Abstract
Background:
The Wireless Innovation for Seniors with Diabetes Mellitus (WISDM) study demonstrated continuous glucose monitoring (CGM) reduced hypoglycemia over 6 months among older adults with type 1 diabetes (T1D) compared with blood glucose monitoring (BGM). We explored heterogeneous treatment effects of CGM on hypoglycemia by formulating a data-driven decision rule that selects an intervention (ie, CGM vs BGM) to minimize percentage of time <70 mg/dL for each individual WISDM participant.
Method:
The precision medicine analyses used data from participants with complete data (n = 194 older adults, including those who received CGM [n = 100] and BGM [n = 94] in the trial). Policy tree and decision list algorithms were fit with 14 baseline demographic, clinical, and laboratory measures. The primary outcome was CGM-measured percentage of time spent in hypoglycemic range (<70 mg/dL), and the decision rule assigned participants to a subgroup reflecting the treatment estimated to minimize this outcome across all follow-up visits.
Results:
The optimal decision rule was found to be a decision list with 3 steps. The first step moved WISDM participants with baseline time-below range >1.35% and no detectable C-peptide levels to the CGM subgroup (n = 139), and the second step moved WISDM participants with a baseline time-below range of >6.45% to the CGM subgroup (n = 18). The remaining participants (n = 37) were left in the BGM subgroup. Compared with the BGM subgroup (n = 37; 19%), the group for whom CGM minimized hypoglycemia (n = 157; 81%) had more baseline hypoglycemia, a lower proportion of detectable C-peptide, higher glycemic variability, longer disease duration, and higher proportion of insulin pump use.
Conclusions:
The decision rule underscores the benefits of CGM for older adults to reduce hypoglycemia. Diagnostic CGM and laboratory markers may inform decision-making surrounding therapeutic CGM and identify older adults for whom CGM may be a critical intervention to reduce hypoglycemia.
Keywords
Introduction
The Wireless Innovation for Seniors with Diabetes Mellitus (WISDM) study evaluated the effect of continuous glucose monitoring (CGM) on hypoglycemia (glucose <70 mg/dL) over 6 months, compared with standard BGM, among older adults ages ≥60 years with T1D. 1 On average, use of CGM reduced hypoglycemia from approximately 73 minutes per day at the trial baseline to 39 minutes per day over the 6-month trial period, with a concurrent reduction in hemoglobin A1c (HbA1c). There were no significant changes in the BGM group. 1 Based on the WISDM trial and other studies,2 -4 contemporary clinical guidelines state that CGM should be considered for glucose monitoring for older adults with T1D, 5 and specifically to reduce hypoglycemia.
Though there are likely benefits of CGM for all older adults with T1D,3,5 it is known that estimates of average treatment effects of the primary outcome in a trial setting can mask the effects that individual participants may experience. Pre-specified subgroup analyses in the WISDM study showed the treatment effect of CGM, in terms of reduction in hypoglycemia, was greater among participants with increased baseline hypoglycemia and glycemic variability as measured by the coefficient of variation (%CV), and lower detectable C-peptide levels. 1
Yet, conventional subgroup analyses are limited in discovering heterogeneous treatment effects as they require moderating markers to be decided a priori. 6 Given that older adults are extremely diverse in terms of biopsychosocial profiles, clinical needs, historical diabetes self-management experiences, and preferences for care,7,8 an entirely a priori approach may not capture patient markers that were not pre-specified as previously recognized as potential moderating markers of a treatment response.
An alternative approach is to use rigorous machine learning methods from the field of precision medicine that estimate a “decision rule,” a mathematical function that maps patient-level information (demographic characteristics, clinical biomarkers, other measures) to a recommended treatment or intervention to optimize (ie, maximize or minimize) an outcome of interest. 9 Decision rules offer an entirely data-driven approach to discovery of moderating markers, i.e., the patient-level variables that are predictive of the within-individual difference in effect of 2 or more treatments, 9 and link therapies to subgroups of patients likely to show favorable responses.9,10
In this post hoc analysis, our objective was to use a data-driven approach to explore the moderating markers (such as baseline CGM glucose management indices and participant characteristics) that are associated with heterogenous effects of CGM on hypoglycemia in older adults with T1D. The significance of this work is to inform future hypotheses by uncovering relationships between moderating markers and differential effects of CGM on hypoglycemia, and to support shared decision-making surrounding the utility of CGM for individual older adults living with T1D.
