Abstract
Aim:
An objective assessment of small nerve fibers is key to the early detection of diabetic peripheral neuropathy (DPN). This study investigates the diagnostic accuracy of a novel perception threshold tracking technique in detecting small nerve fiber damage.
Methods:
Participants with type 1 diabetes (T1DM) without DPN (n = 20), with DPN (n = 20), with painful DPN (n = 20) and 20 healthy controls (HCs) underwent perception threshold tracking on the foot and corneal confocal microscopy. Diagnostic accuracy of perception threshold tracking compared to corneal confocal microscopy was analyzed using logistic regression.
Results:
The rheobase, corneal nerve fiber density (CNFD), corneal nerve branch density (CNBD), and corneal nerve fiber length (CNFL) (all P < .001) differed between groups. The diagnostic accuracy of perception threshold tracking (rheobase) was excellent for identifying small nerve fiber damage, especially for CNFL with a sensitivity of 94%, specificity 94%, positive predictive value 97%, and negative predictive value 89%. There was a significant correlation between rheobase with CNFD, CNBD, CNFL, and Michigan Neuropathy Screening Instrument (all P < .001).
Conclusion:
Perception threshold tracking had a very high diagnostic agreement with corneal confocal microscopy for detecting small nerve fiber loss and may have clinical utility for assessing small nerve fiber damage and hence early DPN.
Clinical Trials:
NCT04078516
Introduction
Diabetic peripheral neuropathy (DPN) affects more than 50% of people with diabetes mellitus and is associated with considerable morbidity due to painful diabetic neuropathy and foot ulceration as well as increased mortality.1 -5 Currently, advocated screening methods for the detection of DPN, for example, monofilament testing or vibration perception thresholds only diagnose advanced disease or large fibers, for example, nerve conduction testing. 6 Small nerve fiber abnormalities occur early and may precede large fiber dysfunction in DPN. 7 Quantitative sensory testing can be used to assess small Aδ- and C-fiber function but have limited precision and reproducibility. 8 More objective methods like corneal confocal microscopy (CCM) and skin biopsies with quantification of intraepidermal nerve fiber density (IENFD) are considered the gold standard for evaluating small fiber damage in DPN.9 -11 However, they require advanced equipment and expertise for analysis and evaluate structural rather than functional abnormalities.
We have recently developed a novel perception threshold tracking technique, which uses weak electrical currents to selectively stimulate peripheral large (Aβ) and small nerve fibers (Aδ). 12 It differed between people with and without diabetes and correlated with thermal thresholds obtained from quantitative sensory testing. 12 However, the limited accuracy and reproducibility of thermal thresholds compared to CCM or skin biopsies, limits the strength of this comparison.
In this article, we aimed to (1) compare the outcomes of perception threshold tracking and CCM, (2) investigate the diagnostic accuracy of perception threshold tracking for the detection of small fiber neuropathy using CCM as a reference, and (3) investigate the relationship between structural and functional measures of small nerve fiber damage.
Methods
Study Design and Participants
The study was conducted between August 2019 and February 2022 in participants from the “MEDON” (Methods for Early Detection Of diabetic peripheral Neuropathy)-cohort (clinicaltrials.gov: NCT04078516).12 -14 This population is described in details elsewhere, but in short, the original cohort consisted of 80 participants equally divided into four groups: participants with T1DM and painful diabetic peripheral neuropathy (PDPN), participants with T1DM and DPN, participants with T1DM and no DPN, and healthy controls (HCs) without diabetes or pain.12 -14 The four groups were matched 1:1:1:1 on age (±2 years) and sex. Painful diabetic peripheral neuropathy was diagnosed based on the Douleur Neuropathique 4 Questionnaire (DN4) score ≥4, and DPN was diagnosed according to the clinical Toronto consensus (neuropathic symptoms, decreased distal sensation, or unequivocally decreased or absent ankle reflexes).15,16 The population was extensively screened to exclude other causes of neuropathy including vitamin deficiencies, hematologic or immune diseases, thyroid, or parathyroid disease, long-term kidney disease, previous alcohol or drug abuse, previous chemotherapy, severe or long-term viral infection, and active cancer. Subjects with current eye-infections, corneal abrasions, a history of bilateral refractive surgery, or anterior segment trauma which might affect the cornea, were excluded. From the original 80 participants, nine were excluded due to previous refractive surgery.
