Abstract
Production of the YSI 2300 STAT PLUS Glucose and
Introduction
The YSI 2300 STAT PLUS Glucose and
The Problem
There are many reasons why it would be better for industry if all of these companies settled on the same comparator instrument. The instrument could be a new-generation YSI instrument (assuming that the company decides to seek FDA clearance and is able to meet applicable regulatory requirements) or another point-of-care (POC) instrument that can handle a large number of specimens, as is the case with the YSI instruments. An alternate comparator might be embraced by the diagnostics industry if it is either currently cleared by FDA or if its manufacturer goes through the process of successfully obtaining FDA clearance. In either case, testing labs would become familiar with a single comparator and would become more skilled with its use, instead of having many types of glucose analyzers (for different companies’ trials). The use of a single industry-wide comparator instrument would also make it easier for consumers to compare performance of glucose monitoring products because most or all would be tested against the same comparator method. In order for stakeholders to identify an industry-wide comparator, it will be necessary to define a mandatory level of accuracy and precision so that candidate instruments can be judged for performance.
An additional aspect to consider in selecting a comparator method is the traceability chain of the method. FDA recommends that a comparator method be traceable to a higher order (eg, an internationally recognized reference material and/or method) and that the traceability chain include as few stages as possible to reduce bias.
Method
We reviewed the clinical chemistry literature for articles where blood glucose monitors, continuous glucose monitors, or noninvasive glucose monitors were assessed for accuracy to see which instruments are being used as a comparator. We also spoke with clinical chemists, endocrinology lab directors, and anaesthesiologists in the United States and Europe about which instruments they were using or would recommend using for this purpose and what they saw as advantages of specific products. We then created a list of candidate industry-wide comparator instruments that might replace the YSI 2300 as the dominant comparator product for testing accuracy of glucose monitors. We only considered instruments that are currently on the market. Those included are examples of laboratory chemistry analyzers and blood gas machines. There may be others in each category and we are presenting these as examples. We are not endorsing any particular instrument, but we are providing examples of technologies that might replace the YSI 2300.
Results
Seven FDA-cleared benchtop or portable glucose monitoring instruments were identified (including the YSI 2300). All are based on a glucose oxidase enzyme method except for the Hemocue Glucose 201+, the GEM Premier 5000, and the Roche Integra Cobas 400 plus, which use, respectively, glucose dehydrogenase, hexokinase, and hexokinase enzyme methods. These two enzymes are resistant to many interfering substances that can affect glucose oxidase-based systems. There is little information in the medical literature of the accuracy of all these instruments compared to each other and to reference methods (see Table 1). We also heard about four additional non-FDA-cleared benchtop glucose analyzers that are frequently used outside the United States as a comparator for glucose measurements or within the United States for purposes other than glucose monitor registration (see Table 2). The list in Table 2 is not meant to be comprehensive, but it contains the instruments which we heard about from experts in this field. There is little information in the medical literature of the accuracy and precision of any of these instruments compared to each other or to reference methods. Some of these candidate comparator methods are handheld POC devices, and FDA has generally recommended against the use of handheld POC devices as comparator methods. Rather, FDA has generally recommended that the comparator method should be an FDA-cleared nonstrip laboratory glucose measurement method, since these are usually more accurate and precise.
Features of FDA-Cleared Candidate Industry-wide Comparator Instruments for Assessing the Accuracy of Blood Glucose Monitors.
