Abstract
We evaluated the performance of the interoperable Cambridge hybrid closed-loop app with FreeStyle Libre 3 glucose sensor, and YpsoPump insulin pump in a real-world setting. Data from 100 users (63 adults [mean ± SD age 41.9 ± 14.0 years], 15 children [8.6 ± 5.2 years)] and 22 users of unreported age) for a period of 28 days were analyzed. Time in range (3.91- 10.0mmol/L) was 72.6 ± 11.1% overall. Time below range (<3.9mmol/L) was 3.1% (1.4-5.1) (median [interquartile range]). Auto-mode was active for 95.8% (91.8-97.9) of time. This real-world analysis suggests that the performance of Cambridge hybrid closed-loop app with this glucose sensor is comparable to other commercially available hybrid closed-loop systems.
Introduction
Hybrid closed-loop (HCL) technology can improve both glycemic control and quality of life in adults and children living with type 1 diabetes when compared to other therapies. 1 A number of HCL systems are commercially available. Most systems are compatible with just one continuous glucose monitoring device and one insulin pump limiting user choice. 2
FreeStyle Libre 3 (Abbott Diabetes Care, USA) is a recently approved continuous glucose monitor. 3 This glucose sensor was integrated with the interoperable Cambridge HCL app (CamAPS FX, CamDiab, UK) and launched in Germany making the app the first commercially available system to integrate with more than one compatible glucose sensor and more than one compatible insulin pump allowing users flexibility to personalize their HCL ecosystem. It is also the first closed-loop system integrated with this particular sensor. In the present analysis, we evaluate real-world outcomes from adult and children users of this HCL app and glucose sensor to assess its performance during free living.
Methods
Design
We retrospectively analyzed data from the first 100 commercial users for a period of 28 days from 23 January to 20 February 2023. All users were based in Germany and all consented to their data being used for this analysis. Year of birth was self-reported by users who agreed to share this information.
Cambridge HCL System
The adaptive model predictive control algorithm residing on this app automatically calculates the insulin infusion rate every 8 to 12 minutes, which is communicated wirelessly to the insulin pump. The closed-loop algorithm has a default target glucose level of 104 mg/dL, which is adjustable between 79 and 198 mg/dL across different times of day and night. This system is Conformité Européene (CE) marked use by individuals with type 1 diabetes aged one year and older including pregnancy.
Data Analysis
All users had a two-week run-in period to allow for familiarity with the system and data collection began after the run-in phase. The data were automatically streamed from the user’s phone to the cloud data repository.
Analyses were performed for all users, as well as by two age-groups (≤18years, 19 years and older). The following endpoints were calculated: mean sensor glucose, standard deviation and coefficient of variation (CV) of sensor glucose, percentage of time spent in range (70-180 mg/dL), below range (<70 mg/dL and <54 mg/dL) and above range (>180 mg/dL and >300 mg/dL), percentage of time spent in auto-mode and the number and percentage of participants achieving >70% time in range. Glucose metrics were calculated using the GStat software (version 2.3, University of Cambridge, UK). Statistical analysis was performed using Statistical Package for Social Sciences (SPSS) version 25. Normally distributed data are presented as mean ± standard deviation and non-normally distributed data are presented as median (interquartile range).
Results
Data were collected from 100 users; 63 adults, 15 children and 22 users without a documented age. Of those who had a documented age, mean age was 35.8 ± 18.3 years (range 3-76 years, Figure 1).
Bar chart of participants’ age.
Glucose endpoints and auto-mode usage overall and by age category are shown in Table 1. Mean sensor glucose was 148.2 ± 19.9 mg/dL, and overall was slightly higher in children (154.8 ± 17.8 mg/dL) than in the adult users (146.9 ± 21.0 mg/dL). Time in target range 70 to 180 mg/dL in the overall cohort was 72.6 ± 11.1% (67.6 ± 9.7% in children and 73.9 ± 11.4% in adults) (Table 1). Special consideration was given to young children (aged 3-14 years) who presented similar outcomes to the general pediatric cohort across all outcomes (Table 1).
Glycemic Metrics of 100 Real-World Users.
