Abstract
Background:
The aim of this single-center observational study was to assess the real-world performance of first- and second-generation automated insulin delivery (AID) systems in a cohort of children and adolescents with type 1 diabetes over a one-year follow-up.
Methods:
Demographic, anamnestic, and clinical data of the study cohort were collected at the start of automatic mode. Data on continuous glucose monitoring metrics, system settings, insulin requirements, and anthropometric parameters at three different time points (start period, six months, 12 months) were retrospectively gathered and statistically analyzed.
Results:
Fifty-four individuals (55.6% of females) aged 7 to 18 years switching to AID therapy were included in the analysis. Two weeks after starting automatic mode, subjects using advanced hybrid closed-loop (AHCL) showed a better response than hybrid closed-loop (HCL) users in terms of time in range (P = .016), time above range 180 to 250 mg/dl (P = .022), sensor mean glucose (P = .047), and glycemia risk index (P = .012). After 12 months, AHCL group maintained better mean sensor glucose (P = .021) and glucose management indicator (P = .027). Noteworthy, both HCL and AHCL users achieved the recommended clinical targets over the entire study period. The second-generation AID system registered longer time spent with automatic mode activated and fewer shifts to manual mode at every time point (P < .001).
Conclusions:
Both systems showed sustained and successful glycemic outcomes in the first year of use. However, AHCL users achieved tighter glycemic targets, without an increase of hypoglycemia risk. Improved usability of the device may also have contributed to optimal glycemic outcomes by ensuring better continuity of the automatic mode activation.
Keywords
Introduction
In recent years, technology has acquired a paramount role in the management of type 1 diabetes (T1D). The relationship between technology and diabetes is characterized by continuous progress aimed to set up advanced devices that allow the achievement of ambitious clinical targets as well as the improvement of quality of life of people with diabetes. 1 Currently, the efforts of researchers are addressed to the introduction of the artificial pancreas, which means a technological device able to fully simulate the physiological function of the endocrine pancreas. Automated insulin delivery (AID) systems are therapeutic tools that get closer to the idea of the artificial pancreas. 2 The first commercially available AID system was the MiniMed 670G (Medtronic, Northridge, CA), which granted regulatory approval in 2018 for people > seven years. 3 The MiniMed 670G uses an algorithm that is based on the proportional integral derivative (PID) approach, which automatically modifies the basal insulinization rate according to the expected interstitial glucose values measured by the integrated glucose sensor. This AID system, which is also known as hybrid closed-loop (HCL), targets a glucose value of 120 mg/dl, which can be increased to 150 mg/dl. 4 More recently, a second-generation AID system, namely, advanced hybrid closed-loop (AHCL), has been developed that includes several PID approach enhancements due to the combination with fuzzy logic algorithm.5,6 The MiniMed 780G (Medtronic, Northridge, CA), currently available in more than 40 countries worldwide, is characterized by the capability to integrate automatic correction boluses and individualize different target set points (100, 110, and 120 mg/dl).7,8 The MiniMed 780G can be prescribed to people seven years or older and allows the user to increase the target threshold to 150 mg/dl in some circumstances, especially in the case of physical activity.
An open-label multicenter, randomized crossover trial showed that AHCL (MiniMed 780G) use was associated with a reduction of time spent in hyperglycemia without increasing hypoglycemia in adolescents and young adults with T1D compared with HCL (MiniMed 670G). 9 However, since the time of approval of MiniMed 780G, very few data are available on the differences in the performance of these two devices in real-world settings. 10
The aim of the present study was to assess clinical outcomes in children and adolescents with T1D using HCL and AHCL over a one-year follow-up.
