Abstract
People with diabetes admitted to hospital are at risk of diabetes related complications including hypoglycaemia and diabetic ketoacidosis. Point-of-care (POC) tests undertaken at the patient bedside, for glucose, ketones, and other analytes, are a key component of monitoring people with diabetes, to ensure safety. POC tests implemented with a quality framework are critical to ensuring accuracy and veracity of results and preventing erroneous clinical decision making. POC results can be used for self-management of glucose levels in those well-enough and/or by healthcare professionals to identify unsafe levels. Connectivity of POC results to electronic health records further offers the possibility of utilising these results proactively to identify patients ‘at risk’ in real-time and for audit purposes. In this article, the key considerations when implementing POC tests for diabetes in-patient management are reviewed and potential to drive improvements using networked glucose and ketone measurements are discussed. In summary, new advances in POC technology should allow people with diabetes and the teams looking after them whilst in hospital to integrate to provide safe and effective care.
Introduction
Point-of-care (POC) technologies are the cornerstone of diabetes self-management and have evolved over the last decades to be an essential component of maintaining clinical safety in people with diabetes admitted to hospital. 1 People with diabetes admitted to hospital are a unique patient subgroup who, irrespective of the cause for the admission, will require additional monitoring for the duration of their admission. In-patients with diabetes are at risk of developing several diabetes complications that include severe hypoglycemia, diabetic ketoacidosis (DKA), and hyperosmolar hyperglycemic state. 2 These harms may relate to the primary reason for the admission, for example, a patient with sepsis in a catabolic state may be at intrinsic risk of DKA but, more concerningly, some complications may develop due to medication errors, untimely administration of medications, inadequate monitoring and in some circumstances, the transfer of decision-making about treatment from the patient to health care professionals.
Point-of-care measurements help facilitate decision-making about diabetes management, but beyond this, have huge potential to drive improvements in in-patient safety both in real time and retrospectively. We review the current available POC technology to support in-patient diabetes care, parameters that are required to ensure quality in POC implementation and the emerging use of POC measurements to drive quality care provision.
What Are POC Technologies?
A device that undertakes a chemical analysis of blood, urine, or other body fluid at the patient bedside or away from the routine laboratory is termed a POC device or test.3,4
Point-of-care technologies to support diabetes management include glucometers and ketone meters that use capillary whole blood, urine dipsticks for the measurement of ketones and in some cases, remote automated platforms for the measurement of glycated hemoglobin (HbA1c), and urine dipstick analyses which may be manual or semi-automated (Table 1).