Assessment of Peripheral Nerves
After inclusion, neurological examination was undertaken alongside the Michigan Neuropathy Screening Instrument (MNSI). 17 Neuropathic pain severity (over the last four weeks) was derived from the PainDETECT questionnaire as average and peak pain intensity rated on a visual analog scale (VAS) ranging from 0 to 10, where 0 is no pain and 10 is the most intense pain imaginable. 18 Quantitative sensory testing and conventional nerve conduction studies were performed as previously described and reported.12 -14
Perception Threshold Tracking
Perception threshold tracking of small nerve fibers was performed following our previously published protocol using the method of limits.12,19 In short, the participants were electrically stimulated using a custom-made pin electrode (Aalborg University, Denmark) in an area of 2 to 3 cm2 proximal to the second toe on the dorsum of the right foot. The electrode consists of a concentric stainless-steel ring electrode with an area of 8.8 cm2 serving as the anode, and a printed circuit board with 16 stainless steel pin electrodes placed in a circle serving as the cathodes. The pins were all blunted with a diameter of 0.2 mm. The electrical stimuli were delivered using a DS5 electrical stimulator (Digitimer Ltd, UK) and controlled by a protocol implemented in a custom-made program (LabBench Io, Inventors Way, Denmark). 19 Participant responses were captured using a custom-made handheld response button (Inventors Way, Denmark), a personal computer, and a data acquisition card (LabBench Io, Inventors Way, Denmark). The perception threshold was estimated using square impulses with varying durations (0.1, 1, and 50 ms) and intensities. The rheobase (the lowest current intensity of infinite duration that results in perception of the impulse) and the chronaxie (the minimal pulse duration required to double the strength of the rheobase) (not reported) were derived from the corresponding strength-duration curve.12,20 Owing to the nature of the electrode, the electrical stimulation is almost exclusively present near the terminals of the small nerve fibers in the epidermis, without reaching the large nerve fibers in the dermis, and thus preferentially stimulate the small nerve fibers. 12
Corneal Confocal Microscopy
All participants underwent in-vivo corneal confocal microscopy (IVCM) using a Heidelberg Retinal Tomograph III Rostock Cornea Module (Heidelberg Engineering GmbH, Heidelberg, Germany). One-hundred images from the corneal apex with a resolution of 400 × 400 µm were acquired using a volume scan. In participants with unilateral anterior segment trauma or refractive surgery only the nonaffected eye was examined. Two authors (JR and SC) blinded for participant ID, each selected three to four representative images, totalling six to eight images per participant. Selection criteria were good contrast of the nerves compared to the background, limited motion artifacts, limited pressure lines, limited image overlapping (maximum 20%), en-face alignment, and proper focus. 21
Manual morphometric analysis was conducted using CCMetrics (M.A. Dabbah, Imaging Science and Biomedical Engineering, University of Manchester, Manchester, United Kingdom) to obtain corneal nerve fiber length (CNFL), corneal nerve branch density (CNBD), corneal nerve fiber density (CNFD), and corneal nerve tortuosity.22,23 Corneal nerve fiber length was defined as the total length of all nerve fibers per frame (mm/mm2). Corneal nerve branch density was defined as the number of primary branches from the main nerves (no./mm2). Corneal nerve fiber density was defined as the number of main nerve fibers taking up more than 50% of the total frame length (no./mm2). The tortuosity coefficient was derived from the main nerve fibers. 22
Statistical Analyses
Categorical data are expressed as a percentage of participants and compared using chi-square or Fisher’s exact tests. Continuous data are expressed as mean ± standard deviation (SD) when data were normally distributed and as medians with interquartile ranges (IQR) when data were not normally distributed. Normally distributed data (visually defined by QQ-plots) were compared using parametric tests (Student’s t tests, analysis of variance [ANOVA]), while non-normally distributed data were compared using nonparametric tests (Kruskal-Wallis H tests followed by Mann-Whitney U-tests, and Spearman’s rank correlation). The significance level was set at α = .05. All relevant comparisons were corrected using Bonferroni corrections. Logistic regressions were used to generate receiver operating characteristic (ROC) curves, estimate the area under the curve (AUC), and calculate sensitivities, specificities, positive predictive values, negative predictive values for each parameter (see Figure 1 for ROC curves and online supplementary material for probability cut-off graphs). When using CNFL, CNBD, or CNFD as reference, the cut-off points for an abnormal result was determined as the lower fifth quantile derived from the published normative values. 24 Only participants with diabetes were used for the ROC curves.