Methods
In this single-center, observational, retrospective study, we enrolled children and adolescents with T1D using MiniMed 670G or MiniMed 780G. The study was exempt from ethical committee approval since it used unidentifiable data that are routinely collected at our diabetes center. Study participants were recruited based on the following inclusion criteria: T1D diagnosis according to the latest International Society for Pediatric and Adolescent Diabetes (ISPAD) guidelines, 11 age 7 to 18 years, HCL or AHCL therapy for at least one year at the time of enrollment, and informed consent from children and their parents to access continuous glucose monitoring (CGM) data remotely. Exclusion criteria included the coexistence of skin disorders affecting glucose sensor wearing, concomitant medical treatments known to interfere with glycemic levels, and daily sensor use < 70%. The choice of the device (HCL or AHCL) used by each study participant was closely related to the commercial availability in our country at the time of AID therapy initiation. Before starting the automatic mode, regardless of the type of system, all children and their caregivers received extensive training, which consisted of two 90-minute visits with an involvement of all diabetes team figures (ie, physicians, nurse, dietician, and educator) and Medtronic® technical staff. Main topics of this training were carbohydrate (CHO) counting, bolus wizard function use, interpretation, and CGM data sharing. Demographical, anamnestic, and clinical data (ie, age, gender, diabetes duration at the start of HCL or AHCL therapy, treatment type previously practiced, mean value of glycated hemoglobin (HbA1c) in the year before beginning HCL or AHCL system, anthropometric parameters, pubertal stage) were collected. Continuous glucose monitoring metrics of the 15-day period prior to AutoMode activation were also gathered, if available. The observational period was divided into: start period (T0—first 15 days of automatic mode), time 1 (T1—six months after automatic mode activation), and time 2 (T2—12 months after automatic mode use). At each observation-time point, anthropometric parameters, CGM metrics, and system settings were gathered. The following glucose control evaluators were considered: %time below range (TBR) < 54 mg/dl, %TBR 54 to 70 mg/dl, %time in range (TIR), %time above range (TAR) 180 to 250 mg/dl, %TAR > 250 mg/dl, coefficient of variation (CV), glucose management indicator (GMI), mean and standard deviation (SD) glucose sensor value, and glycemia risk index (GRI). The mean value of HbA1c over the 12-month study period was also considered. Regarding system settings, automatic mode and sensor use expressed in percentages, total daily dose (TDD) of insulin and its distribution between basal and bolus amount, active insulin time, and total number of exits from the automatic mode were collected. Anthropometric parameters and pubertal stage were identified by reviewing electronic medical records, data on glycemic indicators and system settings were extracted from the specific web-cloud platform (https://carelink.minimed.eu/app/login).
Numerical data were expressed as mean and SD, and categorical variables as absolute frequencies and percentages. Post hoc comparisons of data collected at each observation-time point between HCL and AHCL groups were performed using the Mann-Whitney test. To evaluate significant differences in HbA1c mean value, body mass index (BMI) z-score, percentage of automatic mode use, and TDD between T0 and T2 within each group, the Wilcoxon signed-rank test was applied. The relative changes of TIR, TAR, and CV were also calculated to estimate the percentage of variation of these glucose control parameters from the initiation to the end of the study. The percentages of subjects achieving the main glucose targets (ie, TIR, TBR, and GMI) were evaluated for both groups at each time point and compared by chi-square test and Fisher’s exact test, when appropriate. The estimated sample size ensuring a statistical power > 0.80 was of 45 subjects according to a preliminary power analysis for the most relevant CGM metrics. A P-value smaller than .05 was considered as threshold for significant values.
Results
Our retrospective analysis included 54 children and adolescents switching to AID therapy, with a mild prevalence of females (55.6%). Mean age was 12.1 ± 3.1 years, and duration of disease was 4.6 ± 2.6 years. Fifteen children (27.8%) were prepubertal. Twenty-four (44.4%) individuals were previously on multiple daily injections therapy, while 30 (55.6%) were already using an insulin pump. The study cohort was divided as follows: 24 subjects (44.4%) were on HCL therapy and 30 (55.6%) used an AHCL system. Age, gender, duration of disease, BMI, pubertal stage, previous therapy, and mean HbA1c of the previous 12 months were similar among the HCL and the AHCL groups. No significant variations of BMI were observed within both the groups throughout the study period. Baseline characteristics of the study cohort are summarized in Table 1.
Summary of Demographic and Clinical Characteristics of the Study Cohort.
