Abstract
Background
CamAPS FX is a hybrid closed-loop smartphone app used to manage type one diabetes. The closed-loop algorithm has a default target glucose of 5.8 mmol/L (104.5 mg/dL), but users can select personal glucose targets (adjustable between 4.4 mmol/L and 11.0 mmol/L [79 mg/dL and 198 mg/dL, respectively]).
Method
In this post-hoc analysis, we evaluated the impact of personal glucose targets on glycemic control using data from participants in five randomized controlled trials.
Results
Personal glucose targets were widely used, with 20.3% of all days in the data set having a target outside the default target bin (5.5-6.0 mmol/L [99-108 mg/dL]). Personal glucose targets >6.5 mmol/L (117 mg/dL) were associated with significantly less time in target range (3.9-10.0 mmol/L [70-180 mg/dL]; 6.5-7.0 mmol/L [117-126 mg/dL]: mean difference = −3.2 percentage points [95% CI: −5.3 to −1.2; P < .001]; 7.0-7.5 mmol/L [126-135 mg/dL]: −10.8 percentage points [95% CI: −14.1 to −7.6; P < .001]). Personal targets >6.5 mmol/L (117 mg/dL) were associated with significantly lower time (<3.9 mmol/L [<70 mg/dL]; 6.5-7.0 mmol/L [117-126 mg/dL]: −1.85 percentage points [95% CI: −2.37 to −1.34; P < .001]; 7.0-7.5 mmol/L [126-135 mg/dL]: −2.68 percentage points [95% CI: −3.49 to −1.86; P < .001]).
Conclusions
Discrete study populations showed differences in glucose control when applying similar personal targets.
Introduction
CamAPS FX (CamDiab, Cambridge, UK) is an interoperable hybrid closed-loop smartphone app used to manage type one diabetes (T1D). The app receives sensor glucose data from the Dexcom G6 CGM system (Dexcom, San Diego, CA, USA), and the algorithm adjusts insulin administration via a compatible insulin pump (Dana RS or Dana i; Sooil, Seoul, South Korea, or Ypsopump; Ypsomed, Burgdorf, Switzerland). The closed-loop algorithm has a default target glucose level of 5.8 mmol/L (104.5 mg/dL), but users can select personal glucose targets, adjustable between 4.4 mmol/L (79 mg/dL) and 11.0 mmol/L (198 mg/dL) in 0.1 mmol/L (1.8 mg/dL) increments, which can be set differently for each half-hour block of the 24-hour period.
In this post-hoc analysis, we aimed to evaluate the impact of user-selected personal glucose targets on glycemic control.
Methods
We analyzed data from participants with T1D using CamAPS FX in five randomized controlled trials across different demographic cohorts: very young children aged one to seven years, 1 children and adolescents aged six to 19 years, 2 adolescents aged 10 to 17 years using closed-loop from diagnosis, 3 adults aged ≥18 years, 4 and older adults aged ≥60 years. 5 All studies received regulatory and ethical approval; participants/guardians signed informed consent. Selection of personal targets was determined by the user and could be adjusted throughout the study to suit the needs of the user.
We included 18 484 days of data from 185 participants for whom the closed-loop system was enabled for at least 70% of the 24-hour period. Data of each day were binned into 0.5 mmol/L (9.0 mg/dL) bins (4.0-11.0 mmol/L [72-198 mg/dL]) according to the average of the personal glucose targets applied that day. Only bins with a minimum of 14 days of data were included in the analysis. Data from each target bin were compared using analysis of variance with the post-hoc Tukey test for pairwise comparisons with the default target bin (5.5-6.0 mmol/L [99-108 mg/dL]). Statistical analyses were performed using SPSS (Version 27.0; IBM Corp., Armonk, NY, USA). P values <.05 were considered statistically significant.
Results
Personal glucose targets were widely used, with 20.3% of all days in the data set having a personal target outside the default target bin (5.5-6.0 mmol/L [99-108 mg/dL]). Over 95% of customized personal targets were set above the default target. Personal targets were used most frequently for very young children (>25% of days) and then older adults (>20% of days), while older children and adolescents used this functionality the least (<10% of days).
