Abstract
Project ECHO® is a telementoring workforce development model that targets under-resourced communities lacking access to specialty care. The model builds virtual communities of practice, including specialists and community primary care professionals (PCPs) to combat clinical inertia and health disparities. While the ECHO model has gained global recognition, implementation of the model related to diabetes is lagging compared to other specialty conditions. This review highlights diabetes-endocrine (ENDO)-focused ECHOs using data reported in the ECHO Institute’s centralized data repository (iECHO) and the learning collaborative for diabetes ECHOs. It also describes the implementation of diabetes ECHOs and their evaluation. Learner and patient-centered outcomes related to diabetes ECHOs are reviewed. Program implementation and evaluations have demonstrated utility of the ECHO model for diabetes programs to (1) address unmet needs of diabetes care in the primary care setting, (2) improve knowledge and confidence in managing complex diabetes and change provider prescribing habits, (3) improve patient outcomes, and (4) address diabetes quality improvement practices in primary care. More studies with broader collaboration among sites are needed to evaluate the model related to diabetes, especially applied to addressing therapeutic inertia, adoption of diabetes technology, and reducing health disparities.
Introduction
Advances in diabetes technology and therapeutics are not reaching the majority of people with diabetes (PWD). Fewer than 40% of adults are achieving the American Diabetes Association Standards of Care hemoglobin A1c (HbA1c) goals and more are considered “poorly controlled” (HbA1c > 9%).1 -5 While utilization of continuity of care by endocrinologist and diabetes specialty teams has demonstrated improvement in patient outcomes, there is and will continue to be a shortage of endocrinologists.6 -9 Additionally, a multitude of structural inequalities, including housing instability, food insecurity, educational disparities, and limited employment opportunities, disadvantage many with diabetes and negatively impact its prevalence and outcomes.10,11 For now, and the foreseeable future, the majority of adults with diabetes are and will be treated by primary care professionals (PCPs) and in community health centers (CHCs) rather than by endocrinologists and in diabetes specialty centers. 