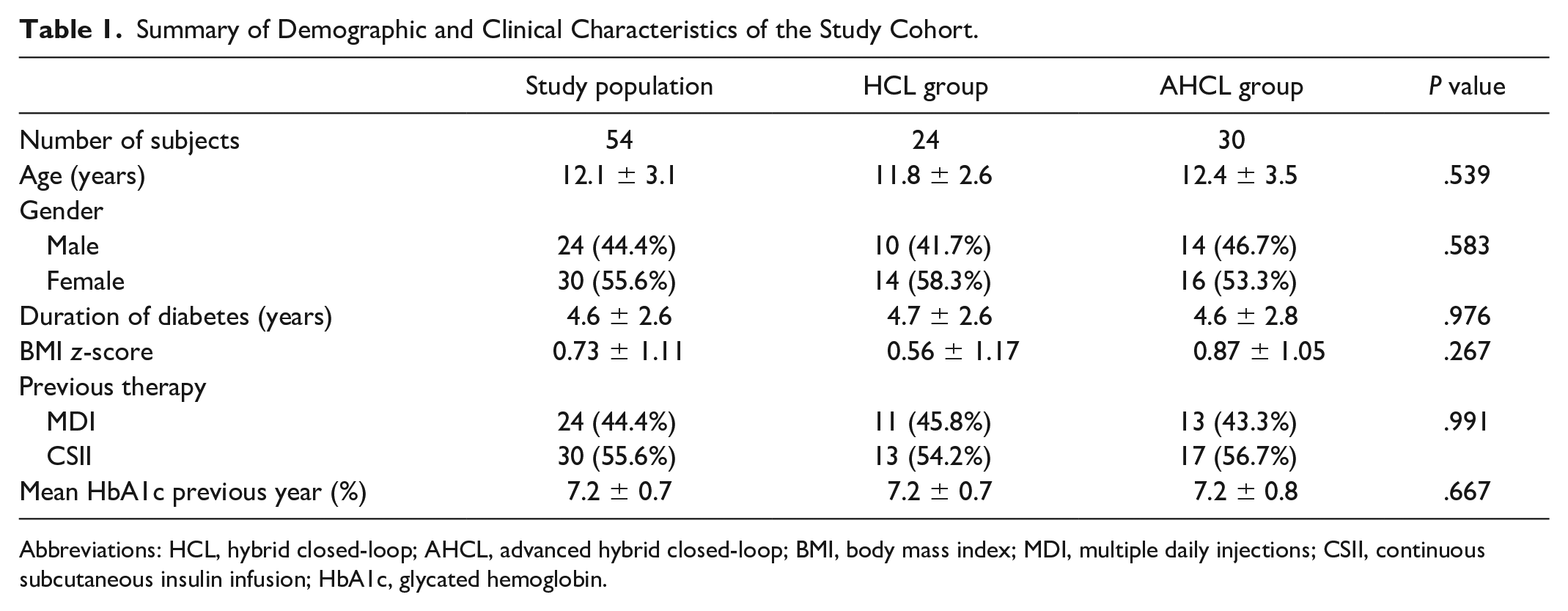
Abbreviations: HCL, hybrid closed-loop; AHCL, advanced hybrid closed-loop; BMI, body mass index; MDI, multiple daily injections; CSII, continuous subcutaneous insulin infusion; HbA1c, glycated hemoglobin.
Glucose Control Indicators
All glucose control indicators, including TIR (62.1% vs 67.8%, P = .190), %TAR 180 to 250 (25.9% vs 23.5%, P = .412), %TAR > 250 mg/dl (9.1% vs 5.9%, P = .061), and sensor mean glucose (162.4 vs 154.6 mg/dl, P = .312) were not different between groups during the 15-day period of manual use before switching to automatic mode.
