Abstract
Glycemic control in hospitalized patients with diabetes requires accurate near-patient glucose monitoring systems. In the past decade, point-of-care blood glucose monitoring devices have become the mainstay of near-patient glucose monitoring in hospitals across the world. In this article, we focus on its history, accuracy, clinical use, and cost-effectiveness. Point-of-care devices have evolved from 1.2 kg instruments with no informatics to handheld lightweight portable devices with advanced connectivity features. Their accuracy however remains a subject of debate, and new standards for their approval have now been issued by both the International Organization for Standardization and the Clinical and Laboratory Standards Institute. While their cost-effectiveness remains to be proved, their clinical value for managing inpatients with diabetes remains unchallenged. This evidence-based review provides an overall view of its use in the hospital setting.
In the United States, the annual estimated cost of treating diabetes in 2012 was $245 billion, a 41% increase from 2007. The largest component (43%) related to inpatient care. 1 A 5-country EU study estimated that in 2010, the total direct costs of care for people with diabetes were highest in Germany (in part due to the greater diabetes population) at €43.2 billion, followed by the UK (€20.2 [£13.8] billion), France (€12.9 billion), Italy (€7.9 billion) and Spain (€5.4 billion). In all countries, the majority of these costs were related to inpatient care—33.7%, 58.2%, 37.2%, 56.9%, and 35.8%, respectively. 2
It is now quite clear that both hyperglycemia and hypoglycemia in hospitals are associated with poorer outcomes such as increased morbidity, mortality and length of stay.3-11 Due to the promise of early trials in critically ill patients,12,13 tight glycemic control (TGC) regimens in hospitals were endorsed by a number of professional organizations including the American Diabetes Association (ADA). 14 Subsequent studies and meta-analyzes of randomized controlled trials of TGC in critically ill patients15-17 have generated conflicting results with evidence pointing to higher mortality rates possibly attributable to higher hypoglycemic rates. As a result, the ADA now recommends a glucose range of 140-180 mg/dl (7.8-10 mmol/l) in critically ill inpatients and premeal glucose target <140 mg/dl (7.8 mmol/l) with random glucose target <180 mg/dl (10 mmol/l) in insulin treated non–critically ill inpatients, and less stringent targets for those with severe comorbidities. 18
In hospitals, control of blood glucose (BG) in narrow therapeutic ranges requires appropriate near patient glucose monitoring techniques that need to be rapid, accurate and cost-effective. The reference standard remains central laboratory testing (CLT) of plasma glucose. However, its remote location and high turn-around time makes point-of-care testing (POCT) for BG an ideal alternative. POCT also known as extralaboratory or near-patient testing has been defined as testing that is performed near or at the site of a patient with the result leading to a possible change in the care of the patient. 19 It is now the standard bedside glucose monitoring technique in most institutions.
The first use of POCT is found in papyrus documents dating back to 1550 BC, which depict Egyptian physicians using ants to determine glycosuria in patients suspected of having diabetes. Jules Maumene was the first to develop a simple urine reagent “strip” in 1850 made of sheep’s wool containing stannous chloride. 20 In 1883, George Oliver, published “Bedside Urine Testing” and promoted reagent papers for testing urine sugar. 21 Subsequently in 1908, Stanley Benedict devised an improved copper reagent, which with modifications became the mainstay of testing urine sugar in diabetes for over 50 years. 22 In 1957, the pursuit for a more convenient method culminated in a “dip and read” urine reagent strip, Clinistix, 23 and Kohn illustrated that it could also give approximate results for BG. 24
The major breakthrough came in 1965 when an Ames research team under Ernie Adams developed the first BG test strip, the Dextrostix, a “visually interpreted” paper reagent strip that used the glucose oxidase (GO) reaction. 25 At the same time, Boehringer Mannheim, a German company developed another “visually interpreted” BG test trip called Chemstrip bG. Later Anton Clemens at Ames developed an instrument, the Ames Reflectance Meter, to produce quantitative BG results from the Dextrostix; this became available in 1970. 26 The Ames Reflectance Meter weighed 1.2 kg, cost approximately $495 and was available only in clinics and emergency departments, thus heralding the beginning of the era of POCT for BG for clinicians but not for patients. However, over the next 40 years, technology evolved into the small, lightweight, portable meters (as small as to weigh only 9 grams) for use by patients and at the hospital bedside. POCT for BG (both hospital and outpatient use) now occupy the lion’s share of the global multibillion-dollar POCT market. 27 In this article, we review POCT for BG in hospitals from a clinical, technical and economic point of view by providing narrative evidence-based synthesis of the literature.
