Abstract
Background:
Nocturnal hypoglycemia (NH) remains a major burden for people with type 1 diabetes (T1D). Daytime physical activity (PA) increases the risk of NH. This pilot study tested whether cumulative daytime PA measured using a smartphone-based step tracker was associated with NH.
Methods:
Adults with T1D for ≥ 5 years (y) on multiple daily insulin or continuous insulin infusion, not using continuous glucose monitoring and HbA1c 6 to 10% wore blinded Freestyle Libre Pro sensors and recorded total daily carbohydrate (TDC) and total daily dose (TDD) of insulin. During this time, daily step count (DSC) was tracked using the smartphone-based Fitbit MobileTrack application. Mixed effects logistic regression was used to estimate the effect of DSC on NH (sensor glucose <70, <54 mg/dl for ≥15 minutes), while adjusting for TDC and TDD of insulin, and treating participants as a random effect.
Results:
Twenty-six adults, with 65.4% females, median age 27 years (interquartile range: 26-32) mean body mass index 23.9 kg/m2, median HbA1c 7.6% (7.1-8.1) and mean Gold Score 2.1 (standard deviation 1.0) formed the study population. The median DSC for the whole group was 2867 (1820-4807). There was a significant effect of DSC on NH episodes <70 mg/dl. (odds ratio 1.11 [95% CI: 1.01-1.23, P = .04]. There was no significant effect on NH <54 mg/dl.
Conclusion:
Daily PA measured by a smartphone-based step tracker was associated with the risk of NH in people with type 1 diabetes.
Keywords
Introduction
Nocturnal hypoglycemia (NH) is a major burden for people living with type 1 diabetes (T1D). At least a third of all people with T1D experience an episode of NH every week. 1 An episode of NH lasts, on average, for about 100 minutes. 2 NH significantly impacts the daytime functioning and well-being of people with T1D. 3 Also, NH is associated with a large emotional burden, typically manifesting as fear and anxiety about future episodes and frustration about being unable to predict or prevent NH. 4 Daytime physical activity (PA) is associated with a higher risk of NH.5,6 Further moderate-intensity PA reduces symptoms and hormone response to subsequent hypoglycemia. 7 Not surprisingly, most people with T1D do not meet the recommended PA targets.8,9 Furthermore, prolonged and severe NH may even lead to death in people with T1D and is thought to be one of the mechanisms for sudden unexplained death in young people with T1D. 10
Real-time continuous glucose monitoring (RT-CGM) reduces NH in people with T1D on multiple daily insulin (MDI). 11 RT-CGM integrated with insulin pumps containing predictive algorithms to automate basal insulin delivery has further reduced the risk of NH. 12 However, access to RT-CGM is limited, and more than half of those living with T1D globally still rely on MDI for their diabetes management. 13 Despite the availability of specific recommendations for insulin dose adjustments to minimize hypoglycemia, multiple challenges remain for people with T1D. 14 People with T1D must estimate their risk of NH and make adjustments to their insulin dosing or consume additional carbohydrates to avoid an episode of NH.14,15 Major factors affecting NH risk include insulin dose and timing, carbohydrate intake, daytime PA, and alcohol intake; an imbalance between these factors results in NH.16,17 However, many people with T1D lack optimal self-care strategies to avoid and treat NH. 18 Even among those trained in optimal diabetes self-care strategies like carbohydrate counting and insulin dose adjustments, quantifying PA duration and intensity can be challenging. Studies have shown that self-assessment of PA can deviate by −100% to +200% from objectively measured activity. 19 Further, simulation studies suggest that current recommendations are inadequate to prevent late post-exercise or NH. 20
The use of activity trackers allows objective quantification of daily PA. Prior studies done mostly in controlled settings have demonstrated that increased PA, as measured by wearable accelerometer devices, is associated with a higher risk of NH.21,22 Although the availability of wearable commercial activity trackers has increased, they may not be acceptable to all, as it necessitates wearing yet another device to manage a life-long condition. On the contrary, a smartphone-based activity tracker is less intrusive and immediately available to most people with T1D due to ubiquitous smartphone use. Smartphone-based activity trackers can be as accurate as wearable activity trackers in controlled settings. 23 To date, there is little evidence that activity measured using smartphone-based activity trackers in the free-living condition is associated with the risk of NH.
We did a pilot observational study to test our hypothesis that a higher daytime PA quantified using a smartphone-based activity tracker is associated with a higher risk of NH in T1D independent of total daily dose (TDD) of insulin dose and total daily carbohydrate (TDC) intake.