As shown in Table 2, the recommended clinical targets were achieved at every time point, regardless of the type of device. However, after two weeks of AutoMode, use a significant difference of TIR (70.3% vs 75.2%; P = .016), %TAR 180 to 250 (22.8% vs 19.1%; P = .022), sensor mean glucose (152.2 vs 145.6 mg/dl; P = .047), and GMI (7.0% vs 6.8%; P = .036) was observed between the HCL and the AHCL subpopulations. After 6 months on AutoMode, sensor mean glucose (151.5 vs 143.1 mg/dl; P = .017), GMI (6.9% vs 6.7%; P = .012), and %TAR 180 to 250 (22.1% vs 17.7%; P = .048) were significantly better in AHCL group. After 12-month follow-up, differences of sensor mean glucose (151.3 vs 143.7 mg/dl; P = .021) and GMI (6.9% vs 6.7%; P = .027) were still present (Figure 1). Glycemia risk index was comparable between the two devices, except for the first two weeks of AutoMode, when the AHCL system showed a significantly lower index (P = .012). No differences in HbA1c levels were detected at any time point between groups.
Comparison of Clinical Data, Glucose Metrics, Insulin Requirements, and AutoMode Use Between HCL and AHCL Users at the Start Period, 6 Months, and 12 Months.
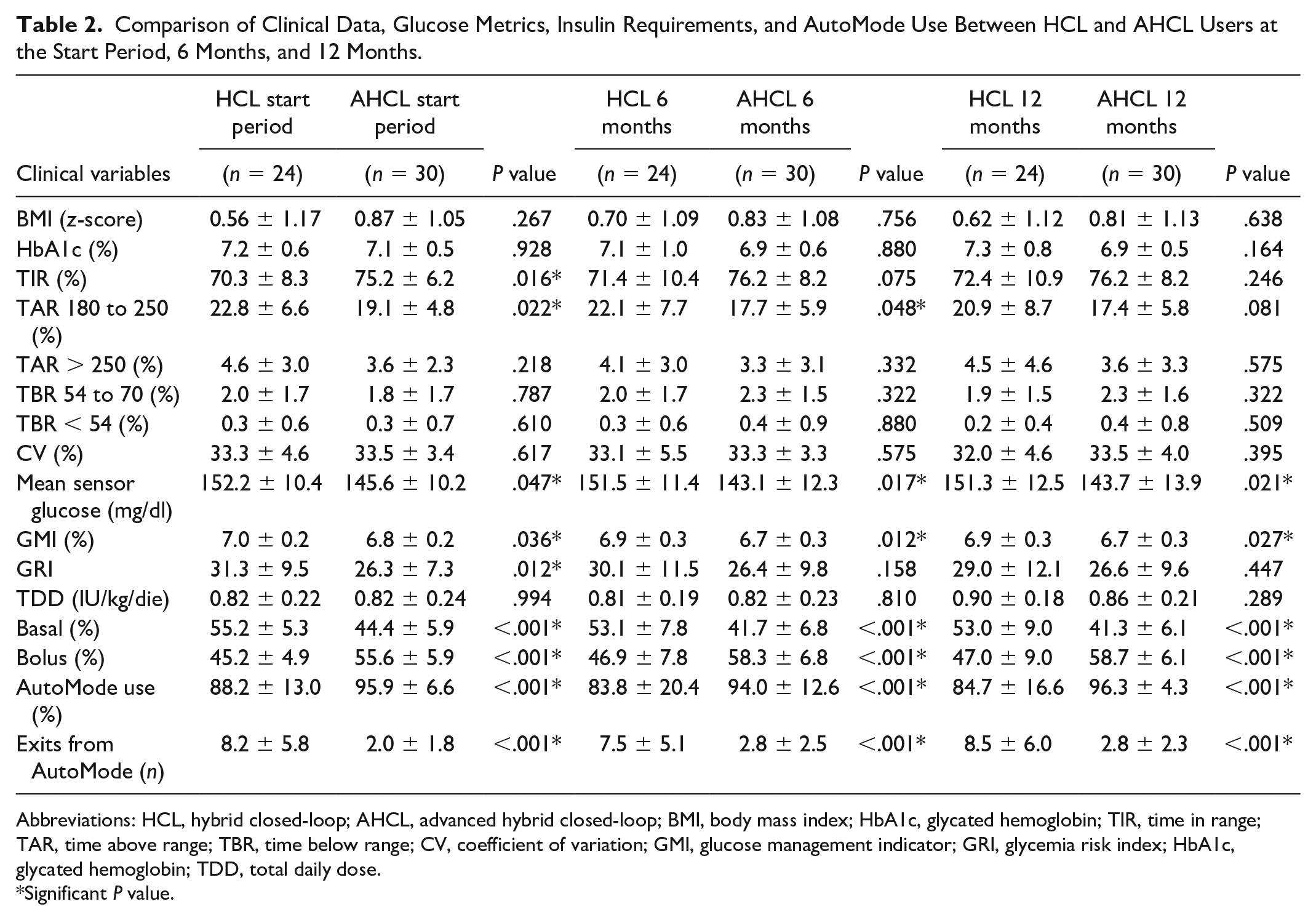
Abbreviations: HCL, hybrid closed-loop; AHCL, advanced hybrid closed-loop; BMI, body mass index; HbA1c, glycated hemoglobin; TIR, time in range; TAR, time above range; TBR, time below range; CV, coefficient of variation; GMI, glucose management indicator; GRI, glycemia risk index; HbA1c, glycated hemoglobin; TDD, total daily dose.
