Abstract
Background:
Pen needles and autoinjectors are necessary for millions of patients needing injectable drug treatment but pose economic and environmental burdens. A durable device with a multiuse needle could reduce cost and improve user experience. This study explores a novel robust needle tip (EXP) designed for multiple uses and durability against hooking.
Method:
Needle robustness was investigated through a structural analysis. Furthermore, EXP and control needles (NF30, NF28) were evaluated in an in-vivo porcine model as pen needles or embedded in autoinjectors to study the resulting increase in skin blood perfusion (SBP). The SBP was assessed by laser speckle contrast analysis (LASCA) of 192 randomized and blinded needle insertions.
Results:
Forming a 33 µm hook against a hard surface requires 0.92 N for the NF30 control needle and 5.38 N for EXP. The EXP did not induce more tissue trauma than the NF30. There was a positive relation between needle diameter and SBP (P < .05). Furthermore, the presence of an autoinjector shield and applied force of 10 N was found to significantly reduce SBP for worn EXP needles (P < .05) compared to insertions without autoinjector shield.
Conclusions:
The investigated robust needle EXP is on par with the single-use needle NF30 in terms of tissue trauma, which is further reduced by combining the needle with a needle shield. These results should encourage the innovation and development of durable, reusable injection systems with pharmacoeconomic and environmental value and a simplified and enhanced user experience for patients.
Introduction
Although pen needles (PN) and autoinjectors (AI) for parenteral drug administration come with a significant financial and environmental cost, they are a crucial necessity for the millions of patients who require injectable drugs. The development and launch of multiuse needles could potentially revolutionize the market for subcutaneous needles and parenteral drug delivery devices. From a usability perspective, multiuse needles in AI devices (i.e. no needle handling) may provide a more user-friendly, simple experience, and eliminate the risk of needle-stick injuries. From a pharmacoeconomic and environmental perspective, it will have a positive impact by reducing the number of needles needed.
The novel needle design (EXP) investigated in this study has been designed to increase the mechanical robustness, that is, the needles ability to be used multiple times, and prevent deformation of the needle tip. 1 Mechanical robustness is accomplished by making the second and third bevel angles larger to form a more curved needle point with increased section thickness compared to conventional needles, see Figures 1 and 2. Prebending of the needle tip reduces the risk of hook formation (distortion of the needle tip). Prebent needles are typically used for penetration through membranes and in combination with implanted infusion ports. They are desired because of their anticoring properties. 2 To the best of the authors’ knowledge, no needles with a prebend design are used for subcutaneous administration of drugs yet.3,4
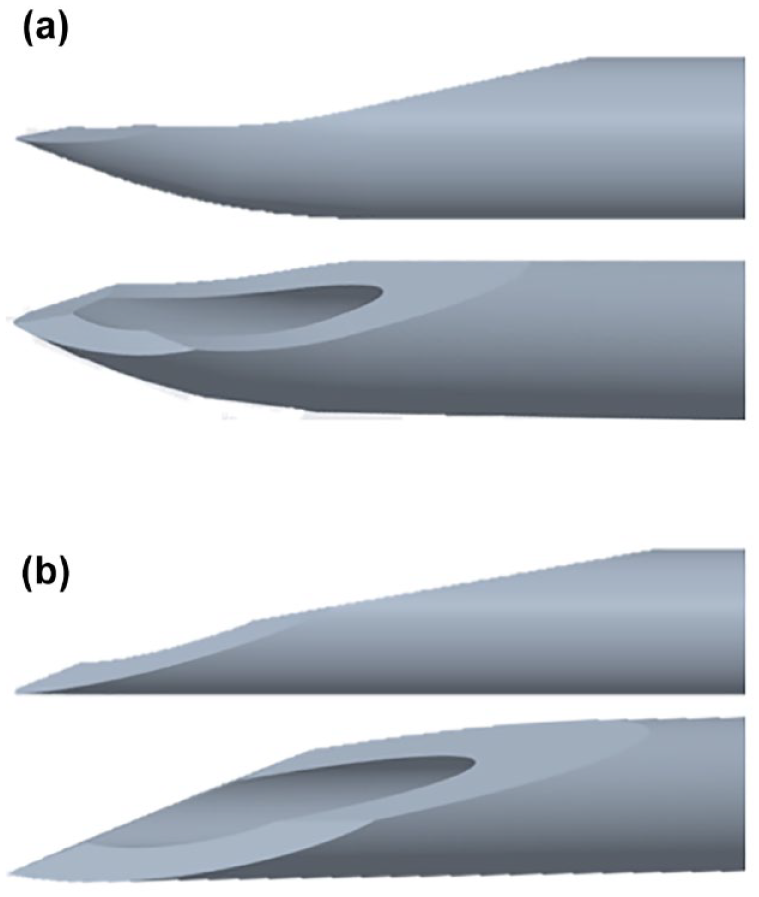
(a) EXP needle tip design with robust 3-bevel prebend needle tip. (b) NF30 standard 3-bevel single-use needle tip design.

Top: photographs of EXP needle (left) and NF30 needle (right). bottom: photographs of AI shields Ø15 and Ø30.
Early laboratory penetration force (PF) testing (results not included in this article) through polyurethane rubber (PUR) showed that the EXP needle with its larger bevel angles has a higher PF, that is, the force required to penetrate the material compared to a conventional already marketed NF30 needle of the same gauge. The higher PF for the EXP needle is not a concern in terms of dexterity as the needle PF is still negligible (<1 N) compared to the force it requires to activate a typical injection device, either by button or shield-activation, which often requires forces around 6 to 12 N. However, it is important to evaluate the performance of the EXP needle to ensure that the new and mechanically robust needle design does not compromise the injection experience. Pain and discomfort caused by needle insertions have been investigated clinically in various evaluation methods, such as, bleeding, PF, intensity, and quality of pain in human subjects.5–7 Præstmark et al8,9 have shown a correlation between pain perception and tissue trauma measured through skin blood perfusion (SBP) in humans and, furthermore, a correlation between SBP in humans and pigs. Thus, since SBP measurements on pigs are likely to predict SBP in humans, the performance and pain perception of EXP can be evaluated preclinically in this study in a porcine model. Changes in SBP have previously been used to quantify the local effect of glucagon, 10 the skin trauma induced by wounds, 11 implanted devices, 12 and needles.13,14
Needle-induced tissue trauma has been examined for various needle designs8,9 but has not been explored for a needle design intended for multiuse yet. Due to the novelty of EXP designed with multiuse in mind, it is relevant to also investigate the effect of needle wear on skin trauma.