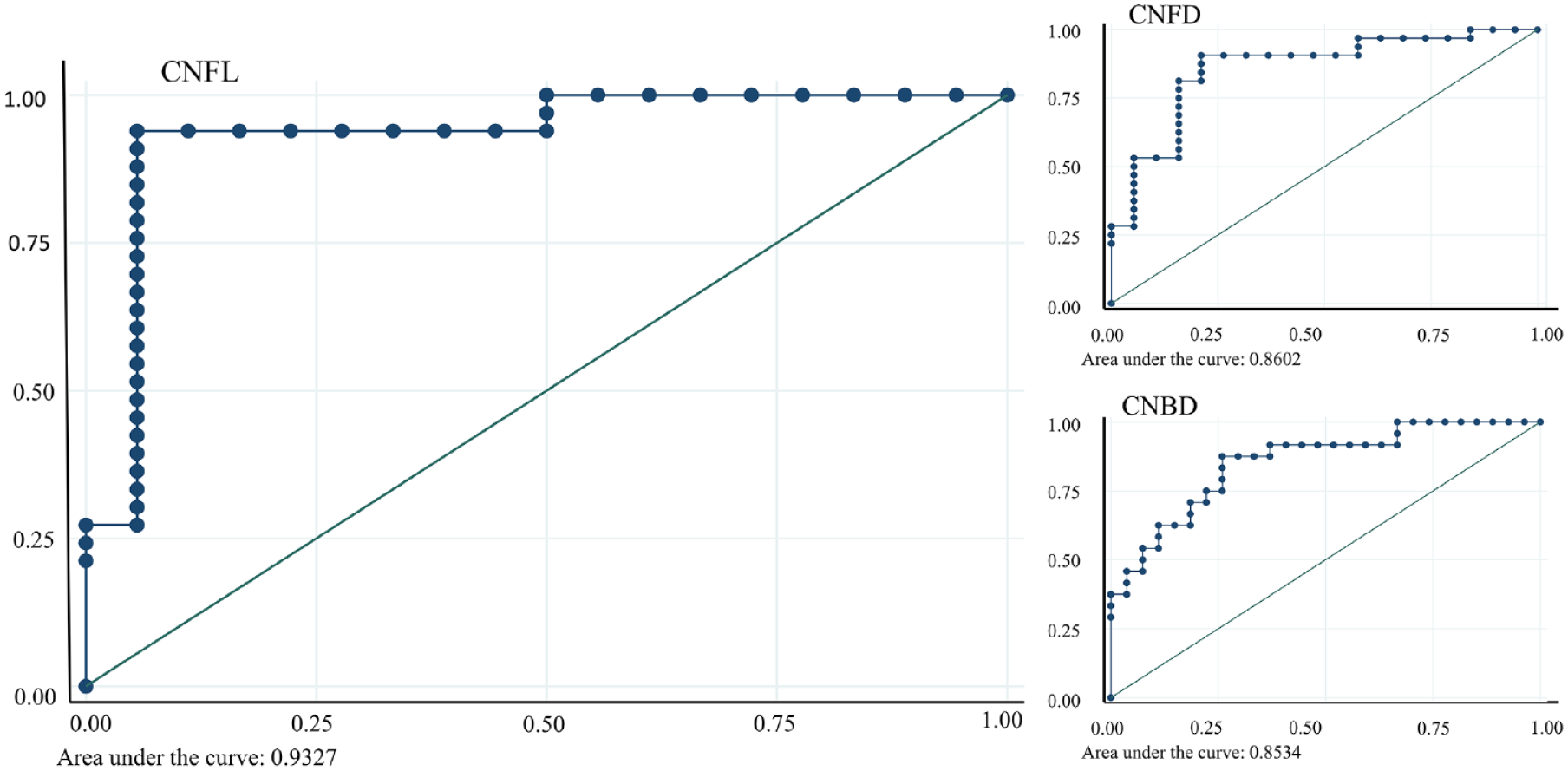
Receiver operating characteristic curves for the rheobase of perception threshold tracking compared to the corneal nerve fiber length, corneal nerve fiber density, and corneal nerve branch density.
Results
Demographics and Characteristics
Participants with complete data were divided as follows: T1DM + PDPN (n = 19), T1DM + DPN (n = 14), T1DM − DPN (n = 19), and HCs (n = 19). There was a significant difference between the groups for hemoglobin A1c, sural nerve conduction velocity and amplitude, cold and heat detection thresholds, pain scores, MNSI, and DN4-scores (all P < .001; Table 1).
Demographics and Test Results for Participants in T1DM + PDPN, T1DM + DPN, T1DM − DPN, and Healthy Controls.
