Abstract

Type 2 diabetes (T2D) is characterized by insulin resistance and progressive loss of pancreatic beta-cell function. Chronic hyperglycemia has negative effects on beta-cell function and insulin secretion: a phenomenon known as glucotoxicity. 1 As beta-cell dysfunction results in further hyperglycemia, reversing this vicious cycle is important in managing T2D.
Closed-loop systems offer a novel method of glucose control in T2D, comprising a continuous glucose monitor, insulin pump, and control algorithm which modulates subcutaneous insulin delivery in response to real-time glucose levels.
A randomized controlled trial comparing fully automated closed-loop insulin delivery with CamAPS HX (CamDiab, Cambridge, UK) with standard insulin therapy demonstrated superior glycemic control, without increasing risk of hypoglycemia in 26 outpatients with T2D. 2
The present post hoc analysis aims to characterize changes in insulin requirements associated with improvements in glycemic control during the eight-week closed-loop period, which may provide insights into glucotoxicity and timing of its reversal.
For each participant, average daily insulin requirement over each two-week period was calculated as a percentage of their average daily insulin requirement over the eight-week closed-loop period. Mean glucose over consecutive two-week periods was calculated. One participant with <14 days of closed-loop use was excluded. Repeated measures one-way analysis of variance compared mean glucose levels and insulin requirements between two-week periods, with post hoc Tukey’s test. Outcomes were calculated using GStat software, version 2.3 (University of Cambridge, UK) and statistical analyses performed using SPSS Statistics, version 28 (IBM Software, Hursley, UK).
Data were analyzed from 1349 days in 25 participants (7 females, mean ± SD age: 58 ± 10 years, diabetes duration: 18 ± 8 years, baseline HbA1c: 75 ± 15 mmol/mol).
Mean glucose levels (Figure 1a) fell significantly from weeks 1-2 to weeks 5-6 of closed-loop (9.3-8.8 mmol/L, P = .04) and this was sustained in weeks 7-8. Associated with this improvement in glucose control, relative insulin requirements (Figure 1b) peaked at weeks 3-4 (108% of 8-week period average) followed by a non-significant trend to decrease in weeks 5-6 and 7-8 (down to 104% and 91% of 8-week period average, respectively, P = .08). Absolute total insulin dose (median [IQR]) peaked at 133 [76, 217] units at weeks 3-4, falling to 122 [64,197] units at 5-6 weeks and 103 [61, 160] units by weeks 7-8 (a decrease of 8% and 23%, respectively).
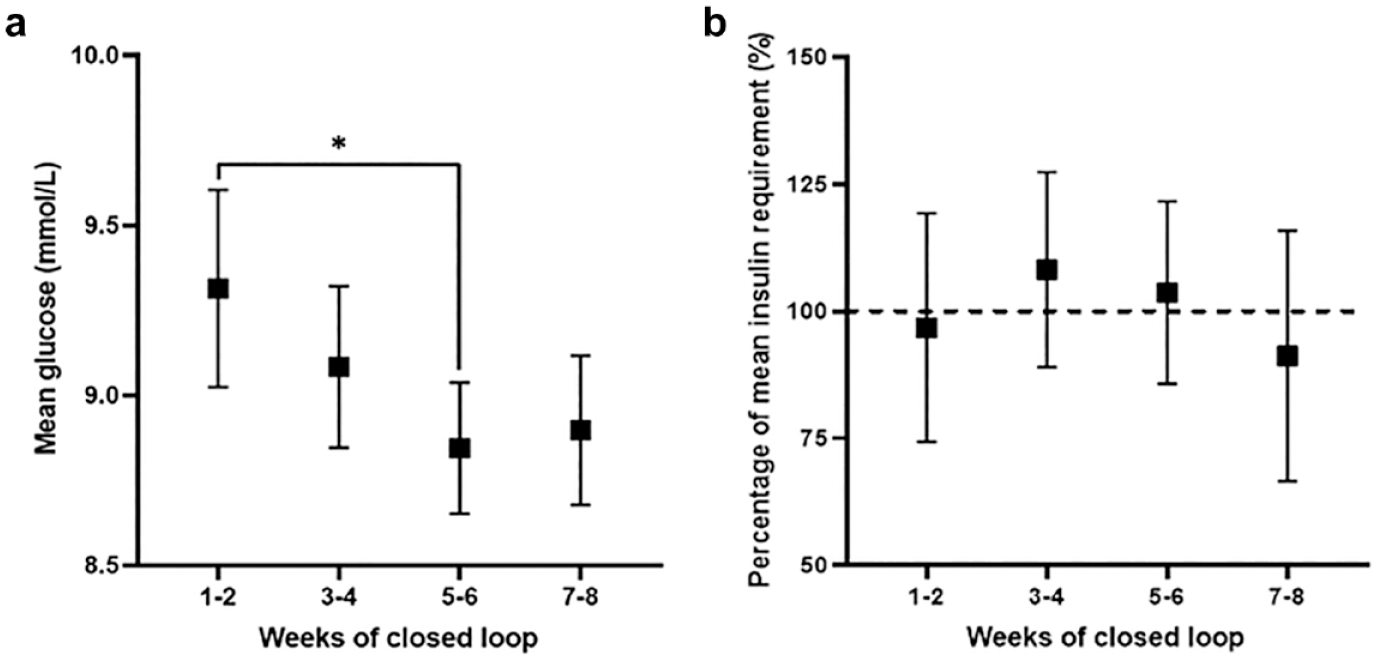
Two-weekly mean glucose (a) and average daily insulin requirement relative to average daily insulin requirement over the whole eight-week closed-loop period (b). Data are mean ± SD (n = 25).