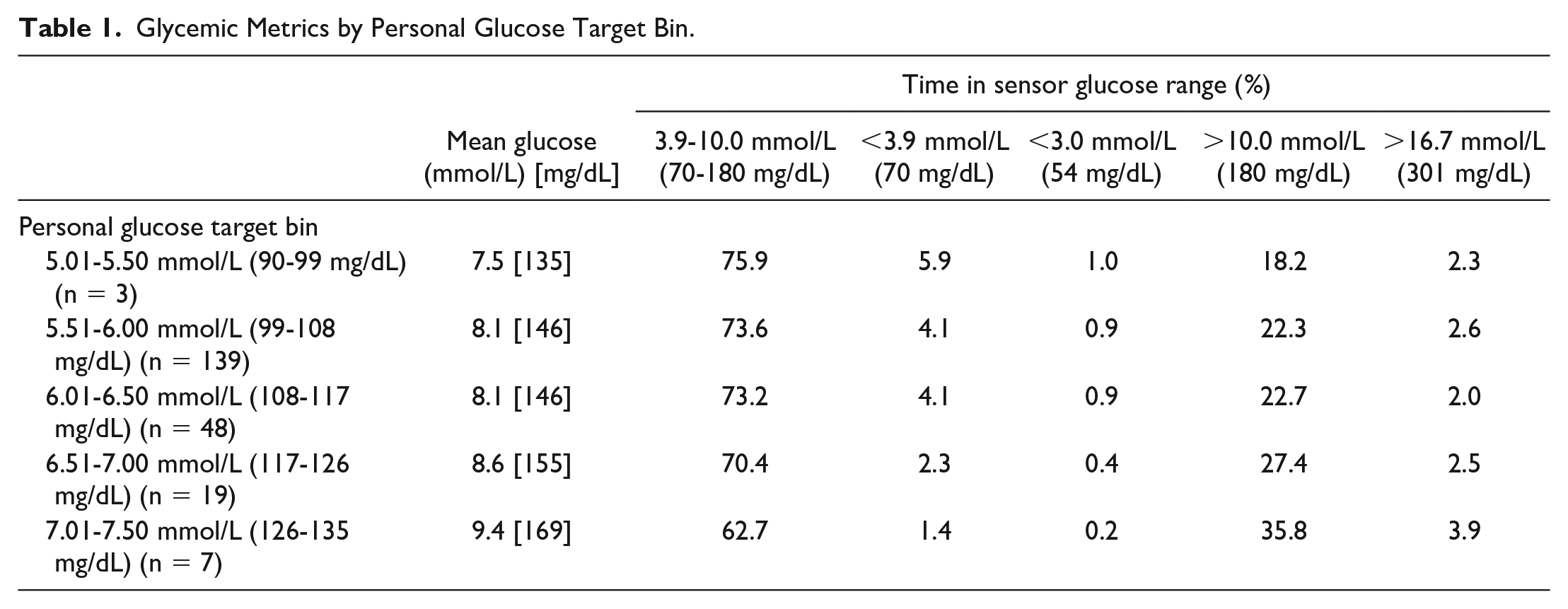
The mean glucose level increased significantly with a higher personal target (6.5-7.0 mmol/L [117-126 mg/dL] bin: mean difference 0.59 mmol/L [10.6 mg/dL; 95% CI: 0.41-0.77 mmol/L, 7-14 mg/dL; P < .001]; 7.0-7.5 mmol/L [126-135 mg/dL] bin: 1.36 mmol/L [24.5 mg/dL; 95% CI: 1.08-1.65 mmol/L, 19-30 mg/dL; P < .001]) and was significantly lower with a lower personal target (5.0-5.5 mmol/L [90-99 mg/dL] bin: mean difference −0.57 mmol/L [10.3 mg/dL; −1.00 to −0.14 mmol/L, −18 to −3 mg/dL; P < .01]) than when the default target was applied (Table 1 and Figure 1).
Glycemic Metrics by Personal Glucose Target Bin.