12 CHCs, which include federally qualified health centers (FQHCs), care for a higher proportion of socially and medically vulnerable populations, where 91% of patients are in or near poverty and where the burden of diabetes is double that of the general population. 13 Consequently, from a population health and “medical home” perspective,14 -16 efficient and targeted approaches to increase access to evidence-based diabetes care and address diabetes health disparities for PWD should target primary care and CHCs.
The Project ECHO® (Extension for Community Healthcare Outcomes) model is one example of a novel telehealth modality that can be used to achieve these goals and target clinicians caring for medically underserved populations with diabetes (Figure 1). It is not traditional telemedicine with 1 clinician and 1 patient;17,18 rather, it is the formation of a community of practice where potentially many patients benefit as a clinician’s knowledge increases and the clinicians involved gain the collaboration and expertise of their colleagues (Figure 1).
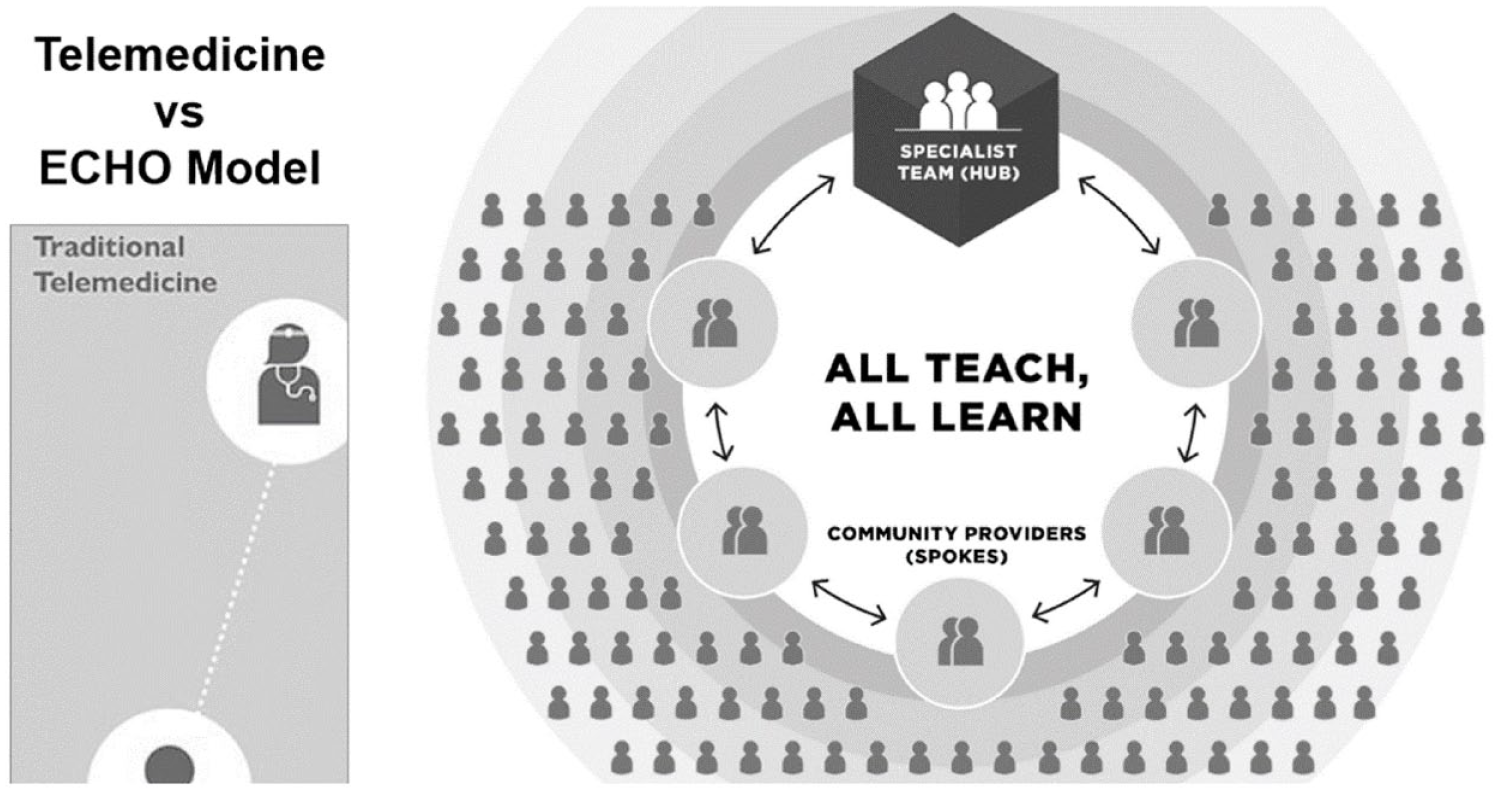
Traditional telemedicine vs. ECHO model.
Disparities in diabetes care, especially in the use of newer medications and diabetes technology, continues to be a barrier to optimizing glycemic control and addressing beyond glycemic targets. Racial and socioeconomic status (SES) disparities in diabetes technology utilization have been well documented in diabetes specialty centers across the United States through the T1D Exchange Registry.19,20 With regard to type 2 diabetes clinical inertia, especially in those with cardiovascular disease, there has been little increase in prescribing of diabetes medications with cardiovascular indications such as glucagon-like peptide-1 receptor agonists (GLP-1 RAs) and sodium glucose co-transporter-2 inhibitors (SGLT2-Is), despite guidelines recommending early use. Their use is even less in those without commercial insurance, of lower income and in Asian, Black, and Hispanic populations.21 -23 The ECHO model is one way of combating this clinical inertia while also targeting health disparities by amplifying diabetes knowledge.