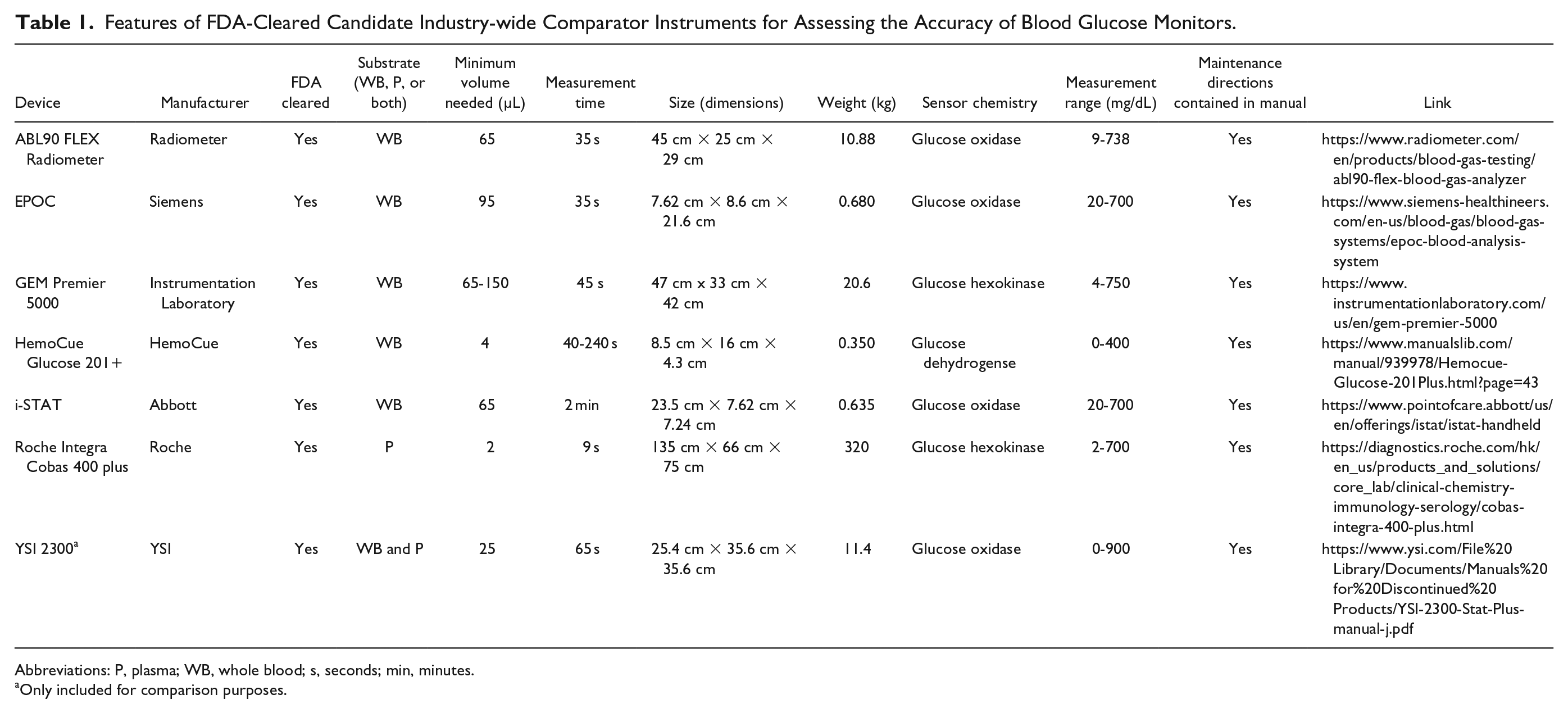
Abbreviations: P, plasma; WB, whole blood; s, seconds; min, minutes.
Only included for comparison purposes.
Features of Non-FDA-Cleared Candidate Industry-wide Comparator Instruments for Assessing the Accuracy of Blood Glucose Monitors.
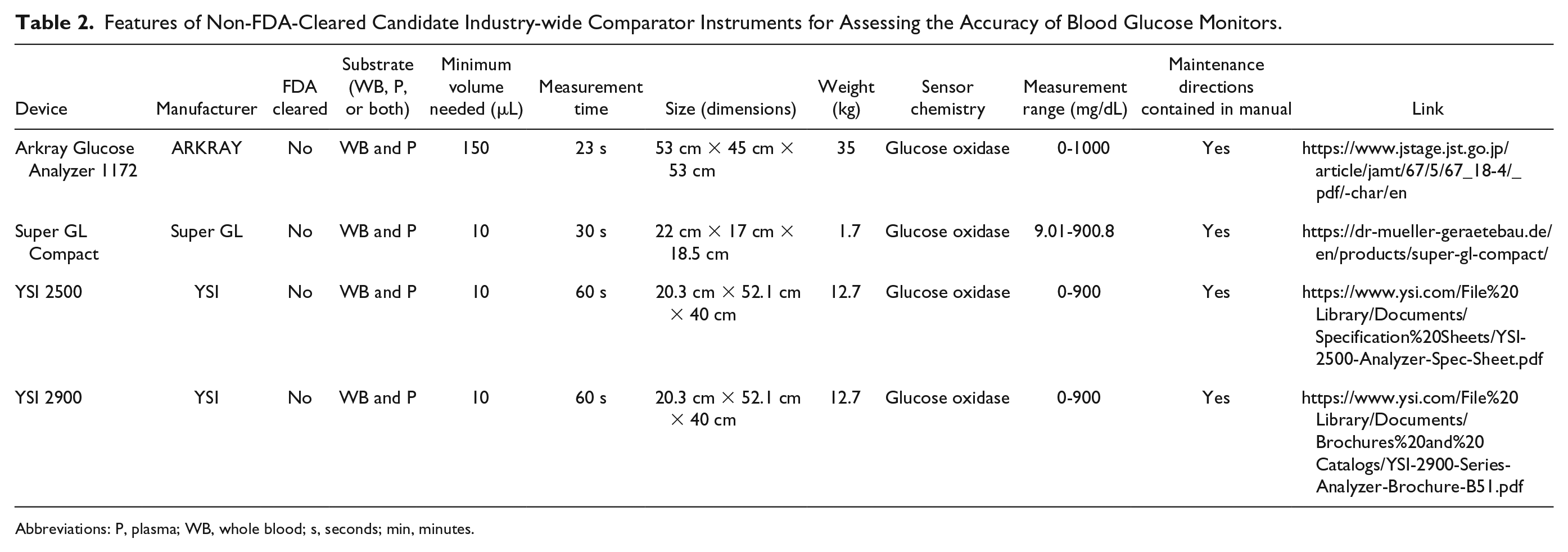
Abbreviations: P, plasma; WB, whole blood; s, seconds; min, minutes.
Description of FDA-Cleared Candidate Glucose Measurement Instruments
ABL90 Flex Radiometer
The ABL90 Flex Radiometer is a high-throughput device for venous and arterial whole blood (WB) that has a fast measuring time of 35 seconds. It can measure blood gases as well as glucose concentrations. 4 The ABL90 Flex Radiometer is most similar to the YSI 2300 in weight (10.88 kg vs 11.4 kg) and can be incorporated into clinical and hospital settings. ABL90 Flex Radiometer uses glucose oxidase sensor chemistry.
EPOC
Siemens’ EPOC has two key features. It has a fast measurement time of 35 seconds. It is a light handheld device for WB with a weight of 0.68 kg, making it convenient to use in hospital settings. In a study comparing the EPOC to i-STAT devices, EPOC measurements showed high precision and good correlation (
GEM Premier 5000
The GEM Premier 5000 measures glucose in venous and arterial WB and has a fast measuring time of 45 seconds. It can measure gases in blood as well as glucose concentrations. 6 The GEM Premier 5000 uses glucose hexokinase sensor chemistry.
HemoCue Glucose 201+
