Abstract
Introduction:
Each year, people with diabetes and their insurers or governments spend billions of dollars on blood glucose monitors and their associated components. These monitors have evolved substantially since their introduction in the 1970s, and manufacturers frequently protect original medical devices and their modifications by applying for and obtaining patent protection.
Research Design and Methods:
We tracked the product iterations of five widely used blood glucose monitors—manufactured by LifeScan, Dexcom, Abbott, Roche, and Trividia—from information published by the U.S. Food and Drug Administration (FDA), and extracted relevant U.S. patents.
Results:
We found 384 products made by the five manufacturers of interest, including 130 devices cleared through the 510(k) pathway, 251 approved via the premarket approval (PMA) pathway or via PMA supplements, and three for which de novo requests were granted. We identified 8095 patents potentially relevant to these devices, 2469 (31%) of which were likely to have expired by July 2021.
Conclusions:
Manufacturers of blood glucose monitoring systems frequently modified their devices and obtained patent protection related to these device modifications. The therapeutic value of these new modifications should be critically evaluated and balanced against their additional cost. Older glucose monitoring devices that were marketed in decades past are now in the public domain and no longer protected by patents. Newer devices will join them as their patents expire. Increased demand from people with diabetes and the health care system for older, off-patent devices would provide an incentive for the medical device industry to make these devices more widely available, enabling good care at lower cost when such devices are substantially equivalent in effectiveness and safety. In turn, availability and awareness of older, off-patent devices could help stimulate such demand.
Introduction
Diabetes is a leading cause of morbidity and mortality, affecting 476 million people worldwide 1 including approximately nine million people with type 1 diabetes. 2 Central to the treatment of diabetes is insulin replacement,3,4 as well as various devices that have been developed to monitor blood glucose and determine the proper insulin dose. Continuous glucose monitors (CGMs) and blood glucose meters (BGMs) both use enzymes that react with blood glucose to produce a signal, but the latter uses a fingerstick to obtain an external blood sample while a CGM uses a small electrode implanted under the skin to take periodic measurements from interstitial fluid. By 2010, the market for blood glucose monitoring systems was valued at approximately $4 billion, and the market for the test strips—paper with chemicals designed to assay glucose levels in drops of blood—used in those systems at $3.5 billion. 5
Manufacturers of medical technologies like blood glucose monitoring systems often seek to protect their technology with patents. Patents last 20 years from the date of patent application, are granted by the U.S. Patent and Trademark Office and are required by law to reflect new and nonobvious changes from existing technologies. 6 Patents allow the inventor to bring suit against competitors who make the same device or device feature without authorization. While license agreements are possible, they can be dependent on the patentee’s willingness to license, generally require payment by the competitor and can be more challenging to negotiate when larger numbers of patents are at issue. Prices for newer medical devices can be high during the patent period.
Original devices and changes to existing devices come to the U.S. market through various pathways. Since 1976, Congress has required review of new devices by the U.S. Food and Drug Administration (FDA) before they can be sold. 7 The highest risk devices require clinical testing, and manufacturers must submit to the FDA a premarket approval (PMA) application, which for diagnostics may include sensitivity and specificity determinations. By contrast, moderate risk devices can be FDA-cleared with more limited testing either by being shown to be “substantially similar” to older devices via the 510(k) pathway or via the de novo pathway if no substantially similar products exist for comparison. 8 Subsequent iterations of cleared or approved devices generate additional 510(k) submissions or PMA supplements. 9
To understand how patents and the regulatory system interact in the context of blood glucose monitoring devices, we reviewed the number and types of patents, as well as the regulatory pathways, for five key products used for people with diabetes.
Methods
We focused on five leading devices and related products available in the United States: Freestyle Libre (Abbott); Accu-chek (Roche); LifeScan (LifeScan, a subsidiary of Johnson & Johnson); and Dexcom G6 (Dexcom). In addition, we considered the lower-cost product True Metrix (Trividia Health, formerly Nipro Diagnostics). First, we searched for these manufacturers or product names in the FDA’s publicly available online 510(k) database as of August 2021 and selected results that by their titles appeared to be BGMs, CGMs, or their components (e.g., “Dexcom G5 Mobile Continuous Glucose Monitoring System” or “FreeStyle InsuLinx Blood Glucose Monitoring System”), including test strips. We extracted 510(k) numbers, sponsoring companies, and regulatory clearance dates. The search was repeated in the FDA’s online PMA and de novo databases.