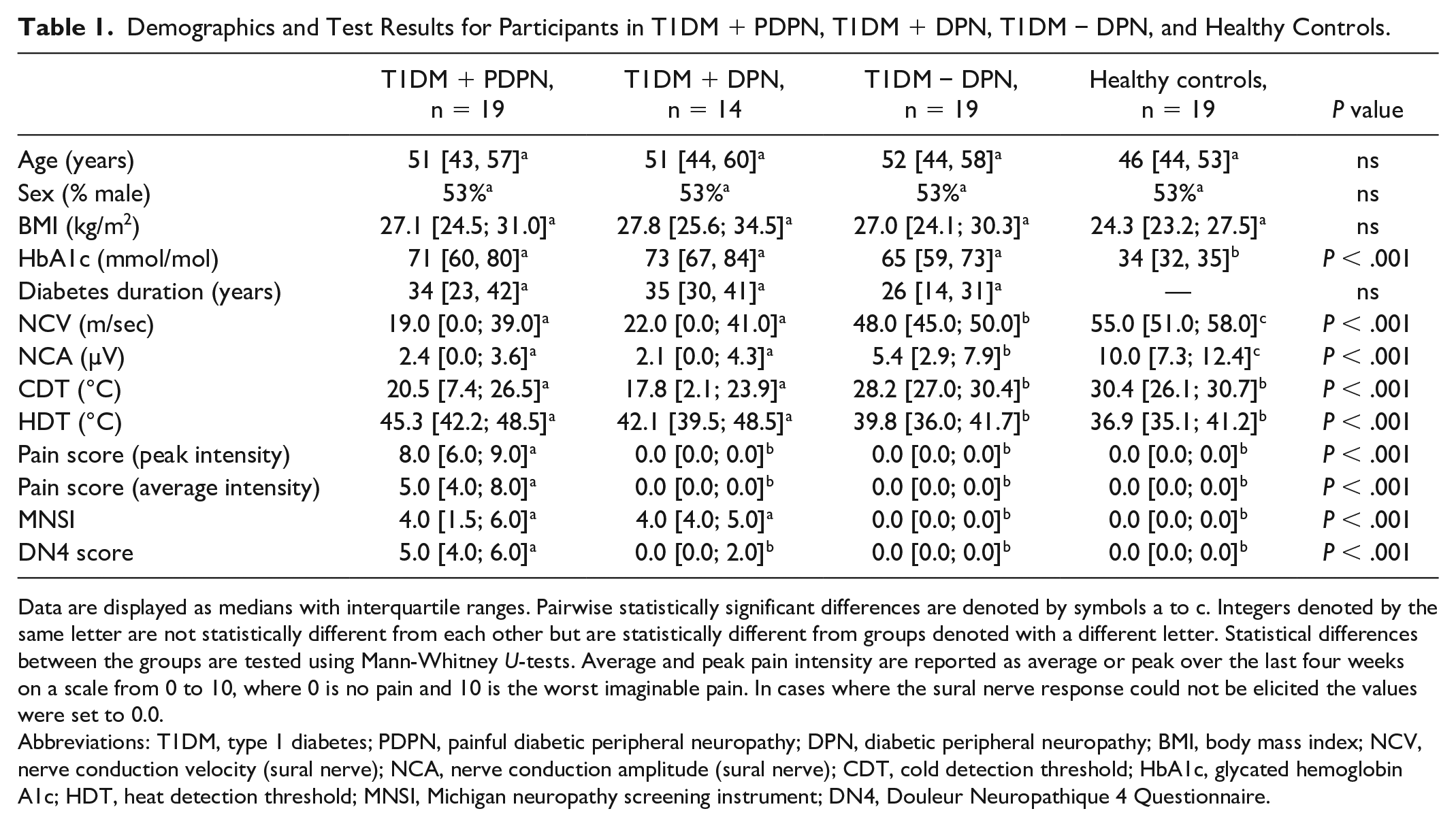
Data are displayed as medians with interquartile ranges. Pairwise statistically significant differences are denoted by symbols a to c. Integers denoted by the same letter are not statistically different from each other but are statistically different from groups denoted with a different letter. Statistical differences between the groups are tested using Mann-Whitney U-tests. Average and peak pain intensity are reported as average or peak over the last four weeks on a scale from 0 to 10, where 0 is no pain and 10 is the worst imaginable pain. In cases where the sural nerve response could not be elicited the values were set to 0.0.
Abbreviations: T1DM, type 1 diabetes; PDPN, painful diabetic peripheral neuropathy; DPN, diabetic peripheral neuropathy; BMI, body mass index; NCV, nerve conduction velocity (sural nerve); NCA, nerve conduction amplitude (sural nerve); CDT, cold detection threshold; HbA1c, glycated hemoglobin A1c; HDT, heat detection threshold; MNSI, Michigan neuropathy screening instrument; DN4, Douleur Neuropathique 4 Questionnaire.
Perception Threshold Tracking and CCM
There was a significant difference in the rheobase, CNFD, CNBD, and CNFL (all P < .001) with no difference in tortuosity (P = .221) between all four groups (Table 2).
Results From Corneal Confocal Microscopy and Perception Threshold Tracking of the Small Nerve Fibers.
