Abstract

Introduction
On October 18, 2022, the U.S. Food and Drug Administration (FDA) cleared the Nova Primary Glucose Analyzer System in a 510(k) substantial equivalence determination (Figure 1). The predicate comparator device was the YSI 2300 STAT PLUS Glucose and Lactate Analyzer (YSI 2300; YSI Incorporated, Yellow Springs, OH, United States). The decision was announced in an assay and instrument summary posted by the FDA on October 24, 2022. 1 According to this report, the instrument is “a benchtop laboratory glucose analyzer that measures blood glucose levels in lithium heparinized venous whole blood or plasma and utilizes a glucose oxidase-based sensor and membrane/cap assembly.” 1 Its indications for use claim can be found in Figure 2. The manufacturer intends for the Nova Primary to fill the need for a glucose reference analyzer to replace the widely used YSI 2300, which is no longer being manufactured. The Nova Primary has not been cleared with a point-of-care claim for patient care; however, we believe it can potentially serve as an alternative comparator method to the YSI 2300 for clinical studies and device submissions to the FDA for determination of glucose accuracy. This method should be sufficiently accurate to not require core laboratory facilities and procedures for reference testing.

Nova Primary Glucose Analyzer System. Figure courtesy of Nova Biomedical Corporation.

Indications for use for the Nova Primary Glucose Analyzer System. 1
Features of the Nova Primary Glucose Analyzer System
The Nova Primary has been available for sale in European Conformity (CE)-regulated countries since January 2021. 2 The device uses a single, reusable glucose electrochemical sensor based on glucose oxidase and has a measurement range of 20 to 900 mg/dL. It is designed to measure a 25-mL sample of either whole blood or plasma, which is internally diluted, like in the YSI. Results are available after approximately two minutes. It uses a color touchscreen display with an icon interface. For calibration, the system uses a single calibrator pack based on radio-frequency identification (RFID) data management to monitor the pack expiration date and number of samples remaining, which eliminates the need for separate reagent bottles and daily monitoring of their levels. The Nova Primary uses a comma delimited (.csv) format data output (which allows data to be saved in a table format) that can be downloaded via the integral USB data port. The analyzer’s dimensions are 17 in × 11 in × 18 in (44 cm × 28 cm × 46 cm). 1
The Nova Primary Glucose Analyzer was found to be substantially equivalent to the YSI 2300 by the FDA using lithium-heparinized venous whole blood or plasma with data submitted by Nova Biomedical Corporation (510K application number K203549). The analyzer performs an automatic two-point calibration every two hours and automatic one-point calibration with each sample. Two levels of quality control are available. One normal and one abnormal level control should be performed every 24 hours or after performing system maintenance following good laboratory practice guidance.
Performance
Within-run precision is ≤2.5% coefficient of variation (CV) for quality control samples, ≤2.9% CV for venous whole blood samples, and ≤1.9% CV for plasma samples over the reportable range of 20 to 900 mg/dL.
Run-to-run precision is ≤2.7% CV for quality control samples, ≤3.3% CV for venous whole blood samples, and ≤3.4% CV for plasma samples.
Linearity was evaluated using three Nova Primary Glucose Analyzers and two YSI 2300 analyzers. Whole blood and venous blood samples containing ten different concentrations of glucose were prepared using a serial dilution method. Each whole blood and plasma sample was analyzed in triplicate on the three Nova Primary test analyzers and on the two YSI 2300 analyzers. The results from each test analyzer were compared to the average of the test results from the YSI 2300 analyzers. The results of least squares linear regression analyses are shown in Figure 3.