Materials and Methods
Design, Setting, and Participants
The WISDM study (ClinicalTrials.gov Identifier: NCT03240432) enrolled 203 older adults with T1D at 22 sites in the United States. Older adults (age ≥60 years) were eligible if they used an insulin pump or multiple daily injections, but they could not have used unblinded CGM as part of T1D management in the past 3 months. Participants were randomized to CGM (n = 103) or BGM (n = 100) for 6 months to study whether CGM could reduce hypoglycemia (% time with glucose <70 mg/dL). 1 The cohort has been extensively characterized, with both treatment groups having balanced baseline characteristics1,11 as per Table 1 and eTable 16 of Pratley et al. 1 Seven patients were lost to follow-up, requested withdrawal from the study, or discontinued the intervention, and 2 patients were missing education data, resulting in 194 older adults for this analysis (CGM [n = 100]; BGM [n = 94]).
Characteristics of Study Participants, Stratified by Decision Rule Subgroup.
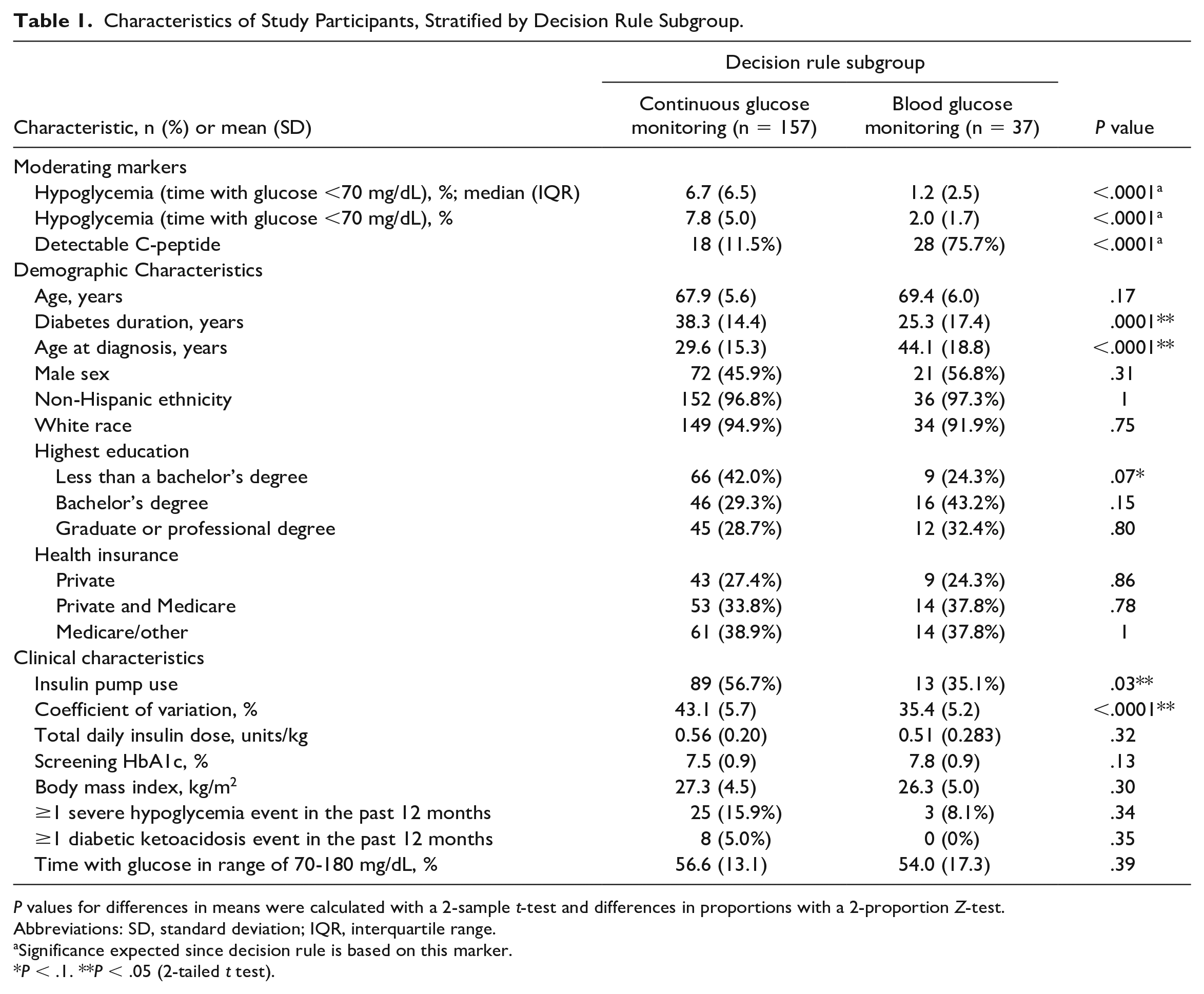
P values for differences in means were calculated with a 2-sample t-test and differences in proportions with a 2-proportion Z-test.
Abbreviations: SD, standard deviation; IQR, interquartile range.
Significance expected since decision rule is based on this marker.
P < .1. **P < .05 (2-tailed t test).
Measures
The primary outcome was CGM-measured percentage of time spent in hypoglycemic range (<70 mg/dL) at follow-up, 1 which used pooled data from approximately 2 weeks prior to randomization, and 1 week prior to the 8-, 16-, and 26-week visits, during which time participants in the BGM arm wore blinded CGM. 1 For the WISDM participants in this analysis, the average baseline time with CGM readings was 16.3 days (fifth percentile of 12.8 days and 95th percentile of 23.8 days) with an average of 21.9 hours of CGM data per day (fifth percentile of 17.0 h and 95th percentile