The continuous quest to improve the user experience of subcutaneous self-administration has led to research within the field of automated delivery devices, also called AIs. In a shield-triggered AI the device and dose delivery are activated when a needle shield is pushed against the skin. Shield-triggered AIs are driven by patient comfort and ease of use and have shown overall preference compared to conventional pen injectors. 15 However, it is unknown if and how shield-triggering influences the tissue trauma and how this is affected by device design variables such as the magnitude of the applied pressure. Since needle-induced skin deflection is influenced by applying a pressurized shield against the skin, 16 it is also relevant to investigate the tissue trauma caused by EXP in combination with AI shields and applied force.
The objective of this study is to explore the effect of using multiuse needles in shield-triggered AI devices. The hypothesis is that the EXP needle performs equally well when compared to the standard NF30 single use needle.
Methods
Needle Preparations
The needles used in this study were custom made by the Novo Nordisk needle manufacturing site in Hjørring, Denmark, who assembled all needle tubes in the same hubs to enable study blinding in the skin trauma studies. All needles were made of the same material and siliconized in the same way. Both EXP and NF30 have the same needle gauge and wall thickness. To mimic in-use wear of needle lubrication, a controlled method using a robotic arm, repeatedly inserting the needles through PUR, was developed. All EXP needles labeled “worn” in this paper were inserted through PUR 75 times, as this has been estimated to mimic needle wear equal to a month of multiple daily injections. 8 The number of penetrations through PUR was based on the correlation between PFs in humans and PUR. 8 An overview of needles and configurations used in the study can be found in Table 1.
Overview of Needles and Autoinjector Shield/Force Configurations.
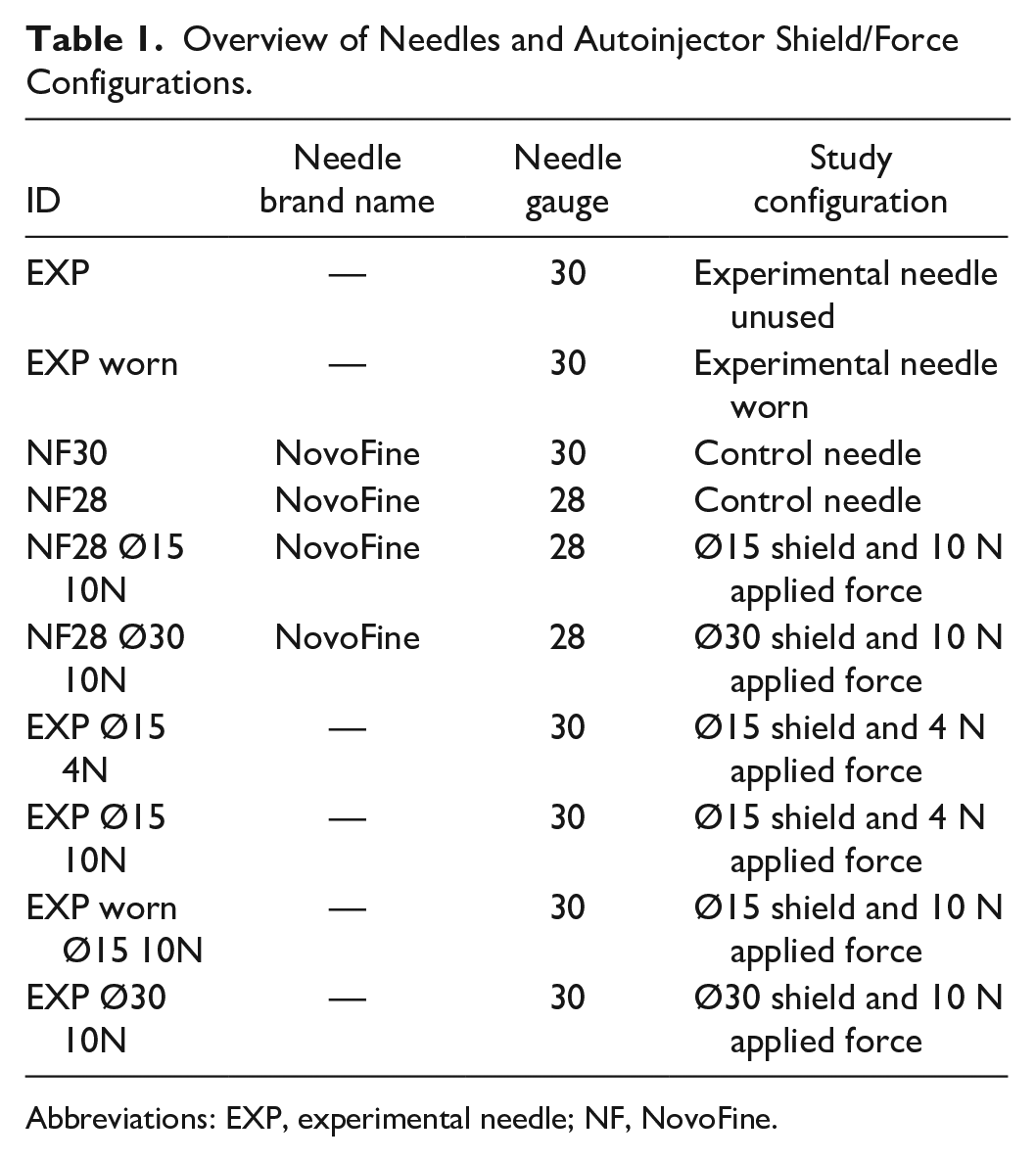
Abbreviations: EXP, experimental needle; NF, NovoFine.