The ECHO Model: Telementoring Primary Care Professionals to Reduce Disparities and Increase Access
This model consists of an interprofessional specialist “hub” team, typically at academic centers, telementoring local frontline PCP teams (“spokes”) via teleconferencing technology, allowing the amplification of care. The spokes provide care for PWD who lack access to routine specialty diabetes care. Hubs provide guided practice using a case-based framework. The model was established in 2003 at the University of New Mexico (UNM) to address health disparities and improve access to treatment for hepatitis C in rural communities. It was developed from several key observations: 1. the majority of patients lack access to or utilization of specialty care and rely on primary care for complex medical care, 2. poor health outcomes in specialty care exist in vulnerable populations as many patients are unable to receive specialty care as frequently as needed, and 3. PCPs lack confidence in managing complex specialty conditions. A landmark study in New Mexico (NM) demonstrated that patients receiving care in the primary care setting leveraging the model had as effective outcomes as patients receiving care at the academic medical specialty center.24,25
As of September 2022, there were over 800 ECHO hubs with outreach to 192 countries to combat disparities for over 70 complex conditions.26 -31 The first diabetes management ECHO was launched in 2009 in Nevada. Since that time, according to data collected by UNM through their iECHO data platform, the program has been adapted and implemented by 33 hub organizations in 58 unique programs, which have held case-based endocrine learning sessions (Figure 2). Over 20 of these hubs participate in a monthly diabetes-related learning collaborative designed to share best practices across ECHOs. 32 Most of these programs target adults living with diabetes and 2 have a specific pediatric focus. Through remote training of PCPs to provide specialty level care locally, the model simultaneously addresses the issues of traditional telemedicine including lack of digital health literacy, access to internet, and specialists, and reimbursement issues, while supporting the primary care core and the “Medical Neighborhood.”
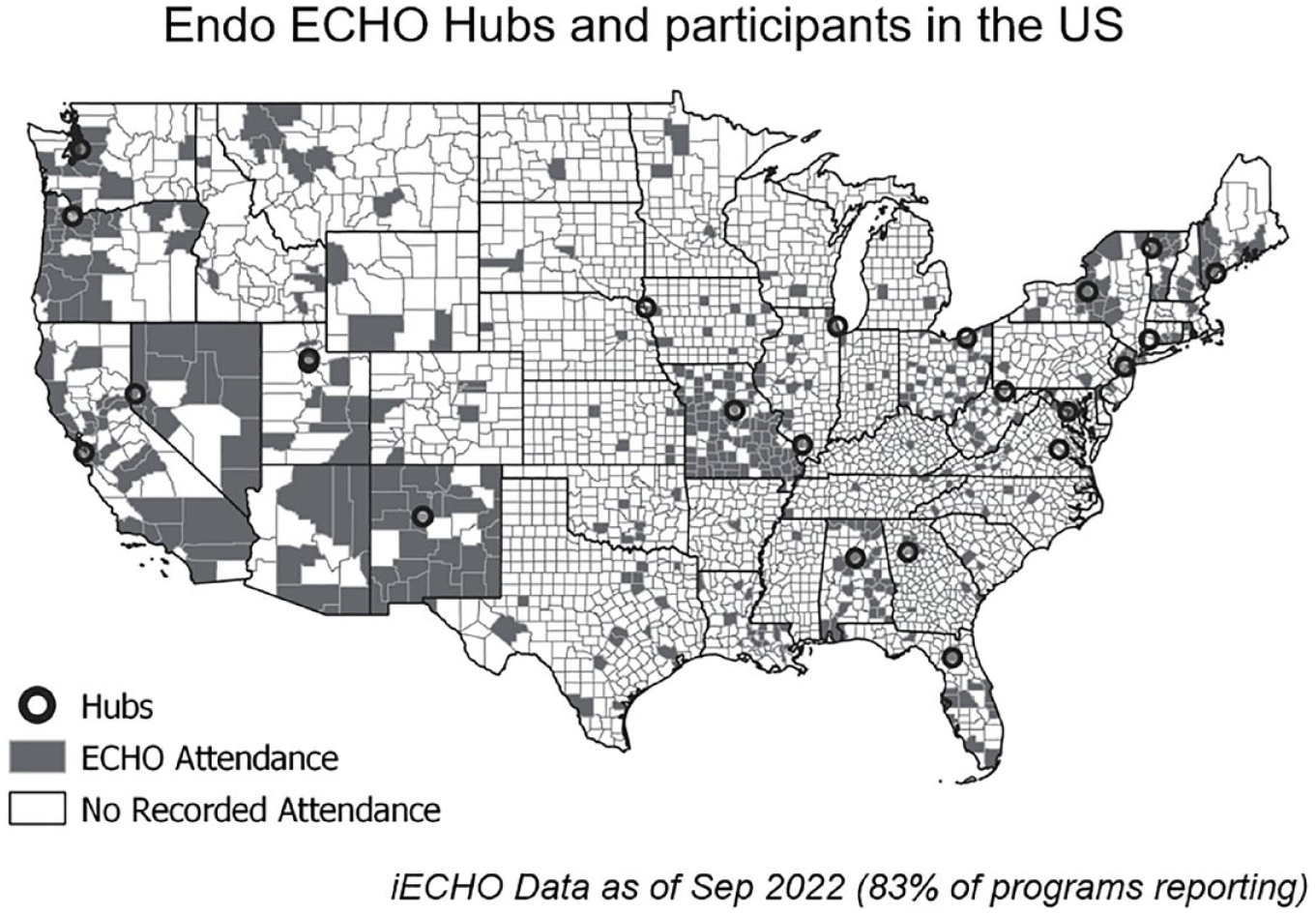
ECHO hubs and regions of diabetes ENDO-ECHO attendance.
