Abstract
Background:
Although exhaled breath condensate (EBC) is a promising noninvasive sample for detecting respiratory analytes such as glucose, current EBC collection methods yield inconsistent results.
Methods:
We developed a custom EBC collection device with a temperature-based algorithm to selectively condense alveolar air for reproducible EBC glucose detection. We characterized the condensate volumes and the corresponding glucose concentrations. We performed a pilot study demonstrating its use during oral glucose tolerance tests.
Results:
The novel device selectively captured alveolar air resulting in slightly higher and less variable glucose concentrations than the overall EBC. Participants with type 2 diabetes demonstrated significantly higher blood plasma-EBC glucose ratios than normoglycemic participants.
Conclusions:
Temperature-based selective EBC collection allows EBC glucose measurement and is a promising sampling method to distinguish patients with and without diabetes.
Introduction
As many as 67% of people with diabetes fail to monitor their blood glucose levels at the frequency recommended by the American Diabetes Association due to pain and inconvenience.1,2 A painless, noninvasive method of monitoring glucose levels can benefit people with diabetes by increasing patient compliance for regular monitoring of glucose levels.
Noninvasive fluids, such as alveolar lung fluid (ALF), contain glucose diffused from the blood.3-7 Alveolar lung fluid can be noninvasively collected in the form of exhaled breath condensate (EBC), which has previously been used to evaluate biomarkers of lung disease.8-10 However, ALF glucose is heavily diluted by water vapor from the anatomical dead space of the upper respiratory tract.11,12
Inconsistencies in EBC collection methods have contributed to a wide range in reported blood-to-breath glucose ratios (from 1000:1 to 50 000:1). 13 A standardized method to separate alveolar air from dead space air may minimize the variability in measured blood-to-breath glucose ratios. Selective sampling of alveolar air can be performed by collecting EBC with high levels of CO2, detected via capnometry. 14 Similarly, we have designed a temperature-based collection system to selectively condense alveolar air. 15 Incorporating a temperature and air flow sensor into a continuously calibrated algorithm, we engineered a device that selectively collects alveolar EBC. Here, we use this novel system and quantify glucose in the condensate. We demonstrate that selectively collected alveolar air reduces variability in glucose concentrations in the EBC. We further demonstrate the functionality of an EBC collection device in a pilot study of oral glucose tolerance tests (OGTTs) on normoglycemic and people with type 2 diabetes.
Methods
Valve Actuation for Selective Collection
We modified our selective alveolar air valve to incorporate more accurate sensing of exhalation temperature and airflow rate. 15 The developed algorithm uses an experimentally derived regression to control the valve based on specific temperature thresholds. The valve is programmed with a sampling rate of 4.05 Hz and opens when the temperature, relative to the start of the exhalation, exceeds 70% of the difference between the beginning and the end of the previous breath. The valve closes after detecting two successive decreases in temperature. The dynamic temperature threshold is updated every 50 milliseconds and adapts to changes in the temperature profile of each exhalation.
Valve and Collection Body Design
The selective collection valve allows for normal inhalation of air from a one-way diaphragm and exhalation to atmosphere when air is not being directed to the cooling plate. When the temperature reaches the threshold, the flow is redirected to a square aluminum cooling plate held at −5°C to 10°C where the exhalate is condensed. The entire collection period is three to four minutes (Figure 1). The OGTT pilot studies at the Indiana University (IU) Clinical and Translational Sciences Institute Clinical Research Center (CRC) (Indianapolis, IN) used dry ice to cool the plate to −20°C. A wiper collected the condensate to one corner of the plate and was pipetted into a microcentrifuge tube for later analyses.

A custom selective EBC collection device pictured with T-valve, embedded Rev. C Wind Sensor, a condensation plate with a collection frame, and a built-in wiper.
Device Feasibility Studies
A feasibility study was conducted with three healthy individuals aged 19 to 22 years (two females, one male) to determine the effects of selective valve actuation on glucose concentration in EBC at different expiratory volumes. Participants in the study did not have any history of diabetes or lung-related diseases. All participants gave written informed consent (approved by Purdue’s Institutional Review Board [IRB]). Participants fasted for two hours before the study.