Structural Analysis of Needle Robustness
A structural analysis was performed numerically in ABAQUS/CAE (Dassault Systèmes, France) to evaluate the robustness of the EXP needle. The robustness was investigated by pressing the needle against a rigid plane and quantifying the force needed to form a 33 μm hook. A comparison was done between the EXP and NF30. A hook size of 33 μm was chosen as it is too small to be seen by the human eye, yet big enough to reduce the quality of the needle below what is considered acceptable. The needle CAD geometry was imported into ABAQUS/CAE from CREO Parametric 4.0 (PTC, United States). A plate was constructed as an analytical rigid shell with a radius of 0.5 mm. Frictionless general contact was used for the contact modeling between needle and plate, since the friction was deemed to have a negligible impact on the results. A kinematic coupling in the Z-direction was set up between a reference point and the last 0.5 mm of the surface of the needle. An assembly was set up to emulate a needle being pressed against a rigid surface, and this was done similarly for both EXP and NF30. A single dynamic explicit step was set up in the analysis, with a step time of 0.004 seconds and a mass scaling factor of
The simulation is based on nominal geometry. Residual stresses from the forming process are not included in the analysis, and the analysis is quasi-static. The physical properties of the material are measured by tensile and compressing testing using tensile bars and assumed unaffected by forming processes.
AI Shield Sizes and Applied Forces
Two circular and closed AI shields were investigated; 15 mm diameter (Ø15) and 30 mm diameter (Ø30), see Figure 2. Applied shield forces of 4 N and 10 N were investigated. The magnitudes of compressive shield forces were measured with a handheld digital Advanced Force Gauge 50 N (Mecmesin, Slinfold, UK, accuracy ± 0.05 N). An overview of shields and configurations used in the study can be found in Table 1.
Laser Speckle (LASCA) Study
Landrace, Yorkshire, and Duroc (LYD) pig skin resembles human skin both anatomically and physiologically. 17 Pigs are tight-skinned animals like humans, and the presence of a dermal collagenous network makes their skin elasticity more like humans than other mammal models. 18 The neck area of female LYD pigs was used for insertions, providing skin and subcutaneous tissue thicknesses similar to humans, while having a diminished presence of panniculus carnosus. These thicknesses were verified using ultrasound imaging before the execution of experiments.
On the day before the experiments, the pigs’ necks were shaved to remove the stiff bristles that otherwise would affect the optical signal. A 10 cm × 10 cm scan area was marked with nine 3-by-3 squares, where each square represented a needle insertion site. The needle was inserted in the middle of the square, see Figure 3. The setup followed the recommendations from previous LASCA studies 19 to ensure minimal operator induced variance. See Table 2 for details of the experimental setup. The study followed the guidelines for the use and care of laboratory animals outlined by Novo Nordisk A/S and Danish laws.

The experimental setup for investigating SBP response around the needle insertion sites. 1: LYD pig with nine marked investigation sites on its neck site. 2: Digital force gauge with AI shield attached. 3: The LASCA scanner is placed above the investigation site. 4: LASCA data are transmitted from the scanner to a PC, where the data are read as arbitrary Pus. Abbreviations: LYD, Landrace, Yorkshire, and Duroc; AI, autoinjectors; LASCA, laser speckle contrast analysis.
Overview of the Experimental Setup Using the Laser Speckle Scanner.

Abbreviations: LASCA, laser speckle contrast analysis; PU, perfusion unit.
LASCA Data Analysis
The SBP data were analyzed with the PeriCam PSI system’s complementary signal processing software: PIMsoft (Perimed Sweden, Stockholm). First, the perfusion unit (PU) data were averaged in 5-second intervals to account for minor movements and breathing variability. Regions of interest (ROIs) were fitted around each needle insertion site, measuring the average of all the pixel PUs within the region. Circular ROIs of 1 cm diameter were chosen for all insertions, as this encompasses most of the signal without underestimating the average PU by inclusion of unaffected skin. Furthermore, time of interests (TOIs) were defined: one time interval for the baseline recording corresponding to the first 3 minutes of the recording and one time interval after insertion (the remaining 15 minutes). The baseline perfusion recording for each ROI was used to make a PU baseline reference. This was then compared to the signal in the 15-minutes block. Thus, the final SBP data were expressed as percentage change in blood perfusion. Blood perfusion graphs were grouped by study parameter and the area under the curve (AUC) from 0 to 15 minutes after the needle insertion was calculated for each parameter.
LASCA Study Statistics
Statistical analyses were performed using SAS JMP (SAS Institute, United States). All models were analyzed as an analysis of variance (ANOVA) fixed effects model. The effects were estimated using least square means (LSM) under the null hypothesis that all effects would be zero. A significance level (α) of 0.05 was used. Fisher’s Least Significant Difference (Fisher’s LSD) was used to indicate significant differences between all pairs of means within a group. Statistics were performed on the grouped log-transformed AUCs.
Results
Structural Analysis of Needle Robustness
The results of the structural analysis are visualized in terms of von Mises stress distribution in the needle and shown for each needle in Figure 4. Due to the differing hook styles caused by the two needle designs, a slightly different evaluation strategy was adopted for the two. For the EXP the hook is produced on the inside of the needle tube. Therefore, the hook measurement was taken from the inside edge toward the center of the needle (marked in red on Figure 4a). The hook formation and stress plots for the EXP is shown in Figure 4c. As for the NF30 needle, the hook is formed on the outside and is therefore measured from the outside edge away from the center of the needle (marked in blue on Figure 4b). The hook formation and stress plots for the NF30 is displayed in Figure 4d.