of 23.7 h). The average follow-up time with CGM readings was 6.9 days (fifth percentile of 5.8 days and 95th percentile of 7.0 days) with an average of 22.5 hours of CGM readings per day (fifth percentile of 17.8 h and 95th percentile of 24.0 h). For our decision rule, we specified a minimization of percentage of time in hypoglycemia as the optimal outcome for each participant. As a secondary analysis, we repeated the analysis using change in CGM-measured percentage of time spent in hypoglycemic range (<70 mg/dL) between baseline and follow-up. 1 Here, a decrease in the percentage of time in hypoglycemia was defined as the optimal outcome.
In estimating the optimal decision rule, we considered the following baseline variables, which were collected from medical records and confirmed by participants 1 : age, diabetes duration, age at diagnosis, sex, highest education, health insurance status, insulin pump use, screening HbA1c, detectable C-peptide levels, history of severe hypoglycemia events, and diabetic ketoacidosis events in the past 12 months. We used 3 variables measured during the baseline period of blinded CGM wear: percentage of time in hypoglycemia (glucose under 70 mg/dL), percentage of time in range (TIR; glucose in range of 70-180 mg/dL), and glycemic variability (defined as the %CV).
Highest education was categorized by “Bachelor’s Degree,” “Less than a Bachelor’s Degree,” “More than a Bachelor’s degree.” Health insurance was split into “Private,” “Private and Medicare,” and “Medicare/other.” C-peptide values were dichotomized into non-detectable (<0.003 nmol/L) and detectable (≥0.003 nmol/L). Severe hypoglycemia and diabetic ketoacidosis were dichotomized to reflect whether an event occurred or not. For characterizing the resulting subgroups produced by the decision rule, we additionally compared the subgroups based on race and ethnicity, total daily insulin dose per kg, and body mass index.
