Abstract
Background:
To meet their glycated hemoglobin (HbA1c) goals, youth with type 1 diabetes (T1D) need to engage with their daily T1D treatment. The mealtime insulin Bolus score (BOLUS) is an objective measure of youth’s T1D engagement which we have previously shown to be superior to other objective engagement measures in predicting youth’s HbA1c. Here, to further assess the BOLUS score’s validity, we compared the strengths of the associations between youth’s HbA1c with their mean insulin BOLUS score and a valid, self-report measure of T1D engagement, the Self-Care Inventory (SCI).
Methods:
One-hundred and five youth with T1D self-reported their T1D engagement using the SCI. We also collected two weeks of insulin pump data and a concurrent HbA1c level. We scored youth’s SCI and calculated their mean insulin BOLUS score using standardized methods. For the analyses, we performed simple correlations, partial correlations, and multiple regression models.
Results:
Youth had a mean age of 15.03 ± 1.97 years, mean time since diagnosis of 8.11 ± 3.26 years, and a mean HbA1c of 8.78 ± 1.49%. The sample included n = 58 boys (55%) and n = 96 families (91%) self-identified as white. Simple correlations between youth’s age, HbA1c, SCI total score, and BOLUS score were all significant. Partial correlation and regression models revealed that youth’s insulin BOLUS score was more strongly associated with HbA1c than the SCI.
Conclusions:
Youths’ BOLUS score has better concurrent validity with HbA1c than the SCI. We should consider reporting the BOLUS score as an outcome metric in insulin pump data reports.
Introduction
Type 1 diabetes (T1D) management requires a combination of both clinician-directed guidance and patient-directed behaviors.1,2 Engagement in daily T1D self-management is necessary to meet goals related to glycemia and treatment outcomes. 3 In the management of T1D, multiple methods of assessing treatment engagement exist (ie, self-report, structured interviews, or electronic monitoring) and each offer different considerations.4,5 For instance, self-report measures and interviews may offer the advantage of being inexpensive to administer and able to provide a comprehensive assessment of T1D self-care (ie, carbohydrate counting, insulin use, glucose monitoring, appointment keeping). Although a disadvantage of these engagement tools is that they may be vulnerable to responder bias.4-6 In contrast, electronic monitoring may offer the advantage of being a low burden, objective assessment tool, although a disadvantage may be their narrower focus on engagement to a specific T1D self-care behavior (ie, glucose monitoring, insulin use). 4 In 2 previous studies, researchers compared the Self-Care Inventory (SCI) and frequency of self-monitoring blood glucose to identify the measure that best-predicted glycated hemoglobin (HbA1c) levels in youth with T1D.6,7 Interestingly, in both studies, the researchers identified youth’s SCI score as the strongest predictor of youth’s HbA1c level after controlling for demographic factors (eg, youth age, race, and sex).6,7 However, we have developed and validated an objective measure of mealtime insulin use in youth with T1D called the mealtime insulin Bolus score (BOLUS) and empirically determined that the BOLUS was a stronger predictor of youth’s current and future HbA1c than frequency of glucose monitoring,8,9 suggesting it is a valid proxy measure of treatment engagement for youth with T1D wherein mealtime insulin boluses are recommended (eg, youth using open and closed-loop insulin pumps and digital insulin pens). To date, no study has directly compared the BOLUS to youth’s SCI score to determine whether one is the stronger predictor of youth HbA1c level. Therefore, our objective in this study was to evaluate how youth’s BOLUS score compared with their SCI total scores in explaining variations in HbA1c. Specifically, we hypothesized that youth’s BOLUS scores would negatively correlate with youth’s mean HbA1c level. We also hypothesized that youth’s BOLUS scores would be a stronger predictor of HbA1c than youth’s SCI total scores.
Methods
We extracted data from the Children’s Mercy Hospital (CMH) database on Type 1 diabetes in Pediatrics (Mercy on TODP), which contains clinical, laboratory, and demographic data documented in the electronic health records of youth withT1D seen at CMH (Kansas City, Missouri) as well as nine different rural outreach facilities since June 1, 1993. For the current analyses, we included a random sample of youth between the ages of 10 to 18 years old and who had a completed SCI, a contemporaneous HbA1c value, and at least 14 days of insulin pump data from the same clinic visit. We excluded youth who did not have a diagnosis of T1D, youth with T1D who were receiving any other medical treatment that could affect their glycemic levels (ie, chronic steroid therapy, immunosuppressive therapy, any documented history of thalassemia affecting the HbA1c level), and youth who were not using an insulin pump.