Methods
We recruited adults (≥21 years) with T1D duration of ≥ 5 years, able to count carbohydrates, with a HbA1c between 6 and 10%. We excluded those with a post-prandial c-peptide of more than 0.6 mcg/L as residual c-peptide is known to reduce the risk of hypoglycemia and possibly attenuate the effect of activity on NH. 24 We excluded those using continuous glucose monitoring, those with an estimated glomerular filtration rate <30 ml/min/m2, and those unable to carry a smartphone during their predominant form of PA (e.g., swimming, contact sports). People using an insulin pump without CGM were eligible to participate. The study was conducted from June 2021 to January 2022 at the Diabetes and Metabolism Centre, Singapore General Hospital. The Central Institutional Review Board (Ref No: 2021/2223) approved the study.
All participants had the smartphone-based activity tracker Fitbit MobileTrack® installed and individual Fitbit accounts created. Fitbit MobileTrack® has been validated against research-grade wearable activity trackers and found reliable. 23 Participants were instructed to always carry their smartphones. Participants wore two blinded CGM sensors, Freestyle Libre Pro®, each lasting 14 days, in total 28 days. An additional sensor was inserted if any sensor provided <7 days of data. People on MDI were required to keep a diary of carbohydrate intake and insulin doses manually or using smartphone applications like mySugr® or Diabetes:M®. Participants were instructed to continue their diabetes self-management as usual. We downloaded step counts, insulin pump data, and CGM data using respective proprietary software. We recorded the predominant type of PA of each participant at recruitment.
The daily step count (DSC, 00:00-00:00 hours) was retrieved from Fitbit accounts. An episode of NH (00:00-06:00 hours, the following day) was defined as lasting for >15 minutes and reported at two levels: <70 and <54 mg/dl. We used the “active minutes” generated by the Fitbit software to assess the intensity of PA. Fitbit calculates active minutes as metabolic equivalents of tasks (MET). The categories “light,” ‘fairly active,’ and “very active” in Fitbit data correspond to light (<3 METs), moderate (3-5.9 METs), and vigorous (≥6 METs) PA.25,26
Statistical Analysis
Each participant contributed between 4 and 27 day-night pairs for analysis. There is no consensus on deciding on a valid day using smartphone-based step trackers. Wrist-worn step trackers are known to overcount steps compared to waist-worn step trackers. 27 A cut-off of 1500 steps has been suggested to define a valid day for wrist-worn step trackers.28,29 Smartphone step tracker apps have shown equivalent accuracy to video recording when carried in a trouser pocket in controlled settings. 30 However in real-life, a smartphone could be carried around the waist pocket or held in the hand while walking. Further, not carrying the smartphone could also lead to underestimating step counts. Hence, we excluded days with <1000 DSC as this is likely due to the person not carrying the phone with him for most of the day or a day with minimal PA. The 1000 steps also coincided with the 25th percentile for all step counts.
The available data consisted of 1006 day-night pairs. We excluded 337 pairs with a DSC ≤1000, 282 due to less than 20 sensor glucose readings for night-time (0000-0600, 24 sensor glucose readings expected), 23 due to zero daily carbohydrates and/or insulin entry, nine as they belonged to a participant with c-peptide >0.6 mcg/L. After exclusions, 355 day-night pairs were available for analysis.
Descriptive statistics was used to summarize the demographic and clinical characteristics of the participants. Frequencies (percentages) were used to summarize categorical variables. The normality of the continuous variables was checked before summarizing them using either mean (standard deviation, SD) or median (interquartile range, IQR). The association between the participant characteristics and whether they had at least one NH episode within the study period was examined using either Pearson’s chi-square test or Fisher’s exact test (for categorical characteristics), or two-independent sample t-test or Wilcoxon rank-sum test (for continuous characteristics) depending on the distribution of the data.
Mixed effects logistic regression was used to estimate the effect of every increment of 1000 steps on the presence of NH (binary variable and defined as a yes if the participant had NH each night) while adjusting for TDC and TDD of insulin, treating only participant as a random effect to account for within-subject correlation. Analysis was done by treating steps as a continuous variable. Additionally, we expressed each day’s step count as a percentage of the individual’s median step count. We assumed that a change from an individual’s usual (median) step count will have a higher impact on the risk of NH than the actual step count. We arbitrarily defined 80% to 120% of the median step count as usual variance and compared it with lower than usual (<80%), higher than usual (>120%-150%) and much higher than usual (>150%). We also estimated the effect of an increment of every 10-minute duration of light, fairly active, and very active minutes on NH.
All analyses were performed using Stata 17 (StataCorp. 2021. Stata Statistical Software: Release 17; StataCorp LLC, College Station, TX). A two-sided P value <.05 was used to declare statistical significance.