ECHO Formats
Diabetes ECHOs implement an “all-teach, all-learn” format, involving not only specialists, but peer mentorship. Programs run continuously, or as a cohort-based model, for 3 to 24 months with session duration ranging 1 to 2 hours, occurring weekly to monthly. Organizations interested in starting a diabetes-focused ECHO are required to complete initial training through UNM or a regional training center (“superhub”). Key components of a diabetes ECHO include a brief evidence-based didactic presentation delivered by an expert and a de-identified case presentation submitted by a PCP. Clarifying questions and best practice recommendations are provided by community peers and hub specialists. Programs serve between 15 and 40 spoke sites, reaching a diverse array of healthcare professionals. Interprofessional hubs typically consist of endocrinologists, behavioral health and diabetes education specialists, pharmacists, and other diabetes support personnel, including integration of PWD with peer support. Participants are offered continuing education credits and, in some cases, maintenance of certification (MOC) as well. Although the model provides medical knowledge, it takes traditional continuing medical education further by supporting the implementation of that knowledge through the development of virtual communities of practice, multidirectional case-based learning and telementoring of participants.
Diabetes-related programming expanded from a regional to a national level to address unmet needs of diabetes disparities in primary care through the “ECHO Diabetes in the Time of COVID-19” series 33 that reached over 4000 live participants from 48 U.S. states and 122 countries. Additional large-scale ECHO webinar series with enduring on-demand content have launched including the University of Washington’s “Cardiometabolic ECHO Project” 34 and the ECHO Diabetes Action Network’s “Addressing Disparities in Diabetes with Project ECHO: A Focus on Diabetes-Related Chronic Kidney Disease (CKD).” 35
Curriculum
Diabetes-related curricula are available across programs through a shared database and modified to meet the needs of each community. Examples of topics for diabetes-related didactics include diabetes practice guidelines, multiple daily injections insulin management, diabetes and CKD, cardiometabolic disease, diabetes technology, beyond A1c targets, motivational interviewing, behavioral health and depression, social determinants of health, insulin affordability, and access to local resources. Specialized cohort-specific diabetes ECHOs have also been implemented for complex diabetes,36,37 type 1 diabetes (T1D), 38 CKD 35 and cardiometabolic disease. 34
While the didactic curriculum varies across hub sites, the key objective of using case presentations is to facilitate best practice sharing among participants. Consensus guiding principles of case presentations and curricula established by the diabetes ECHO learning collaborative include emphasizing the “ABCDES” (Table 1). Although the variability of programming accommodates the needs of the participants as well as the resources of the hubs, a limitation of the model is the heterogeneity of unique programs. There has been no “best practice” established for ECHO such as duration of program, frequency of sessions, length of sessions, core content, number of panelist/experts per session, or amount of participation needed.
The “ABCDES” of the Diabetes ECHO Model.

Abbreviation: ECHO, Extension for Community Healthcare Outcomes.
Diabetes ECHO Participation Growth
All ECHO hubs agree to participate in data sharing with the ECHO Institute at UNM using the iECHO platform. Data pertaining to spoke and hub attendance, total number of sessions, number of case presentations and lectures, and geographic reach of learners are compiled into iECHO. Compliance with the data sharing agreement and entry into iECHO was 83% for diabetes programs. The cumulative number of participants between 12/2009 and 9/2022 was 40,244, reaching 6435 unique learners. The average attendance per session was 22 community learners. In 2022, average learner attendance was 9 sessions. Figure 2 demonstrates the geographic distribution of diabetes or endocrinology ECHOs across the United States and the associated learning communities. Total attendance has seen a marked rise in participation beginning in 2015, coinciding with the launch of the New Mexico (NM) “ENDO ECHO” program in 2014 (Figure 3).