Advantages and Disadvantages of Point-of-Care Testing for Blood Glcuose in Hospitals
Turn-Around Time
Turn-around time refers to the total period from a clinician’s issuance and delivery of order, collection, transportation and analysis of specimen and calculation, verification and delivery of results to review of results. The first and foremost and undisputed advantage of POCT for BG is the rapid turn-around time compared to CLT.28-30 Turn-around time is often <5 minutes with the modern point-of-care BG meters, whereas with CLT it is still 30-60 minutes. This is crucial for physicians and nurses in common hospital scenarios such as hypoglycemia management, adjustment of intravenous insulin infusion and determining insulin dosages in basal bolus regimens. Reliance on laboratory measurements with prolonged turn-around times can adversely affect patient outcomes and render the reason for the test meaningless as they then do not reflect the current status of the patient. While the turn-around time for CLT can be significantly reduced with satellite laboratories, it still would not match that of POCT.31-33
Patient Sample Volume
Point-of-care BG meters require very small volumes of blood for analysis (0.3-1 µL), in contrast to CLT which require 1-3 mL of blood. 34 This is important because repeated venesection is expensive and can render hospitalized patients anemic.35-37 The mean volume of blood loss per patient per admission due to phlebotomy for CLT is between 175 and 200 mL and for those also admitted to intensive care units (ICU) is between 600-700 mL.34-36 In contrast, the mean volume of blood loss per patient per admission due to POCT for BG is only 0.15 ml. 34 Therefore there is a clear advantage in using POCT for BG over CLT for blood conservation. The importance of blood conservation in inpatients cannot be understated as mounting evidence shows that both anemia and repeated blood transfusions are associated with poorer outcomes in critically ill patients. 37
Preanalytical and Postanalytical Factors
Preanalytical factors are independent factors prior to patient sample analysis that affect the outcome of the subsequent analysis where as postanalytical factors influence the outcome of analysis after patient sample testing is completed (Table 1). As a general rule, the greater the number of steps involved in processing of patient test results, the higher the chances of both preanalytical and postanalytical errors. Transportation, multiple user handling, order verification and delayed reporting are all eliminated with POCT. Therefore, it would appear that CLT is more likely to cause both more preanalytical and postanalytical errors compared to POCT.
Preanalytical and Postanalytical Factors That Affect Test Results.