To identify relevant patents for these products, we used the Google Patents advanced search engine as of July 2021 to search for all issued U.S. patents for which the assignee at any point during the history of the patent was one of these manufacturers and that contained either the word “insulin” or “diabetes.” We used a search feature provided by Google Patents to “deduplicate” by publication. We extracted the filing date, the effective filing date (defined as the date of an earlier patent application, if the patent’s claims were asserted as being supported by that earlier-filed U.S. or foreign patent application), and the date the patent was granted. The effective filing date begins the running of the 20-year patent period.
Until 1995, patents lasted 17 years from the date of issuance, and in that year, the law was changed so that patents would expire 20 years from the date of patent application filing. 10 Patents with application filing dates prior to June 8, 1995 that had not yet expired by this date were legislatively set to expire the later of 17 years from the date of grant or 20 years from the effective filing date. 11 Using these rules and assuming no adjustments to the patent period, we identified all patents in force as of July 21, 2021.
Finally, we manually reviewed patent titles to classify each patent into major categories customized to each manufacturer’s portfolio of patents. Categories were designed to separate the major invention types associated with a given device. For example, LifeScan’s patents clustered around five invention types: test strips, sample volume/sensor, analyte concentration/measurement, interface/input/communication, and calculation of dose/schedule.
Results
We found 378 products made by the five manufacturers of interest, including 124 devices cleared through the 510(k) pathway, 251 approved via the PMA pathway or via PMA supplements, and three for which de novo requests were granted. We identified 8038 patents potentially relevant to these devices, 2462 (31%) of which likely expired by July 2021.
LifeScan
LifeScan was associated with 44 BGM or CGM devices and related products all cleared under the 510(k) pathway. The first device was the Glucochek Reflectance Meter (1981), which included a colorimeter, photometer, and spectrophotometer.12,13 The most recent FDA-cleared device in 2020 was the OneTouch Verio Reflect Blood Glucose Monitoring System, which consisted of the device, test strips, lancets, and solutions. According to the FDA, differences between the OneTouch Verio and its most immediate predicate device included its blood glucose algorithm (claimed to have reduced sensitivity to interferences), inclusion of motivational messaging, a more ergonomic design, and backlight on the meter display. 14
We found 377 potentially relevant patents assigned to LifeScan, with effective filing dates from August 13, 1986, to June 28, 2018. About one-half (212, 56%) were estimated to have not yet expired at the time of data extraction. Unexpired patents related to analyte concentration or measurement (45%), interface/input/communication (14%), test strips (11%), sample volume/sensor (6%), and calculation of dose/schedule (5%) (Figure 1).