Statistical Analysis
We estimated and evaluated a series of potential decision rules that estimate an optimal glucose monitoring modality for each WISDM study participant (CGM vs BGM) to minimize total time in hypoglycemia. As above, we repeated our approach as part of a secondary analysis to maximize the reduction (ie, negative change) in time spent in hypoglycemia from baseline. The decision rules were based on the 14 demographic, clinical, and laboratory measures specified above. We selected statistical approaches for precision medicine that estimate simple, interpretable treatment decision rules which can be applied clinically. 12 We estimated optimal decision rules by implementing policy trees 13 and decision lists. 14 The decision list algorithm results in decision rules in the form of a sequence of “if-then” statements, while the policy tree algorithm results in a decision tree. For both algorithms, the “depth” parameter is synonymous with the number of steps involved in using a decision rule to identify an optimal intervention: For a decision lists, depth is the number of “if-then” statements, and for a policy tree, depth is the number of layers of the tree. While the decision rule provides estimation of optimal therapies, it is not meant to provide deterministic treatment “assignments” or clinical recommendations for actual patients, but rather, to explore subgroups and the markers that define the subgroups, as treatment effects may vary based on their values. We refer to the subgroups as the CGM versus BGM treatment rule subgroups.
Decision Rule Evaluation
We evaluated the clinical benefit of potential decision rules using the value function, a scalar performance measure which estimates the expected outcome in the study population if every participant was to receive their optimal intervention as estimated by the rule. For the “total time in hypoglycemia” outcome, a lower value suggests better performance of the rule, as it is indicative of less time spent in hypoglycemia. For the “reduction in time spent in hypoglycemia” outcome, a higher value suggests better performance, as it indicates a larger decrease in percentage of time in hypoglycemia over the duration of the trial. Estimation of the value of the resultant