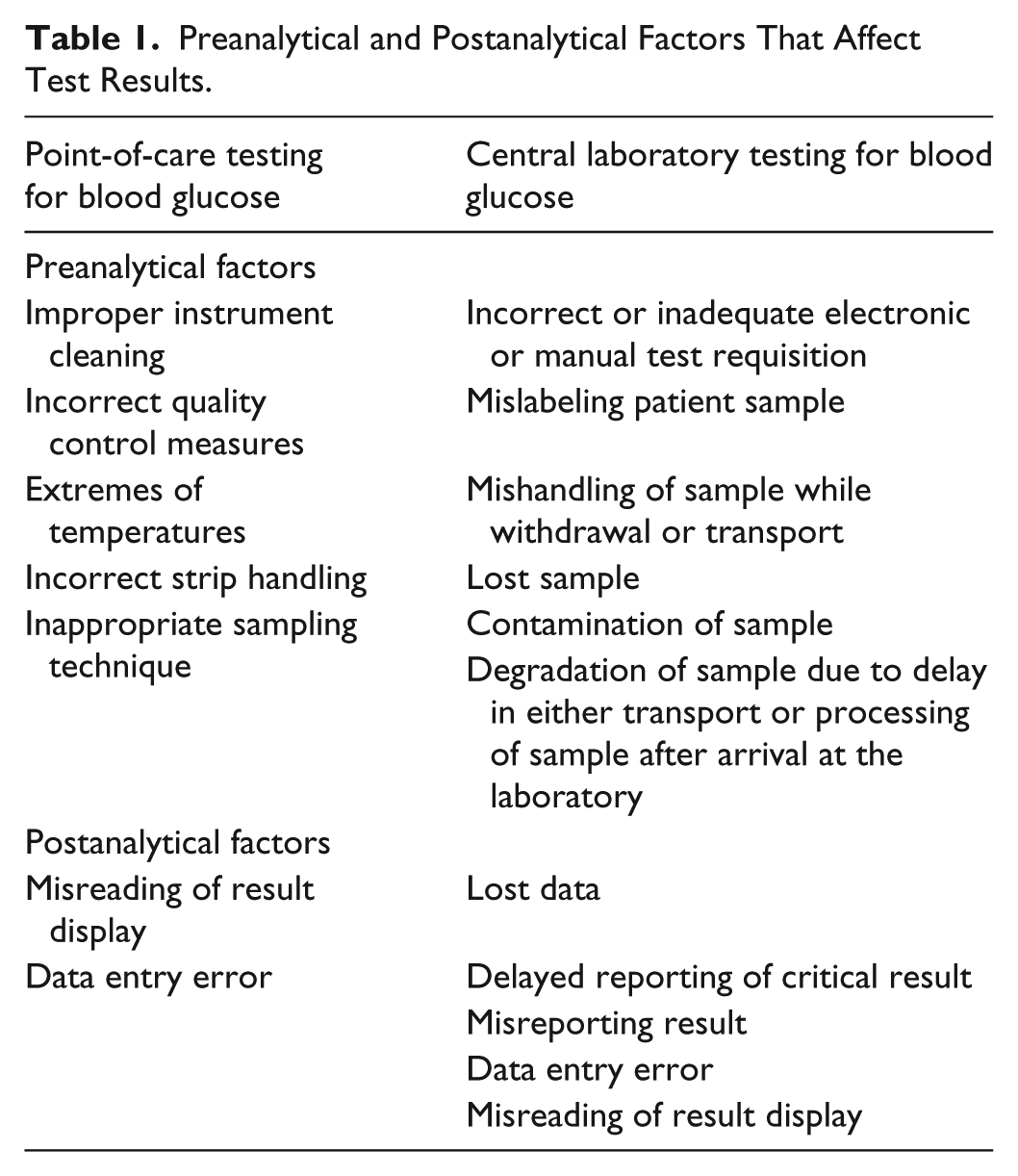
Summaries of error frequencies in CLT have shown that majority of errors were in the preanalytical phase (46-68%) compared to the postanalytical phase (18-47%). Errors in the analytical phase (7-13%) were minimal.28,38 Conversely, the literature is abundant with reports of analytical errors (discussed below) with only few reports of preanalytical and postanalytical errors due to POCT for BG; operator error being far the commonest.39-43 This is because the manufacturers have built into the newer point-of-care BG meters, a number of control processes to minimize the probability of preanalytical and postanalytical errors and to detect errors should they occur. They include bar-coded strips that prevent use after expiry date and prevent incorrect calibration, individually wrapped test strips to prevent reagent exposure due to failure to recap bottles of test strips, detection of correct blood volume to prevent errors due to insufficient blood volume, software operator identification lockout to prevent unauthorized and untrained user access, positive patient identification such as ability to scan bar-coded patient wrist bands to prevent incorrect patient identification, and internal quality control checks like optimal operational temperature range, and so on. 41
Analytical Factors
The accuracy and precision of point-of-care BG meter results in hospitalized patients with diabetes (especially critically ill patients) have been a subject of major debate; in particular the extent to which patients can be harmed from inaccurate results.39,44-47 Unfortunately, as of today, there is no internationally recognized gold standard reference method for the measurement of BG. Plasma glucose assayed in CLT is the reference method usually employed. Almost all point-of-care BG meters use whole blood for measurement as it is impractical to use plasma at the bedside. To avoid clinical misinterpretations, the International Federation of Clinical Chemistry and Laboratory Medicine (IFCC) has recommended that all point-of-care BG meters report in plasma values using a constant factor of 1.11 for conversion. 48 This formula assumes that the patient has a hematocrit of 43%, a plasma concentration of 93% and a red blood cell water concentration of 71%. All these vary significantly in the inpatient population and from day to day in individual inpatients and therefore inherently become a potential source for error. Most point-of-care BG meters now report only in plasma glucose values except a few, for example, HemoCue B glucose analyzer.12,13 Therefore, hospitals using POCT for BG monitoring should set targets that reflect plasma rather than whole BG to reduce risk of hypoglycemia.