Significant P value.
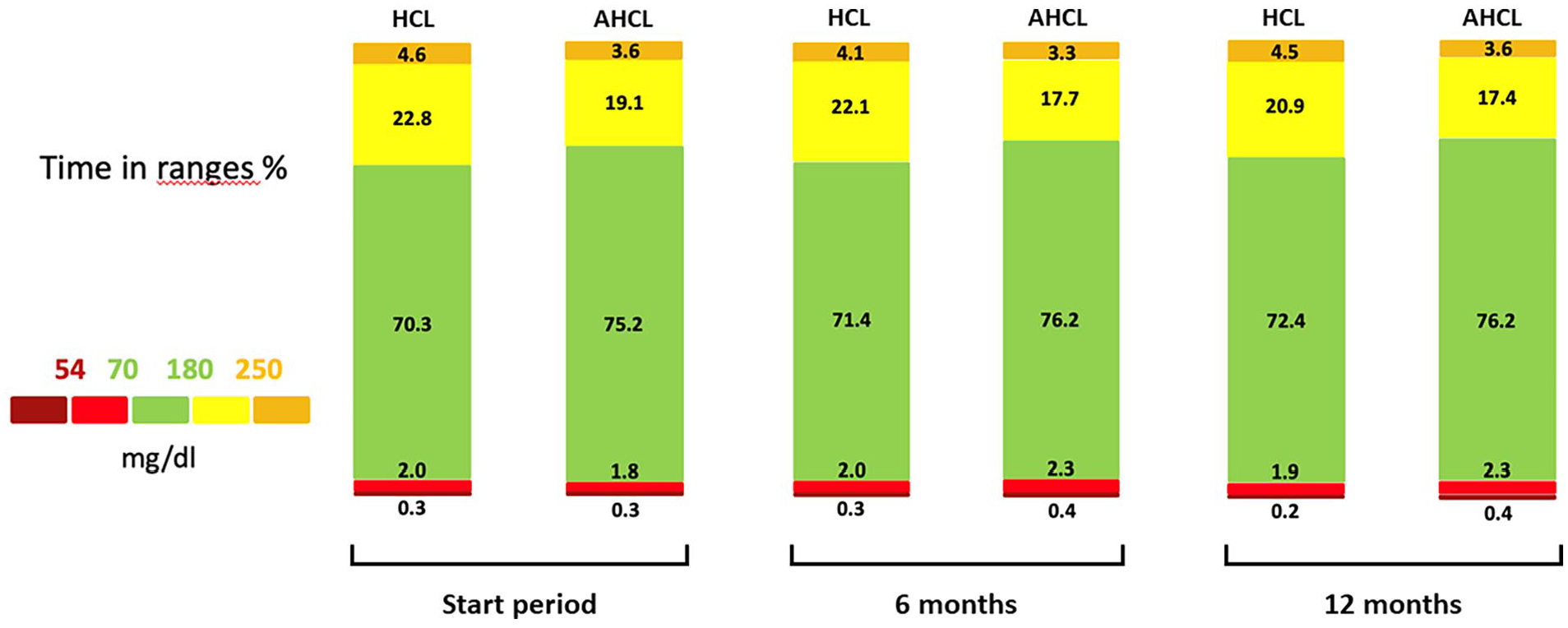
Data on the main glucose metrics at the start period, 6 months, and 12 months in the HCL and AHCL groups.