Results
Twenty-seven participants were recruited, of which one was excluded due to persistent c-peptide >0.6mcg/L after 5 years of diagnosis (0.84mcg/L, paired random glucose 11.7 mmol/L), indicating a non-T1D diagnosis. Baseline characteristics of the study cohort are given in Table 1. Overall, the cohort had a median age of 27 years, a median diabetes duration of 16 years, was predominantly female (65.4%), and had a mean body mass index of 23.9 kg/m2. Fourteen participants (53.8%) used insulin pumps. The predominant type of self-reported PA at recruitment was walking (96.2%) followed by weight training (26.9%). Nocturnal awakenings due to hypoglycemia before recruitment were common with 53.8% waking up at least once per week. Hypoglycemia awareness was good with a mean Gold score 31 of 2.1 (range 1-4), and 73.1% reported a hypoglycemia detection threshold of > 3 mmol/L on the Dose Adjustment for Normal Eating (DAFNE) Hypoglycemia Score. 32 The median episodes of severe hypoglycemia during the lifetime and in the past year were zero.
Baseline Characteristics.
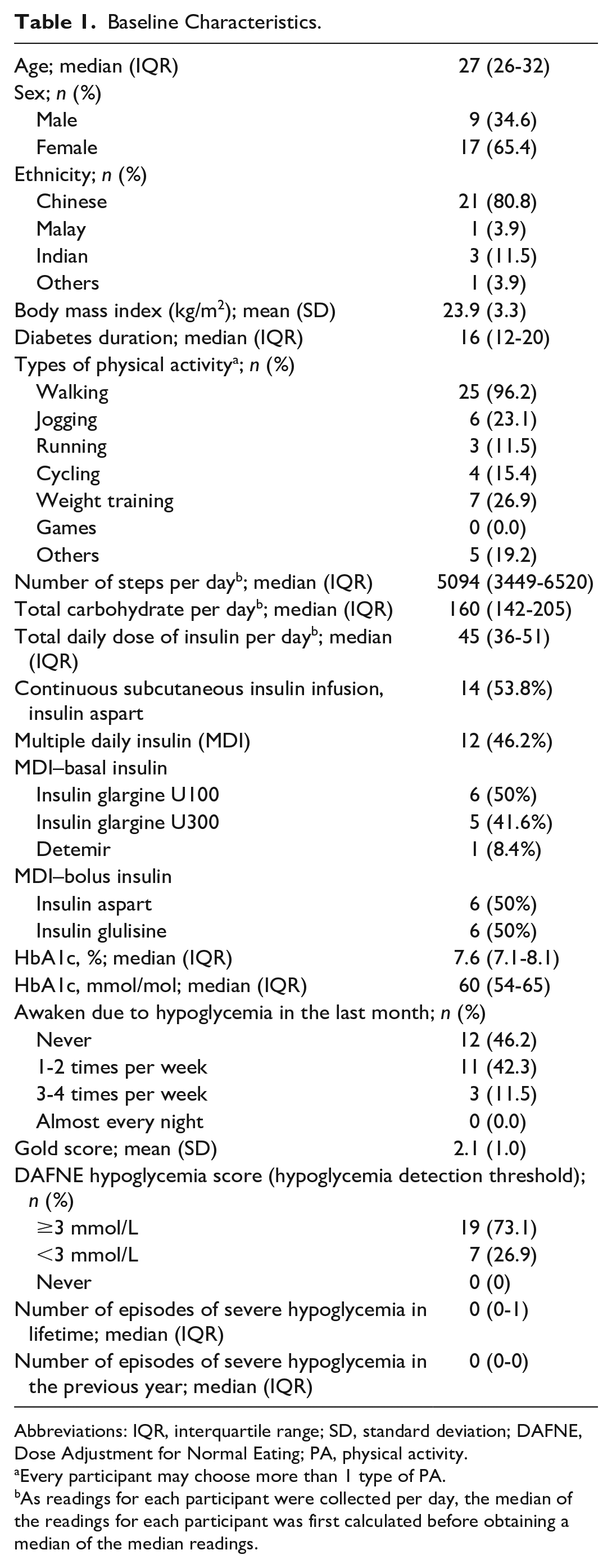
Abbreviations: IQR, interquartile range; SD, standard deviation; DAFNE, Dose Adjustment for Normal Eating; PA, physical activity.
Every participant may choose more than 1 type of PA.
As readings for each participant were collected per day, the median of the readings for each participant was first calculated before obtaining a median of the median readings.