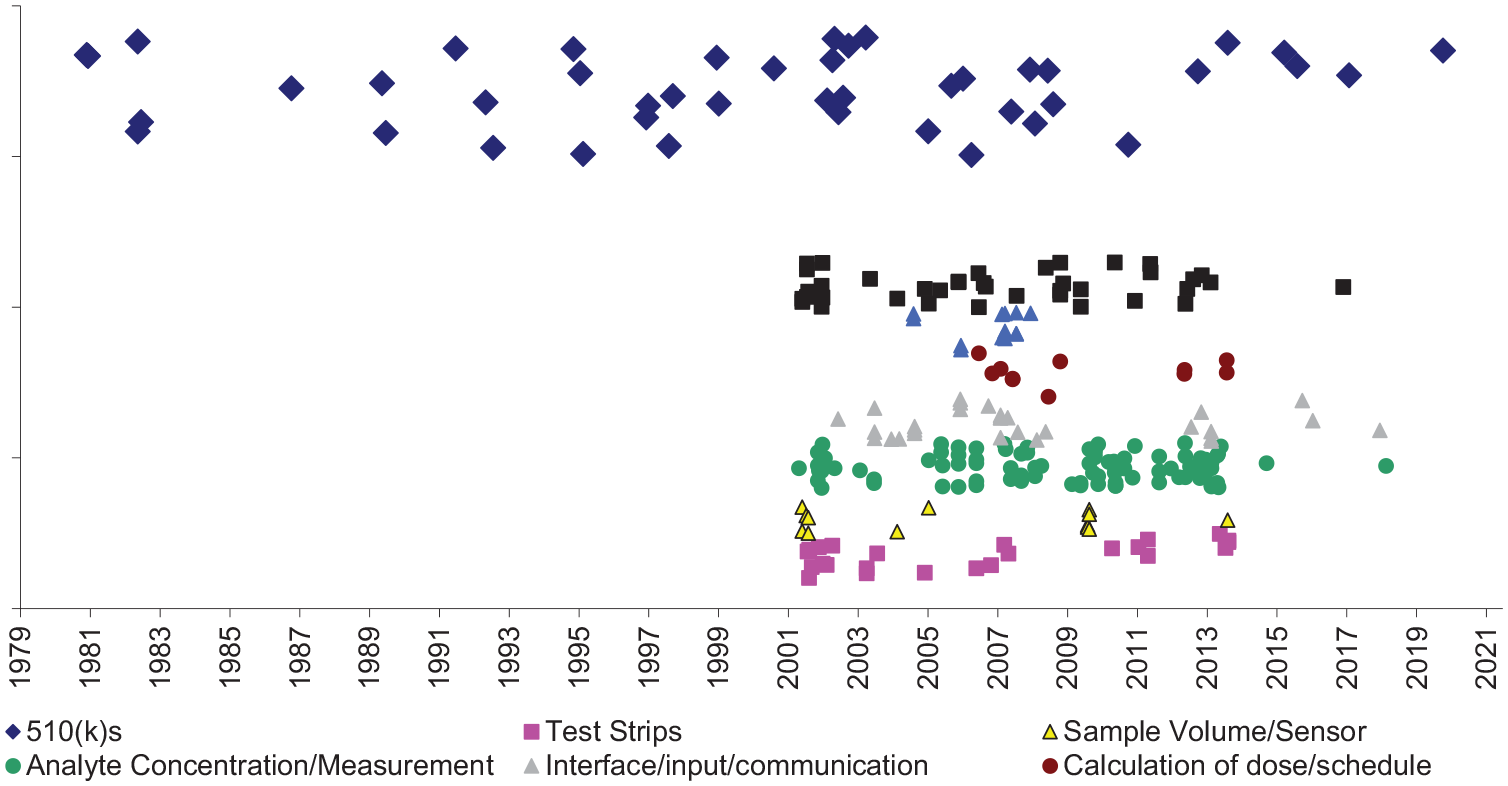
LifeScan insulin delivery system products and potentially related patents.
Dexcom
From 2006 to 2020, Dexcom obtained FDA authorization for 143 CGM devices and related products, including three de novo devices, 10 devices cleared through the 510(k) pathway, and 130 PMAs or PMA supplements. The first device, the STS Continuous Monitor (510(k), 2006) was an externally worn sensor that continuously tracks and reports glucose values and trending information in real-time for up to 72 hours. A recently FDA-authorized device was the August 2021 clearance of the Dexcom G6 Glucose Program Continuous Glucose Monitoring System, which provides information through the patient’s smart phone. In December 2022, FDA approved the Dexcom G7.
Among 633 potentially relevant patents assigned to Dexcom, effective filing dates spanned April 15, 1986, to October 30, 2017. Nearly all (607, 96%) had not yet expired. The most common categories were analyte sensors (68%), interface/input/communication (8%), membranes for implantable analyte sensors (8%), and insulin delivery systems (6%) (Figure 2).
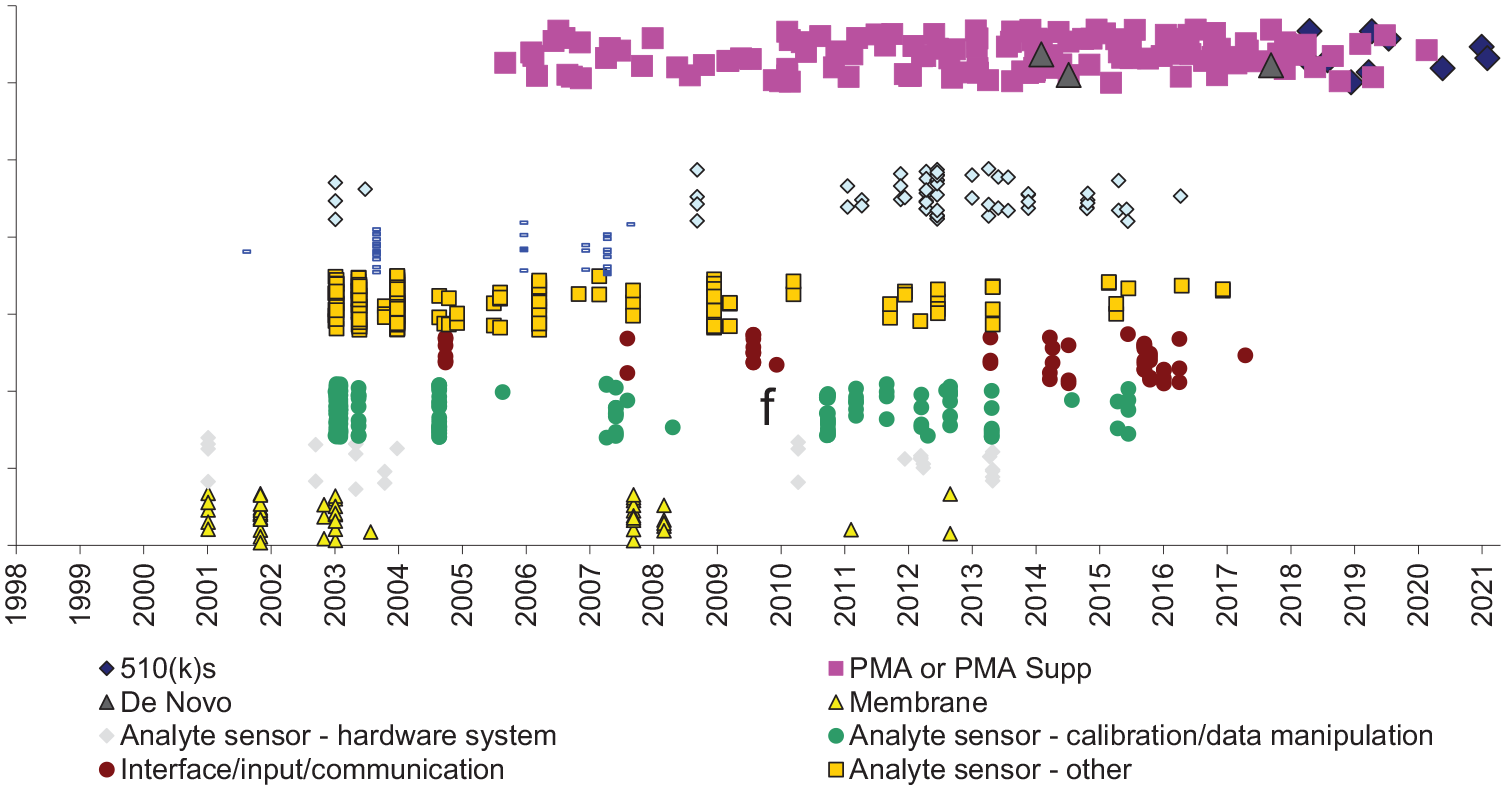
Dexcom insulin delivery system products and potentially related patents.
Abbott