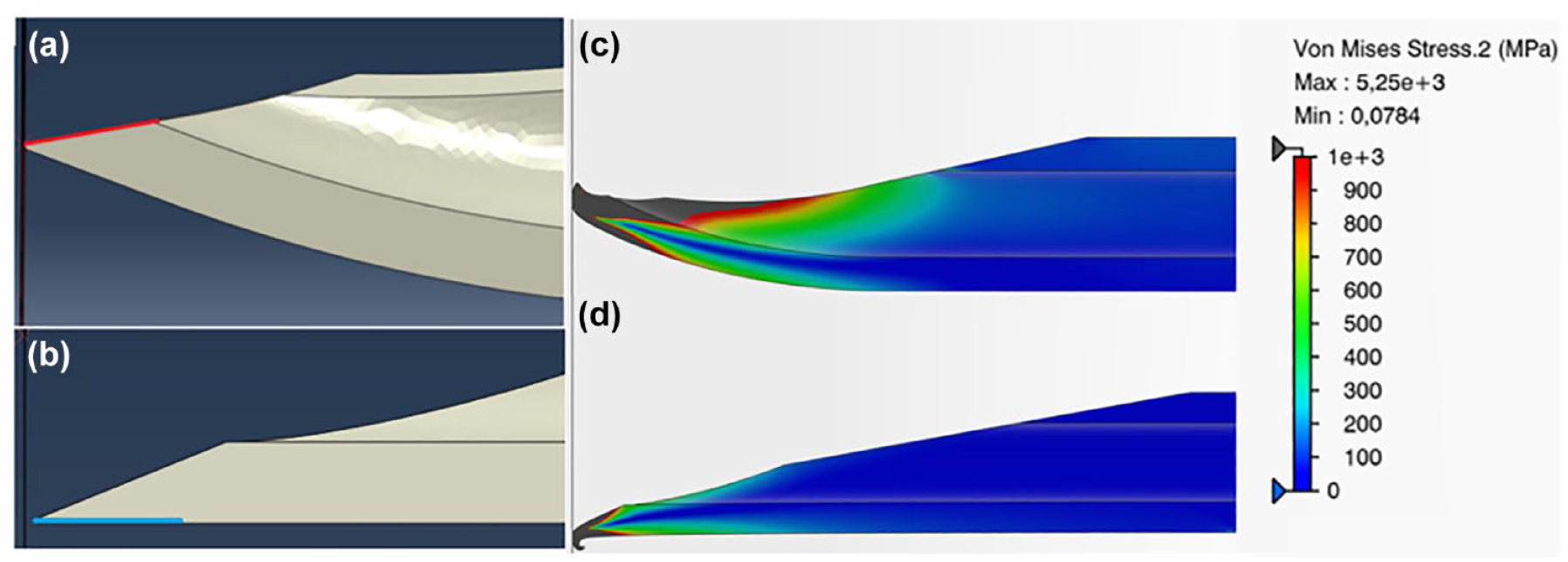
(a) Needle hooking measuring edge for EXP (red), (b) Needle hooking measuring edge for NF30 (blue), (c) Von Mises stress plot for EXP, and (d) Von Mises stress plot for NF30.
The force required to form the 33 µm hook is evaluated from the plot of the reaction force seen in Figure 5. Here, the hook on the NF30 needle was formed at 0.92 N, whereas it was formed at 5.38 N for the EXP needle. This means that the tip of the EXP needle is approximately six times stronger and less likely to form a hook, compared to the NF30 needle.
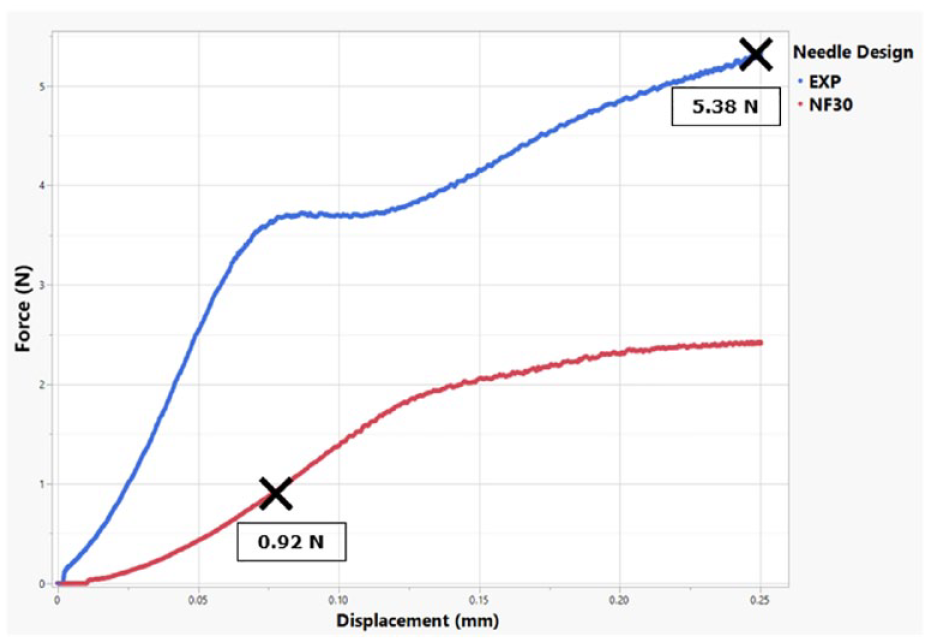
Reaction force plot for EXP needle and NF30 needle. Black crosses mark the point where a 33 µm hook is formed.
Skin Trauma
In total, 225 needle insertions were performed on both neck sides of 19 LYD pigs (weights of 48-112 kg). Out of these needle insertions, 33 were excluded from the final data set. The exclusions included measurements from three pigs + six additional data points; one pig where the scanner failed midway, one pig that necessitated supplement of additional sedation and, thus, obstructed the ongoing recording, and finally one pig where no signal could be measured by the LASCA scanner. The six additional needle insertions were excluded due to either being significant outliers or subjected to operator errors. The final data set compromises 192 needle insertions. The data are presented as the SBP after needle insertion (AUC ± standard error of mean [SEM]) normalized by the baseline SBP.