The median (IQR) of individual median DSCs over the study period was 2867 (1820-4807), while the DSCs of the cohort ranged from 1026 to 23792. Similarly, the median of individual median TDC was 161 (130-254) grams and TDD of insulin was 45.7 (33.9-54) units. Of the 355 nights analyzed, there were 72 nights with at least one NH <70 mg/dl and 25 nights with at least one NH <54 mg/dl, translating to 0.05 episodes/person-week and 0.02 episodes/person-week, respectively. The median HbA1c was 7.6% (7.1-8.1).
A comparison of the PA parameters showed that nights with at least one NH <70 mg/dl were preceded by a higher median DSC as well as a longer duration of PA in the fairly active or very active intensity. There was no significant difference between PA parameters across nights with and without NH <54 mg/dl. TDC and TDD of insulin were not different between nights with and without NH (Table 2).
Summary of Physical Activity, Daily Carbohydrate Intake, and Insulin Use Stratified by the Presence of Nocturnal Hypoglycemia Episodes During the Study Period.
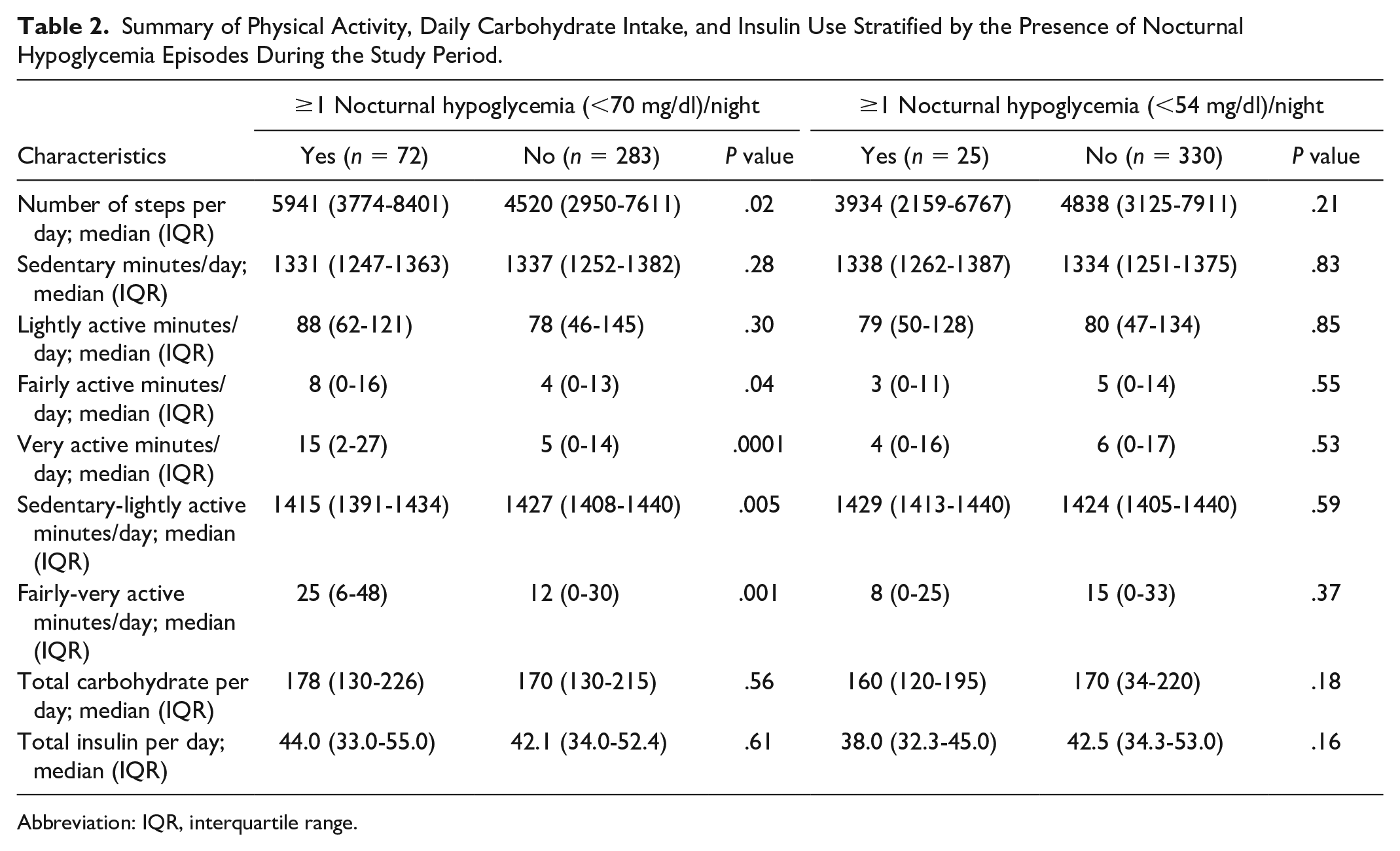
Abbreviation: IQR, interquartile range.