decision rules was carried out over a nested 5 × 5 cross-validation scheme. An initial outer loop was used to split the data into 5 training and test folds, and within the training set, an inner 5-fold cross-validation loop was used for algorithm selection and tuning of the depth parameter. Once the optimal model (algorithm and depth) was selected using the inner loop, for evaluation, its value was estimated on the held-out data using 5-fold cross-validation in the outer loop. For comparison, we estimated the value function for a “CGM-only” rule (ie, all participants receive CGM) through 5-fold cross-validation in the outer loop. As context for the value of the optimal decision rule and the CGM-only rule, we also estimated the value of a “BGM-only” rule.
Subgroup Characterization
Using descriptive statistics, we compared the characteristics of the participants in the CGM versus BGM treatment rule subgroups, as per the optimal decision rule for the primary outcome. P values for differences in means were calculated with a 2-sample t test and differences in proportions with a 2-proportion Z test at the 0.05 and 0.10 significance levels. All analyses were conducted in R, version 3.6. 15
Data Availability and Resource Sharing
The WISDM data are publicly available. All codes to replicate the analyses described herein are available at https://github.com/kushshah1/WISDM-Precision-Medicine.
Results
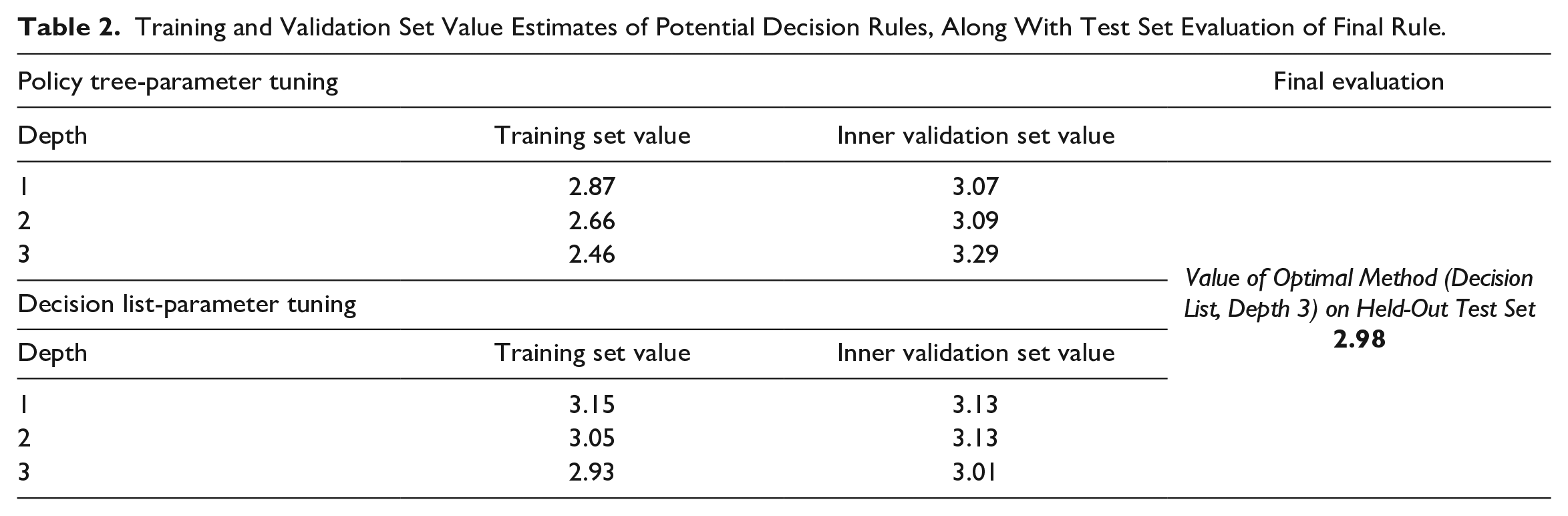
The optimal decision rule was found to be a decision list with a depth of 3 (Table 2). The decision rule is depicted in Figure 1. The first step of the decision rule moved WISDM participants with baseline time-below range >1.35% and no detectable C-Peptide to the CGM subgroup (n = 139), and the second step moved WISDM participants with baseline time-below range of >6.45% to the CGM subgroup (n = 18). The remaining participants (n = 37) were left in the BGM subgroup.
Training and Validation Set Value Estimates of Potential Decision Rules, Along With Test Set Evaluation of Final Rule.
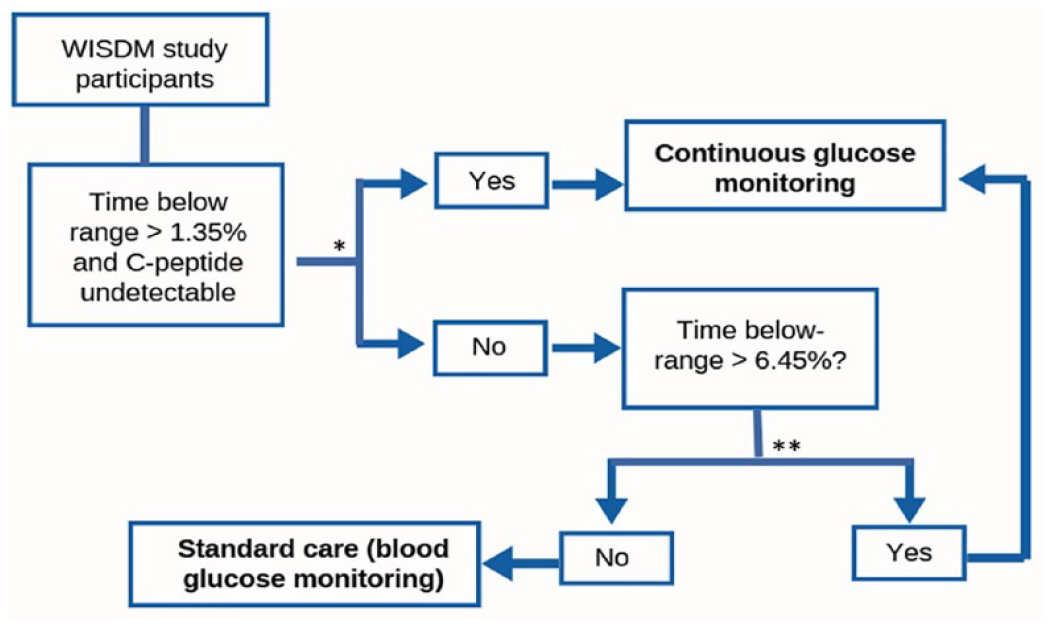
Visualization of the decision rule. Abbreviations: CGM, continuous glucose monitoring; WISDM, wireless innovation for seniors with diabetes mellitus; BGM, blood glucose monitoring.