Key Diabetes-related POC Tests Used in an In-Patient Setting.
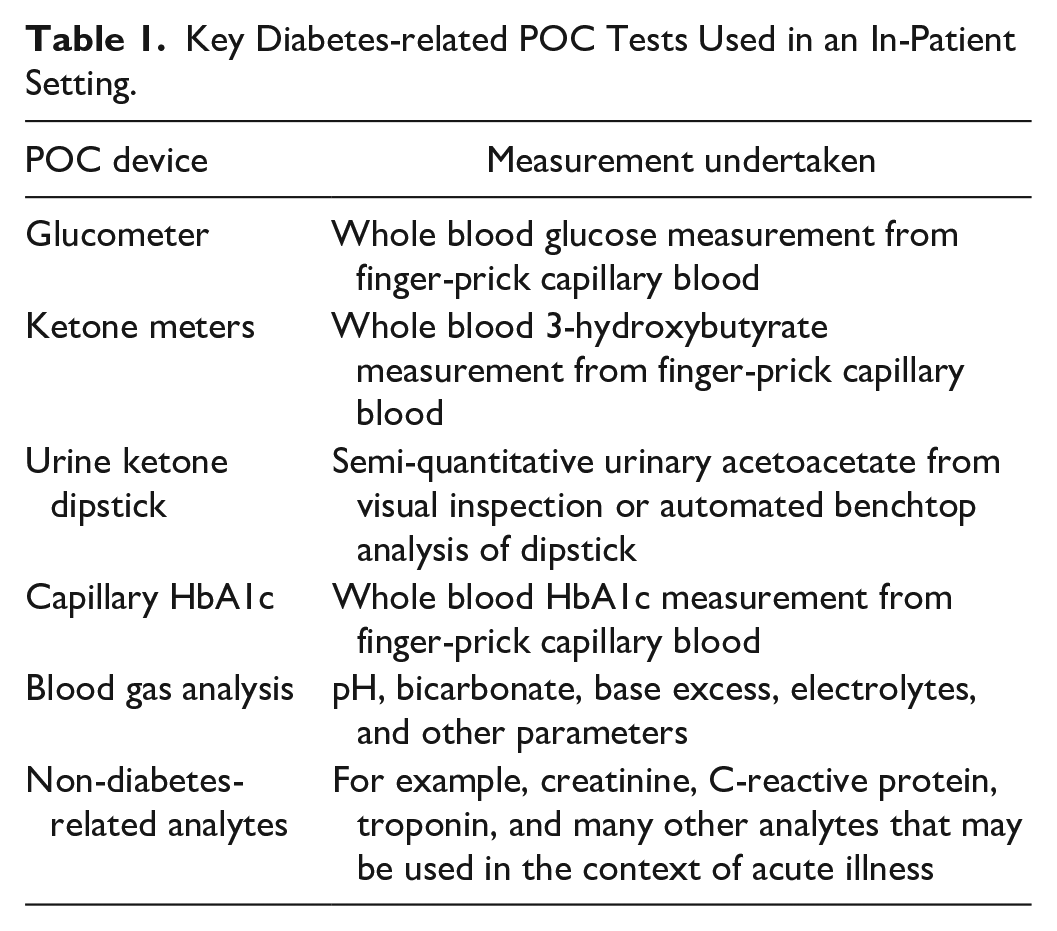
The generic advantages of POC devices in clinical practice are that they enable rapid diagnosis or clinical decision-making on the basis of results obtained in real time, which may then alter clinical pathways for a given patient in a way that routine laboratory analysis cannot 5 (Table 2). The advantages are particularly applicable for people with diabetes in hospital, for example, real-time glucose testing is intrinsic to management of diabetes (either self-management or to guide health care professionals) and the delay of sending blood samples to the laboratory would compromise prompt care and be inconvenient for the patient due to repeated venous blood sampling. Real-time monitoring of glucose (and indeed ketones) also supports reduction in complications such as the development of in-patient hyperglycemic crises or severe hypoglycemia and delivery of fixed rate and variable rate insulin infusions.
Potential Benefits Associated With Point-of-Care Glucose and Ketone Use.

POC Glucose Measurements
The most widely used POC test within an in-patient setting for diabetes is the glucometer. Point-of-care blood glucose allows immediate and reasonably accurate measurement of glucose levels in hospitals, in those with diabetes and also, without if an indication arises. The use of glucometers has a role in management of hyperglycemic emergencies, titration of variable rate intravenous insulin infusions, management of hypoglycemia, and routine monitoring in those with diabetes who are in hospital for another reason. They are, therefore, widely used throughout the hospital system including in critically unwell patients from intensive care units, operating theaters, postoperative recovery rooms, and emergency departments, as well as in general wards.
When selecting a POC glucose device, consideration should be given to the method of glucose analysis as there may be specific interferents, for example compounds in dialysate fluid may interfere with some methods1,6; a full description of assays and considerations for in-patient use has been reviewed elsewhere. 7 It is important to remember that POC glucose devices measure glucose in whole blood rather than laboratory measurements that measure glucose in plasma. A conversion factor that is applied constantly across the measurement range to convert whole blood glucose to plasma glucose assumes a fixed hematocrit, plasma concentration, and red blood cell water concentration.1,7 Thus, in unwell patients, this relationship may be perturbed although newer methods have automated correction for some interferents and parameters that may affect measurement.
Continuous glucose monitoring (CGM) conceptually does not fit with the premise of a POC test as it is an uninterrupted in vivo measure taken from an inserted sensor and continuous measurement could never be achieved in the laboratory. For full details on the use of CGM in hospitalized patients, see our paired publication. 8
POC Ketone Measures
The measurement of ketones is required for the diagnosis, assessment of severity, and monitoring for resolution of DKA. Ketones can be measured in urine or capillary whole blood.