A mixed effects logistic regression model showed a significant effect of DSC on the risk of NH episodes (<70 mg/dl) (For every increment of 1000 steps per day; odds ratio [OR] [95% CI]: 1.11 [1.01-1.23], P = .04, Table 3). When the step counts were expressed as a percentage of individual median step count, the OR for risk of NH <70 mg/dl on days with “much higher than usual” step count, defined as >150% was 1.93 (95% CI: 0.81-4.57, P = .14). On the other hand, on days with “lower than usual” DSC, <80%, the OR for NH was 0.55 (95% CI: 0.25-1.20, P = .11). The relationship between DSC and NH episodes when defined as <54 mg/dl was insignificant after adjusting for TDC and total insulin. TDC intake and TDD of insulin had no significant impact on either NH < 70 mg/dl or NH < 54 mg/dl (data not shown) (Table 4).
Effect of PA on NH (Defined as <70 mg/dl) Adjusted for Total Daily Carbohydrate Intake and Insulin Use Through Mixed Effects Logistic Regression.
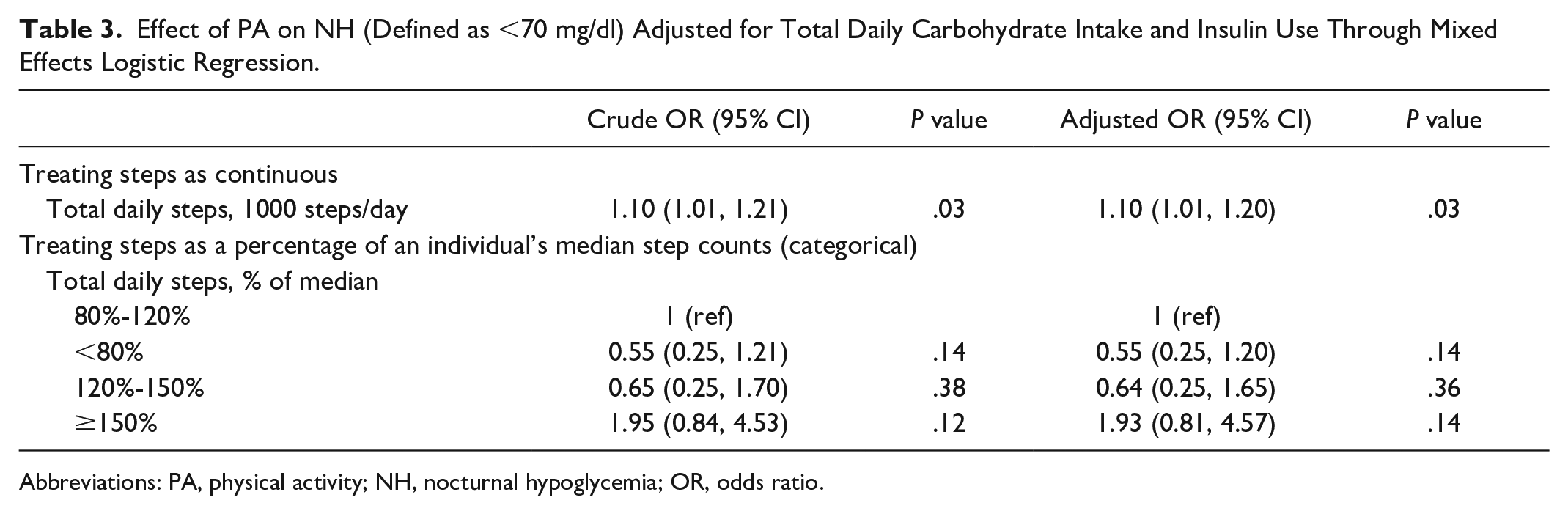
Abbreviations: PA, physical activity; NH, nocturnal hypoglycemia; OR, odds ratio.
Effect of PA on NH (Defined as <54 mg/dl) Adjusted by Total Daily Carbohydrate Intake and Insulin Use Through Mixed Effects Logistic Regression.