From 2000 to 2021, Abbott obtained authorization for 147 BGM or CGM devices or related products, including 26 through the 510(k) pathway and 121 PMAs or PMA supplements. The most recent device estimated to no longer be encumbered by patents was the Freestyle Blood Glucose Monitoring System CAT No 11001 (510(k), 2000), a handheld glucometer. 15 The most recently FDA-cleared device was the FreeStyle Libre 2 Flash Glucose Monitoring System (510(k), 2021), 16 which differed from its predicate by its compatibility with a 14-day sensor only, rather than both a 10- and 14-day sensor.
We found 3023 potentially relevant patents assigned to Abbott with effective filing dates as early as 1955. Nearly two-thirds (1973, 65%) had likely not yet expired, including patents covering interface/input/communication (17%), aspects of analyte sensing (17%), processes of manufacturing (5%), medical devices (15%), and other (34%) (Figure 3).
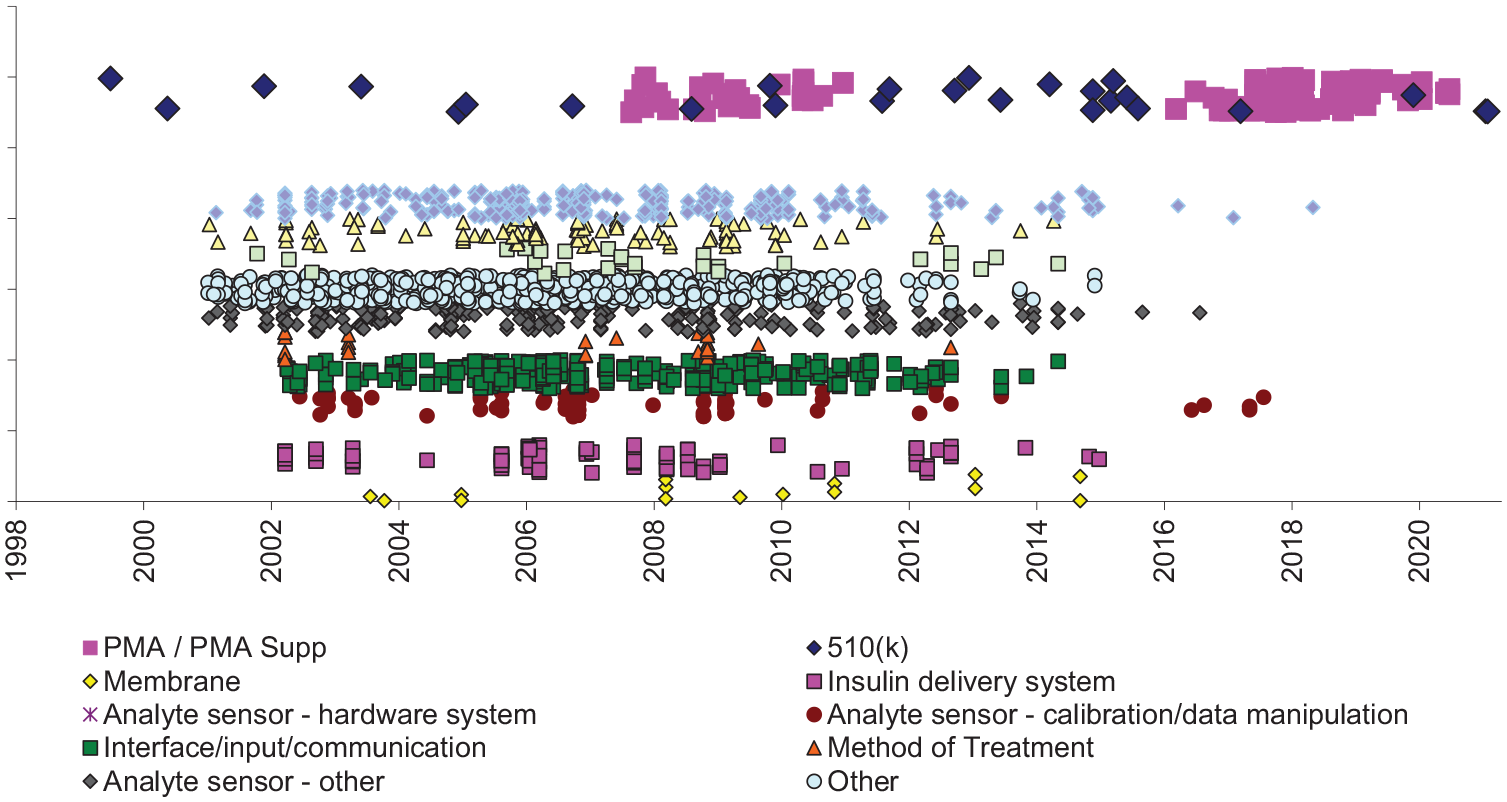
Abbott insulin delivery system products and potentially-related patents.
Roche
From 1999 to 2018, Roche obtained authorization to market 44 BGM or CGM or related devices, all cleared under the 510(k) pathway. The most recent device for which patents were estimated to have expired at the time of our search was the Accu-chek Active System (510(k), 2001), a handheld glucometer. 17 The most recently FDA-cleared device was the Accu-Chek Guide Me Blood Glucose Monitoring System (510(k), 2018).
We found 3993 potentially relevant patents assigned to Roche, with effective filing dates as early as 1951. About two thirds (2778, 70%) of patents had likely not yet expired. The patents estimated to be unexpired related to formulation (45%), medical device–other (17%), process (16%), analyte sensor (12%), and other (14%) (Figure 4).