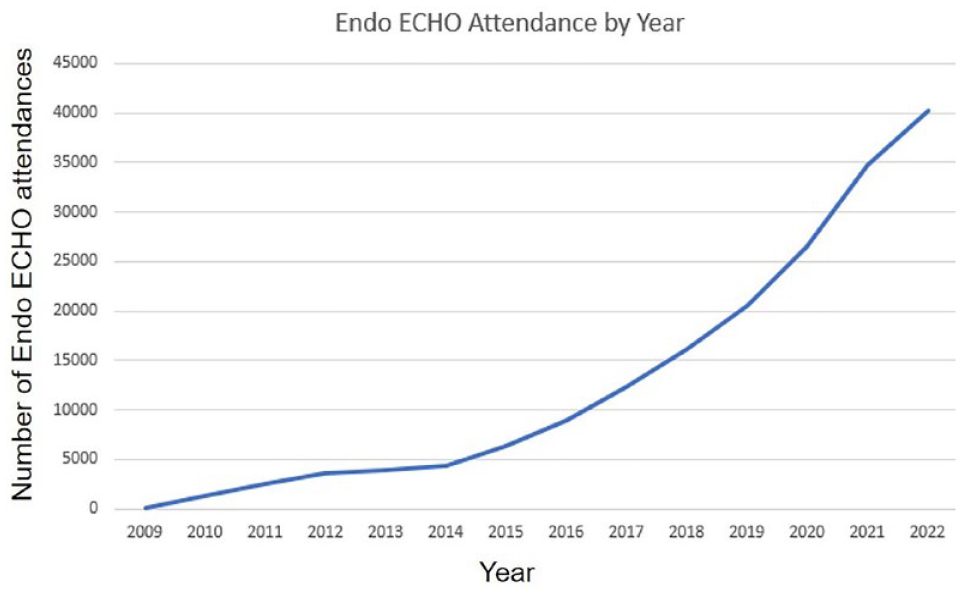
Number of participants in ENDO-ECHO since 2009 through September 2022.
Model to Address Urgent Need of Complex Diabetes in Primary Care
Diabetes-related ECHO programs offer an opportunity to address unmet needs of PWD, and in particular insulin-requiring diabetes. Focus groups (n = 18) in Florida and California were conducted to identify barriers to care among adults with T1D who do not routinely visit an endocrinologist. Barriers identified included cost of insulin, long wait times, distance to see specialists, lack of local support groups, and patient preference for PCP care for their T1D.38,39 These findings from the “ECHO T1D” program add to the growing evidence base of the practical need to support PCPs.
As noted, diabetes technology utilization has been lower in vulnerable T1D populations. Recent registry data collected in specialty and primary care demonstrate similar disparities in primary care40 -42 for technology utilization. These data were further corroborated and used to develop interventions in diabetes-related ECHOs in Florida and California, which showed limited access to an endocrinologist and significant differences in pump use by SES and race/ethnicity. In California, there were significant differences in continuous glucose monitoring (CGM) use by race/ethnicity and education level. 43 These data further highlight the need for equity given the underuse of technology. With regard to T1D insulin management, baseline findings from an ECHO T1D needs assessment highlighted that many PCPs prescribe and oversee insulin dose adjustments (61% in adult and 27% in pediatric populations) despite 58% of PCPs lacking confidence in intensive insulin management in T1D. 44
Improving Clinician Knowledge, Confidence, and Change in Perception of Prescribing Practices in Managing Complex Diabetes
There are several reports describing the impact of the ECHO model on improving knowledge and confidence in complex diabetes management among PCPs. Findings from NM demonstrated significant increases in self-efficacy around complex diabetes management in rural settings, increases in uptake of best practices, reduction in the need for referrals to specialists, and recognition of participating PCPs as local content experts. 45 Diabetes ECHOs in Washington, DC (DC) (George Washington University) 46 and Chicago (University of Chicago), 47 which targeted the urban population, similarly showed PCP increases in self-efficacy and knowledge. In DC, physician assistant students also demonstrated increased knowledge and confidence. 46 A collaborative led by Stanford University and University of Florida Diabetes Institute demonstrated proof of concept that the ECHO model can attract interest from PCPs in a 6-month weekly program for T1D. Pre- and post-surveys demonstrated participants had significant improvement in diabetes knowledge (P ≤ .01) and diabetes confidence (P ≤ .01), with diabetes technology being the most cited change in practice. 38
Three unique diabetes ECHOs representing 4 regions (Chicago, DC, NM, and Washington) evaluated comfort or perception of prescribing practice changes for community PCPs. Self-reported prescription use for newer diabetes medications with cardiovascular indications increased significantly, as did the perceived likelihood of prescribing intensive insulin therapy. Primary care professionals also felt they would be more likely to prescribe CGM technology, with no significant change in use of insulin pumps. 48
Improving Patient Outcomes