Measures
Self-Care Inventory
The SCI 10 is a 14-item self-report survey that measures broad person engagement to T1D self-management (eg, glucose monitoring, insulin use, and clinic visits) over the past month. SCI items prompt respondents to rate their completion of individual behaviors using a 5-point Likert scale with higher scores suggesting greater engagement levels. Scoring for the SCI yields a total score. 11 Of note, we used a version of the SCI for this study that included two modifications. First, the modified SCI replaced the item “reading food labels” with the item “eating the proper foods; sticking to meal plan.” Second, the modified SCI separated the item “eats/meals snacks on time” into two separate items “eats meals on time” and “eating regular snack.” These modifications were not specific to our study; clinic staff had previously made these modifications to enhance the SCI as a clinical assessment tool.
Mealtime Insulin Bolus Score
This validated, objective assessment of T1D engagement uses at least 14 days of insulin pump data to calculate average daily mealtime insulin use. 8 Applying published procedures, 8 youth received 1 point each for a carbohydrate associated bolus occurring between 06:00 and 10:00 hours, 11:00 and 15:00 hours, and 16:00 and 22:00 hours, for a possible maximum of 3.0 points per day. We then averaged these daily values to calculate youths’ BOLUS scores.
Glycated Hemoglobin
We used the point-of-care HbA1c value corresponding to the clinic visit where youth completed the SCI. All point-of-care HbA1c values were analyzed using an Afinion Point-of-Care Analyzer (Alere Technologies, Oslo, Norway) and methods of assay endorsed by the Diabetes Control and Complications Trial (DCCT). 12
Statistical Analysis
A priori, we determined a minimum sample size of n = 100 youth would provide 80% power to detect a correlation of r = .28 between youth’s BOLUS score and HbA1c value. We scanned the data for normality. We used simple correlations to examine the associations between youth’s BOLUS scores, SCI total scores, and HbA1c values. We used partial correlations to examine the associations of youth’s BOLUS and SCI total scores with HbA1c while controlling for the effects of each other. Finally, we used multiple regression models to further evaluate associations between youth’s SCI total scores, BOLUS scores, and HbA1c levels while controlling for the influence of youth age, sex, race, and age at diagnosis.
Results
The final sample included n = 105 youth with T1D. Youth had a mean age of 15.03 ± 1.97 years, with n = 18 (17%) youth less than 13 years old, n = 53 (50%) youth between 13 and 16 years old, and n = 34 (32%) youth greater than 16 years old at the visit from which we extracted their data. Youth had a mean time since diagnosis of 8.11 ± 3.26 years and a mean HbA1c of 8.78 ± 1.49% at the visit from which we extracted their data. The sample included n = 58 boys (55%) and n = 96 families (91%) self-identified as white. Forty-six percent of youth used a continuous glucose monitor (CGM), 71% of youth used an open-loop pump, and 29% used a closed-loop pump. Youth self-reported a mean SCI total score of 37.30 ± 6.03. Their mean BOLUS score was 2.11 ± 0.55. Simple correlations demonstrated significant negative correlations between youth’s HbA1c value, SCI total score, and BOLUS score. Youth’s SCI total score and BOLUS score were also significantly positively correlated (Table 1).
Simple Correlations.
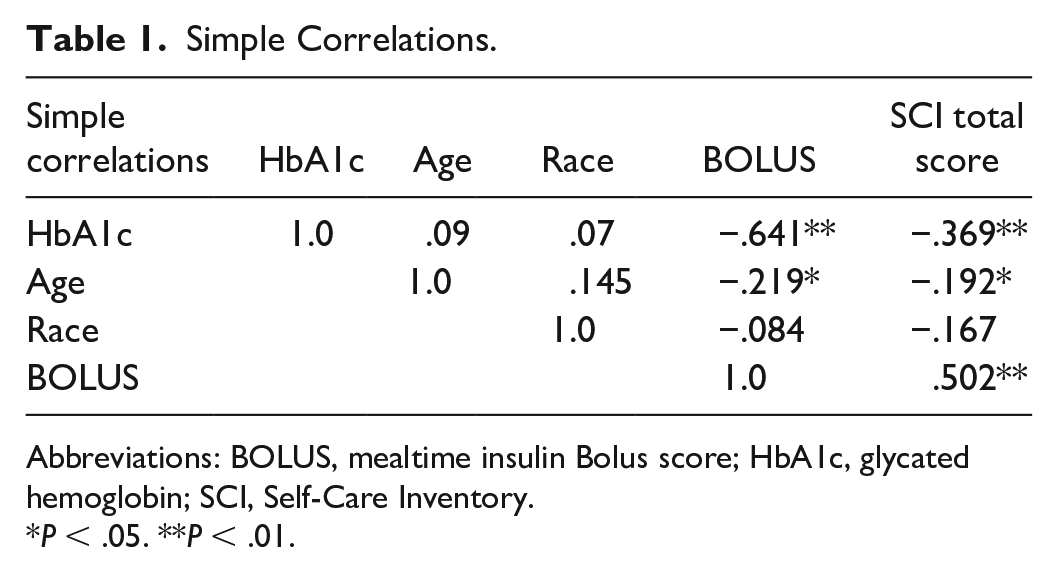
Abbreviations: BOLUS, mealtime insulin Bolus score; HbA1c, glycated hemoglobin; SCI, Self-Care Inventory.