The characteristics of the 157 (81%) WISDM participants in the CGM versus the 37 (19%) WISDM participants in the BGM subgroup are shown in Table 1. The median baseline time spent in hypoglycemia in the CGM subgroup was 6.7% (interquartile range [IQR]: 6.5%) compared with 1.2% (IQR: 2.5%) in the BGM subgroup. The mean proportion of patients with detectable C-peptide levels in the CGM subgroup was 11.5% compared with 75.7% in the BGM subgroup. Compared with the BGM subgroup, the CGM subgroup also had, on average, a longer diabetes duration (38.3 years vs 25.3 years; P = .0001) and younger age at diagnosis (29.6 years vs 44.1 years; P < .0001), in addition to a higher proportion with insulin pump use (56.7% vs 35.1%; P = .03) and larger mean %CV (43.1% vs 35.4%; P < .0001). There were no significant differences in age, sex, or other demographic factors, nor total daily insulin dose, HbA1c, history of hypoglycemia in the preceding 6 months, or history of diabetic ketoacidosis (DKA) in the preceding 6 months.
The optimal decision rule was estimated to result in a total time in hypoglycemia of 2.98% (standard error of the mean [SEM]: 0.32%) across the full study population (Table 2), compared with 3.09% (SEM: 0.24%) with the “CGM-only” rule. For context, use of BGM-only was estimated to correspond with a total time in hypoglycemia of 6.23% (SEM: 0.44%).
The results of the secondary analyses, which focused on the secondary outcome, change in hypoglycemia, are shown in Appendix A. For this outcome, the optimal decision rule was found to be a policy tree algorithm with a depth of one, and the rule suggests that study participants with baseline %CV >34% would experience a greater reduction in hypoglycemia using CGM compared with BGM.
Discussion
We used data from the WISDM study and machine learning methods to discover how patient characteristics are associated with the estimated treatment effects of CGM on hypoglycemia among older adults with T1D. Our results suggest that there are 2 distinct subgroups of the WISDM participants—those with baseline time-below range >1.35% and no detectable C-peptide levels, and those with baseline time-below range of >6.45%—for whom CGM was estimated to result in lower hypoglycemia at the follow-up period compared with BGM. Together, these subgroups represented the majority of the study sample, with distinguishing features including a lower proportion of detectable C-peptide, higher glycemic variability, longer disease duration, and higher proportion of insulin pump use. As such, we elucidate the moderating factors to identify for whom treatment effect is expected to be greater or smaller, and importantly, markers for the subgroups of older adults for whom CGM may be a particularly effective intervention for reducing hypoglycemia.
The benefits of CGM for older adults are both significant and well-established, and CGM now represents standard-of-care for T1D.1,2,5,8,16 -19 This is reflected in our results as well, where the majority of WISDM participants were estimated to benefit from CGM over BGM to minimize time in hypoglycemia. It is also important to note the small difference in mean hypoglycemia reduction with the optimal treatment rule versus CGM-only rule (~2.98% vs 3.09%), which underscores the value of CGM across the population. The decision rule herein also only focused on hypoglycemia, and thus does not focus on other glycemic or patient-oriented benefits associated with CGM. Above all, estimating a decision rule does not provide rationale for restricting an evidence-based therapy; rather, it offers insight into how the same therapy may results in different outcomes for individuals, as well as the subgroups for whom benefits in terms of one outcome may be more versus less pronounced, as well as the physiology and clinical histories that may underlie those differences.