All current glucose measurement systems, including CLT and POCT use indirect enzymatic techniques.44-47 Three enzymatic techniques are in use: namely hexokinase (HK), GO, and glucose-1-dehydrogenase (GDH).44-47 The majority of CLT use HK (some use GO) and is the reference method in most clinical trials due to its higher accuracy. This method has not been used in point-of-care BG meters as HK is less stable than GO or GDH for use in test strips that need to be stored for extended periods of time. While GO is the more classic methodology developed in point-of-care BG meters, a major drawback is that it is susceptible to extremes of hydration or oxygenation. Hyperoxia can underestimate glucose and hypoxia can falsely elevate glucose readings. This is potentially hazardous, especially in critically ill patients. There are 3 subtypes of GDH methods based on the cofactor used—nicotine adenine dinucleotide (NAD), pyrroloquinolinequinone (PQQ), and flavin adenine dinucleotide (FAD). The GDH methods also have its limitations in that the enzyme is less specific for glucose than GO/HK methods and devices using GDH methods can yield falsely high BG readings in the presence of non glucose sugars such as maltose, xylose, or galactose that can be misinterpreted as glucose. On the other hand they are less prone to interference with exogenous substances and blood oxygen compared to GO based systems.
Icodextrin used as a dialysate can cause major problems with systems using GDH-PQQ. During peritoneal dialysis, it enters the blood stream and is hydrolyzed by alpha-amylase to maltose, maltotriose, maltotetraose, and other nonglucose sugars. Point-of-care BG meters using GDH-PQQ can incorrectly interpret these as glucose. 49 From 1997 to 2009, Food and Drug Administration (FDA) received 13 reports of death associated with GDH-PQQ systems with documented interference from nonglucose sugars. Subsequently FDA issued a public health notification in 2009 advising against the use of GDH-PQQ systems for BG monitoring in patients receiving therapeutic nonglucose sugars. 50 The FDA website also lists other products containing nonglucose sugars that can interfere with the GDH-PQQ systems along with the list of test strips that employ this system. 50 The manufacturers of these strips have issued warnings about the interfering sugars. Despite this, there continue to be reports of fatal hypoglycemia and some have called for complete elimination of use of these systems. 51 In 2011, 1 study also reported potential interference between GDH-NAD system and nonglucose sugars but of far less significance than the GDH-PQQ system. 52
Depending on the system and sample used, a number of confounding variables in glucose measurement exist in inpatients (Table 2) including hematocrit, medications, pH, sample type, hemodynamic status, and so on.44-46 It is important to be aware of these variables as they become significant when aiming for TGC. A detailed discussion of these confounding variables, which have been extensively reviewed elsewhere, is beyond the scope of this article.44-47,53 To summarize, arterial and venous BG results from point-of-care BG meters reflects reference plasma glucose more than does capillary BG. The greatest accuracy is when arterial blood is used in arterial blood glucose (ABG) analyzers. Hemodynamic instability, hypoglycemia, and insulin infusion therapy resulting in rapidly changing glucose levels markedly increase the risk of errors in results from point-of-care BG meters. Caution should therefore be exercised when capillary blood is used for testing in point-of-care BG meters for achieving TGC in critically ill inpatients; indeed it could be argued that only arterial blood in ABG analyzers be used in these circumstances.
Confounding Variables in Glucose Management.