The HemoCue Glucose 201+ (HemoCue) is a glucose dehydrogenase-based assay intended for use on WB. In a study comparing this method with the YSI 2300 in 24 subjects, HemoCue glucose measurements were shown to be closely correlated with YSI 2300 glucose measurements, with 98.8% of values in zone A of error grid analysis. 7 When HemoCue Glucose 201+ was tested, 99.0% of glucose measurements were within the system accuracy criteria of ISO 15197:2013. 8 HemoCue only requires 4 µL of sample volume and is a handheld device, making it convenient for portable use. It is the second lightest device in this group with a weight of 350 g. HemoCue uses glucose oxidase sensor chemistry.
i-STAT
Abbott’s POC comparator is the i-STAT. The i-STAT is convenient to use because it is a portable handheld device for WB. Test results can be easily integrated into an electronic health record in a variety of clinical settings. The i-STAT can measure blood gases as well as glucose concentration. 9 It is the third lightest device in this group with a weight of 0.63 kg. iSTAT uses glucose oxidase sensor chemistry.
Roche Integra Cobas 400 plus
The Roche Integra Cobas 400 plus is a high-throughput device of up to 400 tests per hour. 10 Only 2 μL of sample volume is needed per assay, with dead space in the probe requiring additional volume up to 65 to 80 μL. Only plasma (P) or serum glucose samples can be measured. This product is well known for reliability. Roche is the only BG Monitor manufacturer to use its own device (Cobas 6000) as a calibration method. The Roche Integra Cobas 400 plus uses glucose hexokinase sensor chemistry.