Needle tip design
Figure 6 shows that there was no significant difference in SBP increase (AUC [mean ± SEM]) from the SBP profiles seen 0-15 minutes after needle insertion) between the EXP and NF30 when comparing the with similar needle gauge and wall thickness.
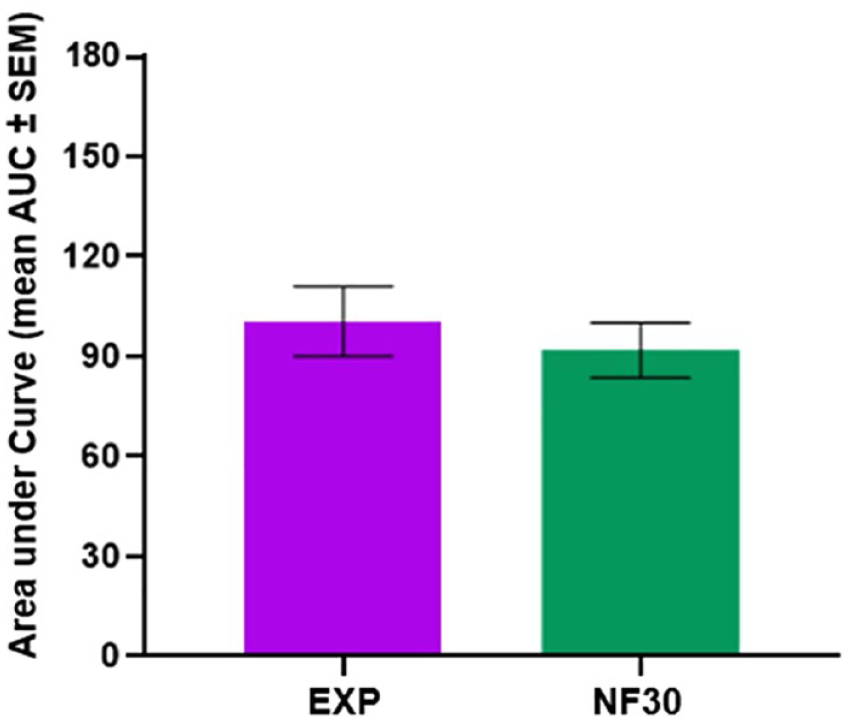
The effect of needle tip design on skin trauma. The AUC (mean ± SEM) from the skin blood perfusion profiles seen 0 to 15 minutes after needle insertion. Statistics are performed on log-transformed data, but represented in this figure as original data.
Needle wear
A slightly larger SBP response (AUC [mean ± SEM]) from the SBP profiles seen 0 to 15 minutes after needle insertion) was observed from the worn EXP needle than the new EXP needle, however not significantly different (Figure 7).
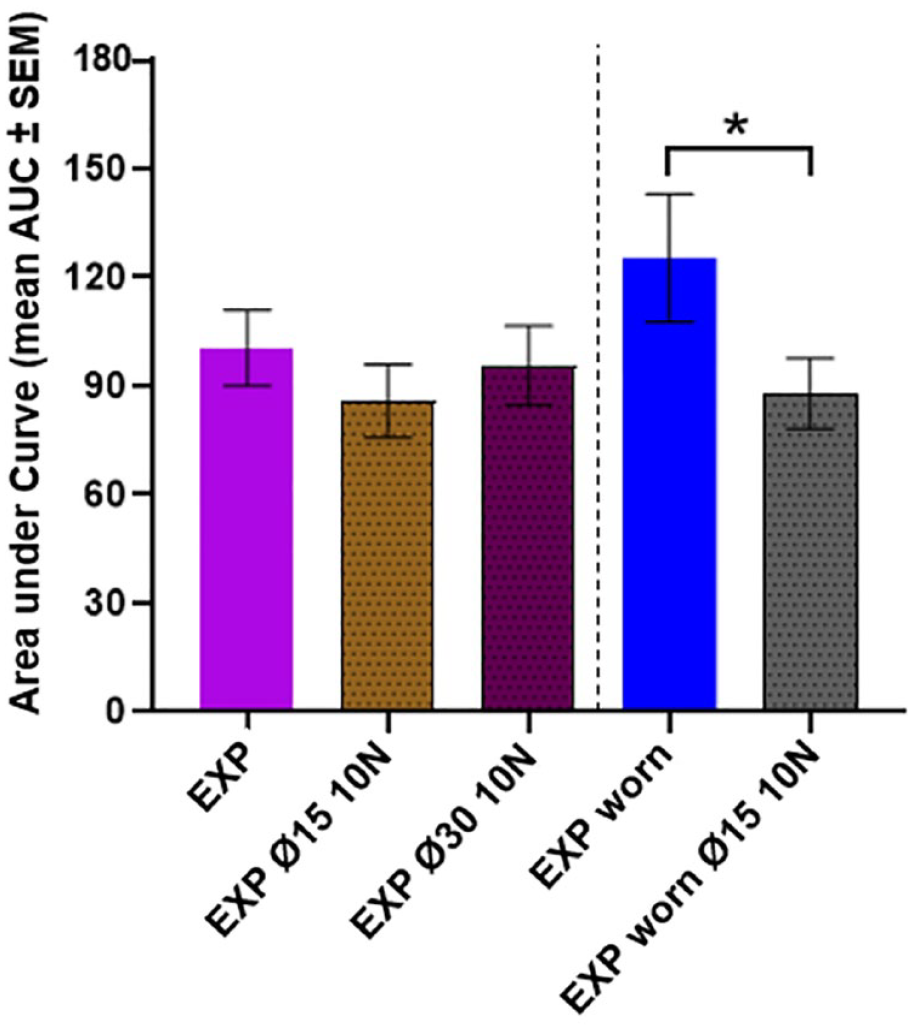
The influence of shield size and needle wear on skin trauma. The AUC (mean ± SEM) from the skin blood perfusion profiles seen 0 to 15 minutes after needle insertion. Vertical line marks results for unworn (left) versus worn needles (right). Statistics are performed on log-transformed data but represented in this figure as original data. Asterisk markings represent statistical significance of P < .05.