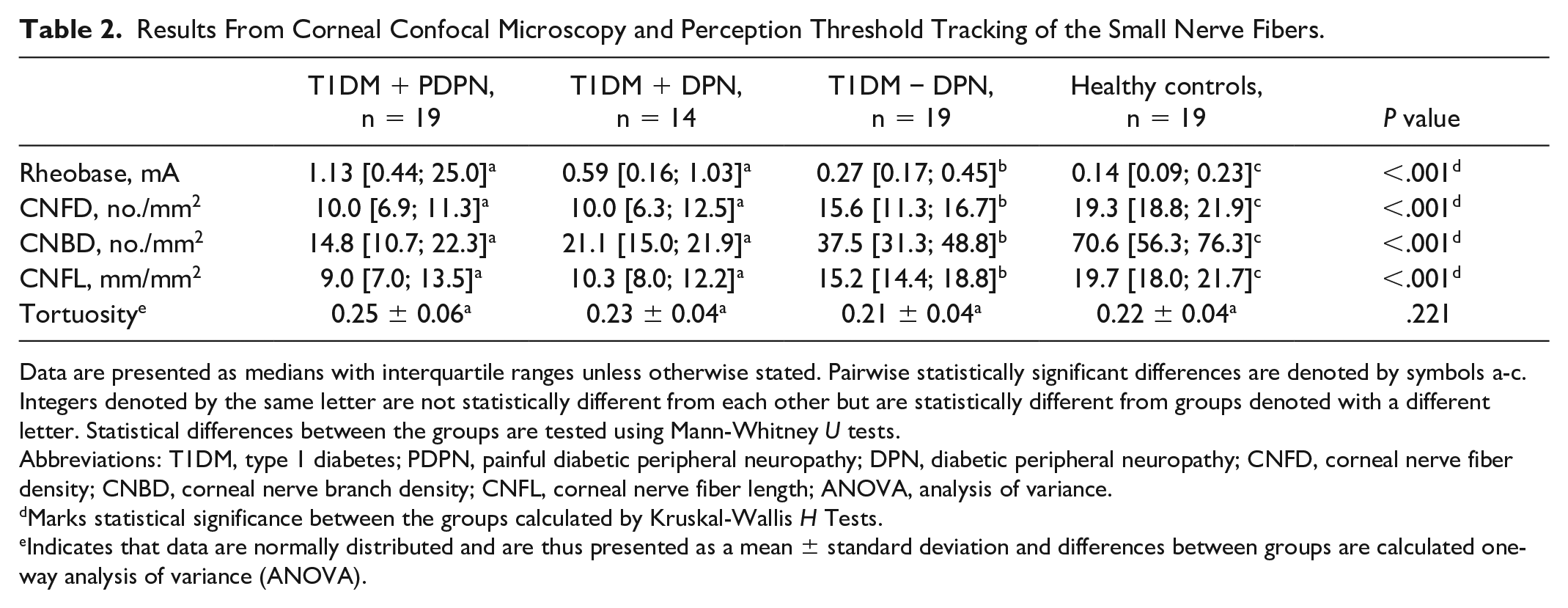
Data are presented as medians with interquartile ranges unless otherwise stated. Pairwise statistically significant differences are denoted by symbols a-c. Integers denoted by the same letter are not statistically different from each other but are statistically different from groups denoted with a different letter. Statistical differences between the groups are tested using Mann-Whitney U tests.
Abbreviations: T1DM, type 1 diabetes; PDPN, painful diabetic peripheral neuropathy; DPN, diabetic peripheral neuropathy; CNFD, corneal nerve fiber density; CNBD, corneal nerve branch density; CNFL, corneal nerve fiber length; ANOVA, analysis of variance.
Marks statistical significance between the groups calculated by Kruskal-Wallis H Tests.
Indicates that data are normally distributed and are thus presented as a mean ± standard deviation and differences between groups are calculated one-way analysis of variance (ANOVA).
Diagnostic Performance of Perception Threshold Tracking
The sensitivity, specificity, positive predictive value, and negative predictive value of the rheobase (perception threshold tracking) to detect small fiber neuropathy (as defined by CNFD, CNBD, and CNFL) was generally very good with the highest performance against CNFL with an AUC of 0.93, sensitivity of 94%, specificity of 94%, positive predictive value of 97% and negative predictive value of 89% at an optimal cut-off of 0.29 mA (Table 3, Figure 1, Supplementary Figure 1).
Comparison of the Rheobase Derived From Perception Threshold Tracking and Corneal Confocal Microscopy Measurements.