In rural NM, their ECHO reached a population with lower education and income levels than that of the academic medical center. 49 Medically underserved PWD who received care from an ECHO-trained clinician reported significant improvements in their perception of well-being, health-related behaviors, self-care, quality of care, confidence in self-management, and diabetes-related symptoms. 50 Care utilization measures were evaluated through Medicaid claims data and compared to propensity score matched individuals in the community not involved in the program. Patients receiving care from an ECHO-mentored clinician had significant increases in outpatient visits and several prescription-related quality indicators, including appropriate diabetes-related prescriptions. 51 Recent data from New Jersey’s Rutgers program showed cost saving in a Medicaid population (61.9% reduction in inpatient spending (P = .021) and 44.3% decrease in inpatient admissions (P = .001)). Significant changes were seen in those who attended 20 or more of the 64 sessions. 52 University of New Mexico’s program also recently showed a 1.2% initial improvement in A1c among spoke sites but no significant difference from controls (difference 0.2% at 3 months and 0.3% at 12 months). However, in long-term follow-up, PWD treated by ECHO-trained clinicians were more likely to achieve an A1c of less than 8% than community peers. 53 Practitioners in this program participated in more than 25 sessions, but attendance may be attributed to financial compensation for their time. Of note, this program also trained community health workers to provide support for PWD whose clinicians participate in their program, so results are reflective of this dual intervention. Diabetes ECHOs in California, Florida, and Washington, DC are currently also undergoing completion of patient outcome evaluation.
Addressing Quality Improvement Practices
Implementation of the ECHO model also serves as an opportunity to leverage quality improvement (QI) projects for community clinics and clinicians.54 -56 In California and Florida, a multi-site diabetes QI initiative leveraging the model demonstrated vast improvements across structural, process, and outcome measures. 57 Examples of measures demonstrating improvement included clinic having a protocol of identifying “high risk” patients and enrolling them in special care pathways, clinic having a workflow for scheduling “lost to follow-up patients,” number of CGM prescriptions submitted last quarter, number and/or percentage of patients whose most recent A1c was greater than 9%, mean A1c of patients of most recent A1c for the current measurement year for both commercial insurance and Medicaid. Programs have also offered MOC Part 4 (Quality Improvement) to participating spoke and hub team members.
ECHO Replication: The Opportunity
The ECHO model was developed to be a force multiplier by increasing specialty knowledge while improving care and addressing lack of access to specialty care and health disparities at the primary care level. Immersion training is offered through UNM and superhubs for organizations interested in replication in their own communities. 58 A learning collaborative of existing diabetes ECHOs also supports new hubs. This learning collaborative has also facilitated launch of dedicated collaborative programming around T1D (California, Florida) and Diabetes Disparities & CKD (Colorado, New Jersey, Washington State). Continued cross collaboration is needed as demonstrated by the prescribing practices cross-site data collection; multi-site program evaluation at patient, clinician and clinic levels is lacking but will demonstrate best practices and guide replication. In the meantime, diabetes specialists and specialty care centers are lagging behind other specialists and specialties in leveraging this model to explicitly address health disparities and lack of access to specialty care.
