such as insulin glargine, detemir, and degludec, are used primarily to manage fasting plasma glucose levels. 2
Ultra-rapid-acting insulin analogs (URAA), including “faster aspart” (URAsp) and “insulin lispro-aabc” (URLi), represent a recent further development in insulin therapy. These are novel formulations of the RAA insulin aspart (IAsp) and insulin lispro (ILispro), respectively, to enable even faster onset of insulin action and to thereby reduce the lag time between subcutaneous insulin injection and onset and peak of insulin action. Both URAsp and URLi have been approved for treatment of people with diabetes mellitus by the European Medicines Agency (EMA) and the US Food and Drug Administration (FDA) in 2017 and 2020, respectively.
The aim of this report, based on the results of clinical trials as well as the experience with these novel insulin analogs in clinical practice, is to define the groups of people with diabetes mellitus who may benefit from using URAA. The practical, evidence-based recommendations provided here are intended to help health care professionals make optimal use of these new opportunities in insulin therapy for defined groups of people with diabetes mellitus.
URAA in Therapy of People With Diabetes Mellitus
Pharmacology and Pharmacokinetics
Compared with insulin aspart (IAsp), URAsp additionally contains two excipients: niacinamide (vitamin B3, 170 mM) and
Ultra-rapid insulin lispro (URLi) contains two additional excipients: citrate (15 mM) and treprostinil (1 µg/mL). Citrate accelerates the absorption of insulin lispro by increasing local vascular permeability, while treprostinil (a synthetic prostacyclin analog currently used for the treatment of pulmonary arterial hypertension) enhances insulin absorption by inducing local vasodilation.9,10 Treprostinil was not detectable in plasma after subcutaneous dosing of URLi in a cohort of people with T1D. 11 Accordingly, no systemic adverse effects are expected to be associated with this excipient. Similar to the comparison between URAsp and IAsp, URLi has a faster onset and offset of exposure than ILispro, reaching 50% of the maximum concentration 14.0 minutes and the late half-maximum concentration 17.6 minutes earlier in a pooled analysis including studies in healthy subjects and people with T1D or T2D. 12 The URLi exposure in the first 30 minutes was increased almost 3.0-fold compared with ILispro. Likewise, the insulin action in the first 30 minutes was increased 3.1-fold and the offset of the glucose-lowering effect occurred 37.9 minutes earlier in the pooled analysis, again with a similar overall insulin action. 12 Overall, compared with the conventional ILispro formulation, the duration of insulin exposure to the circulation was reduced by 68.2 minutes and the duration of insulin action was reduced by 43.8 minutes with URLi. 12
A direct comparison of URAsp and URLi showed faster pharmacokinetic properties of URLi, with a 1.2-fold higher initial insulin exposure during the first 30 minutes after administration. 13 To conclude, both URAsp and URLi exhibit a faster onset and reduced late insulin action as well as a shorter duration of insulin action which more closely represents the physiologic postprandial insulin response.
Clinical Effectiveness
Several phase III trials (Table 1) were conducted to investigate the clinical effectiveness of URAsp and URLi. Most of these studies were designed to examine noninferiority of URAA in change of glycated hemoglobin (HbA1c) from baseline as a primary outcome compared with the conventional RAA. In these studies and a meta-analysis of eight studies including both URAsp and URLi, URAA were confirmed to be noninferior in glycemic control compared with the standard formulations of insulin aspart and lispro, respectively. 30 In a separate meta-analysis comparing URAsp versus IAsp, URAsp was associated with improved HbA1c and decreased PPG excursions in people with T1D, albeit with modest clinical significance. 31
Overview of phase III trials on ultra rapid-acting insulin analogs (URAA) for the treatment of type 1 or type 2 diabetes.
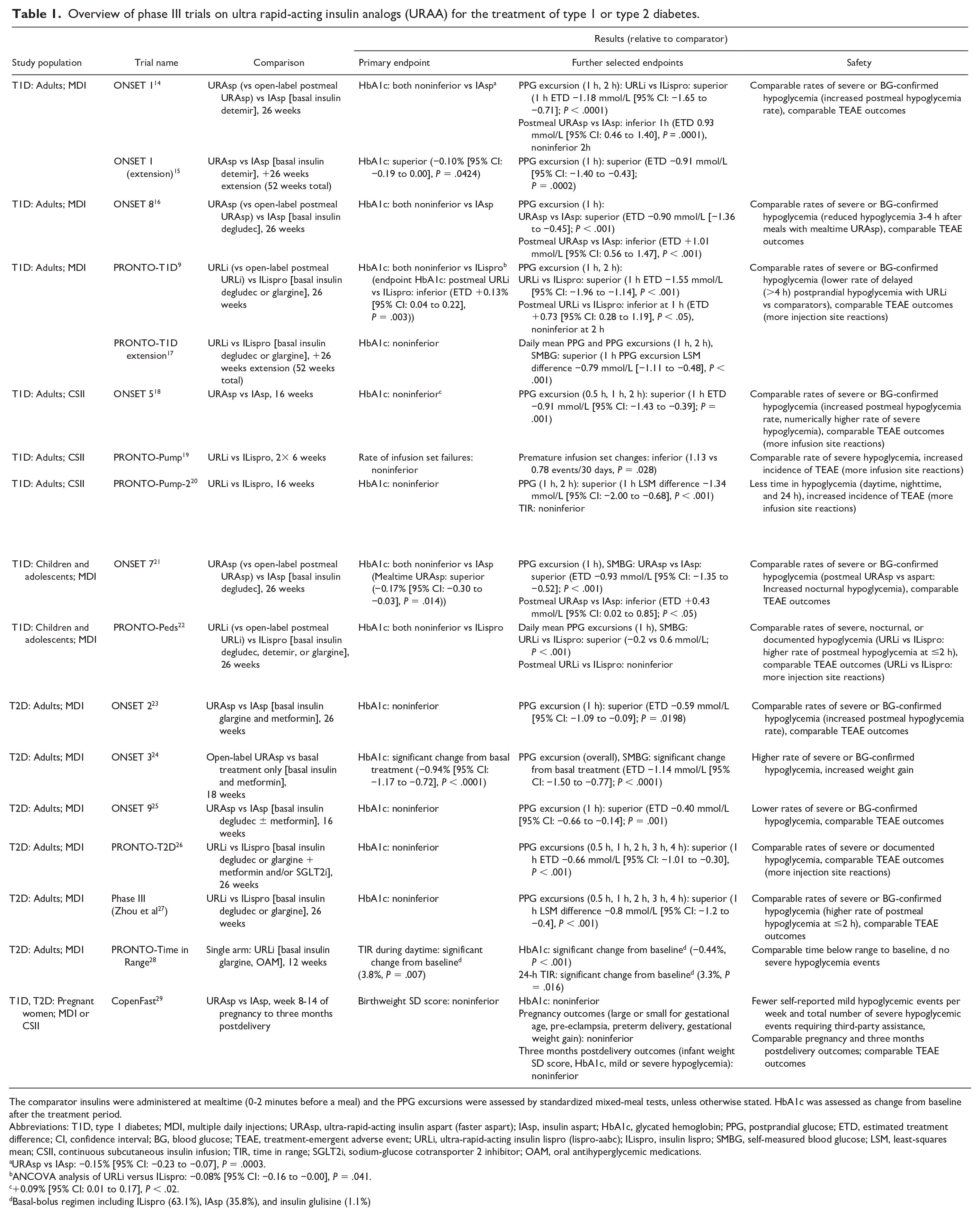
The comparator insulins were administered at mealtime (0-2 minutes before a meal) and the PPG excursions were assessed by standardized mixed-meal tests, unless otherwise stated. HbA1c was assessed as change from baseline after the treatment period.