EXP needle in combination with shield
Worn EXP needles the in combination with needle shield resulted in significantly smaller SBP response than the unworn EXP needles without a needle shield (P < .05). This effect of shield-activation was most pronounced in the initial SBP response, resulting in a 7.11% increase in the SBP after needle insertion compared to 15.8% for bare needle insertions demonstrated in Figures 7 and 8.
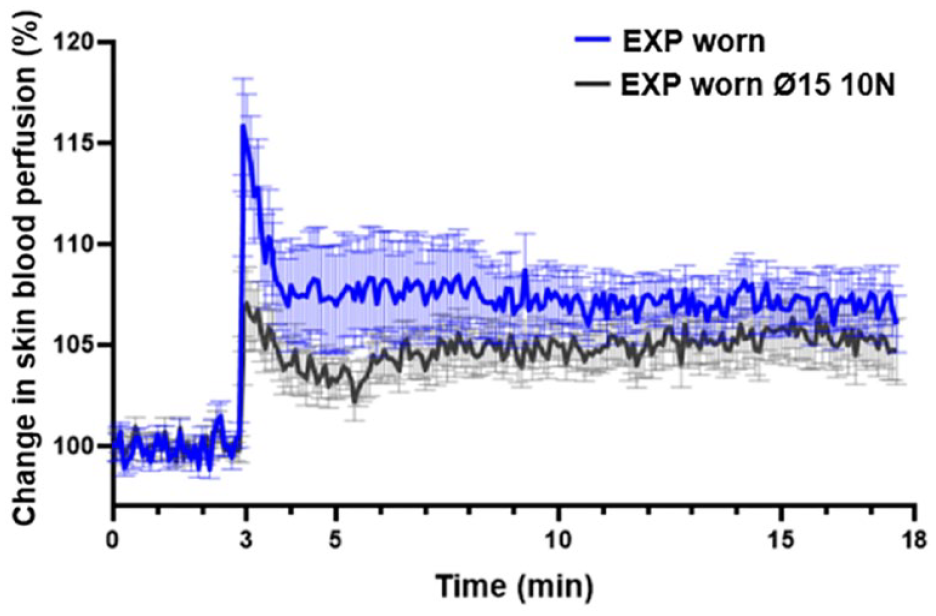
The effect of a Ø15 shield on worn EXP: the change in PU following skin penetration of worn EXP and worn EXP inserted with a shield-activation force of 10 N. Averaged (mean ± SEM) SBP profiles measured 0 to 15 min after needle insertion.
Shield size
There was no significant difference observed between the tested shield sizes (Ø15 and Ø30). However, overall, the smaller shields evoked a slightly lower average SBP response from the needles compared to the larger shields, see Figure 7.
Shield force
When using the Ø15 shield, there were no significant differences between the 4 N and 10 N shield forces; however, it is evident that both of the applied shield forces reduce the SBP response compared to insertions without needle shield and applied force (Figure 9).
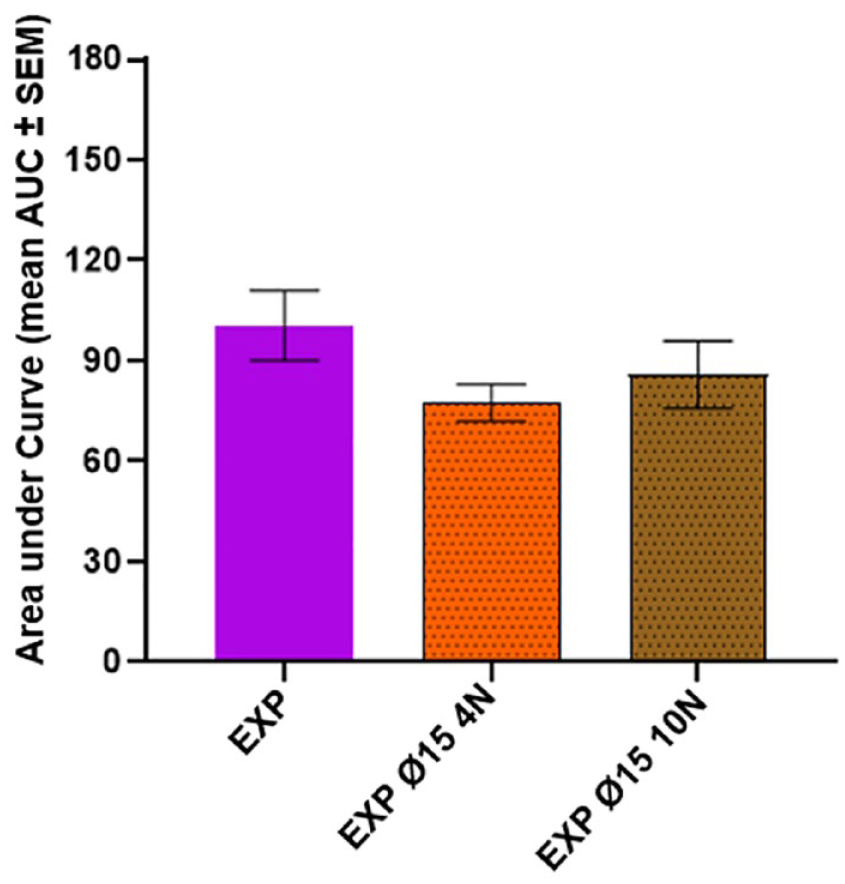
The influence of shield force on skin trauma. The AUC (mean ± SEM) from the skin blood perfusion profiles seen 0 to 15 minutes after needle insertion. Statistics are performed on log-transformed data but represented in this figure as original data.