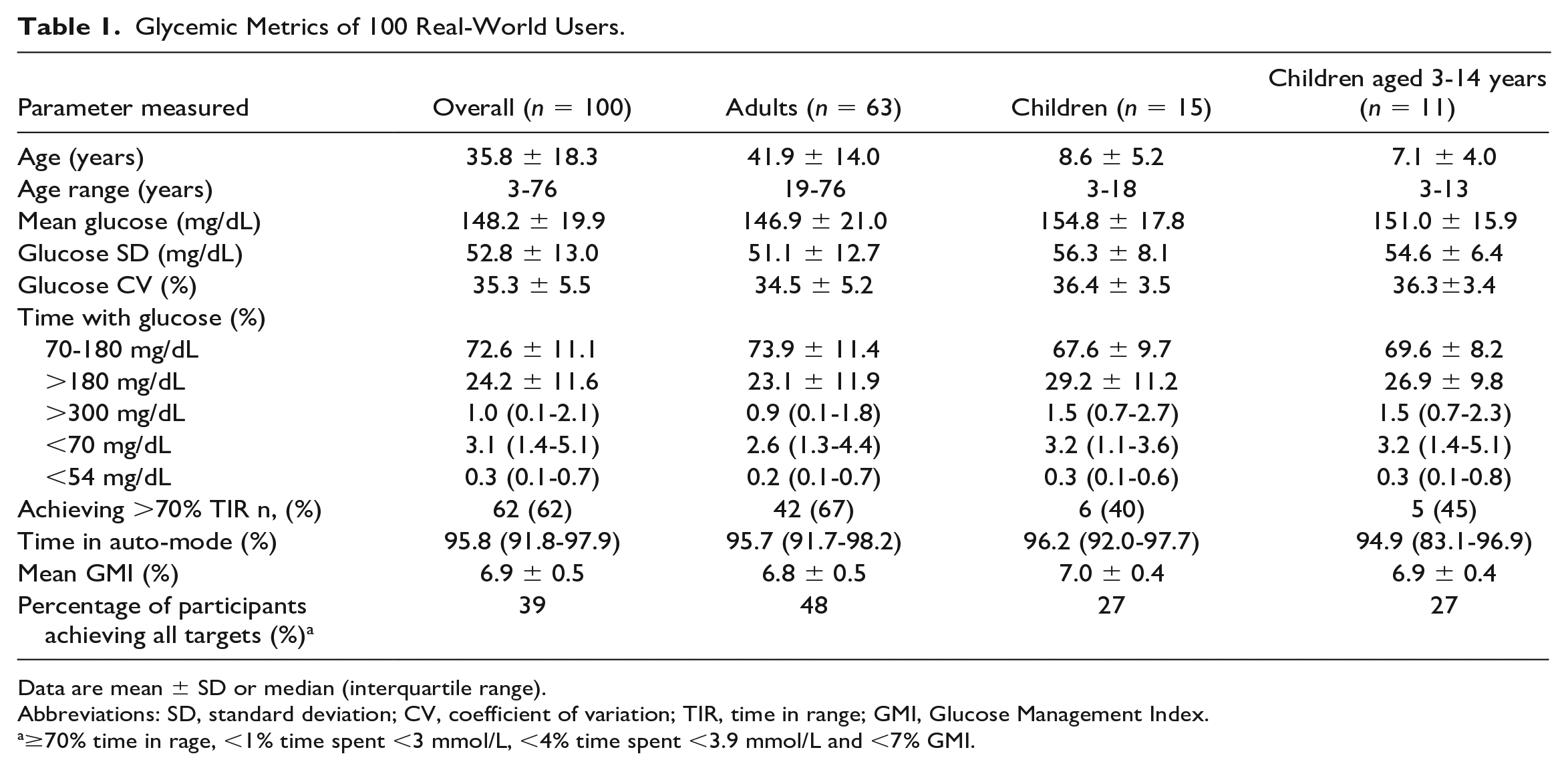
Data are mean ± SD or median (interquartile range).
Abbreviations: SD, standard deviation; CV, coefficient of variation; TIR, time in range; GMI, Glucose Management Index.
≥70% time in rage, <1% time spent <3 mmol/L, <4% time spent <3.9 mmol/L and <7% GMI.
Time spent above target (>180 mg/dL) was 24.2 ± 11.6% overall and higher in the children (29.2 ± 11.2%) than the adult cohorts (23.1 ± 11.9%) (Table 1). Time spent in significant hyperglycemia (>300 mg/dL) was 1.0% (0.1-2.1%) overall.