Abbreviations: T1D, type 1 diabetes; MDI, multiple daily injections; URAsp, ultra-rapid-acting insulin aspart (faster aspart); IAsp, insulin aspart; HbA1c, glycated hemoglobin; PPG, postprandial glucose; ETD, estimated treatment difference; CI, confidence interval; BG, blood glucose; TEAE, treatment-emergent adverse event; URLi, ultra-rapid-acting insulin lispro (lispro-aabc); ILispro, insulin lispro; SMBG, self-measured blood glucose; LSM, least-squares mean; CSII, continuous subcutaneous insulin infusion; TIR, time in range; SGLT2i, sodium-glucose cotransporter 2 inhibitor; OAM, oral antihyperglycemic medications.
URAsp vs IAsp: −0.15% [95% CI: −0.23 to −0.07], P = .0003.
ANCOVA analysis of URLi versus ILispro: −0.08% [95% CI: −0.16 to −0.00], P = .041.
+0.09% [95% CI: 0.01 to 0.17], P < .02.
Basal-bolus regimen including ILispro (63.1%), IAsp (35.8%), and insulin glulisine (1.1%)
Apart from HbA1c, which is the current gold standard for evaluating glycemic control, time in range (TIR) as defined by time with plasma glucose values between 70 and 180 mg/dL 32 is increasingly recognized as an important surrogate marker for glycemic control.33,34 Time in range was shown to significantly improve in people with T1D on URAsp in two observational studies of URAA.35,36 In the single-arm PRONTO-Time in Range study, URLi combined with insulin titration in people with T2D and elevated HbA1c levels showed a significant improvement in daytime (+3.8%) and 24-hour (+3.3%) TIR as well as HbA1c (−0.44%) compared with the prestudy baseline multiple daily injections (MDI) therapy with a RAA. 28 A subanalysis of the PRONTO-T1D trial reported significantly improved TIR during daytime in individuals with T1D using MDI therapy 37 and, importantly, a meta-analysis reported significantly improved TIR with URAA when used in insulin pump therapy. 38
Postprandial glucose excursions are an important component of glycemic control.39,40 Compared with the respective RAA, both URAsp and URLi showed consistently superior effects in terms of reducing 1- and 2-hour PPG excursions after liquid meal testing, which was also supported by the pooled analysis for both T1D and T2D subjects. 30
Safety Outcomes
Overall, URAA did not increase the incidence of serious adverse events in people with T1D or T2D in MDI therapy and had similar safety outcomes to the corresponding RAA. 30 However, the timing of hypoglycemia events differs with URAA use. Several trials reported a statistically significant higher rate of hypoglycemia within the first two hours after a meal,14,15,22,23,27 but a statistically significant lower rate of late postprandial hypoglycemic episodes.9,16,17 Importantly, the overall rate of severe or blood glucose-confirmed/documented hypoglycemia was generally comparable between URAA and the respective RAA, as listed for each study in summarized Table 1. In continuous subcutaneous insulin infusion (CSII) therapy in T1D, URAA resulted in a similar rate of overall hypoglycemic events to RAA,18,19 but a significantly reduced time in hypoglycemia in the PRONTO-Pump-2 study with URLi and in the pooled analysis across all CSII randomized controlled trials (RCTs) for both URAA.20,38
Other reported treatment-emergent adverse events (TEAE) included more frequent mild to moderate infusion or injection site reactions, especially with URLi compared with ILispro.9,17-20,22,26 The incidence of other TEAE events was similar between URAA and RAA for both children and adults with T1D and for adults with T2D. 30
Current Recommendations and Guidelines
The latest 2021 consensus report on the management of T1D in adults by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD) mentions URAA as an alternative that is equivalent to RAA and a preferred option to short-acting (regular) human insulin. While URAA are noted to have an earlier onset and peak of action than RAA, they are not recommended in preference to RAA as they were not superior in terms of HbA1c reduction or rates of hypoglycemia in clinical trials. 41 A similar position can be found in the ADA Standards of Care 2023 and the American Association of Clinical Endocrinology (AACE) Clinical Practice Guideline 2022.42,43 The International Society for Pediatric and Adolescent Diabetes (ISPAD) Clinical Practice Consensus Guidelines 2022 give stronger consideration to URAA, which are seen as potentially beneficial for use in younger individuals. 44
According to the Consensus Recommendations for the Use of Automated Insulin Delivery Technologies in Clinical Practice 2023, RAA continue to be the recommended insulins for automated insulin delivery (AID) systems because of their proven compatibility with most pump technologies. URAA are stated to be at most modestly beneficial or noninferior to RAA in these systems. However, it is noted that URAA may be beneficial for certain individuals, such as those who prefer to administer the bolus immediately before a meal rather than 15 minutes before onset of the meal. 45
Regarding T2D, URAA are currently not explicitly addressed in guidelines and recommendations by medical societies such as the ADA and EASD in their current 2022 Consensus Report on Management of Hyperglycemia in Type 2 Diabetes. 46
Consensus Recommendations
As stated in the previous chapter, URAA provide a faster onset and offset of action compared with RAA and regular insulin and have been shown in clinical trials to provide improved prandial glycemic control. Accordingly, we now provide tailored recommendations for use of URAA in specific groups of people living with diabetes or in specific situations that are relevant to the management of diabetes mellitus below (see Tables 2–12). The recommendations are summarized in Table 13. We used the ADA grading system to indicate the level of scientific evidence for each recommendation. 47 Accordingly, evidence from large, well-conducted RCTs or meta-analyses was graded A, evidence from well-conducted cohort or case-control studies was graded B, evidence from poorly controlled clinical studies was graded C, and E was used for expert consensus or clinical experience in the absence of evidence from clinical trials or in the case of conflicting evidence.