Needle size/diameter
The NF28 needle created a larger SBP response than the NF30 needle (P < .05), demonstrated by the difference in SBP increase seen in Figures 10 and 11.
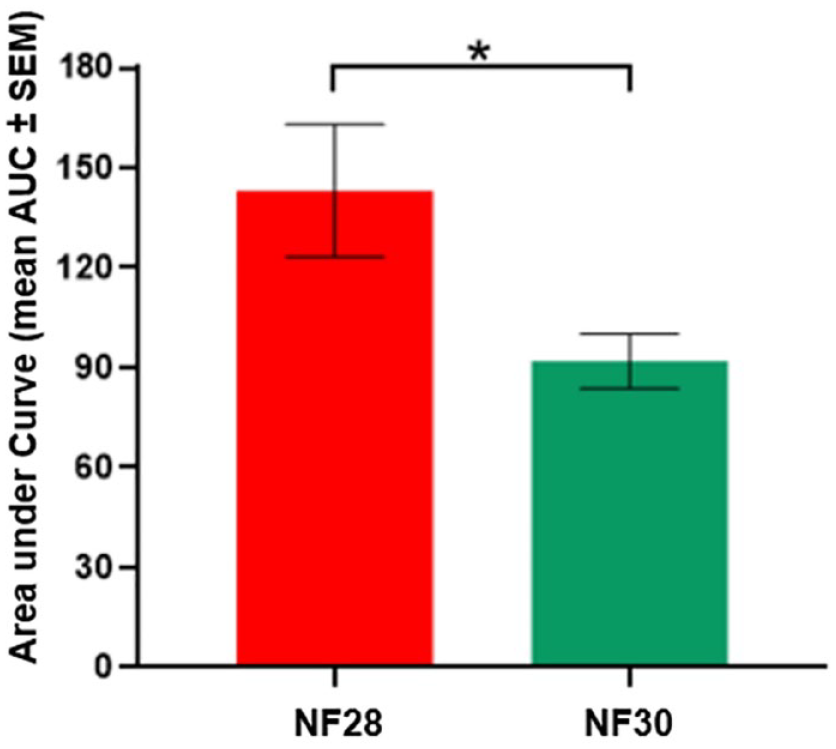
The influence of a needle size/diameter on skin trauma. The AUC (mean ± SEM) from the skin blood perfusion profiles seen 0-15 minutes after needle insertion. Statistics are performed on log-transformed data but represented in this figure as original data. Asterisk markings represent statistical significance of P < .05.
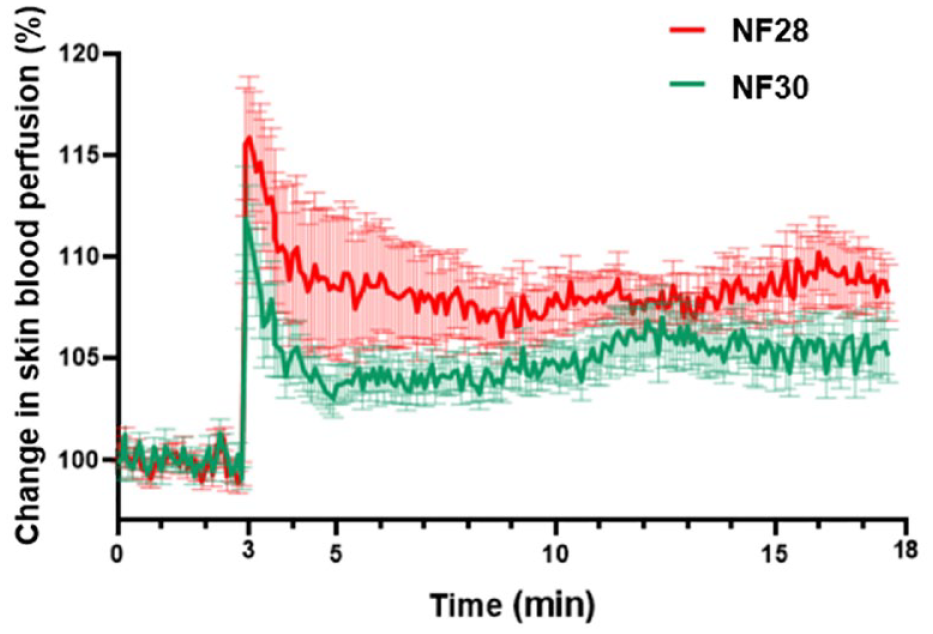
The effect of needle size/diameter: the change in PU following skin penetration of NF28 and NF30 needles. Averaged (mean ± SEM) SBP profiles measured 0 to 15 min after needle insertion.
Discussion
This study suggests that the EXP needle is mechanically robust and can withstand hooks, which should encourage the innovation and development of durable, reusable injection systems with pharmacoeconomic and environmental value and a simplified and enhanced user experience for patients.
A previous study, 8 found that a 150 μm hook caused statistically higher PF, larger SBP, and higher pain intensity compared to a similar needle tip without a hook. This is assumed to be because a hook acts like a fishing hook when the user withdraws the sharp tip from the skin following an injection. The EXP is, as demonstrated in the structural analysis results, much less likely to form a hook should the user accidentally push something against the needle or drop it onto a hard surface in between use. The EXP needle forms an inwards hook that is inside the needle diameter, as opposed to a NF30 hook that protrudes outside of the needle diameter, potentially causing the EXP to act less like a fishing hook on the skin and thus result in less pain for the user. Although these results are exciting, more clinical research investigating microbiological safety, general contamination, and residual drugs in the needles, will need to be conducted before the EXP needle can be documented as safe to reuse.