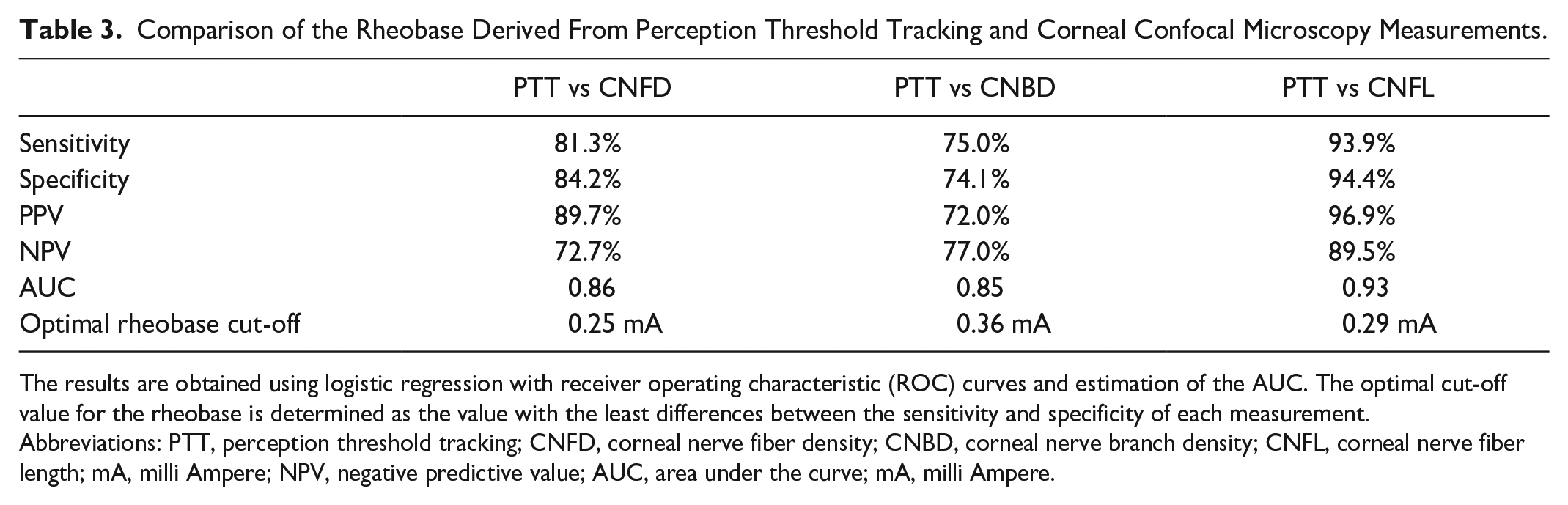
The results are obtained using logistic regression with receiver operating characteristic (ROC) curves and estimation of the AUC. The optimal cut-off value for the rheobase is determined as the value with the least differences between the sensitivity and specificity of each measurement.
Abbreviations: PTT, perception threshold tracking; CNFD, corneal nerve fiber density; CNBD, corneal nerve branch density; CNFL, corneal nerve fiber length; mA, milli Ampere; NPV, negative predictive value; AUC, area under the curve; mA, milli Ampere.
Relationship Between Perception Threshold Tracking and Corneal Confocal Microscopy
There was a significant correlation between the rheobase with CNFD, CNBD, CNFL, and MNSI (all P < .001) (Table 4).
Correlations Between the Rheobase Measured By Perception Threshold Tracking and corneal Confocal Microscopy Measures and Neuropathy Severity Score.