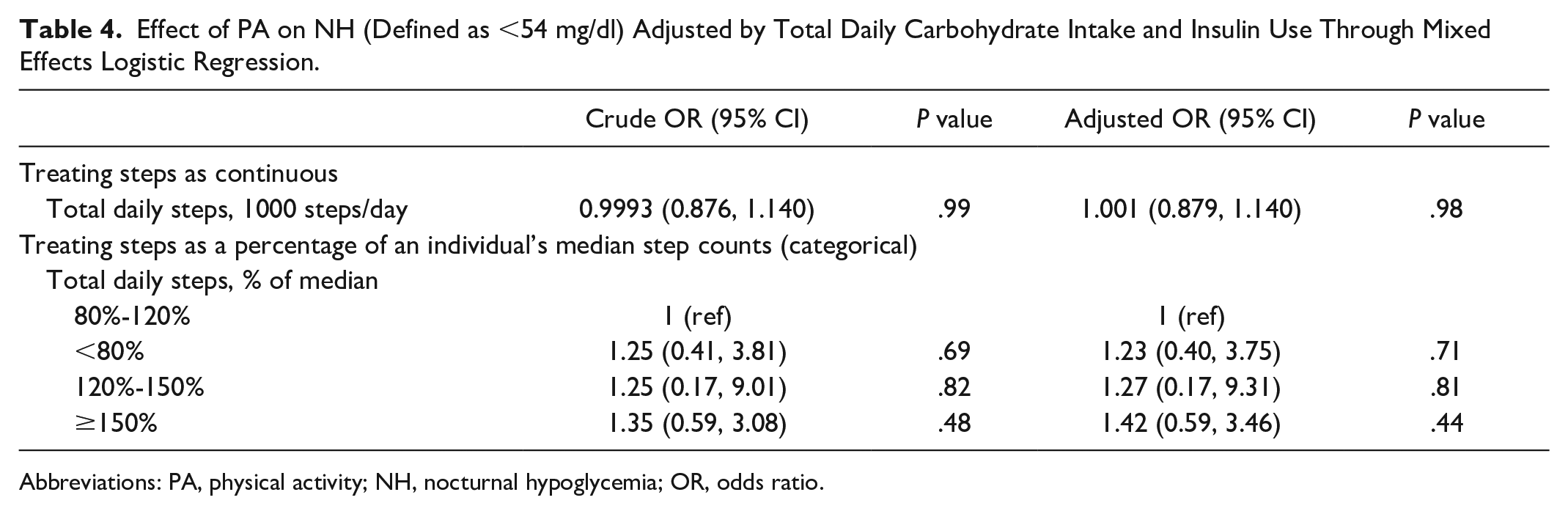
Abbreviations: PA, physical activity; NH, nocturnal hypoglycemia; OR, odds ratio.
We found that increasing duration of intense PA, expressed as fairly active minutes, adjusted OR 1.24 (1.02, 1.50), P = .03, as well as combined fairly and very active minutes, adjusted OR 1.18 (1.06, 1.31), P = .002, increased the risk of NH <70 mg/dl. Fairly and very active minutes correspond to moderate-vigorous PA (MVPA). Duration of varying levels of intense PA did not have any impact on NH <54 mg/dl (Table 5).
Effect of an Increment of Every 10 Minutes of Activity Categorized Based on Intensity, on Nocturnal Hypoglycemia, Adjusted for Total Daily Carbohydrate Intake and Insulin Use.
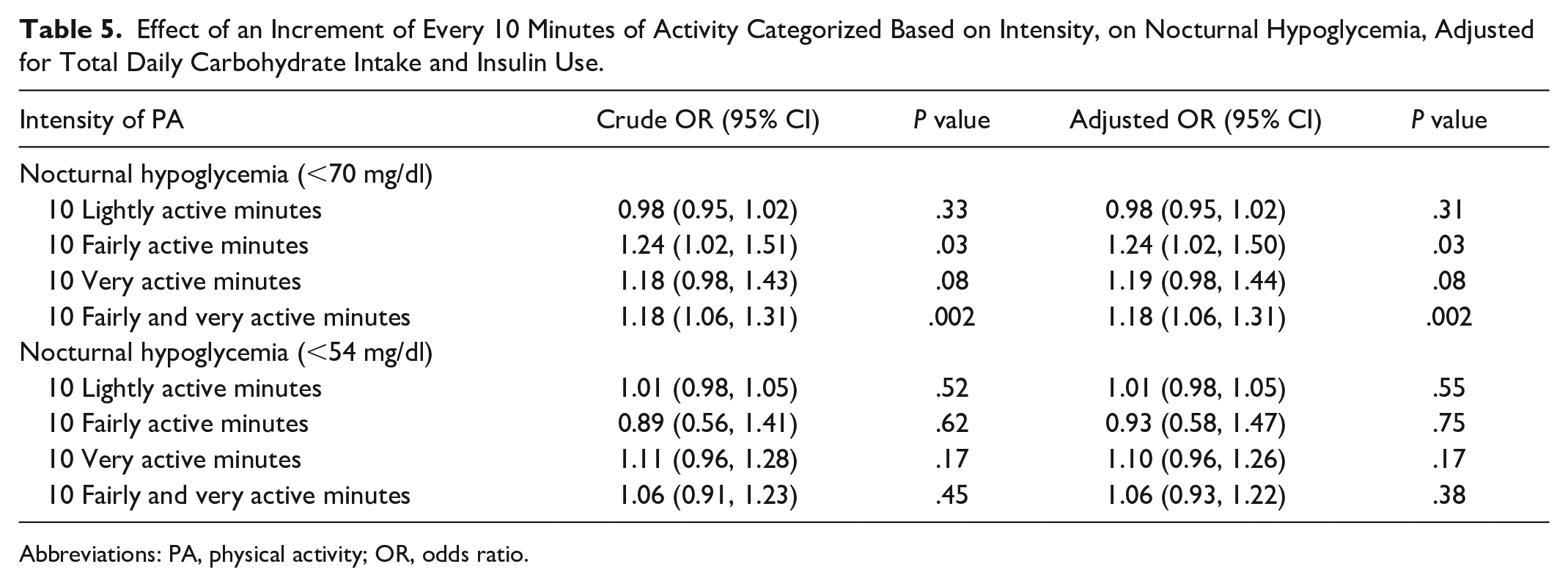
Abbreviations: PA, physical activity; OR, odds ratio.
Discussion
We found that PA measured using a smartphone was associated with NH <70 mg/dl in people with T1D. In particular, a higher DSC and a greater intensity of activity, both measured using a smartphone, were associated with NH<70 mg/dl.
We found a significant association despite lower PA and NH event rates compared to other T1D cohorts. A study using Actigraph, in 44 adults with T1D with a mean (SD) age of 49(9) found an average daily duration of 30 minutes in MVPA. 9 In our study the corresponding median (IQR) for fairly active and very active minutes was only 10 (0, 28) minutes. Our study was carried out during the COVID-19 pandemic with restrictions on the free movement of people. A decrease in total weekly step counts in the general population during the COVID-19 pandemic has been reported 33 ; hence, it may be assumed that our cohort’s activity was lower than usual. The event rates for NH <70 mg/dl and <54 mg/dl in our study (0.05 and 0.02 NH/person-week, respectively) were much lower compared to event rates in the “INRANGE” study (0.22 and 0.08 NH/person-week, respectively), that recruited 343 people with T1D on MDI. 34 Notwithstanding the lower median step count and the NH rates, we found that increasing step count and duration of MVPA measured using a smartphone application was associated with NH. This is similar to other studies using wearable activity trackers showing that MVPA was associated with NH. 5