No statistical difference in average SBP was found between the EXP and the NF30 needles. So even though the EXP had a higher PF in PUR, it did not cause more needle trauma. When comparing the NF28, NF30, and EXP needles, which pairwise had either the same grind design (NF28 and NF30) or the same diameter (NF30 and EXP), the diameter was the only significant contributor to larger SBP response and thus tissue trauma.
Needle-insertion under the influence of an applied shield force reduces the amount of mechanical trauma exerted on the skin compared to needles inserted without applied shield force. This trend was evident for all investigated shields compared to bare needles and was found to significantly reduce the SBP response for worn EXP needles (P < .05). The shield size was not found to cause a statistical difference in the SBP increase. To the best of the author’s knowledge, there is no published research investigating the skin trauma caused by needles integrated in autoinjectors. The shield-activation exerts a compressive force on the skin before needle actuation, which fixates the skin at the injection site, causes a skin strain around the shield, and compresses the tissues underneath the shield tip. Compared to bare needles, it is hypothesized that these effects limit the skin deformation possibility by reducing the boundary displacement phase during needle insertion, where the skin is pushed downward by the needle. Butz et al 16 found that prestress applied to the skin surface minimizes skin deflection. Furthermore, manual needle insertions have previously been found to yield different needle-tissue interactions compared to automated insertions. 20 Results from a recent model describing autoinjector-skin interactions during dynamic needle insertion suggests that the tissue compression caused by the AI shield will regulate the stress field near the skin surface and add strain energy that is available for crack formation during the needle insertion. 21
The forces acting on the needle during insertion include the friction force acting on the needle shaft in the axial direction, the clamping force acting on the needle shaft in the radial direction and the axial cutting force needed to penetrate the tissue acting on the needle tip.22,23 Assuming the same needle geometry and insertion speed for a PN and a needle in a shield-triggered AI, the introduction of a compressive shield force will induce a concentrated pressure around the injection site and distribute the applied load over the shield. The skin will respond with a normal force toward the shield, thus potentially relaxing the magnitude of forces applied on the needle. Decreasing the magnitude of forces acting on the needle by introducing a shield force might aid in the decreased SBP response for the EXP needle in combination with needle shields. In addition, externally applied forces on the skin will compress the skin, the subcutaneous tissue layer, and their blood vessels. Compressed blood vessels have previously been found to decrease the blood flow to the skin, 24 and it is likely that the compressive force from the shield will decrease the SBP to some extent. Capillaries in the dermal skin layer are made of a single endothelial layer and will deform under load, potentially causing them to stretch out. This would reduce the probability of hitting the capillaries with the needle, which otherwise gives a blood perfusion increase.
There was no significant difference observed between the tested shield sizes (Ø15 and Ø30), however the smaller shields evoked a lower SBP response on average compared to the larger shields. This can potentially be explained by the higher force/area relation for the smaller shield. A higher sample size could potentially have detected a significant difference of that magnitude.
Future Implications
Although single-use PN are known to be reused by patients,25–28 needles are still only approved for single use due to the risk of compromised sterility, needle hooking, and an increased risk of infection and tissue trauma. However, this study proves that it is possible to develop mechanically robust needles, which do not compromise on skin trauma and thus pain perception. A recent review, highlights that one of the most important factors to consider from a pain perspective, in development of injection technology, is the needle design. 29 Together with recent findings indicating that needle reuse is not associated with increased infections, 30 this paves the way for initiating the research and development of multiuse needles.
The findings of this study indicate that the novel robust needle design is on par with conventional single-use needle in terms of SBP. Also, shield-activation indicated less variability in SBP results, suggesting a more consistent injection mechanism. Future studies could potentially investigate whether larger needles in combinations with shields could be used without increasing pain and SBP in patients. This will allow for injection of more viscous drugs without compromising the user experience.
Conclusion
A structural analysis of the investigated EXP needle found that it required a six times higher axial force to form the same 33 µm hook as on a standard NF30 needle.
In a randomized, blinded, in-vivo study in pigs the EXP needle was on par with the standard NF30 needle in terms of tissue trauma, which was further reduced by integrating the needle in a shield-triggered injection device. No significant differences in tissue trauma were observed between shield sizes of Ø15 mm and Ø30 mm and shield forces of 4 N and 10 N. Needle insertions performed in combination with needle shields caused the least skin trauma and specifically for worn needles, the application of shields significantly reduced the measured SBP response (P < .05), strongly indicating that EXP is a needle suitable for multiuse. Skin trauma was found to increase significantly with needle diameter (P < .05).
The innovative EXP needle tip design allows for potential multiple uses of the needle while reducing discomfort and skin damage associated with injections.
Footnotes
Acknowledgements
Laboratory technicians Charlotte Gustafsson and Anja Benfeldt from Novo Nordisk A/S for their help with preparation and execution of the animal experiments. Novo Nordisk A/S needle manufacturing site in Hjørring for fabricating all needles in identical hubs to enable study blinding in the skin trauma study. Michael Pedersen and Torben Strøm from Novo Nordisk A/S for their help with the structural analysis. Bente Stallknecht, Jonas Kildegaard, Antonino Azzarello, and Dan Sørensen in their help with preparation of this manuscript.
Abbreviations
AI, autoinjector; AUC, area under curve; DM, diabetes mellitus; EXP, experimental needle; G, gauge; LASCA, laser speckle contrast analysis; LYD, Landrace, Yorkshire, and Duroc; NF, NovoFine; PF, penetration force; PN, pen needle; PU, perfusion units; PUR, polyurethane rubber; ROI, region of interest; SBP, skin blood perfusion; SD, standard deviation; SEM, standard error of mean; TOI, time of interest.
Declaration of Conflicting Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A-SMS is employed as Industrial PhD student at Novo Nordisk A/S. JS and KAFP are full-time employees at Novo Nordisk A/S.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is a part of a Danish Industrial PhD project which is funded by the Danish Innovation Fund and Novo Nordisk A/S.