P < .05. **P < .01.
Partial correlation of the association between youth’s SCI total score and HbA1c value, while controlling for youth’s mean BOLUS score was not significant (r = −.071, P = .475). However, partial correlation of the association between youth’s mean BOLUS score and HbA1c value, while controlling for their SCI total score was significant (r = −.567, P < .000). These results suggest that irrespective of the influence of youth’s SCI total scores on their mean BOLUS scores and HbA1c levels, the BOLUS score remains significantly associated with youth’s HbA1c values.
To further explore the associations between youths’ SCI total scores, BOLUS scores, and HbA1c levels, while controlling for potential covariates, we conducted a series of multiple regression models. In the first model, we included youth age, race, sex, time since diagnosis, and SCI total score as the independent variables and HbA1c as the dependent variable. This model produced R2 = .14, F1,104 = 16.227, P < .000. Moreover, in this model, youth’s SCI total score was the only significant predictor of their HbA1c level (β = −.091, P < .000). In the second model, we included youth age, race, sex, time since diagnosis, and mean BOLUS score as the independent variables and HbA1c as the dependent variable. The second model produced R2 = .411, F1,104 = 71.928, P < .000. Also, in the second model, youth’s mean BOLUS score was the only significant predictor of their HbA1c level (β = −1.73, P < .000). In the third model, the independent variables were youth age, race, sex, time since diagnosis, SCI total score, and mean BOLUS score, with youth HbA1c as the dependent variable. In this stepwise model, once youth’s mean BOLUS score entered the model, all other covariates, including youth’s SCI total scores, were no longer significantly related to youth’s HbA1c level. An interpretation of the unstandardized coefficients of model three suggests every 1-point increase in youth’s BOLUS score may associate with a 1.73% decrease in youth’s HbA1c level.
Discussion
In a previous validation study, we demonstrated that the BOLUS was a stronger predictor of youth’s current and future HbA1c than frequency of self-monitoring blood glucose.8,9 At present, this study contributes to the literature by also demonstrating that the BOLUS is a stronger predictor of youth’s current HbA1c level than self-reported T1D engagement, as measured by a modified form of the SCI. Specifically, in partial correlations, youth’s mean BOLUS score maintained a robust association with HbA1c even when controlling for the influence of youth’s SCI total scores. In addition, in a stepwise multiple regression model that included other potential covariates (ie, age, sex, race, time since diagnosis), results confirmed a strong association between youth’s BOLUS score and their HbA1c level irrespective of the influence of youth’s SCI total score.
While multiple measures of T1D treatment engagement exist, the SCI and frequency of self-monitoring blood glucose are two measures used frequently in research.3-5 Both measures show significant correlations with youth’s current HbA1c levels.5,7 There is also evidence suggesting frequency of self-monitoring blood glucose significantly correlates with youth’s future HbA1c levels up to 12 months. 9 In contrast, the BOLUS score is a relatively new measure of T1D engagement. The BOLUS is unique because it is the only validated, objective measure of T1D engagement that is based on youth’s self-delivery of medication (ie, insulin). 8 The BOLUS can be easily and reliably calculated from youth’s insulin pump records. Moreover, with increasing uptake of CGMs and closed-looped insulin pumps in youth with T1D, the BOLUS may represent a relatively durable objective measure of T1D engagement.
Our results suggest that for every 1-point increase in youth’s insulin BOLUS score, there was a 1.73% decrease in youth’s HbA1c levels (full model and BOLUS only model). In comparison for every 1-point increase in youth’s SCI total score, there was a .09% decrease in youth’s HbA1c levels (SCI model only). This comparison may represent the superiority of using youth’s BOLUS score versus SCI total score when explaining variations in youth’s HbA1c levels. It also may offer a practical and specific treatment target for lowering HbA1c levels in youth with T1D, namely, implementing strategies to promote consistent mealtime insulin use.