Consensus on the Use of URAA in MDI Therapy in T1D.

Abbreviations: URAA, ultra-rapid-acting insulin analogs; MDI, multiple daily injection; T1D, type 1 diabetes; RCT, randomized controlled trial; URAsp, ultra-rapid-acting insulin aspart (faster aspart); URLi, ultra-rapid-acting insulin lispro (lispro-aabc); HbA1c, glycated hemoglobin; RAA, rapid-acting insulin analogs.
Consensus on the Use of URAA in Insulin Pump Therapy in T1D.

Abbreviations: URAA, ultra-rapid-acting insulin analogs; T1D, type 1 diabetes; RCT, randomized controlled trial(s); URAsp, ultra-rapid-acting insulin aspart (faster aspart); URLi, ultra-rapid-acting insulin lispro (lispro-aabc); TIR, time in range; RAA, rapid-acting insulin analogs; HbA1c, glycated hemoglobin.
Consensus on the Use of URAA in AID Therapy in T1D.

Abbreviations: URAA, ultra-rapid-acting insulin analogs; AID, automated insulin delivery; T1D, type 1 diabetes; RCT, randomized controlled trial(s); URAsp, ultra-rapid-acting insulin aspart (faster aspart); URLi, ultra-rapid-acting insulin lispro (lispro-aabc); HbA1c, glycated hemoglobin; RAA, rapid-acting insulin analogs.
Consensus on the Use of URAA in MDI Therapy in T2D.

Abbreviations: URAA, ultra-rapid-acting insulin analogs; MDI, multiple daily injection; T2D, type 2 diabetes; RAA, rapid-acting insulin analogs; RCT, randomized controlled trial(s); URAsp, ultra-rapid-acting insulin aspart (faster aspart); URLi, ultra-rapid-acting insulin lispro (lispro-aabc); HbA1c, glycated hemoglobin.
Consensus on the Use of URAA During Pregnancy With Diabetes Mellitus.

Abbreviations: URAA, ultra-rapid-acting insulin analogs; RAA, rapid-acting insulin analogs; URAsp, ultra-rapid-acting insulin aspart (faster aspart); URLi, ultra-rapid-acting insulin lispro (lispro-aabc); RCT, randomized controlled trial(s); HbA1c, glycated hemoglobin; GDM, gestational diabetes mellitus.
Consensus on the Use of URAA in Children and Adolescents With T1D.

Abbreviations: URAA, ultra-rapid-acting insulin analogs; T1D, type 1 diabetes; RCT, randomized controlled trial(s); URAsp, ultra-rapid-acting insulin aspart (faster aspart); HbA1c, glycated hemoglobin; RAA, rapid-acting insulin analogs.
Consensus on the Use of URAA in Elderly Individuals With T2D.

Abbreviations: URAA, ultra-rapid-acting insulin analogs; T2D, type 2 diabetes; RAA, rapid-acting insulin analogs; RCT, randomized controlled trial(s); URAsp, ultra-rapid-acting insulin aspart (faster aspart); URLi, ultra-rapid-acting insulin lispro (lispro-aabc); HbA1c, glycated hemoglobin.
Consensus on the Use of URAA in Postprandial Hyperglycemia.

Abbreviations: URAA, ultra-rapid-acting insulin analogs; T1D, type 1 diabetes; T2D, type 2 diabetes; RCT, randomized controlled trial(s); URAsp, ultra-rapid-acting insulin aspart (faster aspart); URLi, ultra-rapid-acting insulin lispro (lispro-aabc); RAA, rapid-acting insulin analogs; HbA1c, glycated hemoglobin.
Consensus on the Use of URAA in the Context of an Increased Risk of Late Postprandial Hypoglycemia.

Abbreviations: URAA, ultra-rapid-acting insulin analogs; T1D, type 1 diabetes; RCT, randomized controlled trial(s); URAsp, ultra-rapid-acting insulin aspart (faster aspart); URLi, ultra-rapid-acting insulin lispro (lispro-aabc); CSII, continuous subcutaneous insulin infusion; RAA, rapid-acting insulin analogs.
Consensus on the Use of URAA When Premeal Insulin Administration Is a Challenge.

Abbreviations: URAA, ultra-rapid-acting insulin analogs; URAsp, ultra-rapid-acting insulin aspart (faster aspart); URLi, ultra-rapid-acting insulin lispro (lispro-aabc); HbA1c, glycated hemoglobin; RAA, rapid-acting insulin analogs.
Practical Recommendations When Switching From Rapid-Acting Insulins to URAA.

Abbreviations: URAA, ultra-rapid-acting insulin analogs; CSII, continuous subcutaneous insulin infusion; AID, automated insulin delivery.
Overview of Consensus Recommendations and the Level of Evidence According to the ADA Grading System. 47

American Diabetes Association (ADA) grading system.
Ultra-Rapid-Acting Insulins in Type 1 Diabetes
MDI therapy
Most of the RCT on URAA discussed in the previous chapters were conducted in adults with T1D on MDI therapy. Based on these data, URAA have two major advantages as compared with RAA:
They enable an accelerated onset and offset of postprandial insulin action and thereby result in significantly reduced PPG excursions throughout the studies 30 and a reduced risk of late (>four hours) postprandial hypoglycemia in some people.9,16,17 Regarding overall hypoglycemic events and other TEAE, URAA are noninferior to RAA, 30 except for a higher probability of mild to moderate injection site reactions with URLi. Although data from the ONSET 1 extension study showed slightly better HbA1c control with URAsp compared with IAsp, 15 overall, URAA have been shown to be noninferior to RAA in terms of HbA1c change from baseline in phase III trials.9,14,16-18
URAA can be safely administered up to 20 minutes after the start of a meal, which may offer greater flexibility and convenience compared with RAA, which are recommended to be injected 15 to 20 minutes before a meal. 48 Postmeal administration of URAA was shown to be noninferior to mealtime RAA regarding glycemic control assessed by change from baseline HbA1c levels in RCT for both URAsp and URLi.9,16,21,22 Nevertheless, mealtime URAA was superior to postmeal URAA regarding PPG excursions and is therefore recommended in preference to postmeal administration.