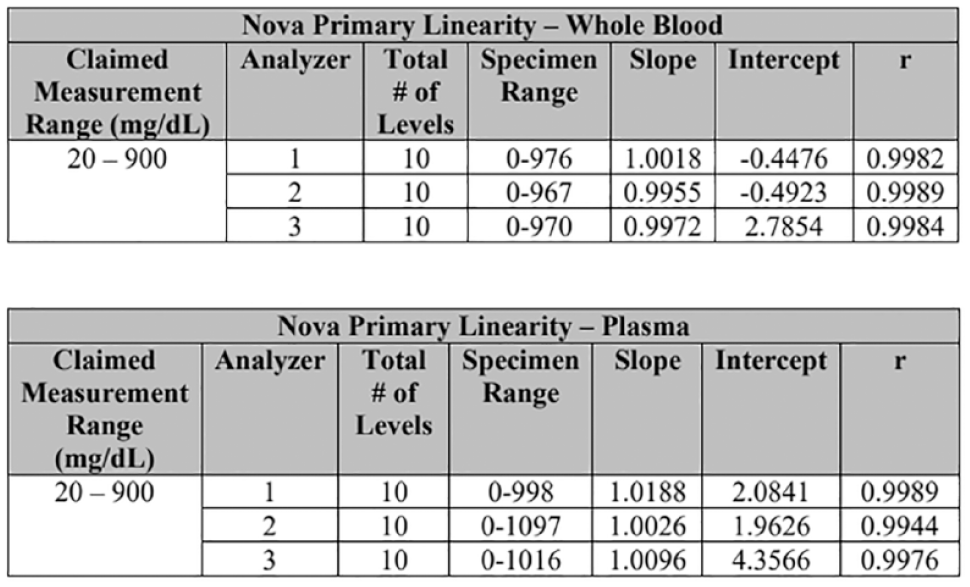
Linearity of the Nova Primary Glucose Analyzer according to its FDA 510(k) substantial equivalence determination decision summary. 1
A comparison study used 174 discarded lithium-heparinized venous whole blood specimens (15 of which were altered) from hospitalized patients. They were analyzed in singlet on three Nova Primary Glucose Analyzers and two YSI 2300 analyzers. The study used two lots of Nova Primary Calibrator Cartridges and two lots of glucose membranes. Each singlet result from the test instrument was compared to the average of the test results from the comparator instruments. Over the course of the study, the results showed singlet slopes of 0.9927 to 1.0083 with intercepts from −3.7428 to 0.3878 (r > 0.9989) over the range of 34 to 871 mg/dL. Furthermore, 170 discarded lithium-heparinized venous plasma specimens (15 of which were altered) were also tested in the same manner. The results showed singlet slopes of 0.9935 to 1.0088 with intercepts from −0.238 to 1.1364 (r >0.991) over the range of 30 to 835 mg/dL.
The End of the Road for the YSI 2300 STAT PLUS Glucose and l -Lactate Analyzer
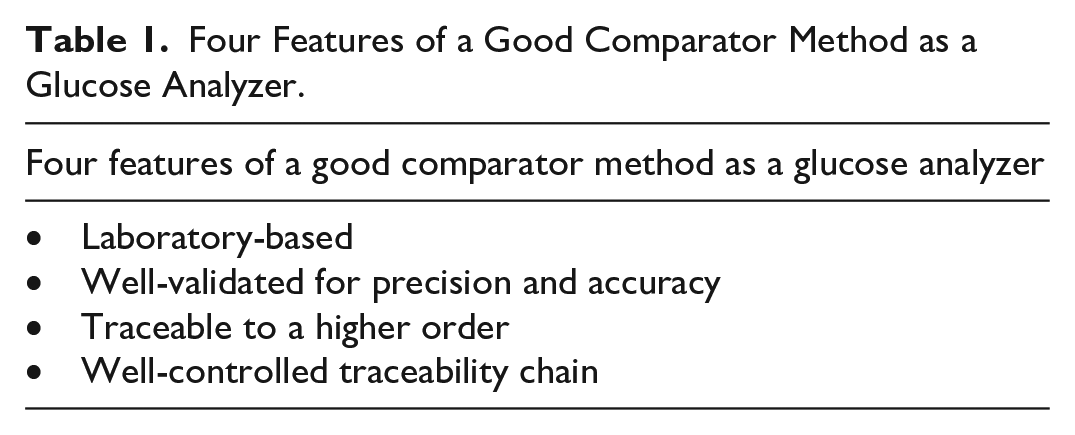
In the United States, the YSI 2300 has been the most widely used benchtop comparator instrument for determining the accuracy of products that measure blood glucose and interstitial fluid glucose, since it was cleared by the FDA in 1991. Many manufacturers of glucose monitoring devices have noted its use to be so widespread that they have treated it like it is actually a regulatory standard for comparison testing of glucose monitoring devices, although it is not. On November 13, 2019, Diabetes Technology Society (DTS) organized a meeting called The YSI Replacement Meeting to discuss replacement of the YSI 2300. There, an official from the FDA reviewed the agency’s position on glucose comparator products. He stated that (1) the FDA had never formally identified a single specific laboratory analyzer (such as the YSI 2300) as the required comparator method for clinical and analytical studies of glucose measurement devices and (2) a good comparator method should have four features. 3 These features are listed in Table 1. The perception that it was necessary to use the YSI 2300 for comparison testing has been a barrier that has discouraged the use of alternate comparator devices. Thus, there was not a large market for other comparator devices until July 2021, when the YSI 2300 was discontinued by its manufacturer, and the market became larger in July 2022, when manufacturers discontinued supporting the product with parts. 4 Its replacement products (which all measure glucose with similar biosensor technology as the YSI 2300) are the YSI 2500, YSI 2900, and YSI 2950 analyzers. The YSI 2500, like the YSI 2300, measures glucose and lactate whereas the YSI 2900 and 2950 analyzers measure other analytes in addition to glucose and lactate. None of these three products have been cleared by the FDA for measuring glucose in human whole blood or plasma like the now-discontinued YSI 2300 was. These newer YSI instruments are intended to measure foods for their glucose and lactate contents (Table 2). 5 Currently, the YSI 2300 is the most widely used device for determining the accuracy of products that measure blood glucose and interstitial fluid glucose. 6 In the United States, most testing laboratories are using discontinued YSI 2300 instruments with replacement parts coming from stockpiles, and there have been questions about what would happen once replacement parts are no longer available from these sources.
Four Features of a Good Comparator Method as a Glucose Analyzer.
Indications for the YSI 2500 Glucose Plus Lactate Analyzer.