When assessing the variation of glucose metrics from the initiation to 12 months of AutoMode use, TIR showed a slight relative improvement (1.8%) within the AHCL group, whereas a relative deflection of 5.8% was observed in the HCL group. Time spent with sensor glucose > 180 mg/dl dropped in both the AHCL and HCL groups by 4.0% and 11.7%, respectively. Finally, CV relatively decreased by 10.6% among the HCL users, while subjects with AHCL maintained their CV after 12 months (+0.3%).
Percentages of individuals achieving recommended targets of TIR, TBR, and GMI over the study period are shown in Figure 2. No significant differences were found at each time point, except for concomitant achievement of TIR, TAR, and GMI at the start period that was obtained by a greater number of AHCL users (70% vs 41.7%; P = .036).
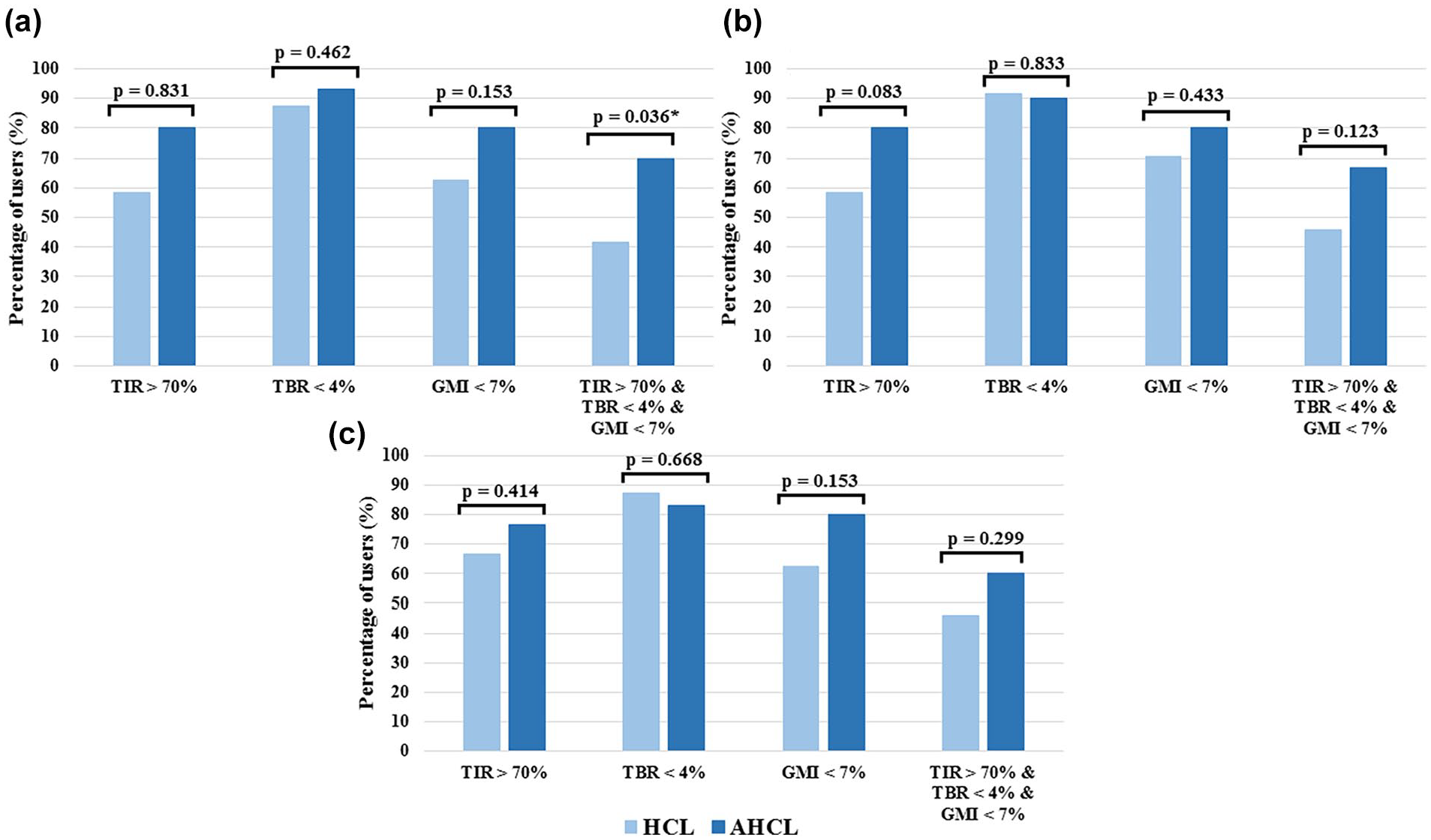
Percentage of HCL and AHCL users achieving the recommended target of TIR, TBR, and GMI at the start period (a), 6 months (b), and 12 months (c).