Beyond the controlled environment of interventional clinical trials, observational studies may be helpful to evaluate the benefit of URAA in the real-world setting, although the results must be interpreted with caution. In this context, the GoBolus study found that people with T1D on stable insulin treatment with MDI and intermittently scanned continuous glucose monitoring (CGM) largely profited from switching from conventional prandial insulins to URAsp 35 : after 24 weeks of treatment, despite no significant change in insulin doses, the HbA1c improved by an absolute 0.19% from baseline, and TIR increased by 46.1 minutes per day on average—a finding that was also confirmed in a similar retrospective study from Belgium. 36 The GoBolus study also included patient-reported outcomes: according to participants’ responses to the Diabetes Treatment Satisfaction Questionnaire (DTSQ), 49 quality of life improved significantly after switching to URAA. Positive feedback was also received from physicians treating people with T1D or T2D. 50
While treatment with URAA did not result in increased incidence of serious adverse events, there was a higher rate of mild to moderate injection site reactions with URLi.9,17
To conclude, URAA are safe and effective for individuals with T1D in MDI therapy, which is confirmed by the results of two meta-analyses, one including RCT of URAsp versus IAsp in MDI therapy only and a second including RCT of URAsp and URLi versus RAA or placebo.
Insulin pump therapy
A rapid pharmacokinetic profile is of particular importance for CSII or insulin pump therapy, which is generally based on rapid-acting insulins. 51 Instead of a single dose of a long-acting insulin with MDI therapy, the pump delivers insulin at a continuous, adjustable rate throughout the day, supplemented by individually set prandial boluses. In this regard, in principle, accelerated insulin pharmacokinetics as offered by URAA are highly desirable to further enhance the more physiologic delivery of insulin via CSII and to thereby improve glycemic control without increasing hypoglycemia. 52
The compatibility, efficacy, and safety of URAA in insulin pumps were investigated in a series of RCT for both URAsp and URLi and also evaluated by a subsequent meta-analysis. 38 Across the single studies, URAA were reported to be compatible with CSII19,53 and to provide a comparable glycemic control as RAA regarding HbA1c levels compared with pretrial RAA CSII treatment.18,20 In the pooled analysis, which also included trials investigating URAA in hybrid closed-loop systems, URAA were reported to increase the 24-hour TIR by 1.1%, equivalent to 110 minutes per week. 38 Interestingly, this result was driven by a lower time in hypoglycemia throughout the 24 hours rather than a decreased time spent in hyperglycemia. However, the use of URAA also significantly increased the odds of unplanned infusion set changes, as reported in the ONSET 5 18 and PRONTO pump studies19,20 and in the pooled analysis across the URAsp and URLi clinical trials. 38 The latter one particularly reported an odds ratio of 1.60 (95% confidence interval [CI]: 1.26, 2.03) for unplanned infusion set changes with URAA, with 398 events reported in 623 individuals using URAA and 324 events in 612 individuals using RAA. 38 The main reason given for this outcome was an increased risk of injection site reactions and pain. This should be considered and discussed when recommending URAA to individuals on CSII. Observational studies of URAsp outside the controlled setting of RCT have also shown that pump settings—which in most RCT were held constant from the pretrial pump insulin—should be adjusted to account for the ultra-rapid pharmacokinetics of URAA.54,55
Therapy with AID systems
AID devices, which combine the benefits of insulin pump delivery with CGM and a control algorithm that regulates insulin delivery based on sensor glucose readings, are an important technological advance. 56 Hybrid closed-loop systems are the first generation of AID systems that allow automated basal insulin adjustments or, in advanced hybrid AID devices, additional delivery of correction boluses. 57 Yet, these systems require user-defined mealtime boluses. 45 The iLet bionic pancreas system, which received FDA clearance in May 2023, is the first AID system approved that does not require calculation of prandial insulin doses, but only meal announcements from the user.
AID has a favorable effect on glycemic control and decreases the risk of hypoglycemia in individuals with T1D.58,59 URAA as even faster-acting insulins may allow the device to respond more quickly to elevated glucose levels and have therefore been investigated in a small number of clinical studies. Both URAsp and URLi showed comparable efficacy and safety to RAA with the hybrid closed-loop Medtronic 670G system in adults.60,61 An improvement in TIR 62 was reported in one study with URAsp in the latter as well as in the MiniMed Advanced Hybrid Closed-Loop system. 63 Using the CamAPS FX, another study found a lower time in hypoglycemia with URAsp compared with IAsp. 64 Two studies specifically compared clinical outcomes in children and young adults with the CamAPS FX or a fully closed-loop system and also showed comparable efficacy of URAsp to IAsp.65,66 Nevertheless, expectations of a significant advantage of URAA over RAA were thus far largely unfulfilled by the results of these trials. Importantly, though, and in analogy to the CSII studies, the control algorithms in the aforementioned trials were maintained from the pretrial RAA, with no explicit adjustments of the parameters for URAA. However, in silico studies have suggested that there is potential for significantly improved TIR by adapting AID settings to the faster pharmacokinetics of URAA.67-69
Regarding safety aspects, despite the small number of available studies on AID systems, a subgroup analysis in the CSII pooled analysis suggested no increased infusion set changes with URAA versus RAA in hybrid closed-loop systems. 38 One study specifically investigated the efficacy and safety of URAsp with the investigational version of the MiniMed 780G in physically active children, and reported comparable outcomes to children using IAsp. 70 However, more studies are warranted to definitively assess the clinical efficacy of URAA in AID. Nevertheless, on the basis of the evidence currently available, URAA provide comparable glycemic control to RAA and are safe for use in AID systems, where approved for such devices.