Urine dipsticks (detecting ketones qualitatively or semi-quantitatively) impregnated with nitroprusside reagent react only with acetoacetate but not 3-hydroxybutyrate. Urine ketone concentrations reflect the total accumulated since the last void and levels measured may, therefore, become uncoupled from real-time changes. 9
Urine ketone measurement has some limitations; there is a paradoxical increase in urinary acetoacetate during resolution of DKA, which is due to the conversion of excess 3-hydroxybutyrate back to acetoacetate, as acidosis resolves; it is also the semi-quantitative with subjective interpretation of the test based on a color change. The lag time between serum and urine metabolic changes and difficulties with serial urine samples in a dehydrated unwell patient have led to some consensus that capillary blood ketone measurement may be superior to urine assessment. 10 This is further supported by the apparent delay in urine dipstick analysis compared with blood ketones, in those presenting acutely with hyperglycemia11,12 and that many acutely unwell patients are unable to provide a urine sample in the emergency department. 13
Capillary Blood Ketones
The measurement of capillary blood ketones (CBK) at the POC has been advocated in several international guidelines. 14 Like all POC tests, the rapid quantitative result obtained in real time allows for immediate changes in management.
There are a limited number of meters capable of measuring CBK using dry-chemistry methodologies, and versions of these meters are available for in-patient monitoring, with connectivity. These methods and older variants have been evaluated against reference enzymatic spectrophotometric assays and generally show good correlation.12,15 -18 Most manufacturers quote linearity up to ketone values of 8 mmol/L, after which a “Hi” reading is reported; however, there is some debate as to whether this is maintained in real-world evaluations.19,20
Most ketone meters also have recommended operational hematocrit ranges. However, dehydration and hyperosmolality are clinical states nearly always seen in DKA, and hematocrits may be well above the usual 60% threshold, stated. As with any test, these limitations should be taken into consideration when the measurement is being made, though it should be noted that bedside ketone monitoring is one parameter of several to aid making the diagnosis.
The cost and lack of availability of widespread CBK analysis mean that in some centers / countries urine ketone testing may still be the favored choice. Point-of-care devices must also be compliant with biochemistry laboratory protocols, to ensure staff training is adequate, that meters undergo rigorous and frequent quality assurance, that there is a facility to perform laboratory ketone testing when meter readings are invalid, and that there is sufficient connectivity to electronic patient records. 21 These standards are a challenge to maintain, but if CBK testing is to become widespread in and outside of hospital, the provision of these services must be taken into consideration. Reassuringly both CBK and serum BOHB have external quality assurance schemes, which laboratories can participate in. There is, therefore, infrastructure in place to ensure quality is maintained.22,23
Other POC Devices
There are many other devices used at the POC that may have relevance for in-patients with hyperglycemia (Table 1). Point-of-care HbA1c is used in many outpatient and general practice services to assist monitoring of people with established diagnoses of diabetes. 24 Current guidelines do not advocate use of POC HbA1c for a diagnosis of diabetes, due to concerns about accuracy though newer devices may fulfill international standards. 25 Evidence for benefit in hospitalized patients is limited to a few small studies that suggest screening hyperglycemic individuals in the emergency room may help stratify treatment approaches and identify undiagnosed diabetes.26,27 Downstream benefits of such an approach have yet to be formally evaluated. Once admitted to hospital, the logical benefits of a POC HbA1c may not be realized given that only a single measurement is needed and a laboratory analysis of venous blood may be turned around in sufficient time.
Blood gas analysis, another POC test used regularly in in-patients with diabetes, can offer measurement of glucose, acid-base status, creatinine, and electrolytes and is often the first set of “chemistry” results available in an acute setting. Venous blood gas analysis is required for the diagnosis of ketoacidosis, based on the pH, bicarbonate, anion gap, and glucose. Glucose levels are usually ascertained using amperometric methods using, for example, glucose oxidase. In one systematic review, the blood gas glucose on heparinized venous samples was more accurate than capillary glucose levels in critically unwell patients, which may not be surprising given changes in peripheral circulation and hematocrit. 28
Implementing POC Devices Effectively
Implementation of POCT devices for clinical applications requires a number of steps, including (1) a comprehensive validation of the method29,30 in conjunction with the biochemistry laboratory POC testing management team; (2) installation of a quality framework; and (3) connectivity to pathology results databases and electronic health records (EHRs).