Exhaled breath condensate samples were collected in triplicate at a 7.5-L expiratory volume with nonselective (0%) and temperature-selective threshold valve actuation (70%) for each subject. Samples were collected in a randomized order and stored in microcentrifuge tubes at −20°C until further use. A sterile, disposable mouthpiece was used for each participant, and they were instructed to breathe normally into the device through their mouth for both inhalation and exhalation. The mouthpiece, collection tray, and valve inlet port were cleaned with 70% ethanol between each sample to prevent contamination of residual glucose. The total volume of condensate, time for collection, and glucose concentration were recorded for each sample.
OGTT pilot study
Oral glucose tolerance test studies with two normoglycemic individuals (N01 and N02) and two individuals with obesity and type 2 diabetes (TD01 and TD02) were performed at the IU CRC. All participants gave written consent to the experimental procedures, which had been approved by IU’s IRB.
Intravenous blood samples were collected at baseline after participants had fasted for 10 hours, and a 75-g glucose drink was administered at T0. Intravenous blood and EBC samples using our in-house device were collected every five minutes for the first 60 minutes, then every 20 minutes for the subsequent 120 minutes. Plasma glucose was sampled using the YSI 2300 STAT-plus glucose analyzer (Yellow Springs Instruments, Yellow Springs OH, USA). For participant T2D01, blood and exhaled breath were sampled every 10 minutes during the remaining 120 minutes. For participant N01, the study was cut short by 80 minutes. The condensate samples were collected into tubes without additives or enzymes and were transported on dry ice and stored at −80°C until analysis within one day of the collection.
Glucose concentration analysis
A commercial fluorometric glucose oxidase assay (Abnova, Taipei, Taiwan) was used according to the manufacturer’s instructions to quantify glucose concentrations in EBC. Fluorescence was quantified on a SpectraMax M5 Microplate Reader (Molecular Devices LLC, San Jose, CA, USA). Before testing the EBC condensate, it was confirmed that residual 70% ethanol did not affect glucose results in this assay.
Statistical Methods
All statistical analyses were performed using GraphPad Prism version 8.2.1 (GraphPad Software Inc, San Diego, CA, USA) and R Studio v1.1.463. A paired t test was performed to determine if the temperature-dependent threshold level resulted in significant effects on the EBC glucose concentration. Coefficients of variation were calculated to determine the extent of glucose variability with each threshold level.
Blood-to-EBC glucose ratios were calculated by dividing the blood glucose concentrations by EBC glucose concentrations. These ratios were then separated into diabetes (n = 49) and normoglycemia (n = 38) sample groups. A two-tailed unpaired Students t test assuming unequal variances was performed with a 95% confidence interval.
Results
Effect of Selective Threshold
The glucose concentration from selectively collected EBC was higher, but not significantly different from those of a nonselective collection (P = .17) at this sample size (Table 1). The glucose concentration coefficient of variation was lower for samples collected with temperature selection versus those without (20% vs 36%). The time to collect EBC did not significantly change between threshold levels (Table 1). Selective collection using the temperature threshold did not have a significant impact on condensate volume (mean difference 16.25 ± 13.55 µL) (Table 1).
Effect of Threshold on Exhaled Breath Condensate Glucose Collection.

Values correspond to mean ± standard deviation from three participants.
OGTT Pilot Studies
Oral glucose tolerance test pilot studies demonstrated highly varied relationships between blood plasma and EBC glucose (Figure 2a and b). The higher early peaks in EBC glucose concentrations among participants with diabetes resulted in significantly higher (P = .01) blood plasma glucose-EBC glucose ratios (5640 ± 4750) averaged over the course of the study than those of normoglycemic participants (3621 ± 2549) (Figure 2b).