Ultra-Rapid-Acting Insulins in T2D
MDI therapy
In the course of T2D, many people require treatment intensification with the initiation of insulin therapy. In countries where access to insulin is guaranteed, it is expected that more than one fourth of people living with T2D are treated with insulin.71,72 Yet, the number of people with suboptimal blood glucose control for whom intensification of therapy would be indicated is thought to be even higher. 73 In a retrospective study, more than 40% of people with T2D and suboptimal glycemic control on oral diabetes therapies declined to use insulin when recommended. 74 The reasons for this therapeutic inertia are many and include fear of insulin side effects and inconvenience of its use. 75 Therefore, the availability of insulins with enhanced user flexibility and improved health benefits may help to overcome these barriers.
Treatment intensification with insulin in T2D is a stepwise approach, generally initiated by introducing basal insulin. If HbA1c target values are not reached with non-insulin glucose-lowering therapies and adequate basal insulin therapy, prandial insulin should be added. 76 Prandial glucose control is an important component of glycemic control.39,40 In this context, several RCTs have investigated the safety and efficacy of URAA in adults with insulin-treated T2D. The PRONTO-Time in Range study reported a significantly increased TIR (both daytime and 24 hours) with URLi and insulin dose optimization compared with prestudy basal-bolus regimen, which was driven by a lower time above range. 28 However, the prestudy baseline treatment included real-time or intermittent scanning CGM in only 22% of participants, and therefore it cannot be excluded that the unblinded CGM during the study period or the insulin dose titration contributed to the positive effect on glycemic control. Other phase III trials directly compared URAA with RAA in people with T2D, with results similar to those described in people with T1D: URAA showed superior PPG control (although to a generally lesser extent), noninferior change in HbA1c compared with baseline, and a similar or lower rate of hypoglycemia.23,26,77 Other TEAE were comparable between treatment and control groups. These results were likewise confirmed in the pooled analysis. 30
Another important benefit of URAA compared with RAA is that it can be safely administered up to 20 minutes after the start of a meal. This may also be reflected in a higher level of treatment satisfaction with URLi compared with the prestudy treatment including RAA, 28 as assessed in the Insulin Treatment Satisfaction Questionnaire (ITSQ) survey of individuals with T2D. 25
Ultra-Rapid-Acting Insulins in Diabetes Mellitus During Pregnancy
Gestational diabetes mellitus (GDM) is a common condition, with a global prevalence of approximately 14% in pregnant women. 78 It is accompanied by an increased risk of preterm delivery, macrosomia, or neonatal respiratory distress syndrome. 79 Accordingly, treatment of GDM was shown to reduce perinatal morbidity 80 and screening for GDM is therefore largely recommended in current guidelines for pregnant women at elevated risk.81,82 Many pregnancy complications such as spontaneous abortion, pre-eclampsia, macrosomia, and neonatal respiratory distress syndrome are even more common in women with T1D and T2D during pregnancy. 83 It is therefore particularly important to ensure optimal glycemic control in all pregnant women with diabetes.
Insulins are the central pillar for diabetes therapy during pregnancy, as other glucose-lowering medications often lack positive safety data or can cross the placenta with potential adverse effects on the fetus. 82 Both MDI and CSII therapy are equally appropriate treatment options for pregnant women with diabetes. 84 An important factor to consider during pregnancy is the changing insulin physiology, with increased insulin sensitivity in the first trimester but greatly reduced sensitivity and increased insulin requirements in late pregnancy. 85 Yet, even a small 5% increase in time above range during the second and third trimesters in women with T1D is associated with an increased risk of neonatal hypoglycemia and neonatal intensive care unit admissions. 86 Therefore, monitoring of both fasting and postprandial blood glucose is recommended during pregnancy in all women with diabetes mellitus 82 and underlines the importance of a good predictability of insulin action to ensure stable glycemic control and to prevent hypoglycemia.
According to data from retrospective studies and a limited number of RCT, IAsp or ILispro is not associated with adverse maternal or fetal outcomes.87-89 Similarly, there are no reported safety issues with the additional excipients in URAA. Large study data in pregnant women are usually lacking and this is also true for RAA 90 ; caution is therefore still warranted. Nevertheless, the available data suggest that IAsp and ILispro in combination with the additional excipients are a safe option for pregnant women with diabetes during pregnancy and lactation. For URAsp, data from two clinical studies in pregnant women are now available. The first was a retrospective study of women with GDM on MDI therapy and reported significantly lower postprandial PPG levels and lower glycemic variability in women using URAsp compared with those using IAsp. 91 The CopenFast trial was an open-label RCT of women with pre-existing T1D or T2D using URAsp in MDI or pump therapy during pregnancy. The results demonstrated comparable fetal growth and HbA1c values but significantly fewer severe hypoglycemic events with URAsp versus IAsp. 29 Other pregnancy or three months postdelivery outcomes as well as TEAE were comparable.
In conclusion, the rapid onset and offset of URAA may offer a benefit to pregnant women with either GDM or pre-existing T1D or T2D by providing better predictability of insulin action and improved postprandial glycemic control without increasing safety outcomes, although definitive data from studies on URLi in pregnancy are currently not available. Bearing in mind the risk of hyperglycemia for both mother and infant in the light of the existing safety data, we support the use of URAA during pregnancy where achievement of PPG control is a priority.