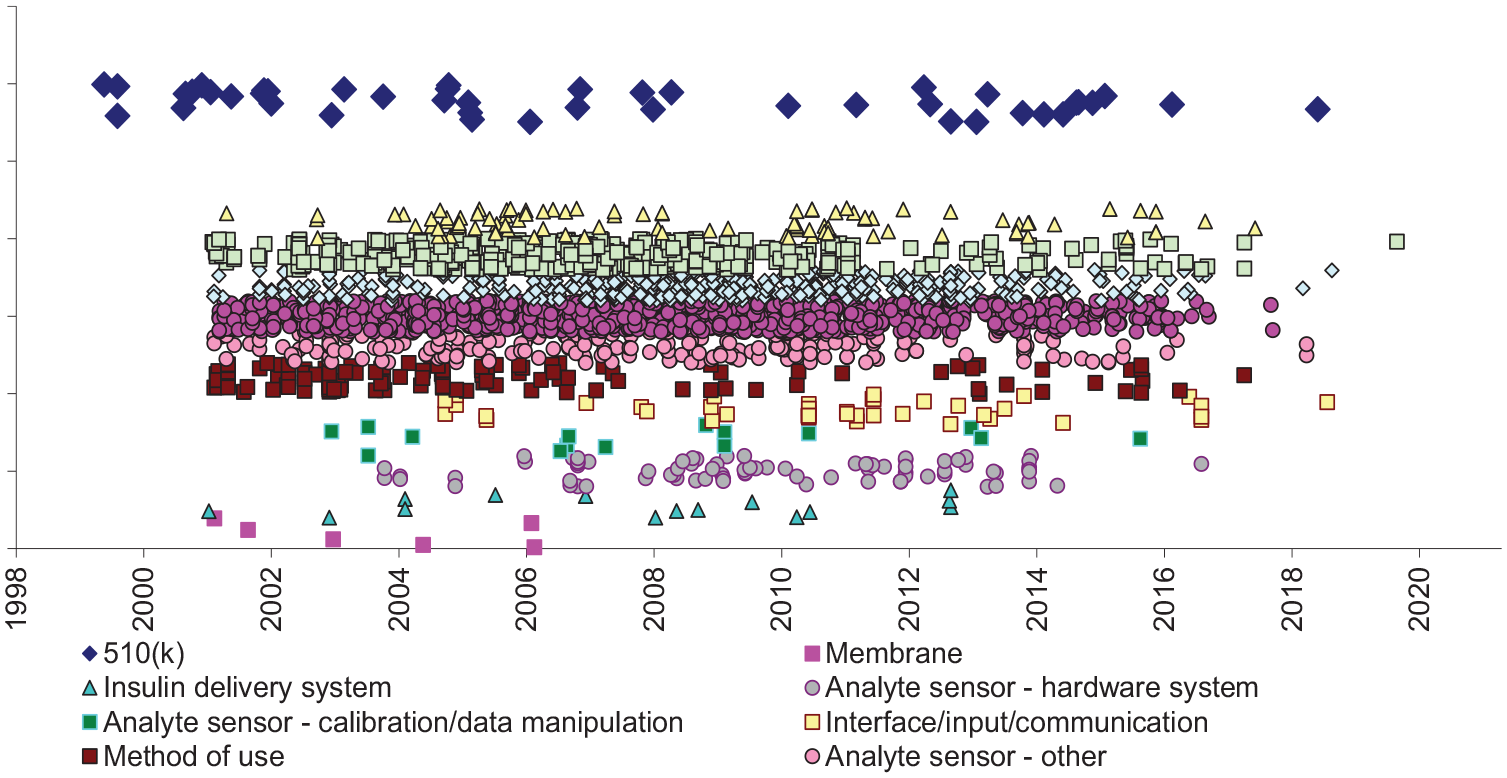
Roche insulin delivery system products and potentially related patents. The oldest Roche products (blue diamonds, left) are likely to unencumbered by patents. Numerous patents have not yet expired.
In the other category, the patents covered types of devices related to but not directly covering analyte monitoring devices. Examples include ampules, tapes, pumps, lancets, test elements, drug reservoirs, diagnostic devices, magazines, and needle tips. The process category included patents with claims in which the process of using an article or element of an article was claimed. 18 The analyte sensor category included patents covering biosensors, electrochemical sensors, test strips, analyte measurement or meters, fluid delivery systems for sensing analyte concentration levels, insertion systems, handheld diabetes management devices, glucose meters, and infusion site interfaces, or methods of using such devices.
Trividia
Trividia was associated with six BGM or CGM devices and related products all cleared under the 510(k) pathway (Figure 5). The first was the True Metrix (2013), a self-monitoring BGM device. The most recent FDA-cleared device was the True Metrix Go self-monitoring BGM device (2016). We found 57 potentially relevant patents assigned to Trividia, with effective filing dates from November 24, 1999, to May 5, 2017. Fifty (88%) were estimated to have not yet expired at the time of data extraction. Unexpired patents related to test strips (32%), system claims (14%), analyte measures (11%), data or communication (5%), and other (25%).
