Abstract

Previous studies showed that remote monitoring of continuous glucose monitoring (CGM) data mitigates hypoglycemia at diabetes camp. 1 However, some camps are still reluctant to permit CGM use, discounting the life-saving capacity of CGM and ignoring that an increasing majority of children are relying on it at home. 2 Camps that allow CGM use are not currently able to conveniently monitor CGM data from multiple campers simultaneously. This observational, non-randomized study evaluated whether multiplex centralized remote monitoring of CGM data efficiently reduces hypoglycemia at diabetes camp.
Insulin-treated campers ages 7 to 18 years were enrolled at a week-long American diabetes camp. All campers performed regular fingerstick testing at camp, which informed treatment decisions. Campers that monitored their glucose with self-monitoring of blood glucose (SMBG) at home were fitted with blinded Dexcom CGM at camp; those who used Dexcom CGM Systems with the capacity to share data at home continued to use their self-supplied systems at camp; shared data were displayed on the Follow app on counselor-carried smart devices and were ported to a centralized multiplex display in the camp medical cabin (Figure 1A); remote monitoring by family members was temporarily paused. Remote monitoring of CGM data informed staff of out-of-range glucose values (<80 mg/dL or >250 mg/dL [day, all campers], <80 mg/dL or >300 mg/dL [overnight, those on pumps], and <80 mg/dL or >350 mg/dL [overnight, those using injections]), prompting testing and interventions outside scheduled intervals. Data were compared between blinded and remotely monitored (unblinded) CGM systems. Campers with <50% CGM data coverage were excluded from analysis.
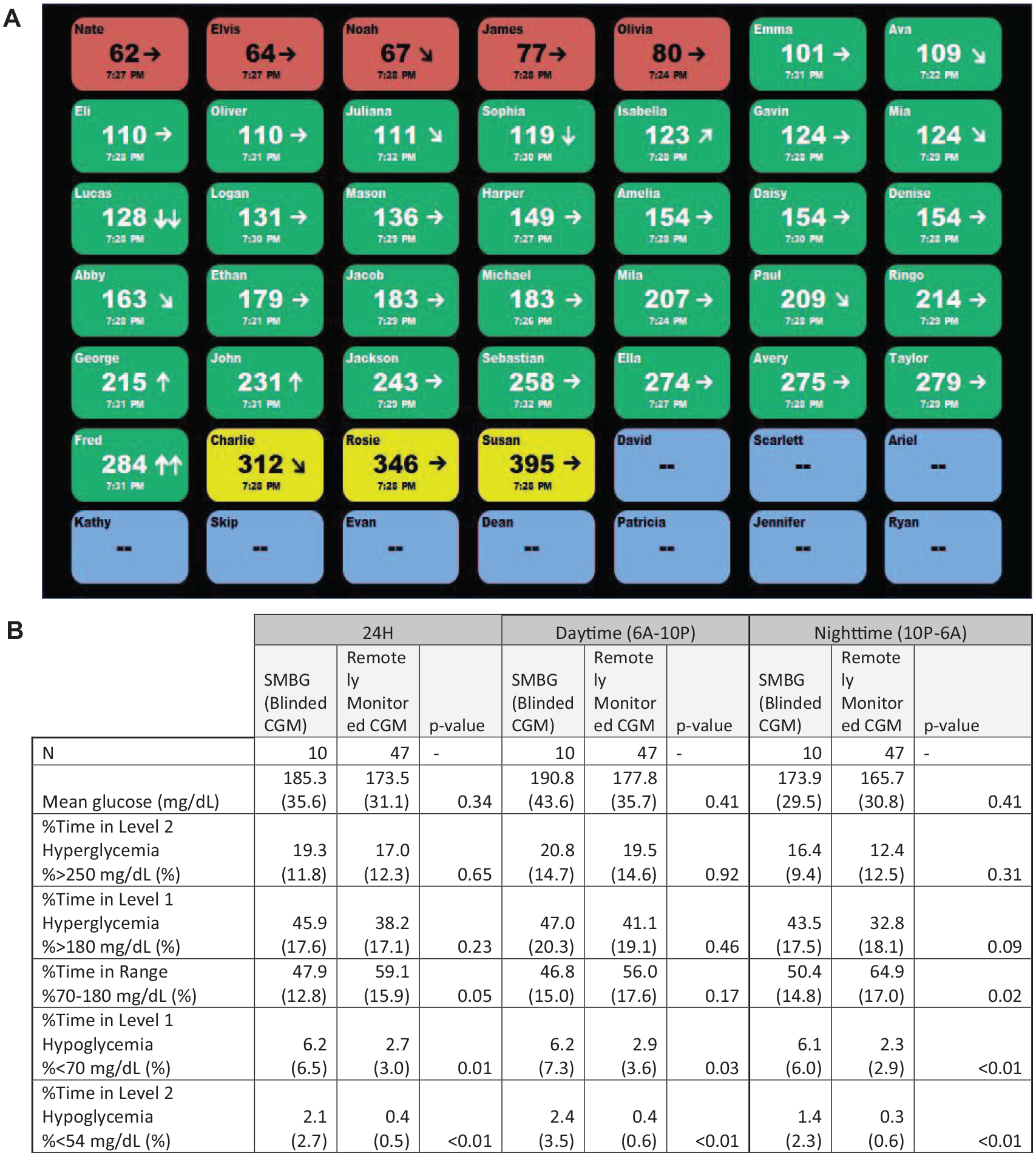
(A) Schematic of the investigational, centralized multiplex display that simultaneously displayed CGM data and trends from up to 49 campers (names shown are fictitious). Red marked campers who were low (according to camp target ranges), green marked those in range, and yellow marked those who were high. Blue were those who had lost connectivity due to smart phones being out of range of the campers. (B) CGM-derived glycemic metrics over 24 hours, during the day (6:00 am-10:00 pm) and overnight (10:00 pm-6:00 am) for campers treated with SMBG alone (wearing blinded CGM) versus those wearing unblinded CGM that were remotely monitored. Data reported are mean (SD).