Ultra-Rapid-Acting Insulins in Special Groups and Situations
Children and adolescents
Children and adolescents with T1D are a vulnerable group where tight plasma glucose control and avoidance of hypoglycemic or hyperglycemic episodes is particularly important for the development of the brain and other organs. 92 At the same time, diabetes management in young children is complicated by a higher variability in insulin requirement 93 with a subsequently increased risk for off-range insulin dosing. The use of both basal and bolus insulins in MDI or prandial insulins in insulin pump therapy is the recommended treatment approach for young people with T1D according to the current ISPAD guidelines, 44 and glucose should be monitored regularly, ideally using CGM. 94
URAA as a novel class of even faster prandial insulins offer an even faster onset and offset of insulin action compared with RAA also in children and adolescents.95,96 Therefore, URAA have been explicitly investigated in this age group in two large RCT, the ONSET 7 trial and the PRONTO-Peds trial.21,22 In both studies, mealtime and open-label postmeal URAsp or URLi were compared with mealtime IAsp or ILispro, respectively, in MDI therapy for 26 weeks. Regarding change from baseline in HbA1c, URAA were noninferior, 22 or slightly superior with an estimated treatment difference of −0.17% (95% CI: −0.30, −0.03) when mealtime URAsp was compared with IAsp. 21 According to a post hoc analysis of the ONSET 7 data, the effect on HbA1c was independent of the participants’ age. 21 Mealtime URAA in both studies significantly decreased PPG excursions at one hour, while postmeal URAA were shown to be noninferior to the respective mealtime RAA in this regard. Also, the rate of severe or documented hypoglycemia and other TEAE was comparable in both trials, except for a higher number of injection site reactions in the PRONTO-Peds trial. 22 Yet, although URAA can be safely administered up to 20 minutes after starting a meal, children should receive the bolus preferentially at the start of the meal. 44 This is also supported by the finding of an increased risk of nocturnal or ≤4 hours postmeal hypoglycemia with postmeal URAA administration.21,22
While URAA have been shown to be safe and effective for children and adolescents in MDI therapy, data on their use in CSII devices are largely lacking. However, a small prospective study conducted in children and adolescents with T1D using URAsp in a sensor-augmented pump documented increased TIR and less time in hyperglycemia compared with IAsp. 97 Three studies using a hybrid or fully closed-loop system in children showed comparable efficacy of URAsp to IAsp.65,66,70 Also, a Portuguese retrospective analysis in pediatric individuals with T1D confirmed an increased TIR and decreased time above range with URAsp, explicitly also in the subgroup of participants using insulin pump therapy. 98
To conclude, based on this evidence as well as the supportive study data from URAA in CSII therapy in adults (see section regarding Insulin pump therapy), URAA can be recommended for use in pediatric MDI as well as CSII therapy, in line with their current approval for the respective use by the EMA (children ≥one year old) and FDA (children ≥two years old). Despite the increasing prevalence of T2D in children, URAA in T2D have only been evaluated for safety and efficacy in adults ≥18 years of age and therefore cannot currently be recommended for children or adolescents with T2D.
Elderly individuals
The group of elderly individuals with diabetes is growing worldwide. It is estimated that in 2019, the prevalence of diabetes among those older than 65 years old was 19%. 99 The treatment of elderly people with insulin requires specific attention to the particular needs of this group, including potential comorbidities, difficulties with insulin administration, unpredictable dietary intake and timing, and a high risk of microvascular and macrovascular complications. 100
The ONSET and PRONTO trials, which investigated the efficacy and safety of URAA in individuals with T2D, have also included participants above 65 years of age, so the results derived from these studies should also be applicable to older individuals. This was confirmed by subgroup analyses of the ONSET 9 and PRONTO-T2D trials demonstrating a similar glycemic control of URAA in individuals ≥65 years compared with younger adults with T2D,101,102 with sustained safety outcomes. Beyond this, the PRONTO-T2D post hoc analysis particularly reported lower 1-hour and 2-hour PPG excursions in elderly people, 101 whereas the ONSET 9 substudy found a lower rate of severe or blood glucose–confirmed hypoglycemia in individuals with T2D ≥65 years. 102
In older people with T1D, the benefit of URAA is less clear. However, the faster onset and offset of URAA is confirmed by data from phase I studies that explicitly compared the pharmacokinetics of URAsp and URLi in younger and older adults ≥65 years with T1D: both studies reported no age-related difference,103,104 underlining the accelerated onset and offset of URAA compared with RAA also in older individuals. Likewise, the studies reported that URAsp and URLi were well tolerated in both age groups. Yet, the age of the participants in the phase III studies of URAsp and URLi, with a mean age between 41.1 16 and 46.4 20 in those with T1D, was markedly lower than the mean age of 57.4 24 to 62.8 28 in those with T2D. However, there was no evidence within the trials of an increased rate of TEAE or altered outcomes in the group of older participants with T2D or T1D. The data in elderly individuals ≥75 years or those with comorbidities, including severe renal or hepatic impairment, are very limited. Therefore, respective individuals should closely monitor glucose levels and adjust the insulin dose accordingly on an individual basis.
In summary, URAA provide better postprandial glycemic control than RAA because of their accelerated pharmacokinetics without compromising safety outcomes in individuals ≥65 years, particularly those with T2D. In elderly people with T1D and those with comorbidities or renal or hepatic impairment, further studies are warranted to definitively confirm the efficacy and safety of URAA.
Postprandial hyperglycemia
Postmeal hyperglycemia is defined by the International Diabetes Federation (IDF) Guideline on management of postmeal glucose as a plasma glucose level >7.8 mmol/L (140 mg/dL) two hours after the ingestion of food. 105 Although evidence from interventional studies is lacking, according to epidemiological studies, high PPG levels are clearly associated with an increased risk for progression of diabetic microangiopathy,106,107 the incidence of major cardiovascular events, and all-cause and cardiovascular death.108,109 Thus, good control of PPG is an important goal of insulin therapy.