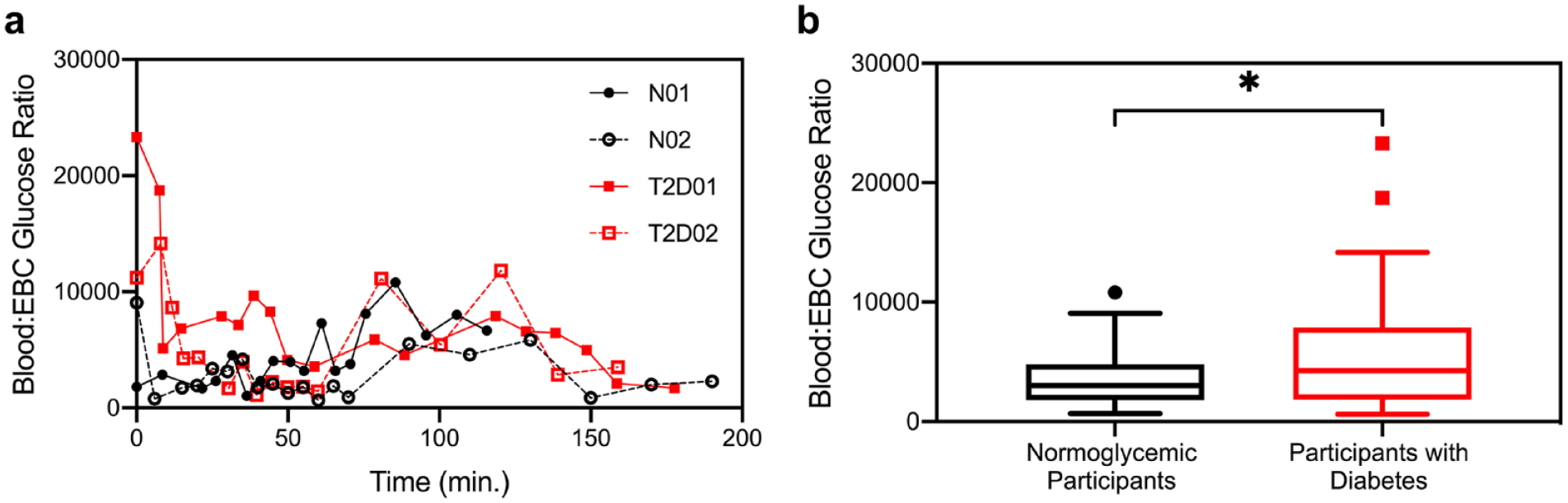
(a) Blood to EBC glucose ratios over time for all participants in an OGTT pilot study. (b) Distribution of blood to EBC glucose ratios for normoglycemic participants (N = 2, n = 19) versus participants with type 2 diabetes (N = 2, n = 24) shown with Tukey box whisker plots depicting lower quartile, mean, and upper quartile with whiskers indicating 95% SD. Abbreviations: EBC, exhaled breath condensate; OGTT, oral glucose tolerance tests.
Discussion and Conclusion
We demonstrated reproducible selective EBC collection from alveolar air. We observed that selective collection of alveolar air has the potential to increase concentration and reduce overall variability in EBC glucose measurements. However, due to the limited sample size, it was not possible to evaluate whether this reduction in variability was significant.
However, this study has limitations. A more stringent temperature selection threshold could be implemented using devices with greater collection efficiency, such as the commercially available R-Tube. Such an approach may enable robust noninvasive analyses of ALF and blood glucose in future studies.
Footnotes
Acknowledgements
We would like to thank the Purdue Stats Department, Tonya Hamilton, Nick Patel, the Bechtel Innovation Center, Purdue Research Manufacturing Services, Norvin Bruns, Dr Yunjie Tong, and Shubhankar Takle.
Abbreviations
ALF, alveolar lung fluid; CRC, Clinical Research Center; EBC, exhaled breath condensate; IU, Indiana University.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: J.C.L. is a co-founder of OmniVis, Inc, EverTrue LLC, and Rescue Biomedical LLC, which have licensed Purdue University technology to develop point-of-care diagnostics and wearable monitoring technologies. At the time of publication, D.T. was an employee of Bristol Myers Squibb, Summit, NJ. All aspects of the study preceded and were independent of this employment. At the time of publication, K.M. was an employee of Eli Lilly and Company, Indianapolis, IN. All aspects of the study preceded and were independent of this employment.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the National Institute of Health grant R21 EB023427-01 and a Pilot and Feasibility Award within the CDMD NIH/NIDDK Grant Number P30 DK097512.