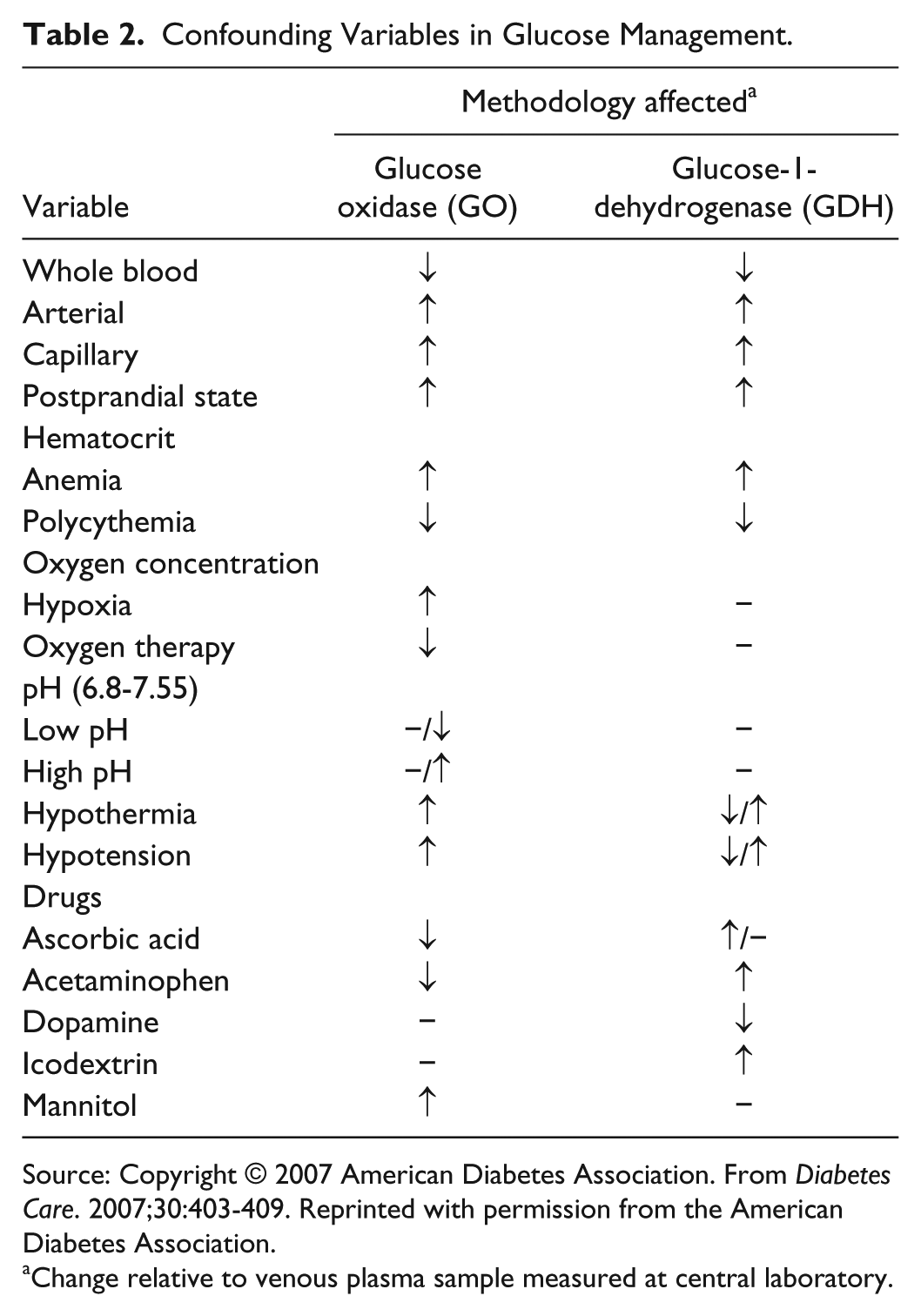
Source: Copyright © 2007 American Diabetes Association. From Diabetes Care. 2007;30:403-409. Reprinted with permission from the American Diabetes Association.
Change relative to venous plasma sample measured at central laboratory.