URAA have clearly shown to decrease PPG excursions at 1 hour, but also other timepoints in individual studies, in all stratified groups enrolled across the ONSET and PRONTO trial programs. As a result, individuals with T1D using MDI9,15,14,16,17 or insulin pump therapy,18,20 including children and adolescents,21,22 and people with T2D, including elderly individuals ≥65 years,23,24,26,27,77 may have improved PPG glucose control with use of URAA compared with RAA. Also pregnant women with T1D or T2D showed improved postprandial glycemic control with URAsp. 29 The superior effect of URAA on 1-hour PPG excursions was likewise confirmed by a meta-analysis of both URAsp and URLi 30 as well as a pooled analysis on URAsp only. 31
For optimal postprandial glycemic control in persons who experience elevated PPG levels, it is nevertheless important to dose URAA at the start of the meal. Although postmeal URAA administration was shown to be noninferior to mealtime RAA regarding HbA1c change from baseline, PPG levels were not improved or even higher in these groups.9,16,21,22 Another caveat is the liquid mixed-meal tests generally used for the assessment of PPG and PPG increments in most of the respective clinical trials, which does not adequately resemble the real-life situation of most individuals with diabetes. Nevertheless, real-world evidence from observational studies confirmed significantly lower PPG or overall time above >10.0 mmol/L with URAsp.35,36
Increased risk of late postprandial hypoglycemia
Hypoglycemia is a common and potentially dangerous adverse effect of insulin therapy, fueled by use of exogenous insulin with pharmacokinetics that imperfectly mimic the physiologic response. 110 The International Hypoglycaemia Study Group has classified hypoglycemia into three levels, with level 1 defined as blood glucose levels below 3.9 mmol/L (70 mg/dL), level 2 with blood glucose levels <3.0 mmol/L (54 mg/dL) indicating severe, clinically important hypoglycemia, and level 3 defined as severe hypoglycemia requiring external assistance for recovery.111,112 Hypoglycemia can have severe neurological and cardiovascular consequences, including death. 113 Particularly, in pediatric insulin therapy, hypoglycemia and diabetic ketoacidosis pose a high risk to the developing brain and are a critical hazard. 114 At the same time, diabetes management in very young children is complicated by their inability to reliably communicate symptoms, and variability in insulin requirements with growth, 93 which increases the risk of imprecise insulin dosing. Older individuals represent another group with a greater risk of hypoglycemia with potentially fatal consequences, especially as their perception of hypoglycemia can be limited by nonspecific or individually less perceptible symptoms. 115
In this regard, with their faster offset of insulin action, URAA may be an appropriate prandial insulin for individuals facing an increased risk of hypoglycemia. In clinical trials, URAA tended to have a lower risk of late postprandial hypoglycemic episodes occurring between 3 and 4 hours or more than 4 hours after a meal,9,16,17 but also showed a higher rate of hypoglycemia within the first two hours after a meal (early postprandial hypoglycemia).14,15,22,23,27 The rate of severe or blood glucose-confirmed/documented hypoglycemia was generally comparable between URAA and the respective RAA, with a significantly lower rate only reported in a single trial conducted in adults with T2D using URAsp. 77 Overall, URAA were confirmed to be noninferior to RAA regarding the overall risk of hypoglycemia, which was supported by the respective meta-analysis. 30 Likewise, in CSII therapy in T1D, URAA resulted in a similar rate of overall hypoglycemic events as RAA,18,19 but a significantly reduced time in hypoglycemia in the PRONTO-Pump-2 study and in the pooled analysis across all CSII RCT.20,38 Also the observational studies resulted in a slight advantage for URAsp, with time spent below 3.0 mmol/L (54 mg/dL) either unchanged 35 or significantly decreased with URAsp use after 12 months in people with T1D. 36 In conclusion, URAA have no disadvantages compared with RAA in terms of the overall risk of hypoglycemia and are superior in this respect when used with pump therapy. They may also be superior to RAA in reducing the risk of late postprandial hypoglycemia, in particular.
The behavioral challenge of premeal insulin administration
A survey of T1D Exchange clinic registry participants found that only 21% of people with T1D followed the recommendation of administering RAA 15 to 20 minutes prior to meal consumption for optimal PPG control. 48 Those who injected insulin during or after meals had significantly higher HbA1c levels 116 and were significantly more likely to experience hypoglycemia than individuals who were able to bolus premeal. 117 The requirement to plan ahead and bolus mealtime 15 to 20 minutes before a meal adds to the burden of living with diabetes and is challenging for people to do repeatedly. Suboptimal mealtime insulin timing in both people with T1D and T2D can be associated with poor glycemic control which leads to an increased risk of microvascular complications.118-120
One factor that may improve outcomes is a greater flexibility in the dosing regimen. 119 In this respect—allowing people with diabetes to inject their prandial insulin just before meals without having to wait several minutes for optimal efficacy—URAA indeed offer greater flexibility to RAA. They are also safe to be administered up to 20 minutes after starting a meal, albeit at the cost of a reduced glucose-lowering effect compared with mealtime administration,9,16,21,22 which leads to the conclusion that this option is less recommendable for general practice. Nevertheless, postmeal timing of URAA was noninferior to mealtime RAA regarding changes of HbA1c from baseline in the respective trials (Table 1). Thus, it is likely that this treatment option will benefit those who previously tended to use RAA after meals. As well as being more convenient, people living with diabetes may be at lower risk of hypoglycemia due to short-term changes in meal composition or size (such as eating a smaller meal than originally expected) with the option of administering prandial insulin following the meals.
Therefore, URAA may fill a gap in the treatment of people who struggle with the rigid demands of premeal insulin injections by offering greater treatment flexibility. This may also be inferred from a reported improvement in quality of life among individuals with T1D after replacing conventional RAA with URAsp, as described in the GoBolus study. 35
Practical Considerations: Switching From Rapid-Acting to Ultra-Rapid-Acting Insulins
In clinical practice, it is important to consider the different pharmacokinetic properties of URAA in comparison with RAA. The glucose-lowering effect occurs more rapidly after injection of URAA class insulins, resulting in a significantly better postprandial glycemic control. 30 However, this advantage is inherently associated with a slightly higher risk of hypoglycemia within the first two hours after the start of a meal.14,15,22,23,27 In addition, while the quicker offset of URAA may help reduce the risk of late postprandial hypoglycemia,9,16,17 it may potentially lead to elevated plasma glucose levels several hours after a meal. 17 Therefore, people with diabetes should be made aware of the different pharmacokinetic properties of URAA and the associated increased risk of hypoglycemia early after the injection. We recommend an increased frequency of glucose monitoring during the first days after initiation of treatment.
URAA contain an identical amount of active insulin to the corresponding RAA, allowing users to maintain their usual dose and dosing regimen for both MDI and CSII therapy. This was common practice in the ONSET and PRONTO study programs. In line, total insulin exposure was comparable between URAA and RAA in pharmacokinetic studies,7,12 and there was no change in mean daily bolus insulin dose according to the meta-analysis. 30
Individualized adjustment of pump parameters is essential in CSII therapy. In the clinical trials, the pump parameters were in fact taken from the pretrial RAA. However, clinical experience with URAsp in CSII therapy highlights the importance of reviewing pump settings when switching to URAA,54,55 and may therefore contain untapped potential for further improving glycemic control with URAA. 121 In this process, regular glucose monitoring, ideally including real-time or intermittent scanning CGM, is strongly recommended.
In contrast to the standardized liquid meal tests used in the clinical trials, the individual variation in meal duration and composition in the daily lives of people with diabetes could impact the individual outcomes of URAA. Particular attention should therefore be paid to individuals who consume extended meals or meals containing a high amount of fat in MDI therapy. Likewise, health care professionals should be cautious when prescribing URAA to individuals with slow gastric emptying or very slow digestion. In these specific cases, the ultra-rapid onset and release of action of URAA may be unfavorable, but further studies are warranted to address this topic.