The present study characterized the association between glucose levels and insulin requirements in adults with T2D using a fully closed-loop system. During the first two weeks, as the adaptive closed-loop algorithm determines individual insulin needs, mean glucose was at its highest while insulin delivery gradually escalates to optimize glycemic control. 3
As mean glucose reduces following initial algorithm adaptation, exogenous insulin requirements start to reduce after weeks 3-4. We hypothesize this may be due to amelioration of glucotoxicity, where lower glucose levels lead to improved beta-cell function and increased endogenous insulin production.
This analysis is limited by the small study size and relatively short duration. Larger, longer studies, including people with a more recent onset of T2D, are required to determine whether closed-loop insulin delivery can result in a significant and sustained reversal of glucotoxicity.
Footnotes
Acknowledgements
Dexcom supplied discounted continuous glucose monitoring systems for the study; company representatives had no role in the study conduct. Supported by National Institute for Health and Care Research Cambridge Biomedical Research Centre. The University of Cambridge has received salary support for MLE from the National Health Service in the East of England through the Clinical Academic Reserve. The funders of the study had no role in study design, data collection, data analysis, data interpretation, or writing of the report. We are grateful to all study participants for their contribution, time and support. The views expressed are those of the authors and not necessarily those of the NIHR, the Department of Health and Social Care, or other funders.
Author Contributions
RL, CKB, and RH designed the analysis. AD, CKB, and RH co-designed the study. AD, CKB, MN, and SH were responsible for screening and enrolment of participants, arranged informed consent from the participants, and provided patient care. RL, CKB, MEW, and RH carried out data analysis. RH, CKB, AD, MN, RL, and MLE contributed to the interpretation of the results. AC coordinated the study. All authors critically reviewed the manuscript. RH and CKB are the guarantors of this work and, as such, had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Abbreviations
IQR, interquartile range; SD, standard deviation; T2D, type 2 diabetes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CKB has received consultancy fees from CamDiab and speaker honoraria from Ypsomed. MN has received travel grant support from Sanofi, Janssen, and Eli Lilly and was previously chair of the Young Diabetologists’ and Endocrinologists’ Forum in the United Kingdom, which uses unrestricted sponsorship from industry partners to deliver educational programs for health care professionals. SH serves as a member of Sigma (Dexcom) and Medtronic advisory boards, is a director of Ask Diabetes Ltd providing training and research support in health care settings, is a consultant at CamDiab, and reports having received training honoraria from Medtronic and Sanofi. MEW reports receiving license fees from B. Braun, patents related to closed-loop, and being a consultant at CamDiab. AC has received consultancy fees from CamDiab. MLE has been a member of advisory panels and/or received speakers fees from Novo Nordisk, Eli Lilly, Abbott Diabetes Care, Medtronic, Ypsomed, Pila Pharma, and Zucara. RH reports having received speaker honoraria from Eli Lilly, Dexcom, and Novo Nordisk, receiving license fees from BBraun, and being director at CamDiab. RL and AD declare no duality of interest associated with the present manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by the NIHR Cambridge Biomedical Research Centre.