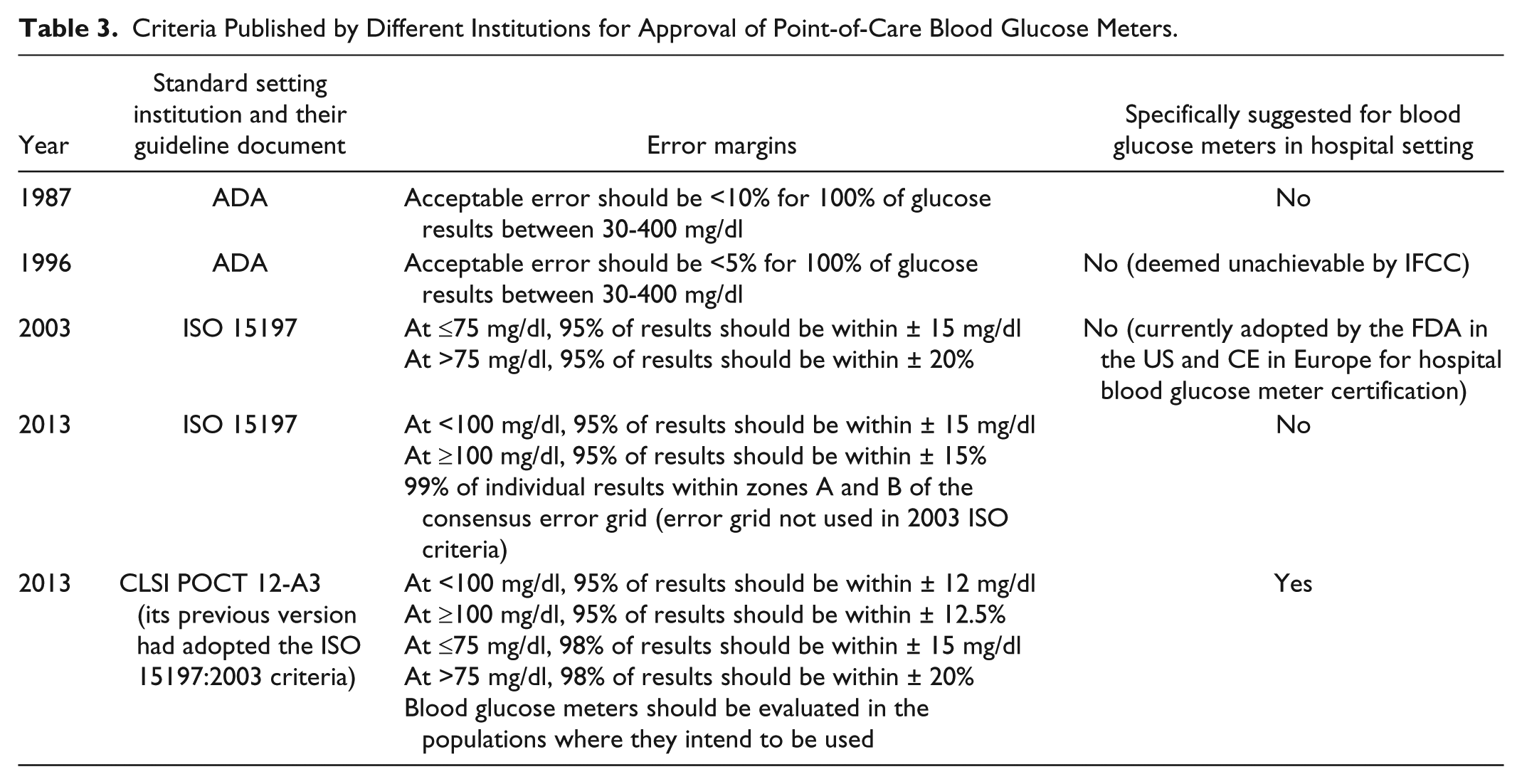
Table 3 summarizes the major published standards for point-of-care BG meters over the years54-56 and the new 2013 published updates.57,58 Significant concerns were expressed with the widely accepted and approved 2003 standards in which the margin for error is large. For example, with the International Organization for Standardization (ISO) 15197:2003 standard, the BG meter could receive approval even though up to 1 clinically unacceptable result may occur once in every 20 measurements. 59 Of greater concern was once approved (by Communauté Européenne [CE] in Europe or FDA in the United States) many of the existing meters (both hospital and outpatient use) failed to maintain the original standard.60-65 Most clinicians and patients are unaware of this. In 2011, 45 expert diabetes technology physicians of the Diabetes Technology Society (DTS) echoed a widely held view that a new FDA approval standard for BG meters was needed to improve patient safety. 66 In 2013, both new ISO and Clinical and Laboratory Standards Institute (CLSI) guidelines were issued. The CLSI standards include more stringent criteria for hospital BG meters where as the ISO standards do not specify this. These new guidelines have not yet been adopted for evaluating hospital BG meters. The European Association for the Study of Diabetes (EASD), ADA, and DTS all agree that more stringent standards are required for evaluating BG systems as well as postmarketing device performance surveillance.67-69 The FDA and CE recognize the need for improved standards and are considering the issue.