Time spent in level 1 hypoglycemia (<70 mg/dL) was 3.1% [1.4-5.1%] overall and was higher in children (3.2% [1.1-3.6%]) than in adults (2.6% [1.3-4.4]%). Time spent in level 2 hypoglycemia (<54 mg/dL) was 0.3% (0.1-0.7%) and was similar in both children and adults (Table 1). Overall, the standard deviation of sensor glucose was 52.8 ± 13.0 mg/dL and the CV of sensor glucose was 35.3 ± 5.5%. The glucose variability endpoints were comparable between adults and children.
Closed-loop usage was high with median 95.8% (91.8-97.9%) of time spent in auto-mode. This was comparable across adults and children.
Discussion
The present retrospective analysis evaluates the performance of an established HCL app with a novel glucose sensor in a real-world setting. Mean time in range and median time in hypoglycemia (<70 mg/dL) overall exceeded the targets set out by the American Diabetes Association4,5 and the International Society for Pediatric and Adolescent Diabetes. 6
The glycemic outcomes seen in this real-world data analysis are comparable to data from RCTs in adults and children.1,7 The data in the present analysis are also consistent with the results from real-world reports of adult and pediatric cohorts using other commercially approved HCL systems.8-15 Rates of level 1 hypoglycemia varied between studies and tended to be higher in studies with younger participants. 14
A review of one real-world study of mixed adult and pediatric populations did demonstrate some differences compared to our study. Higher rates of time in range and lower levels of time above range and time below range were demonstrated in more than 1000 participants using another HCL system for seven weeks. 16 Many of the differences observed are potentially explained by the younger participants included in our analysis. The hybrid closed-loop system evaluated in this study s is approved for use in children one year and older and participants as young as three years were included in this dataset, while the youngest participant was 14 years old in the above real-world analysis. 16 While the inclusion of younger participants in real-world evidence is important, the differences in insulin sensitivity, unpredictable activity and food intake and high variability in insulin requirements means cohorts of different ages are not directly comparable.
We also evaluated the key parameter of CV. In our cohort, only the adult group had a CV <35% variation. We observed the rates of 34.8 ± 5.2% in our adult cohort and other real-world adult studies report rates of 31-33%8,10,17 between seven weeks and six months of hybrid closed-loop use. Randomized controlled trials (RCTs) of patients with suboptimal control have reported CV rates as high as 40%. 18 Dedicated pediatric real-world and RCTs demonstrated CV rates of 37.5% 15 and 47% 19 respectively. In mixed real-world studies of adult and pediatric populations, CV rates were similar to those observed in our population at 33% 9 ; however, rates tended to be higher in younger versus adult patients. 8
Limitations of this analysis are the small number of participants and short period of follow-up data, the lack of information about users’ demographics and prior glycemic control. Despite this, this study has a number of strengths. It is the first real-world evaluation of a recently approved continuous glucose monitor in a hybrid closed-loop system and includes a broad range of participants, including very young children who are often under-represented in studies.
Conclusion
In conclusion, the present analysis contributes to the real-world evidence for hybrid closed-loop systems and supports the use of interoperable systems to improve user experience. The combination of our hybrid closed-loop app and novel glucose sensor was on average able to achieve recommended glycemic targets while also providing greater treatment option for people with type 1 diabetes.
Footnotes
Abbreviations
CV, coefficient of variation; GMI, Glucose Management Index; HCL, hybrid closed-loop; SD, standard deviation; SPSS, statistical package for social sciences.
Author Contributions
CN wrote the first draft of the manuscript. CN, MEW, and SH were involved in verifying and analyzing the data, performed the statistical analyses, and were involved in writing and editing the article. RH, MEW, and SH were involved in the design and execution. RH was involved in writing and editing the article. HA critically reviewed the manuscript. CN and MEW are the guarantors of this work and, as such, had full access to all the data in the analysis and take responsibility for the integrity of the data and the accuracy of the data analysis.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RH reports receiving speaker honoraria from Eli Lilly, Dexcom, and Novo Nordisk, receiving license fees from BBraun, declares consulting fees from Abbott Diabetes Care; patent issued in closed-loop field, being director and stockholder at CamDiab. CN declares support for attending meetings from Novo Nordisk and Sanofi, speaker honoraria from Boehringer Ingelheim and receiving research grants from Novo Nordisk. HA is a consultant at CamDiab. MEW declares patents related to closed-loop and being a consultant at CamDiab. SH reports receiving speaker honoraria from Medtronic, Sanofi, Ypsomed and Dexcom and consulting fees for CamDiab. She is a director of Ask Diabetes Ltd providing training and research support in health care settings.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