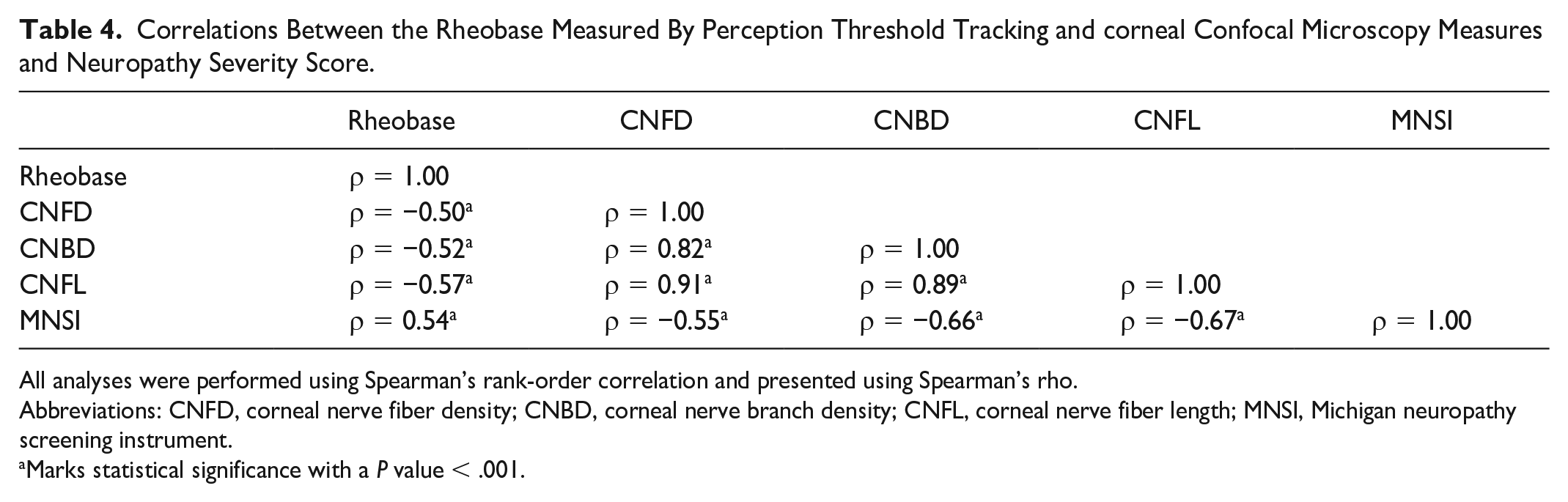
All analyses were performed using Spearman’s rank-order correlation and presented using Spearman’s rho.
Abbreviations: CNFD, corneal nerve fiber density; CNBD, corneal nerve branch density; CNFL, corneal nerve fiber length; MNSI, Michigan neuropathy screening instrument.
Marks statistical significance with a P value < .001.
Discussion
The perception threshold tracking technique is a novel small nerve fiber test with a unique ability to selectively stimulate small sensory nerve fibers in the skin. We now demonstrate a close relationship and high diagnostic agreement between perception threshold tracking and CCM, an acknowledged structural marker for small fiber damage. 25 Due to the novelty of perception threshold tracking, no studies have previously made a similar comparison. In participants with type 1 diabetes, a significant correlation was demonstrated between conventional threshold tracking of motor nerves, a measure of peripheral nerve excitability and axonal ion channel dysfunction and CCM parameters.26,27 In another study of participants with type 2 diabetes, there was a significant relationship between conventional threshold tracking and CCM, with a corresponding decrease in the recovery cycle, suggesting an abnormality in voltage-gated potassium channels. 28 Thus, this sensory perception threshold tracking technique may provide important insights into voltage-gated ion channel function on sensory nerves.
Functional tests of small fiber damage have been a corner stone for the diagnoses of small fiber neuropathy. Indeed, the German Research Network on Neuropathic Pain (DFNS) developed a comprehensive standardized protocol to enable deep-phenotyping and subtyping of neuropathic pain.29,30 However, quantitative sensory testing has repeatedly failed as a clinical endpoint in clinical trials.31,32 Thus, it has been proposed that objective measures of corneal nerve and intraepidermal nerve fiber pathology may have diagnostic utility for small fiber neuropathy. 33 However, the invasive nature and technical requirements for skin biopsies and limited availability and extensive image analysis of CCM have limited their wider adoption. 6 The sensory perception threshold tracking technique presented in this article is a simple, rapid (<5 min) measure of peripheral sensory nerve fiber function, feasible for large-scale screening.
There is no consensus as to whether there are functional or structural measures of small fiber damage which differentiate painful from painless neuropathy.34,35 Several recent studies with large cohorts have shown greater corneal nerve fiber loss in people with diabetes and painful compared to painless diabetic neuropathy and a relationship with the severity of pain, whilst others have shown increased nerve branching or have failed to establish such a relationship.21,36,37 Similarly, some studies using conventional threshold tracking have associated altered axonal excitability with PDPN, while others have reported altered sodium conductance and ion-channel function, alteration in Na+/K+ pump function, and membrane depolarization in DPN irrespective of the presence of neuropathic pain.38 -40 Recently, a large multicentre study reported no difference in axonal excitability when comparing those with painless to those with PDPN, concluding that electrophysiological measures targeting the small nerves are needed to rule out axonal excitability changes in PDPN. 41 One such method is microneurography, which is capable of assessing the function of small cutaneous C-fibers, and have previously associated increased spontaneous activity of cutaneous C-fibers with PDPN.42,43 Microneurography is however limited by invasiveness and vast technical requirements and time-consumption, while simultaneously being unable to assess small cutaneous Aδ-fibers. 44
In this study, there was no difference in CCM measures or perception threshold tracking between people with painful and painless diabetic neuropathy, possibly due to the small cohort size. Also, the selectivity of perception threshold tracking declines as the required current intensity rises in those with more severe neuropathy, ultimately reaching a point where the acquired threshold is likely the threshold of the large (Aβ) fibers rather than the threshold for the small (primarily Aδ) fibers. 12 Threshold tracking may be more useful in differentiating people with PDPN grouped into irritable and non-irritable sub types, which may provide insights into peripheral ion-channel composition using computational modeling alongside perception threshold tracking.45,46 Such analyses would however require more extensive and time-consuming perception threshold tracking protocols including pulses relevant for assessing, that is, threshold electrotonus. 27
Based on this study, it would appear that DPN is associated with comparable changes to both nerve fiber structure and function, although further studies are needed to confirm these findings.