Method validation
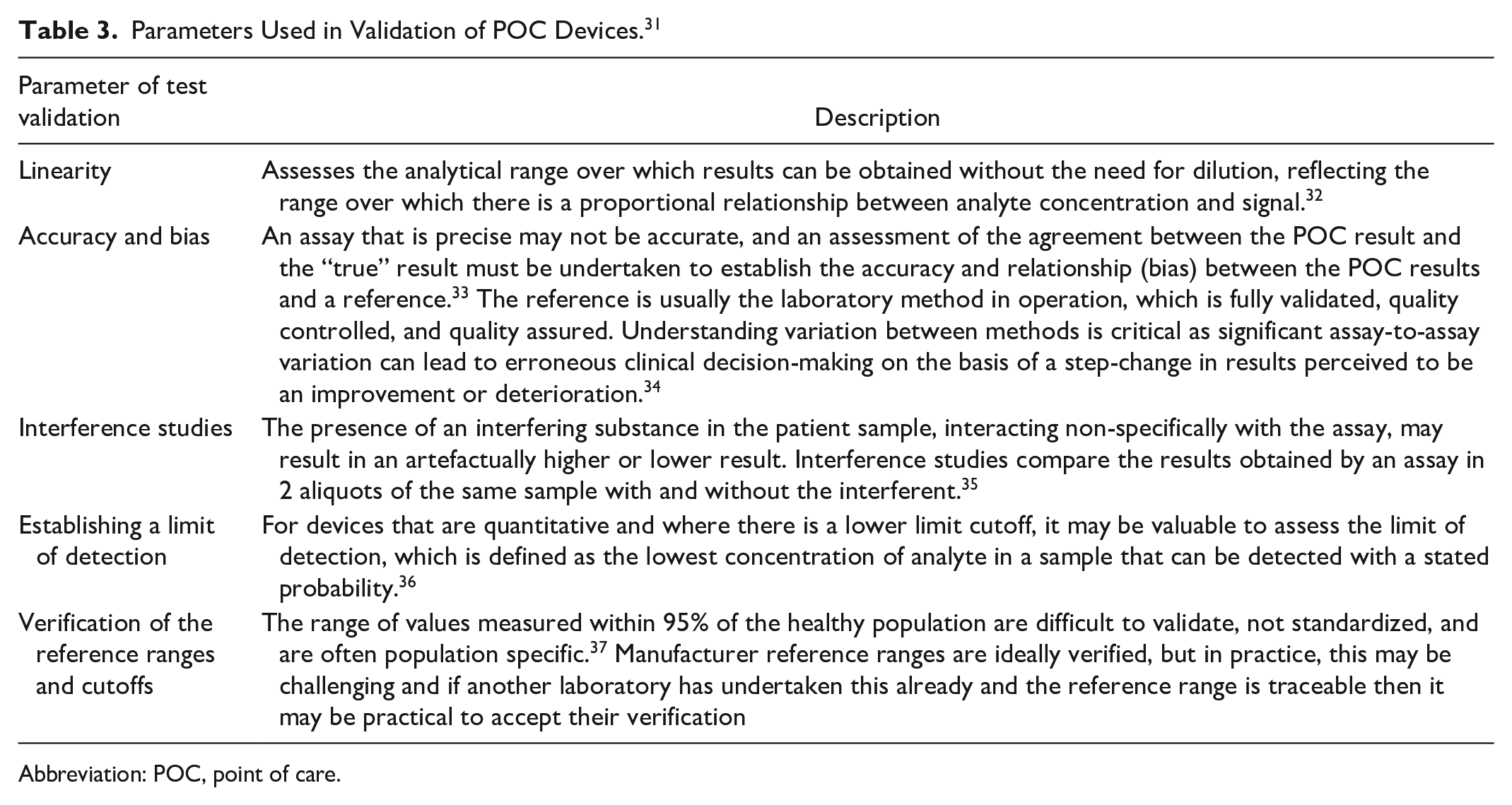
Validation experiments should assess several aspects of the device performance and are important to understand the parameters in which a POC device can be used accurately and safely (see Table 3). Validation and verification are applied to any new laboratory analytical methodology as part of formal acceptance testing but are equally applicable to POC and aim to verify the manufacturer performance specifications of the method. Providing performance specifications for in vitro diagnostics is a legal requirement that manufacturers must fulfill and for laboratories to be fully accredited, validation or verification of these specifications must be undertaken. 37
Parameters Used in Validation of POC Devices. 31
Abbreviation: POC, point of care.