Furthermore, despite the benefits and value of CGM for older adults, there remains a proportion of patients who do not use therapeutic CGM as part of their T1D self-management.4,20,21 The decision to initiate CGM may be shaped by baseline glycemic management and priorities, 4 perceived benefits and burdens of the technology, 20 or the impact of age-specific challenges to integrating technology into care. 22 As adults get older, managing the device and data can be perceived as challenging, and preferences surrounding technology use may evolve. 18 Understanding heterogenous treatment effects and markers thereof may aid providers in identifying patients for whom CGM technology is critical to mitigate the risk of hypoglycemia. Providing more granular, individualized estimation of the value of CGM may help providers to find ways to balance clinical outcomes and individual preferences through shared decision-making.
The decision rule suggests that 2 groups of study participants would experience a lower proportion of time spent in hypoglycemia by using CGM compared with BGM: (1) those with baseline hypoglycemia >1.35% and with no detectable C-peptide, and (2) those with baseline hypoglycemia >6.45%. The decision rule thus underscores an important link between baseline hypoglycemia and risk for future events, which may be mitigated by CGM use. It is worth noting that in a clinical setting, any duration of hypoglycemia greater than 1% may indicate a need for CGM for the reduction in non-severe hypoglycemia and prevention of severe episodes in the future, 23 and 2023 Standards of Care in Diabetes recommend CGM to reduct hypoglycemia among older adults with T1D. 5 An inverse association between age at diagnosis and pump use has been described previously, 24 and one possible explanation between the higher proportion of insulin pump use in the subgroup may reflect existing efforts to mitigate the burden of diabetes management, though this association cannot be considered causal.
Together, this subgroup of patients was also distinguished by a longer disease duration and higher glycemic variability at baseline, which may reflect, together, a phenotype of very long-standing T1D. An association between glycemic variability and severe hypoglycemia has been reported in older adults previously.25,26 A high CV (>36%) has also been associated with duration of time spent in non-severe hypoglycemia among older adults with T1D. 3 Interestingly, the a-priori subgroup analysis of the WISDM study showed that among participants in the CGM arm, a higher baseline %CV was associated with a greater treatment effect. 1 The results of the decision rule analysis are related, but not redundant, with that finding, as the subgroups reported herein are generated from the algorithm comparing predicted effects between CGM and BGM, for each individual within the data set, to assign them to an optimal therapy group. Our finding that demographic features, markers of socioeconomic position, and HbA1c levels were not significantly different across subgroups is also consistent with the main trial analyses, which did not find other baseline characteristics, including age (<70 vs ≥70 years), socioeconomic status, presence of cognitive impairment, or HbA1c value to interact with the treatment effect (on hypoglycemia). 1
Our secondary analysis showed that the moderating factors may shift based on the outcome the decision rule aims to optimize, where baseline glycemic variability greater than 34% was a marker in determining patients for whom CGM was estimated to produce the greatest reduction in hypoglycemia. A point of interest is that the %CV threshold from the entirely data-driven decision rule aligns closely with existing cutoffs for glycemic variability to minimize risk of hypoglycemia, 27 where a %CV of <34% has been specified as an optimal threshold to reduce such episodes27 -29 and a %CV target of <33% for “additional protection against hypoglycemia” in insulin-treated diabetes. 30 An association between glycemic variability and severe hypoglycemia has been reported in older adults previously.25,26
Future studies can confirm the utility of hypoglycemia and C-peptide as a marker for heterogenous effects of CGM on hypoglycemia in older adults. A potential implication of the decision rule is a role for laboratory values (ie, C-peptide) and diagnostic CGM to inform decision-making surrounding therapeutic CGM, particularly for older adults who do not wish to permanently integrate new technology into their T1D self-management routines. Future studies may also investigate decision rules for CGM to optimize other glycemic outcomes, such as TIR. To understand the degree to which moderating markers generalize across age, it may be important to evaluate markers of CGM effects on hypoglycemia in different age groups, including published clinical trials in adolescents and young adults, such as the CGM Intervention in Teens and Young Adults with T1D (CITY) trial.