In this study, the diagnostic agreement between perception threshold tracking and CCM were highest when comparing the rheobase of perception threshold tracking with CNFL from CCM. Corneal nerve fiber length is the most established and well-examined of the corneal parameters and based on our results it might also be the most reliable. In fact, perception threshold tracking correctly classified all participants with clinically established neuropathy (T1DM + PDPN or T1DM − DPN) when compared to CNFL.
We acknowledge that the small numbers of participants studied may limit our ability to differentiate different subtypes of DPN, especially painful DPN. The perception threshold tracking technique also loses its selectivity at very high current intensities, which may impact on the correlations in this article but does not impact on sensitivity, specificity, positive, and negative predictive value. The CCM values in the controls were slightly lower than previously published normative values, which may have impacted on the identification of abnormal values. 24 The very high diagnostic agreement between the two different methods may reflect the highly selected population used in this study, and it remains unknown if the diagnostic agreement will remain this high in other populations.
Conclusion
Perception threshold tracking is a rapid measure of small nerve fiber function and appears to have clinical utility as a method for neuropathy screening given that CCM identifies early small nerve fiber damage. 1Further validation of the method is required against skin biopsies at the site of threshold testing in a larger, randomly selected, cohort.
Supplemental Material
sj-docx-1-dst-10.1177_19322968231157431 – Supplemental material for Diagnostic Accuracy of Perception Threshold Tracking in the Detection of Small Fiber Damage in Type 1 Diabetes
Supplemental material, sj-docx-1-dst-10.1177_19322968231157431 for Diagnostic Accuracy of Perception Threshold Tracking in the Detection of Small Fiber Damage in Type 1 Diabetes by Johan Røikjer, Suganthiya Santhiapillai Croosu, Benn Falch Sejergaard, Tine Maria Hansen, Jens Brøndum Frøkjær, Chris Bath Søndergaard, Ioannis N. Petropoulos, Rayaz A. Malik, Esben Nielsen, Carsten Dahl Mørch and Niels Ejskjaer in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
JR wrote the manuscript, conducted most examinations, researched data, and contributed to the idea and study design. NE, CM, JF, and TH contributed to the idea and study design, reviewed the manuscript, and conducted critical editing of written text. SC assisted conducting some of the examinations, contributed to the idea and study design, reviewed the manuscript, and conducted critical editing of written text. BS, CS, and EN conducted the CCM examinations and conducted critical editing of written text. RM and IP assisted CCM image selection and analysis, contributed with expert knowledge and insight, and conducted critical editing of the written text. Each author is accountable for his own contribution, disclosure of potential interests, and approved the final version of the manuscript. NE is the guarantor and is responsible for all aspects of the manuscript. CM is part of the Center for Neuroplasticity and Pain (CNAP), supported by the Danish National Research Foundation (grant no. DNRF121). No specific grant was received to undertake this study.
Abbreviations
AUC, area under the curve; BMI, body mass index; CCM, corneal confocal microscopy; CDT, cold detection threshold; CNAP, Center for Neuroplasticity and Pain; CNBD, corneal nerve branch density; CNFD, corneal nerve fiber density; CNFL, corneal nerve fiber length; DFNS, German Research Network on Neuropathic Pain; DN4, Douleur Neuropathique 4 Questionnaire; DPN, diabetic peripheral neuropathy; HbA1c, glycated hemoglobin A1c; HC, healthy controls; HDT, heat detection threshold; IENFD, intraepidermal nerve fiber density; IQR, interquartile ranges; IVCM, in-vivo corneal confocal microscopy; mA, Milliampere; MNSI, Michigan neuropathy screening instrument; NCA, nerve conduction amplitude; NCV, nerve conduction velocity; NPV, negative predictive value; PDPN, painful diabetic peripheral neuropathy; PPV, positive predictive value; PTT, perception threshold tracking; ROC, receiver operating characteristic; SD, standard deviation; T1DM, type 1 diabetes mellitus; VAS, visual analog scale.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study is funded by Aalborg University Hospital and Aalborg University.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