Next, we explored the relationship between step counts and NH by expressing activity measured as step counts and percentage change from an individual’s median step count over the study period. The latter was chosen as we anticipated that a change from the usual activity is likely more important than the actual step count. We found that the risk of NH doubled when the DSC was >150% of the individual’s median step count, although not statistically significant. This suggests that a change from the usual activity is likely important. This concept is also supported by the fact that in athletes, who generally have a higher baseline PA, only vigorous PA increase their risk of NH while in non-athletes both moderate and severe PA increase risk of NH.5,21 It is plausible that people with a high baseline DSC or PA might have already made the necessary adjustments to their insulin regimen or carbohydrate intake to minimize the risk of NH from their usual level of PA. Thus, a relative change from an individual’s usual PA rather than the absolute quantity of PA seems to be important to predict a person’s risk of NH.
The discordance in the relationship between PA and NH defined at <70 mg/dl and <54 mg/dl was an interesting finding. There are a few possible explanations for this finding. Firstly, our cohort had a relatively good awareness of hypoglycemia. The average Gold score was 2.1, and 73.1% could detect hypoglycemia above 3 mmol/L. It is likely that people woke up from sleep and self-treated for hypoglycemia before the glucose threshold of <54 mg/dl occurred. We did not collect data on nocturnal awakenings and treatment of hypoglycemia to test if this was true. Secondly, self-restriction of PA by those with a higher risk of NH could explain the lack of an association between step count and NH less than 3 mmol/L. People with T1D are known to limit moderately vigorous PA compared to those without diabetes. 9 Thirdly, since we recruited people who could do carbohydrate counting and given that >50% were on insulin pump therapy this group was likely more motivated and engaged in their T1D care. This could suggest that on days with significantly higher PA, they might have proactively made changes to minimize NH<54 mmol/L. Finally, since the level of PA and the NH event rates were much lower than those of other T1D cohorts, it is likely that our study was underpowered to detect the effect of PA on NH <54 mg/dl.