Glycated hemoglobin mean values showed no differences between the two subpopulations over the study period, and no significant within-group variations in HbA1c levels were detected compared with the 12-month period before AID system use.
Insulin Requirements and AutoMode Use
No differences of total daily insulin requirements were detected between HCL and AHCL users over the entire observation period. During the initiation period, total daily insulin dose was slightly higher in HCL pubertal individuals than prepubertal children (0.70 vs 0.86 IU/kg/day, P = .057). Limitedly to the HCL group, an increase of daily insulin dose from the initiation to 12 months was detected (P = .030). When considering the distribution of TDD into basal delivery and boluses, a greater amount of boluses (P < .001) at the start period, 6 months, and 12 months was detected in AHCL group, whereas basal distribution was prevalent among HCL users (P < .001). The percentage of automatic correction boluses in the AHCL group was 29.2 ± 9.0, 26.6 ± 11.1, 34.6 ±13.2 at each time period, respectively.
Among HCL users, active insulin time was set at 3 hours in the majority of individuals at each time point (58.3% at the start period, 58.3% at 6 months, and 54.2% at 12 months), while the use of two hours increased from 8.3% at the initiation to 20.8% at 12 months. In the AHCL group, percentage of users adopting active insulin time of two hours consistently increased from 23.3% at the start period to 60.0% at 12 months.
The MiniMed 780G showed a greater percentage of time spent in AutoMode in comparison with its predecessor at every time point (P < .001), with an overall lower number of exits due to any reason (P < .001). No variations of time spent in AutoMode were revealed by within-group analyses across the study period.
Discussion
According to our data, first-generation HCL and AHCL showed both successful results in pediatric subjects with T1D, allowing the majority of them to achieve the recommended glycemic targets according to the latest International Consensus on Time in Range. 12
The benefits of AID systems on glycemic control have been widely demonstrated in both experimental8,9,13 and real-world settings.14,15 Better performances of these devices compared with traditional continuous subcutaneous insulin infusion therapy have been also observed under particular conditions, including the recent global health challenge represented by the COVID-19 pandemic.16-18
In our study, the effectiveness of the AHCL system seems to be slightly greater than the standard HCL therapy, as witnessed by better levels of TIR, %TAR 180 to 250 mg/dl, and mean sensor glucose reached by the MiniMed 780G users. The achievement of more ambitious CGM metrics has a remarkable clinical relevance. Currently, these glucose control parameters are universally recognized as predictors of development or progression of microvascular complications in people with diabetes. 19
The overall lower time spent with high glucose, specifically in the range between 180 and 250 mg/dl, might suggest a better performance of the AHCL for the management of mealtime. In support of this hypothesis, a post hoc analysis of a randomized crossover trial comparing the two systems showed a lower post-meal mean glucose in the AHCL arm, mainly attributable to a better glucose response in the late post-prandial phase. 20 Benefits of AHCL therapy on the management of mealtime has been also observed in subjects with impaired gastrointestinal function, such as those with diabetic gastroparesis. 21
In our study, mean sensor glucose showed the widest and most persistent gap between the two groups among all glucose metrics. Mean glucose is currently recognized as a glycemic indicator that holds a direct correlation with the risk of long-term microvascular complications. 22 In addition, an analysis of aggregated data from several clinical studies on subjects with T1D using CGM systems revealed a strong linear correlation with time spent in target glucose values and in hyperglycemia, making mean glucose a very sensitive indicator to assess the efficacy of therapeutic interventions on people with diabetes. 23