Quality assurance framework
The aim of the POC quality framework is to ensure an analytical test that is removed from the laboratory environment, and undertaken by non-laboratory staff, still retains the quality aspects of the laboratory, and has safeguards in place to prevent incorrect results from being reported.
Pathology laboratories operate standards of practice that ensure quality and confidence in results reporting. These practices are benchmarked against standards outlined by accreditation services derived from international standards ISO 15189 29 and ISO 22870, 38 such as the UK accreditation service standards (UKAS). 39 The standards cover all aspects of undertaking a test from factors before a test is undertaken (pre-analytical), the analytical process itself and actions after a result is validated to being reported (post-analytical).29,40 Recognizing the expanding repertoire of POC analytes, devices, provision outside of laboratories, and potential pitfalls of incorrect implementation, the Medicine and Healthcare products Regulatory Agency (MHRA) produced guidance on the management of in vitro POCT devices. 41 The key themes are as follows:
to have a clear management structure with responsibility for delivery of the service, taken in the form of a POC committee and POC manager (Procurement of POC without input from local laboratories is not recommended.
to maintain quality of the testing process through adequate training of users, clear documentation of operating procedures and competency assessments
to maintain and evaluate quality of the analytical method through appropriate internal quality control and external quality assessment schemes
to identify and react appropriately to process failures using documented procedures
to ensure connectivity of results to electronic health care records
Connectivity
Connectivity via interfacing software (middleware) of data derived from the POC device to laboratory information management system (LIMS, the pathology laboratory database of blood results) and EHRs is a necessary component of the quality framework that, at a basic level, ensures data integrity, recording, and linkage (see Figure 1). Thus, organizations where this chain of linkage is broken may be at higher risk of clinical incidents, for example hospitals with paper-based notes and POC devices that are not connected to LIMS may require practitioners to hand-write results from the device into notes, which leaves no digital record and has a much greater potential for human error.