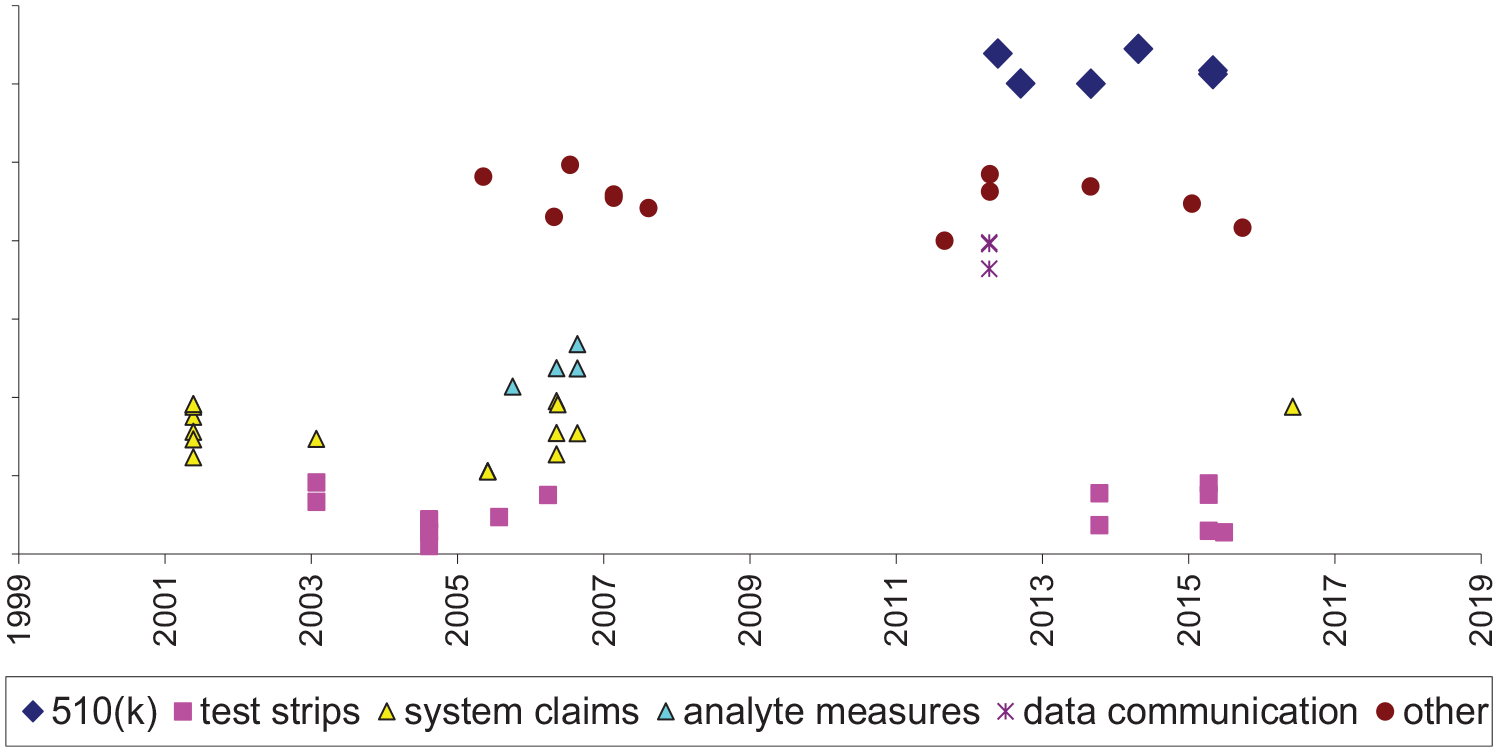
Trividia insulin delivery system products and potentially related patents.
Discussion
In this review of products and associated patents of five leading blood glucose monitoring systems and their associated devices, we identified 8038 patents potentially relevant to these devices, 2462 (31%) of which likely expired by July 2021. The large number of patents is suggestive of these companies’ strategies to patent numerous incremental changes to BGM and CGM devices and related products over time.
A number of versions of FDA-cleared or FDA-approved BGM and CGM devices made by LifeScan, Abbott, and Roche are likely no longer encumbered by patents and therefore available for copying by other manufacturers. Production of these patent-free products—because they are no longer burdened with the need to license or pay patent royalties—could help to increase access to safe and effective diabetes technologies within the United States and globally and could be accomplished by industry, government, or nonprofit manufacturers. There is likely to be even less patent protection of BGM and CGM devices in lower-income countries around the world, potentially expanding the range of unpatented product options available for royalty-free manufacture and use in those settings. 19 To the extent that patents substantially increase prices, their expiration can be expected to significantly decrease the total cost of bringing a product to market. Once patents expire on BGM and CGM devices, patents covering associated products originally used in those devices, such as test strips, will also likely expire because all patents are subject to a 20-year term. Such unpatented test strips may cost only about $0.15 per strip to be manufactured. 20 However, manufacturers are likely to be willing to manufacture off-patent devices and test strips only if health care practitioners and people with diabetes are willing to prescribe and use older technology in return for a lower price.
Newer devices often contain changes intended to improve some aspect of their function. For example, since the 1970s, BGM devices have become smaller, easier to use, and more accurate; offering larger display screens, auto-calibration, and connectivity to smart phones or other personal devices; and requiring less blood and fewer user inputs. 21 Such features may not be available with older, off-patent devices.