What is Needed Now
There had been concern in the glucose diagnostics community among manufacturers of products that measure blood glucose as well as reference laboratories that test the accuracy of these products that no suitable comparator product is now being manufactured for use in the United States. Such a candidate product would have to be an FDA-cleared nonstrip benchtop instrument for measuring small volumes of whole blood or plasma with good accuracy and human factors. Furthermore, such a product’s method would have to be traceable to a higher metrological order (such as an internationally recognized reference material and/or method), and the traceability chain must include as few stages as possible to reduce bias and imprecision.7,8 Ideally, such a product should exhibit both low bias and low imprecision, so that results from different devices of that product would be highly comparable. This was not always the case with YSI 2300 instruments. 9 The data submitted for FDA clearance of the Nova Primary looks promising with respect to both bias and imprecision, and the Glucose Calibrators, Controls, and Standards used for clearance studies were reported to be traceable to the National Institute of Standards and Technology (NIST) Standard SRM-917. For glucose accuracy studies, while correction of bias is feasible through recalibration, 10 regulatory authorities might not accept retrospective recalibration of comparator results. Finally, fast turnaround is also a requirement for a comparator instrument used by a testing laboratory. This is because in many cases of assessing the accuracy of a glucose measurement device, a test subject’s glucose concentration is manipulated, sometimes to borderline risky hypoglycemic levels. It is critically important to know an accurate glucose concentration in as close to real time as possible to determine when it is time to rescue the subject with a glucose treatment.
At the 2019 DTS YSI Replacement Meeting, there was widespread interest in encouraging manufacturers of benchtop glucose analyzers to build suitable products for this purpose and obtain FDA clearance. At the same time, there was a general reluctance for the glucose measurement industry to once again fall into a situation where only a single product would be used by everyone, which has been the case with the YSI 2300. Rolf Hinzmann from Roche Diabetes Care presented eight reasons why only one single comparator method should not be endorsed by the glucose measurement community (Table 3). Thirteen factors that are relevant to a manufacturer of a glucose measuring device or a testing laboratory in choosing a comparator method were presented at the conclusion of the 2019 meeting. They are listed in Table 4.
Reasons Why Stakeholders Would Not Endorse Only a Single Product for Determining and Documenting the Accuracy of Comparator Glucose Measurement Devices.

Thirteen Factors That Are Relevant in Choosing a Comparator Method.

Conclusions
The availability of a now-FDA-cleared comparator instrument will be a boon to the further development of increasingly accurate blood tests, minimally invasive tests, and eventually noninvasive tests for measuring glucose accurately. Manufacturers of other benchtop products might eventually seek FDA clearance for their comparator methods in the future. These future candidates may or may not offer greater accuracy or more attractive features compared to the Nova Primary, but at this time, no other instrument with similar capabilities is both cleared and available for sale. We expect to eventually see published results of studies conducted by independent investigators assessing the accuracy of the Nova Primary compared to reference methods known to have even greater accuracy than the YSI 2300 predicate system that was used in this clearance procedure. Thus, the news of clearance of the first post-YSI 2300 comparator system means that the process of reference testing for glucose measuring devices in the United States has advanced beyond the end of one era and into the beginning of a new era.
Footnotes
Acknowledgements
The authors thank Guido Freckmann and Stefan Pleus for their helpful advice. The authors also thank Annamarie Sucher-Jones for her expert editorial assistance.
Abbreviations
CE, European Conformity; CV, coefficient of variation; DTS, Diabetes Technology Society; EU, European Union; FDA, United States Food and Drug Administration; NIST, National Institute of Standards and Technology; Nova Primary, Nova Primary Glucose Analyzer; RFID, radio-frequency identification; YSI 2300, YSI 2300 STAT PLUS Glucose and Lactate Analyzer.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DCK is a consultant for EOFlow, Fractyl Health, Integrity, Lifecare, Rockley Photonics, and Thirdwayv. AMY, JH, and JHN have nothing relevant to disclose.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