Consistent with previous research, we did not observe an association between youth race and youth mean BOLUS scores 8 or SCI total scores. 7 Interestingly, with simple correlations, we did observe significant negative associations between youth’s age with their BOLUS score and SCI total score (r = −.219, P = .024, r = −.192, P = .049, respectively). These simple correlations follow patterns previously observed in the literature suggesting a tendency for T1D engagement levels to be lower among older adolescents.1,2 Still in our regression models, with the addition of the two T1D engagement measures (ie, BOLUS score and SCI total score), youth age was not a significant predictor of HbA1c. This may be a positive treatment finding in that it suggests it may be possible to help youth with T1D to achieve lower HbA1c levels by promoting higher T1D engagement regardless of their age.
Recently, Adolfsson et al 13 conducted an observational study to test whether use of a connected insulin pen affected glycemic levels in a sample of adults with T1D. The study included 94 adults with T1D newly introduced to a connected insulin pen and using CGM. During routine clinic visits, adults and their provider reviewed their insulin bolusing activities and CGM; however, between visits, adults with T1D were unable to download and view their insulin bolusing activities. The researchers found a significant increase in time within the target range and a 43% reduction in missed mealtime bolus doses among adults with T1D after completing five clinic visits with their provider. Thus, the researchers concluded that adults benefited from the objective insulin data made available by the connected insulin pen and the opportunity to evaluate their past insulin dosing activities with their provider. 13 To date, the BOLUS has not been evaluated as a treatment target, although we believe it would be possible to use the BOLUS as a teaching tool in routine diabetes care and would encourage a future trial to evaluate its influence on youth glycemic levels in a real-world study.
A strength of this study is the sample, which is relatively large and representative of our overall clinic population. Youth completed the SCI, we extracted insulin pump records to calculate the BOLUS score, and we extracted HbA1c levels all from a routine diabetes clinic visit, which may enhance the generalizability of our findings. Also, we believe that we more rigorously tested the strength of the associations between our T1D engagement measures and youth HbA1c levels by focusing on adolescents who, based on their age (83% 13 years or older), we expect were likely relatively self-reliant in managing T1D, and by including potential confounding variables (viz, youth age, sex, race, and time since T1D diagnosis) in our models. One study limitation is our limited inclusion of youth of color (ie, 9.6%). In the future, it will be important to see whether the study results replicate in a sample that includes a higher proportion of youth of color with T1D. Another limitation is our focus on cross-sectional associations between youth’s T1D engagement measures and HbA1c levels. As a follow-up, it would be important to see how youth’s SCI total scores and BOLUS scores compare when predicting HbA1c levels into the future, as this might provide an indication of the stability of these associations. In this study, we used HbA1c as the sole metric of youth glycemic levels, although other glucose metrics exist and are clinically meaningful. 14 Therefore, another direction for future BOLUS validation studies is to compare associations between the youth’s BOLUS scores and other measures of T1D engagement with time within the target range, time outside of the target range, and glycemic variability. 14 Relatedly, there is a need for a study to validate the BOLUS in youth using the newer closed-loop and automated insulin delivery systems to determine whether the strength of the association (β = −1.73) between youths’ daily BOLUS score and HbA1c levels persists. Also, as the BOLUS has never been tested in adults with T1D, a future BOLUS validation study should examine its validity in a large and diverse sample of adults living with T1D. Finally, a limitation exists because this study used a modified version of the SCI versus the standard version. Although we believe the content of the two versions was largely identical (ie, only modified two items), a future study should test the existing SCI-Revised form vs the BOLUS.
To summarize, in a cross-sectional study of youth with T1D, we show that youth’s BOLUS scores demonstrate greater capacity to explain variance in HbA1c than youth’s modified SCI total scores. Therefore, for research and clinical implications, we conclude that the BOLUS is likely a more accurate measure of T1D engagement than the modified SCI and a tool that researchers and providers should consider using going forward. Furthermore, we recommend that researchers conduct a follow-up study to confirm our study results in a large racially heterogeneous sample of youth with T1D and consider a longitudinal, prospective study to compare the stability of the associations between youth’s HbA1c levels with their SCI total score or BOLUS score.
Footnotes
Abbreviations
CGM, continuous glucose monitor; BOLUS, Mealtime Insulin Bolus Score; HbA1c, glycated hemoglobin; SCI, Self-Care Inventory; T1D, type 1 diabetes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Mark A. Clements is Chief Medical Officer of Glooko, Inc and has received grants or contracts from Dexcom, Abbott Diabetes Care, National Institutes of Health, JDRF, the Emily Rosebud Foundation, Eli Lilly, Tolerion, and Garmin. Dr Susana R. Patton has received grants from the National Institutes of Health.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