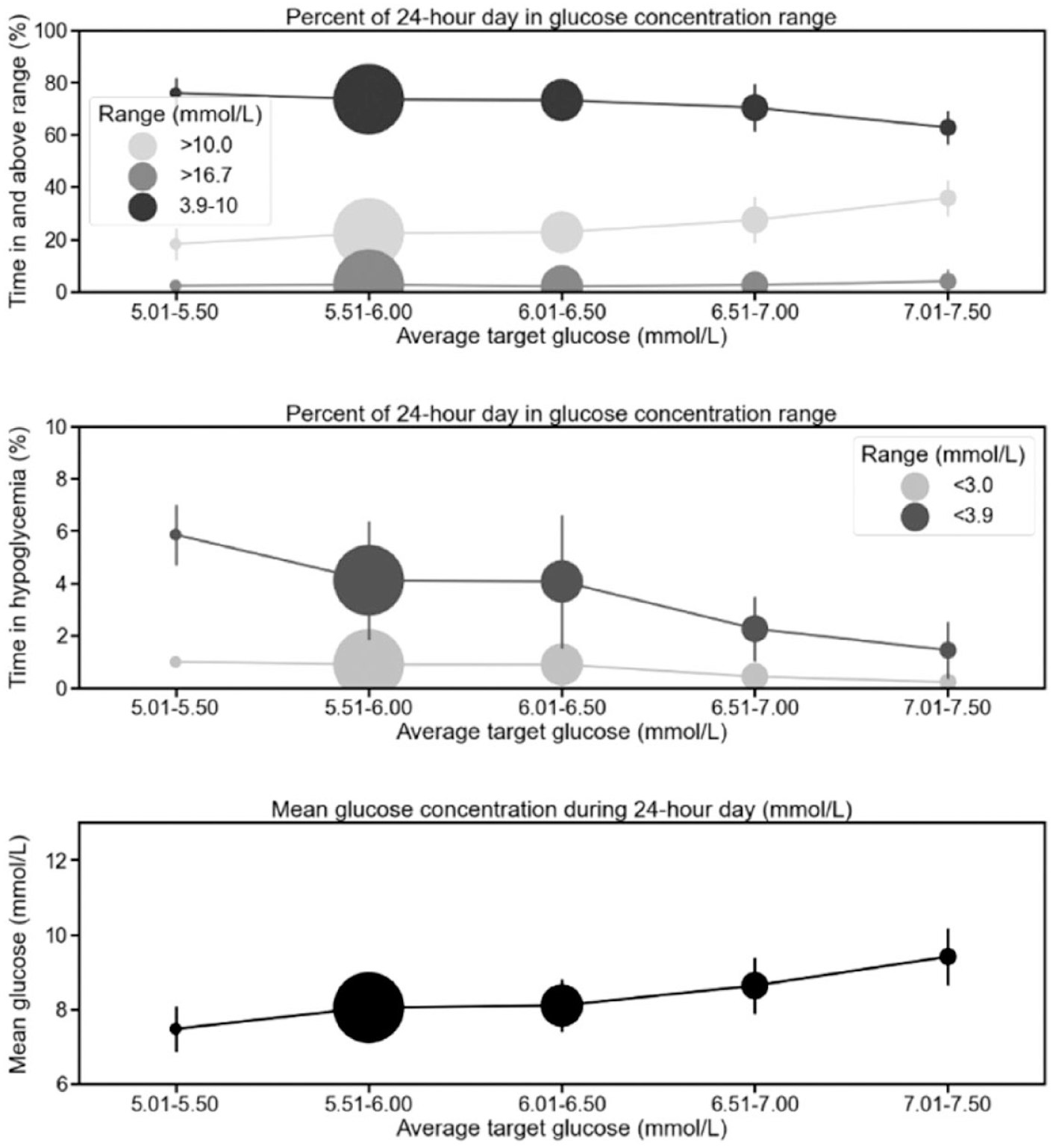
Glycemic metrics across different personal glucose target bins. The circle size reflects the n number with average personal glucose targets within the bin.
Personal glucose targets above 6.5 mmol/L (117 mg/dL) were associated with significantly less time in target range (3.9-10.0 mmol/L [70-180 mg/dL]; 6.5-7.0 mmol/L [117-126 mg/dL] bin: mean difference −3.2 percentage points [95% CI: −5.3 to −1.2; P < .001]; 7.0-7.5 mmol/L [126-135 mg/dL] bin: −10.8 percentage points [95% CI: −14.1 to −7.6; P < .001]) (Table 1), but the time in target only dropped below the recommended 70% when the personal target was set >7.0 mmol/L (126 mg/dL). The reduced time in target with higher personal targets occurred in parallel with significantly increased time above 10.0 mmol/L (180 mg/dL) (6.5-7.0 mmol/L [117-126 mg/dL] bin: mean difference 5.1 percentage points [95% CI: 3.0-7.2; P < .001]; 7.0-7.5 mmol/L [126-135 mg/dL] bin: 13.5 percentage points [95% CI: 10.2-16.9; P < .001]).