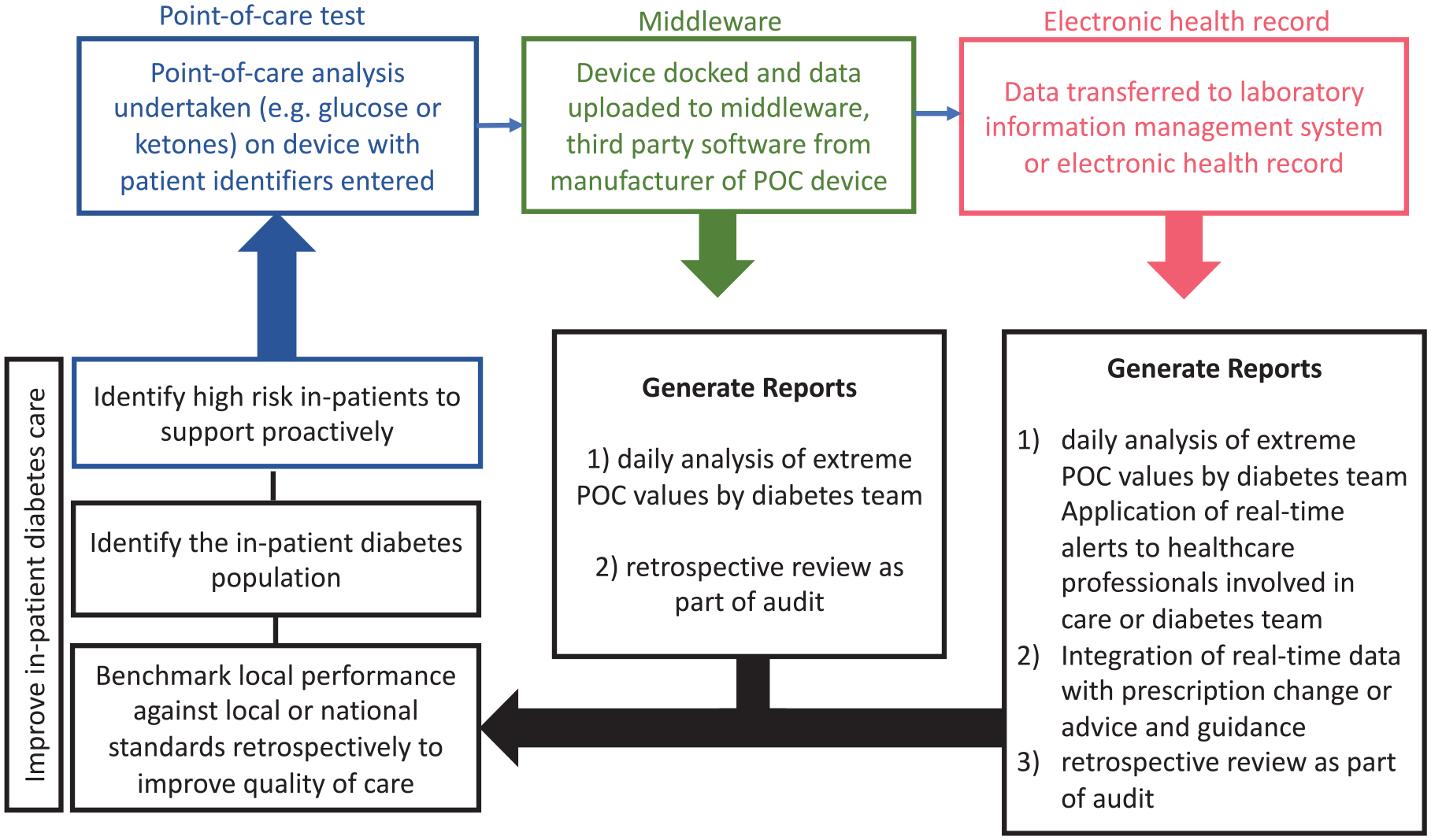
Flow chart depicting flow of results in a fully networked system for POC glucose measures and potential for generating reports.
Thus, total connectivity of the device to LIMS to EHR, or in some cases to LIMS and EHR independently, via middleware, is to be encouraged41,42.
Using Networked POC Measurements to Drive Clinical Effectiveness
While connectivity of glucose and ketone meters to the EHR is an important component of maintaining clinical governance around point-of-care results, the increasing sophistication of data interrogation in EHRs offers the tantalizing possibility of using these glucose measures in real time for patient benefit.
In broad terms, there are 2 ways in which glucose measurements in the EHR can be used. The first is in real time to identify patients who have extreme glucose values for example hyper- or hypoglycemia. Searching for these individuals proactively could then lead to input from dedicated diabetes in-patient teams. At a more rudimentary level, identification of glucose values beyond set thresholds is a useful way of identifying who has diabetes in the in-patient population, a stipulation from national drives to improve quality of care, for example in the United Kingdom, Getting it Right First Time. 43 Of course real-time proactive identification is only useful if a there are teams to identify such excursions and enough staff to make useful clinical interventions. One envisages that proactive identification may in the future limit the need for diabetes team referrals or at least streamline the pathway.
The second utility of networked measurements is for retrospective audit. In England, for example, all hospitals are mandated to submit data on in-patient diabetes harms such as in-patient severe hypoglycemia. 44 Interrogation of glucose readings and ketone readings could facilitate identification of in-patients experiencing harms or indeed excursions in glucose measurements retrospectively. These data could then be used to benchmark against audit standards but also reviewed serially to drive improvements as part of an audit cycle, in a given local service.