A strongly significant difference between the two systems was detected in the insulin delivery data, which showed a more aggressive trend of the AHCL through a higher amount of boluses despite an unchanged total daily insulin dose. The different distribution of basal delivery and boluses is mainly related to the different functioning of the two systems. The MiniMed 780G has been set up with some important changes compared with the first-generation device by implementing the former PID controller with key features of a fuzzy logic artificial pancreas algorithm, including automatic correction boluses delivered up to every five minutes, and the possibility to choose between three target set points (100, 110, and 120 mg/dl). 9 The availability of different glucose targets, ranging from a stricter threshold (ie, 100 mg/dl) to a temporary target (ie, 150 mg/dl) suitable for physical activity, allows to minimize glycemic excursions and obtain tighter metabolic outcomes. 24 Despite the increased aggressiveness of the new system, the risk of hypoglycemia is not affected according to our data. Consistently, a retrospective study by Arrieta and associates on a large cohort of subjects using the MiniMed 780G, demonstrated that the use of stricter device settings, such as a glucose target of 100 mg/dl and an active insulin time of two hours, does not increase the time spent with low glucose. 24
Another positive finding of our study is related to the observed trend of BMI, which did not seem to be affected by the use of any of the two devices as it remains stable over the entire study period. A similar analysis of anthropometric parameters among young individuals using AHCL systems over a one-year period was carried out by Seget and colleagues, with comparable results. 25
Finally, our AHCL group was found to being able to maintain the automatic mode more steadily, with a reduced number of overall switches to manual mode than HCL subjects. This finding is related to the innovations included in the new algorithm, whose conditions for AutoMode exits have been optimized to guarantee a better continuity of the system, while decreasing user burden. Indeed, one of the most relevant criticisms for the first-generation HCL lies in the need of a constant engagement from the user, who is often required to fulfill glucose calibrations and finger-sticks to keep the closed-loop system working properly. 26 Repeated exits from the automatic mode due to this issue represent a serious hindrance to the achievement of optimal device performance. As demonstrated by several real-world studies on AID systems effectiveness, which evaluated predictors of clinical targets, the percentage of sensor and AutoMode use strongly correlates with successful glycemic outcomes.15,27,28
Limitations of the study include the relatively small sample size and lack of previous glucose data except for HbA1c values, as not all subjects included in the study wore a glycemic sensor prior to the AID use. On the other hand, the study duration is the main strength. To our knowledge, this is the first comparison between standard HCL and AHCL over one-year period in real-life settings in a pediatric population. Furthermore, the uniformity of the education received by each individual prior before starting AID, allowed by the monocentric design, reduced the risk of bias.
Conclusions
In conclusion, our study revealed successful and long-lasting results of both standard HCL and AHCL systems on glycemic control in children and adolescents with T1D over the first year of use. However, the AHCL system enabled tighter glycemic targets to be achieved without any negative implications, especially in terms of risk of hypoglycemia. Improved usability of the device may also have contributed to optimal glycemic outcomes by ensuring better continuity of the automatic mode use.
Footnotes
Abbreviations
AHCL, advanced hybrid closed-loop; AID, automated insulin delivery; BMI, body mass index; CGM, continuous glucose monitoring; CHO, carbohydrate; CV, coefficient of variation; GMI, glucose management indicator; GRI, glycemia risk index; HbA1c, glycated hemoglobin; HCL, hybrid closed-loop; PID, proportional integral derivative; SD, standard deviation; T1D, type 1 diabetes; TAR, time above range; TBR, time below range; TDD, total daily dose; TIR, time in range.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