Eighty campers were enrolled; 17 were fitted with blinded CGM and 63 continued to use their Dexcom CGM systems with Share at camp. Of these, 10 and 47, respectively, met data-coverage inclusion criteria and were analyzed. Mean ± SD age overall and for those in the blinded and unblinded groups was 12.2 ± 2.4, 11.4 ± 3.1, and 12.4 ± 2.2 years, respectively. All participants had T1D. All participants in the blinded group were treated with multiple daily injections (MDI). Only 23.4% (11/47) participants in the unblinded CGM group were treated with MDI while the remainder used pumps; 38.3% (18/47) of those were using automated insulin delivery (AID)/predictive low glucose suspend systems.
Centralized remote monitoring of CGM data was associated with significant improvement in time in range (TIR) overnight, manifested by a reduction in level 1 hyperglycemia, and with significantly less level 1 and level 2 hypoglycemia across the day and night compared to SMBG (blinded CGM) (Figure 1B). Remotely monitored campers spent 62% less time in level 1 and 79% less time in level 2 hypoglycemia overnight than those wearing blinded CGM. Hypoglycemia was not significantly different between remotely monitored campers who used AID systems vs standalone CGM (not shown). Centralized remote monitoring facilitated 73 interventions overnight that occurred outside the scheduled twice nightly SMBG measurements. These findings demonstrate the feasibility of centralized multiplex remote monitoring with modern CGM systems in a diabetes camp setting. Primary limitations are the lack of randomization and the small N in the blinded CGM group.
Footnotes
Acknowledgements
The authors thank Margi Battin and Tara Winkelman for their essential role in conducting the study, Shant Tokatyan and Charlotte Blanck for their help in developing and deploying the centralized remote monitoring dashboard, and Drs Christy Chao and John Welsh for their assistance in preparing the manuscript. Dexcom is a registered trademark of Dexcom, Inc. in the United States and/or other countries
Abbreviations
AID, automated insulin delivery; CGM, continuous glucose monitoring; MDI, multiple daily injections; SMBG, self-monitoring of blood glucose; TIR, time in range; T1D, type 1 diabetes.
Prior Publications
Presented, in part, at the 13th International Conference on Advanced Technologies & Treatments for Diabetes (ATTD) in February 2020, at the Diabetes Education and Camping Association (DECA) conference in February 2020, and at the American Diabetes Association (ADA) 80th Scientific Sessions in June 2020.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Sarah Puhr and Tomas Walker are employees of Dexcom, Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this research has been supported by a grant from Dexcom, Inc.