The findings from our study have many potential implications. Current guidelines on adjustments to insulin require people with T1D to assess both the duration and intensity of exercise. 14 Self-assessment of exercise is often inaccurate and subject to bias, 20 while activity trackers provide a more objective and quantitative measure of the accumulated daily activity and its intensity. These data could be incorporated into predictive algorithms to guide self-management adjustments to prevent NH. To date, complex algorithms incorporating PA data detection and prediction from activity trackers in closed-loop insulin delivery have been shown to reduce the risk of hypoglycemia. 35 Machine learning algorithms have shown that this is also feasible in people on MDI. 36 However, we believe this is the first time that activity measured through a smartphone-based activity tracker has been demonstrated to be associated with NH in free-living people with T1D. Harnessing this technology, paired with the ubiquitous use of smartphones globally, would mean greater access to self-management tools, particularly for those without access to advanced technologies like CGM or closed-loop insulin delivery.
The findings from our study need to be confirmed in larger-scale studies. On a practical level, there are multiple potential applications for T1D care. Firstly, it suggests that activity tracking using a smartphone-based step tracker could inform a person about the risk of NH. Commercially available diabetes self-management smartphone applications that can access the step count data could display this information more meaningfully for the user to interpret and take action.37,38 DSC expressed as a percentage of a person’s median step count provides a more detailed quantitative estimation of the increase in activity from baseline to inform about NH risk. This information could trigger actions like taking a bedtime carbohydrate snack, reducing bedtime basal insulin, or setting a temporary basal reduction at bedtime for insulin pump users to reduce the risk of NH. Determining the thresholds above which such recommendations are made requires more data and remains to be discerned. Secondly, insulin pump algorithms could integrate accelerometer data to improve hypoglycemia prediction and insulin suspension. Accelerometer data could come from smartphone-based applications, wearable devices, or even an inbuilt accelerometer within the pump. Studies have already explored the possibility of accelerometer-augmented insulin pump suspension algorithms to prevent exercise-induced hypoglycemia with encouraging simulation results. 39
Our study had several limitations. The research study was carried out during the COVID-19 pandemic, which restricted the free movement of people; hence, the median step counts documented in our study were lower than usual. This might have led to the much lower NH event rates in our study. Due to the lower activity and lower event rates than reported from other T1D cohorts, it is possible that we have underestimated the effect size of activity measured using a smartphone on NH. We had missing data due to technical issues with the Fitbit MobileTrack App and Freestyle Libre Pro sensors. We did not perform any imputation for missing data. Instead, we eliminated day-night pairs without adequate data from the analysis as described in the methods. The sample size of this pilot study was small. The predominant form of PA in our study was walking. Hence, the findings may not be directly translatable to other forms of PA.
Conclusion
Smartphone-based measurement of step count and activity intensity is associated with NH in free-living adults with T1D.
Footnotes
Abbreviations
BMI, body mass index; CSII, continuous subcutaneous insulin infusion; DAFNE, dose adjustment for normal eating; IQR, interquartile range; MDI, multiple daily insulin; MET, metabolic equivalents of task; MVPA, moderate vigorous physical activity; NH, nocturnal hypoglycemia; OR, odds ratio; PA, physical activity; RT-CGM, real-time continuous glucose monitoring; SD, standard deviation; T1D, type 1 diabetes; TDC, total daily carbohydrate; TDD, total daily dose.
Author Contributions
Daphne Gardner: Methodology, Investigation, Resources, Writing—review and editing, Visualization, Supervision, Project administration.
Hong Chang Tan: Methodology, Formal analysis, Writing—review and editing, Supervision, Project administration, Funding acquisition.
Gek Hsiang Lim: Methodology, Formal analysis, Resources, Data curation, Writing—review and editing.
May Zin Oo: Investigation, Resources, Data curation, Writing—review and editing, Project administration.
Xiaohui Xin: Methodology, Formal analysis, Resources, Writing—review and editing, Funding acquisition.
Andrew Kingsworth: Methodology, Visualization, Writing—review and editing.
Pratik Choudhary: Conceptualization, Methodology, Data curation, Writing—review and editing, Visualization, Funding acquisition.
Suresh Rama Chandran: Conceptualization, Methodology, Software, Validation, Formal analysis, Investigation, Resources, Data curation, Writing—original draft, review, and editing, Visualization, Supervision, Project administration, Funding acquisition.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Singapore General Hospital Research Grant (SRG-OPN-03-2021).