Conclusions
Currently available ultra-rapid-acting insulins are novel prandial insulin analogs based on either insulin aspart or insulin lispro that offer significantly accelerated onset and offset of insulin action compared with conventional rapid-acting insulins. Each of these insulin analogs has been shown to provide sustained glycemic control in terms of HbA1c levels, but clinically and statistically significantly lower PPG excursions. This clinical advantage was not associated with an increased overall risk of hypoglycemia or other treatment-emergent adverse events, except for a higher risk of mild to moderate injection site reactions. In insulin pump therapy, ultra-rapid-acting insulins likewise provided superior PPG control as well as overall improved TIR. Emerging evidence from clinical practice suggests that the results from clinical trials can be extrapolated to the real-life situation of people living with diabetes.
The ultra-rapid-acting insulin analogs URAsp and URLi are safe and effective prandial insulins that more closely mimic the physiologic insulin response than conventional insulin aspart or lispro. These insulins represent an important, clinically useful new option for many people with T1D and T2D.
Footnotes
Acknowledgements
The authors would like to thank Carina Mauersberger, PhD, from Sciarc GmbH for excellent medical writing and editorial assistance.
Abbreviations
AID, automated insulin delivery; CGM, continuous glucose monitoring; CSII, continuous subcutaneous insulin infusion; GDM, gestational diabetes mellitus; HbA1c, glycated hemoglobin; IAsp, insulin aspart; ILispro, insulin lispro; MDI, multiple daily injection; PPG, postprandial glucose; RAA, rapid-acting insulin analog(s); RCT, randomized controlled trial(s); T1D, type 1 diabetes mellitus; T2D, type 2 diabetes mellitus; TIR, time in range; URAA, ultra-rapid-acting insulin analog(s); URLi, ultra-rapid-acting insulin lispro (lispro-aabc); URAsp, ultra-rapid-acting insulin aspart (faster aspart).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: FG has served as an advisor for AstraZeneca, Boehringer Ingelheim, Eli Lilly, Merck Sharp & Dohme, Medtronic, Novo Nordisk, Roche Diabetes Care, Sanofi; has served as a research investigator for Eli Lilly, Novo Nordisk, Roche Diabetes Care; and has received grants from Eli Lilly and Roche Diabetes Care. TB served on advisory panels of Novo Nordisk, Sanofi, Eli Lilly, Boehringer Ingelheim, Medtronic, and Indigo Diabetes. TB received honoraria for participating on the speaker’s bureau of Eli Lilly, Novo Nordisk, Medtronic, Abbott, Sanofi, Aventis, AstraZeneca, and Roche. TB’s institution received research grant support from Abbott, Medtronic, Novo Nordisk, Sanofi, Novartis, Sandoz, and Zealand Pharma, Slovenian Research Agency, the National Institutes of Health, and the European Union. RMB has received research support, has acted as a consultant, or has been on the scientific advisory board for Abbott Diabetes Care, Ascensia, Bigfoot Biomedical, Inc., CeQur, DexCom, Eli Lilly, Embecta, Hygieia, Insulet, Medtronic, Novo Nordisk, Onduo, Roche Diabetes Care, Tandem Diabetes Care, Sanofi, United Healthcare, Vertex Pharmaceuticals and Zealand Pharma. RMB’s employer, nonprofit HealthPartners Institute, contracts for his services and he receives no personal income for these activities. TF has served on speaker panels for Amarin, AstraZeneca, Boehringer Ingelheim, Berlin Chemie, Cipla, Daiichi-Sankyo, Eli Lilly, Fortbildungskolleg, MSD, Novartis, Novo Nordisk, Sanofi, and Santis and has served on advisory panels for AstraZeneca, Atrogi, Bayer, Cipla, Diabetes Akademie Bad Mergentheim, Eli Lilly, Eysense, Fortbildungskolleg, Novo Nordisk, Pfizer, Sanofi, Remynd, and Roche. JBG receives research support from Merck Sharp and Dohme Ltd., Eli Lilly and Company, Boehringer Ingelheim, and Roche; and is a consultant for AstraZeneca, Novo Nordisk, Pfizer, Bayer, Anji, Boehringer Ingelheim, Eli Lilly and Company, Valo, and Vertex. CM serves or has served on the advisory panel for Novo Nordisk, Sanofi, Merck Sharp and Dohme Ltd., Eli Lilly and Company, Novartis, AstraZeneca, Boehringer Ingelheim, Roche, Medtronic, ActoBio Therapeutics, Pfizer, Imcyse, Insulet, Zealand Pharma, Avotres, MannKind, Sandoz, and Vertex. Financial compensation for these activities has been received by KU Leuven; KU Leuven has received research support for CM from Medtronic, Imcyse, Novo Nordisk, Sanofi, and ActoBio Therapeutics; CM serves or has served on the speakers’ bureau for Novo Nordisk, Sanofi, Eli Lilly and Company, Boehringer Ingelheim, AstraZeneca, and Novartis. Financial compensation for these activities has been received by KU Leuven. CM is president of EASD. All external support of EASD is to be found on ![]() . HWR served as a consultant for and/or has received research support from Bayer, Boehringer Ingelheim, Eli Lilly, Inversago, Medtronic, Merck, MSD, Novo Nordisk, Pacira, and Sanofi. OS is founder and CEO of Sciarc GmbH. OS served on speaker panels and/or on advisory panels for Abbott, Bayer, Boehringer Ingelheim, Eli Lilly, Glooko, LifeScan, Lilly, Mannkind, Sanofi, and Woerwag. EGW has received personal fees from Abbott, AstraZeneca, Dexcom, Eli Lilly, Embecta, Glooko, Insulet, Medtronic, Novo Nordisk, Roche, Sanofi, and Ypsomed and research support from ABCD, Abbott, Diabetes UK, Embecta, Insulet, Novo Nordisk, and Sanofi.
. HWR served as a consultant for and/or has received research support from Bayer, Boehringer Ingelheim, Eli Lilly, Inversago, Medtronic, Merck, MSD, Novo Nordisk, Pacira, and Sanofi. OS is founder and CEO of Sciarc GmbH. OS served on speaker panels and/or on advisory panels for Abbott, Bayer, Boehringer Ingelheim, Eli Lilly, Glooko, LifeScan, Lilly, Mannkind, Sanofi, and Woerwag. EGW has received personal fees from Abbott, AstraZeneca, Dexcom, Eli Lilly, Embecta, Glooko, Insulet, Medtronic, Novo Nordisk, Roche, Sanofi, and Ypsomed and research support from ABCD, Abbott, Diabetes UK, Embecta, Insulet, Novo Nordisk, and Sanofi.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The publication has been funded by an unrestricted educational grant from Eli Lilly and Company.