It may be possible to conduct relatively low-cost epidemiological research using large healthcare databases to understand whether similar clinical outcomes (including objective clinical outcomes such as amputations or blindness, and surrogate endpoints such as HbA1c or time-in-range) can be achieved with the older products. For less critical outcomes, such as convenience or reduced discomfort, it will be important for policymakers and patients to consider how much these outcomes are worth and to what extent people with diabetes should bear the higher cost of improved devices that yield clinical outcomes that are similar to properly used older devices. Comparative clinical trials measuring the extent to which newer devices are superior to older ones could be conducted, and the results clearly communicated to people with diabetes and caregivers, but doing so would be a substantial and costly undertaking, and for ethical reasons would have to be limited to products for which clinical outcomes are expected to be similar.
This study represents one of few studies of device patents, in contrast to a large literature on drug patents. One reason might be that key patents on traditional small molecule drugs are publicly listed by the FDA, while no similar publication is available to ascertain patents protecting medical devices. The lack of a device patent register can increase risk for competing businesses, which must avoid infringing the patents of other parties. Congress should consider enacting a patent listing requirement for medical devices analogous to that for drugs, allowing the FDA to include patent information in its existing Device Registration and Listing database. 22
Using such a resource, researchers with expertise in intellectual property could clarify which device technology is (or will soon be) freely available without the need for permission from the holder of the now-expired patents. Physician-scientists could evaluate which off-patent devices adequately satisfy patient needs, including consideration of when compatible off-patent peripheral products (such as test strips) are available. Social scientists could explore why physicians and people with diabetes tend to prescribe or use newer patented products rather than less-expensive off-patent products that perform similarly. Insurers and integrated health care systems could establish policies to promote the use of high-value older devices, being sure to pass along a substantial share of the savings to people with diabetes who choose to use them. For example, an insurer might contract with a low-cost manufacturer to guarantee availability of an off-patent device, then offer people with diabetes the option of using the lower-cost device, passing along much or all of the savings. In low- and middle-income countries, the public sector might invite competitive bidding to procure large volumes of off-patent products at low prices. If successful, these approaches could be expanded to other diabetes technologies, such as insulin pumps and hybrid closed loops.
One limitation of this study is that it did not consider variables that could potentially shorten patent terms such as terminal disclaimers, 23 the nonpayment of maintenance fees, and judicial or administrative invalidations, nor did we consider patent term extensions and adjustments. 24 We also excluded patent applications, even though some of these will likely mature into issued patents. Another limitation is that our search terms may have been both over- and under-inclusive. We included only patents the assignee of which included, at any time point, one of the five named companies; it is possible patents were assigned to a different entity and licensed to one of the manufacturers in our study or were held by subsidiaries or other affiliates or by suppliers of components used in the five devices. We did not systematically examine the predicate devices listed in 510(k) product documentation. Therefore, it is possible that additional similar products marketed by other manufacturers are off-patent and available for low-cost manufacture. Another limitation is that our method of identifying patents was intentionally broad to avoid missing relevant patents, but included a small number of patents that did not appear to be relevant to any of the five devices or their associated products. Finally, our paper does not provide an economic model for calculating expected savings from the use of off-patent devices.
Conclusion
Manufacturers of BGM and CGM devices frequently modified their products and obtained patent protection related to these device modifications. Physicians, patients, and policymakers should critically evaluate manufacturer claims that newer products outperform older ones and carefully weigh therapeutic value against additional cost. Some older glucose monitoring devices that were marketed in decades past are now likely in the public domain, and newer devices will join them as their patents expire. Increased demand from people with diabetes and the health care system would provide an incentive for the medical device industry to make older, off-patent devices more widely available.
Footnotes
Author Contributions
All authors approved the final manuscript. JJD conducted data analysis (patent, regulatory submission search), manuscript design/conception, and first-draft writing. VVdW contributed to patent categorization, and VVdW, DB, and ASK contributed to manuscript revisions.
Abbreviations
BGM, blood glucose monitor; CGM, continuous glucose monitor; FDA, Food & Drug Administration; PMA, premarket approval; USPTO, United States Patent and Trademark Office.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article, other than as disclosed in the Funding section, below.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by Health Action International’s Addressing the Challenge and Constraints of Insulin Sources and Supply (ACCISS) project, which in turn receives funding from the Helmsley Charitable Trust. DB is a co-leader of ACCISS. JJD and ASK also receive grant support from Arnold Ventures and the Commonwealth Foundation. The Helmsley Charitable Trust, Arnold Ventures, and the Commonwealth Foundation had no role in the conception, drafting, editing, or submission of this manuscript.