Lower personal targets were associated with increased time with glucose levels <3.9 mmol/L (70 mg/dL; 5.0-5.5 mmol/L [90-99 mg/dL] bin: mean difference −0.57 percentage points [95% CI: −1.00 to −0.14; P < .005]) although the data set was relatively small (Table 1). Only personal targets >6.5 mmol/L (117 mg/dL) were associated with significantly lower time <3.9 mmol/L (70 mg/dL) (Figure 1) (6.5-7.0 mmol/L [117-126 mg/dL] bin: −1.85 percentage points [95% CI: −2.37 to −1.34; P < .001]; 7.0-7.5 mmol/L [126-135 mg/dL] bin: −2.68 percentage points [95% CI: −3.49 to −1.86; P < .001]). No personalized targets were associated with significant differences in time with glucose levels <3.0 mmol/L (<54 mg/dL) (which was ≤1.0% across all bins).
There was no significant difference in glycemic outcomes with personal targets between 5.5 and 6.0 mmol/L (99-108 mg/dL) and between 6.0 and 6.5 mmol/L (108-117 mg/dL) suggesting that adjusting the personal target by >1 mmol/L (18 mg/dL) may be required for clinically meaningful changes to glucose outcomes.
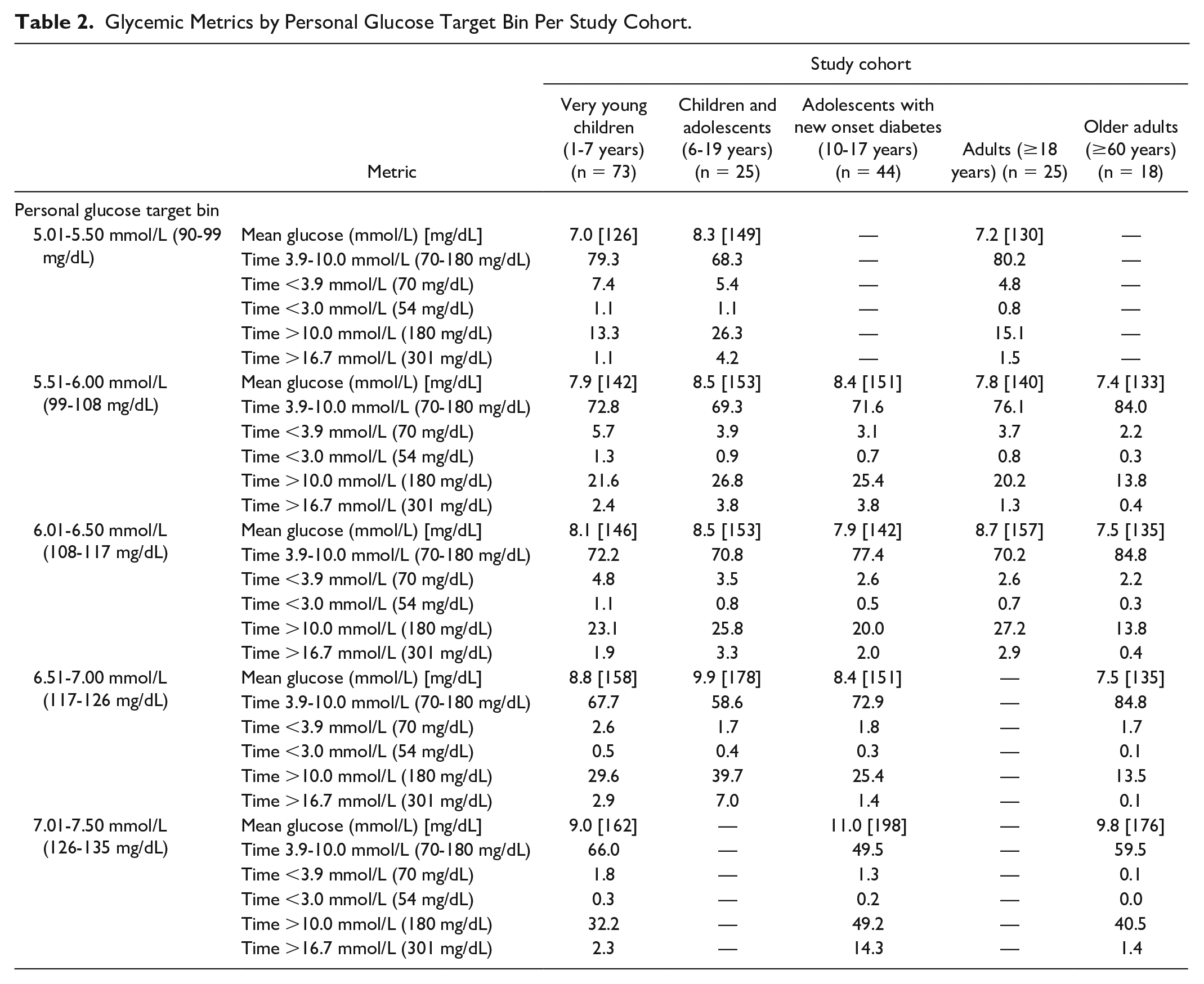
Glucose metrics when different personal targets are applied are shown by study cohort in Table 2.