One of the most significant limitations of the study relates to the fact that the WISDM study participants do not reflect the same level of heterogeneity of the larger population of older adults with T1D. Not only is the sample a “young” older adult population, including individuals between 60 and 65 years of age, but this study population is also reflective of robust, highly-educated, and majority non-Hispanic White older adult patients. The value of the precision medicine-based statistical methods applied herein may be greater in the setting of a wider population, including variability in cognitive and physical function, living status, and diabetes self-management.
Other limitations of the study include its exploratory nature, small sample size, and the focus on hypoglycemia only. The WISDM study demonstrated treatment effects for hyperglycemia (glucose levels >180 mg/dL, >250 mg/dL, and >300 mg/dL), mean glucose concentration, and glycemic variability 1 ; each of these outcomes comprise important clinical benefits for patients. Several potential important variables were lacking for the analysis; data on cognitive status were not included, in addition to physical function, other behavioral or psychosocial measures such as impaired awareness of hypoglycemia, or living situation. Their inclusion may change the decision rule. 31
The strengths of this analysis include the application of novel machine learning methods from the field of statistical precision medicine. These methods are entirely data-driven and thus permit the discovery of subgroups of interest with characteristics/features or a combination thereof that were not defined a priori. Though benefits of CGM for older adults are both significant and well-established, our results may serve as the basis for future studies to explore heterogenous treatment effects and their markers across a range of outcomes, in more diverse patient populations, and considering variables with known relevant to the clinical care of older adults, such as frailty status, cognitive impairment, and living situation. In the meantime, the results may also aid providers in identifying patients for whom technology is critical to mitigate the risk of hypoglycemia and support enhanced shared decision-making surrounding the individualized benefits of CGM for individual older T1D adults.
Supplemental Material
sj-docx-1-dst-10.1177_19322968221149040 – Supplemental material for Estimation of a Machine Learning-Based Decision Rule to Reduce Hypoglycemia Among Older Adults With Type 1 Diabetes: A Post Hoc Analysis of Continuous Glucose Monitoring in the WISDM Study
Supplemental material, sj-docx-1-dst-10.1177_19322968221149040 for Estimation of a Machine Learning-Based Decision Rule to Reduce Hypoglycemia Among Older Adults With Type 1 Diabetes: A Post Hoc Analysis of Continuous Glucose Monitoring in the WISDM Study by Anna R. Kahkoska, Kushal S. Shah, Michael R. Kosorok, Kellee M. Miller, Michael Rickels, Ruth S. Weinstock, Laura A. Young and Richard E. Pratley in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors are grateful to Lauren Kanapka, MSc, for her help in acquiring the WISDM data and guiding analyses.
Abbreviations
%CV, coefficient of variation; BGM, blood glucose monitoring; CGM, continuous glucose monitoring; T1D, type 1 diabetes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RSW participated in multicenter clinical trials through her institution, sponsored by Insulet, Medtronic, Eli Lilly, Novo Nordisk, and Boehringer Ingelheim, and has used donated DexCom CGMs in projects sponsored by NIH and the Leona M. and Harry B. Helmsley Charitable Trust. KMM has used donated DexCom CGMs in projects sponsored by NIH and the Leona M. and Harry B. Helmsley Charitable Trust. REP reports consulting fees from Bayer AG, Corcept Therapeutics Incorporated, DexCom, Hanmi Pharmaceutical Co., Merck, Novo Nordisk, Pfizer, Sanofi, Scohia Pharma Inc., and Sun Pharmaceutical Industries, and grants/research support from Hanmi Pharmaceutical Co., Janssen, Metavention, Novo Nordisk, Poxel SA, and Sanofi. All funds are paid directly to Dr Pratley’s employer, AdventHealth, a nonprofit organization that supports education and research.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: ARK is supported by the National Center for Advancing Translational Sciences, National Institutes of Health (grant no. KL2TR002490). The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH. The post hoc study was supported by a grant from the Diabetes Research Connection (PI Kahkoska). The WISDM study was supported by the Juvenile Diabetes Research Foundation (grant no. 3-SRA-2017-425-M-R) and the Leona M. and Harry B. Helmsley Charitable Trust; DexCom provided the CGM devices and sensors.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