YSI 2300
The YSI 2300 can be used on both WB and P. Only 25 μL of sample volume is needed per assay, and each measurement takes 60 seconds. It is currently widely used in the United States for new product clearance. YSI 2300 uses glucose oxidase sensor chemistry.
Description of Non-FDA Cleared Candidate Glucose Measurement Instruments
Arkray Glucose Analyzer 1172
Arkray Glucose Analyzer 1172 (GA-1172) can measure glucose from both WB and P samples, similar to the YSI 2300 and Super GL Compact. In this group, it has the second fastest measuring time of 23 seconds. The GA-1172 is being used as a calibrator instrument by many Japanese companies and is currently only available in the Japanese market. Arkray Glucose Analyzer 1172 uses glucose oxidase sensor chemistry.
Super GL Compact
Many testing labs in Europe are currently using the Hitado Super GL Compact Analyzer (Super GL) for comparison studies when they do not intend to seek future marketing authorization through FDA. During glucose clamp studies, it is useful to measure blood glucose quickly and accurately, as well as for registration studies outside the United States. 11 Unlike the YSI 2300 and 2900D, the Super GL can determine hematocrit values as well as analyze glucose from WB samples. Super GL can use both WB and P samples, similar to the YSI 2300. It requires only 10 μL of sample volume for each measurement and has the third fastest (in this group) measurement time of 30 seconds. When Super GL Compact was tested, 99.5% of glucose measurements were within the system accuracy criteria of ISO 15197:2013. 8 Super GL uses glucose oxidase sensor chemistry.
YSI 2500 and 2900
YSI 2500 and 2900 are the next generation product following the YSI 2300. YSI claims that YSI 2500 and 2900 are manufactured with the same measurement technology as the YSI 2300. However, YSI 2500 and 2900 are not FDA cleared. Similar to YSI 2300, YSI 2500 and 2900 can use both WB and P samples, and only need 10 µL of sample volume. YSI 2500 is limited to glucose and lactate measurements while YSI 2900 can measure up to 13 analytes, including glucose. The measurement time decreased from 65 to 60 seconds from the YSI 2300, and the weight of both increased from 11.4 to 12.7 kg. They are the closest comparators to the YSI 2300 in the current market in terms of product features. YSI 2500 and 2900 both use glucose oxidase sensor chemistry.
Discussion
The YSI 2300 has been a popular comparator instrument for testing glucose measuring devices since it was cleared by the FDA in 1991. 12 Over these 28 years, it has achieved a dominant position in the comparator industry. Now, it is time for stakeholders in assessing the accuracy of glucose monitoring devices to look elsewhere for a product or products that can serve this same function. The best candidates will be FDA-cleared laboratory glucose measurement methods with robust traceability that are inexpensive, accurate, precise, fast, easy to use, and able to measure WB as well as P. No candidate replacement product will be the best in all of these areas. It is time to consider alternate methods for glucose comparison testing in a post-YSI 2300 world.
Conclusion
It will be helpful for the glucose monitoring industry if a consensus can be reached in two topics. First, what is the necessary accuracy and precision required for comparator glucose monitoring instruments? Second, which instrument, if any, would be the best industry-wide comparator for determining the performance of glucose monitoring products? Such a consensus should be developed by experts from (1) manufacturers of glucose monitoring instruments, (2) FDA, (3) academic clinical chemistry, (4) academic endocrinology, (5) testing lab officials who currently use the YSI-2300, and (6) representatives of professional diabetes and clinical chemistry organizations. We are at a historic time with a need to replace an industry-wide workhorse that has served as a comparator for 28 years. If the glucose monitoring community does not act quickly, then the currently standardized process for testing the performance of glucose monitors might devolve into disarray after YSI 2300 instruments are no longer available.
Footnotes
Acknowledgements
The authors thank Annamarie Sucher for her expert editorial assistance.
Abbreviations
GA-1172, Arkray Glucose Analyzer 1172; HemoCue, HemoCue Glucose 201+; m, minutes; P, plasma; POC, point-of-care; s, seconds; Super GL, Hitado Super GL Compact Analyzer; WB, whole blood; YSI, YSI Incorporated; YSI 2300, YSI 2300 STAT PLUS Glucose and l-Lactate Analyzer.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JH, JN, and MR have nothing to disclose. DCK is a consultant to Abbott, EOFlow, Fractyl, Know, Lifecare, Novo, and ThirdwayV. He also owns stock in ThirdwayV.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