Glycemic Metrics by Personal Glucose Target Bin Per Study Cohort.
Discussion
Discrete study cohorts showed differences in glucose control when applying similar personal targets. The burden of hypoglycemia was greatest in very young children even when higher personal targets were used (Table 2), reflecting the challenges of diabetes management in this population. 1
Older adults were able to achieve >80% time in range with the application of higher personal targets up to 7.0 mmol/L (126 mg/dL), a threshold at which there was only 1.1% of time in hypoglycemia (<3.9 mmol/L [70 mg/dL]), suggesting that higher personal targets may be beneficial in this population.
A similar trend of reduced time spent in the target glucose range when higher algorithm target glucose settings are applied has also been reported with the Omnipod (Insulet Corporation, Massachusetts, US) closed-loop system and in real-world data from the Medtronic 780G (Medtronic, Northridge, California, US) closed-loop systems.6,7 The Medtronic 780G settings that predicted the highest time in range were an active insulin time of two hours and the lowest glucose target of 5.6 mmol/L (100 mg/dL).
Strengths of our analysis include the use of data from participants across a wide range of age demographics and from multicenter, multinational clinical trials. Limitations include the retrospective analysis and that personal targets were averaged over the 24-hour period. Selection of personal targets was determined by the user, which may reflect individual differences in assessment of hypoglycemia and hyperglycemia risk. The largest cohort of participants were very young children who have the greatest hypoglycemia burden; however, this was accounted for in the analysis.
In conclusion, personal targets are a well-accepted and useful tool to individualize glucose control to suit the needs of the user. Our analysis allows users and healthcare professionals to understand the impact of personal target adjustments on glucose control and the recommended glycemic targets. 8
Footnotes
Acknowledgements
The authors thank the study volunteers for their participation in the studies and acknowledge support by the staff at the Addenbrooke’s Wellcome Trust Clinical Research Facility. They also thank Josephine Hayes, Matthew Haydock, Nicole Ashcroft, and Alina Cezar (University of Cambridge), as well as Meena Murthy and Lidiya Varghese (Cambridge Clinical Trials Unit), for the administrative support.
Abbreviations
CGM, continuous glucose monitor; HCL, hybrid closed-loop; T1D, type 1 diabetes; UK, United Kingdom; USA, United States of America.