Is POC Connectivity Clinically Useful in Diabetes?
A seminal study first investigating the use of “networked glucose” measurements to improve clinical care was undertaken over a 3-year period 45 ; the investigators used a “glucose management system” to interrogate extreme POC glucose values on a daily basis and to make changes to insulin doses, using a virtual prescription. Using an outcome of the proportion of patient-days hyper-hypo or at target over 12 months, the study found that the proportion of patients with hyperglycemic reduced by 39% from 6.6 per 100 patient-days pre-intervention to 4.0 per 100 patient-days during the intervention. Similarly, proportion of days in hypoglycemia reduced by 36%. Similar results were obtained in other studies, showing reproducibility of the concept 46 and with different study designs, for example, an Australian study of ~10 000 blood glucose measurements was also able to identify the percentages of people experiencing hypoglycemia and hyperglycemia episodes and interrogate patterns, 47 concluding that “glucometric” analysis supported by networked glucose meters could provide detailed in-patient data and enable benchmarking for safety and patient safety of diabetes patient
Glucose outcomes are of course important but rely on timely intervention from staff. In the same study of a glucose alert system, 47 the ability of a visual real-time alert for out-of-range blood glucose measurements provided by networked blood glucose meters was investigated over 4 months and the primary outcome was documented nursing and medical staff action in response to adverse glycemia . They found an increase in clinical staff response with the glucose alert system rather than standard care and concluded that there was scope for this to improve rates of hyperglycemia in hospital setting.
The most benefit is likely to come from integrated systems that join up analysis of glucose measurements with decision support. A study that trialed a comprehensive decision support system incorporated analysis of POC glucose measurements in the EHR to identify glucose excursions and inappropriate insulin prescriptions. They found use of the clinical decision support system reduces the odds of the events studied 48 and the use of diabetes dashboards has proved helpful in some services as a tool to facilitate in-patient diabetes care during the coronavirus-19 pandemic. 49
Conclusion
Point-of-care technologies remain a critical component of the management of patients with diabetes in hospital. The need for appropriate infrastructure to support implementation of these devices is paramount as erroneous results can lead to incorrect clinical decision-making. Connectivity that ensures POC measurements are recorded in EHRs and/or LIMS is a requisite for ensuring appropriate governance but beyond this offer the ability to use these data to drive improvements. As we look forward to how POC technologies can drive improvements in in-patient diabetes care, such systems will become an inevitable standard of care, but the resource, infrastructure, and staffing to embed these as a standard will be necessary.
Footnotes
Acknowledgements
SM is funded by a Wellcome Trust career development fellowship and supported by the National Institute for Health Research (NIHR) Biomedical Research Centre at Imperial College Healthcare NHS
Abbreviations
CBK, capillary blood ketone; DKA, diabetic ketoacidosis; EHR, electronic health record; LIMS, laboratory information management system; POC, point of care.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AL has received payments for speaking and advisory boards from Insulet, Dexcom, Abbott Diabetes Care, Novo Nordisk, Sanofi, and Institutional Research Support from Abbott Diabetes Care, and Novo Nordisk. DF is the national leader for the UK diabetes care accreditation program and has received speaker honoraria from AstraZeneca, Novo Nordisk, and Sanofi Diabetes. SM is appointed to the Board of Trustees at the Diabetes Research & Wellness Foundation and is in receipt of funds from Dexcom for an investigator-initiated research study. GR has received personal fees from Abbott Diabetes Care, Sanofi Aventis, and Eli Lilly. PC has received personal fees from Abbott Diabetes Care, Dexcom, Diasend, Eli Lilly, Insulet, Medtronic, Novo Nordisk, Roche, and Sanofi Aventis. KD is the chairman of the Joint British Diabetes Societies for In-patient Care and has received speaker fees, travel or taken part in advisory boards for AstraZeneca, Sanofi Diabetes, Boehringer Ingelheim, Lilly, and Novo Nordisk. PA has no conflicts of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