Funding opportunities for ECHO are becoming more common place. The 2016 Expanding Capacity for Health Outcomes Act, 59 led to the 2019 ECHO ACT, 60 which reported modest but positive effects from the model. 61 In 2020, the U.S. congress authorized a 5-year, $50 million ECHO grant program. The Oregon ECHO Network was one of the first organizations to leverage this funding for expansion of diabetes-related programming. 62
Expansion of diabetes ECHOs can be used to address the diabetes public health crisis, particularly in the United States where 37 million Americans, or 1 in 10 people, are living with diabetes 63 and where this burden is doubled at CHCs. 64 The model is being used for other chronic diseases to address health disparities and diabetes is lacking. For example, HIV, which affects approximately 1.2 million in the United States 65 has 26 U.S. programs 66 while the UNM ECHO Institute program tracking site only lists 23 diabetes programs. 66 Diabetes-related ECHOs are not growing proportionally to the need and prevalence of the disease. Increased awareness of this model, promising data on the benefit of diabetes-related ECHOs and available funding, highlight the need and opportunity for replication of this model.
Limitations and Further Needs for ECHO Evaluation
Despite increasing funds available for ECHO, and more outcomes on the overall impact of the model for diabetes and other diseases, data are still limited. Additionally, one of the largest barriers to replications is that what influences the model’s effectiveness remains unclear. Standardization for best practices, including in diabetes, remains to be determined; there is ongoing evaluation to synthesize what influences the development of competencies in healthcare professionals, 67 which should help direct future programs. While there are advantages to ECHOs tailoring their approach to the needs of the community, this does introduce heterogeneity across programs in terms of length and frequency of sessions, open versus closed participation, continuous versus cohort-based, and specific curricular content. Additional studies are needed to determine the optimal “dosage” of programming in relation to outcomes. Finally, although this model is believed to help address health disparities due to many participating spokes being FQHCs caring for underserved populations, how and how much it helps address health disparities is another important avenue to explore.
Conclusions
Application of the ECHO model in diabetes is a promising approach to overcome health disparities and limited access to specialists. It empowers PCPs to combat therapeutic inertia among PWD and expand access to diabetes technology. Evidence suggests the model increases participant satisfaction and confidence in managing complex diabetes and that interest in this continuing education model is growing. There are also reports of cost savings and improved clinical outcomes, but more outcomes data and multi-site collaboration are needed. The model can be used to leverage diabetes QI initiatives in clinics and explicitly address diabetes disparities. ECHO is an example of a model that can help address the urgent need of the growing burden of diabetes in primary care and especially among vulnerable populations.
Footnotes
Acknowledgements
None.
Abbreviations
CGM, continuous glucose monitoring; CHCs, community health centers; CKD, chronic kidney disease; COVID, coronavirus disease 2019; CVD, cardiovascular disease; DC, Washington, DC; ECHO, extension for community healthcare outcomes; ENDO, endocrine; GLP-1 RAs, glucagon-like peptide-1 receptor agonists; HbA1c, hemoglobin A1c; NM, New Mexico; PCPs, primary care professionals; PWD, people with diabetes; QI, quality improvement; SES, socioeconomic status; SGLT2-Is, sodium glucose co-transporter-2 inhibitors; T1D, type 1 diabetes; UNM, University of New Mexico; US, United States.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Ehrhardt has been on an advisory board for Dexcom and Novo Nordisk and received investigator-initiated grants from Dexcom and educational grants from Merck and Novo Nordisk.
Dr. Weinstock has participated in multicenter clinical trials, through my institution, sponsored by Insulet, Medtronic, Eli Lilly, Novo Nordisk, Kowa and Boehringer Ingelheim, and have used devices donated by Tandem (insulin pumps) and DexCom sensors in clinical research projects.
Dr. Gabbay is on the advisory board of Lark, Vida, and Sweetch.
Dr Cuttriss does consult for The Leona M. and Harry B. Helmsley Charitable Trust and Cecelia Health.
Dr. Desimone, Dr. Thomas, Dr. Peek, Dr. Bouchonville, Linda Baer and Tracy Zou have no disclosures.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: MERCK foundation Bridging the Gap: Reducing Disparities in Diabetes Care.
Norvo Nordisk and Merck Educational Grant.
Leona M. and Harry B. Helmsley Charitable Trust.
NIDDK P30 grant: NIDDK 2P30 DK092949.