Author Contributions
M.F., C.K.B., J.W., and R.H. codesigned the analysis. M.F. performed data analysis. M.F. and C.K.B. wrote the manuscript. C.K.B., J.M.A., H.T., L.L., M.L.E., M.E.W., and R.H. codesigned the clinical studies. R.H. designed the control algorithm. C.K.B., J.W., J.M.A., S.H., M.E.W., A.T., C.d.B., F.M.C., E.F.-R., S.E.H., T.M.K., B.R.-M., A.G., T.L.R., R.E.J.B., D.E., N.T., L.D., N.D., L.B., H.T., L.L., M.L.E., and J.K.M. provided patient care for participants in the studies. All authors critically reviewed the manuscript. M.F., C.K.B., J.W., and R.H. are the guarantors of this work and, as such, had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: C.K.B. has received consulting fees from CamDiab and speaker honoraria from Ypsomed. J.W. has received speaker honoraria from Ypsomed. J.M.A. has received consulting fees from CamDiab. S.H. serves as a member of Sigma (Dexcom) and Medtronic advisory boards; is a consultant for CamDiab and a director of Ask Diabetes Ltd., providing training and research support in healthcare settings; and reports having received training honoraria from Medtronic, Sanofi, and Ypsomed. M.E.W. reports receiving license fees from B. Braun, patents related to closed-loop systems, and being a consultant at CamDiab. C.d.B. reports having received speaker honoraria from Medtronic and has served on the EU psychology e-learning board of Medtronic. E.F.-R. reports having received speaker honoraria from Medtronic, Eli Lilly and Company, Novo Nordisk, and Sanofi and serving on the advisory board for Eli Lilly and Company. S.E.H. has received speaker honoraria from Medtronic, Eli Lilly, Ypsomed, and Insulet. T.M.K. reports having received speaker honoraria from Eli Lilly, Merck Serono, and Novo Nordisk. B.R.-M. has received speaker honoraria from Abbott Diabetes Care, Eli Lilly, Medtronic, Novo Nordisk, Roche Diabetes Care, Sanofi, and Menarini and has been on the advisory boards of Eli Lilly, Roche Diabetes Care, and Abbott Diabetes Care. T.L.R. has received consultancy fees from Abbott Diabetes Care and speaker honoraria from Novo Nordisk. R.E.J.B. reports having received speaking honoraria from Eli Lilly and Springer Healthcare and sits as an unpaid member of the Novo Nordisk UK Research Foundation grant and selection committee. L.D. has received honoraria for taking part in an Interactive Advisory and Advocacy Forum on CGM Use in Pediatric Clinical Practice from Dexcom and has received conference fees from Novo Nordisk. H.T. reports having received research support from Dexcom and speaker honoraria from Eli Lilly and Dexcom. L.L. reports having received speaker honoraria from Animas, Abbott, Insulet, Medtronic, Novo Nordisk, Roche, and Sanofi; was on advisory panels for Animas, Abbott, Novo Nordisk, Dexcom, Medtronic, Sanofi, and Roche; and received research support from Novo Nordisk and Dexcom. M.L.E. reports having received speaker honoraria from Eli Lilly and Company, Novo Nordisk, Abbott Diabetes Care, Medtronic, AstraZeneca, and Ypsomed and acting on advisory boards for Medtronic, Novo Nordisk, Zucara Therapeutics, Pila Pharma, and Abbott Diabetes Care. J.K.M. is a member of the advisory boards of Abbott Diabetes Care, BD, Boehringer Ingelheim, Eli Lilly and Company, Medtronic, Novo Nordisk AS, Prediktor A/S, Roche Diabetes Care, and Sanofi; received speaker honoraria from Abbott Diabetes Care, AstraZeneca, Dexcom, Eli Lilly and Company, Menarini Diagnostics, Novo Nordisk A/S, Roche Diabetes Care, Servier, and Ypsomed; and is a cofounder and shareholder of decide Clinical Software Ltd. R.H. reports having received speaker honoraria from Eli Lilly and Company, Dexcom, and Novo Nordisk; receiving license fees from Medtronic; receiving patents related to closed-loop systems; and being the director at CamDiab. M.F., A.T., F.M.C., A.G., D.E., N.T., N.D., and L.B. have no conflict of interest to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by the National Institute for Health Research EME Grant (14/23/09); National Institute of Diabetes and Digestive and Kidney Diseases (UC4DK108520 and 1DP3DK112176-01); JDRF; Helmsley Trust (#2016PG-T1D046); National Institute for Health Research Cambridge Biomedical Research Center; Horizon 2020 (grant agreement no. 731560); and Wellcome Trust Strategic Award (100574/Z/12/Z). The University of Cambridge has provided salary support for M.L.E. from the National Health Service in the East of England through the Clinical Academic Reserve. R.E.J.B. received salary support from the National Institute for Health Research (NIHR) Oxford Biomedical Research Center. The views expressed are those of the authors and not necessarily those of the funders. No sponsor had any role in the study design, data collection, data analysis, data interpretation, or writing of the manuscript.