Criteria Published by Different Institutions for Approval of Point-of-Care Blood Glucose Meters.
Cross-Infection
One of the concerns with POCT for BG in hospitals is the risk of cross infection including Hepatitis B.70,71 Hospital acquired infection significantly increases length of stay and cost of hospitalization.72,73 Louie et al in a multicenter study across 12 hospitals reported that the prevalence of blood contamination was 30.2 ± 17.5% in non-ICU BG meters and 48.2 ± 30.2% in ICU BG meters; an increase of 100 operators increased the odds of contamination by 6%. 74 The prevalence of bacterial contamination in unused test strips in opened vials (not individually foil packaged) ranges from 25.7 to 80%.75-77 This includes highly pathogenic organisms like methicillin-resistant Staphylococcus aureus and Staphylococcus hemolyticus. 77 However test strips that are individually foil packaged have very low prevalence of bacterial contamination of 3-7%, similar to that of newly packaged strips at the factory.75,77
Reasons for cross infection include operator errors such as lack of sufficient disinfection. The FDA and Centers for Disease Control and Prevention (CDC) recommend that point-of-care BG meters should not be shared between patients in hospital. If dedicating individual point-of-care BG meters to a single patient is not possible, the devices should be properly cleaned and disinfected after every use as described in the device labeling. 78 CDC also recommends that unused supplies taken to a patient’s bedside for BG monitoring should not be used for another patient because of inadvertent contamination. 79 Therefore, staff using point-of-care BG meters should handle test strip vials with clean gloves, designate each vial of test strips to a single patient and discard any remaining unused strips. However 1 study estimated the annual additional cost of dedicating a vial of glucose test strips ($0.50 per strip) to each patient and discarding unused strips in opened vials at $83 350 from 25 strip vials and $202 940 from 50 strip vials. 80 This has enormous financial implications for any hospital. It should be more economical to use individually foil packaged test strips but there are no studies to show this.
Staff Training and Quality Control
Management of training and competency for staff using point-of-care BG meters is no easy task especially in large institutions where the number of personnel to be trained may number in the thousands. Each institution must therefore develop its own training module, often with support from the manufacturer. From our experience we suggest that this also includes a program for retraining at regular 6- to 12-month intervals, especially in units of low-volume POCT. Most point-of-care BG meters require regular quality control checks. They are designed such that the device/software will not permit use unless it has been performed and the results are in range. The frequency of quality control checks are outlined by regulatory authorities for that country in addition to the manufacturer’s guidelines. 81
Connectivity
Achieving connectivity was hailed as the “millennium challenge for point-of-care testing.” 82 Then, point-of-care BG meters in hospital did not interface with computerized hospital information systems (HIS) such as the laboratory information systems (LIS) and electronic medical records (EMR). Only a small fraction of data was captured electronically. To find solutions to this problem, the Connectivity Industry Consortium (CIC) was formed in 2000 by the Industry Liaison Committee of the American Association for Clinical Chemistry (AACC). Currently the CLSI has taken over as the chartered standards maintenance organization. In 2001, the POCT1-A standard was published in the US that was then jointly upgraded by both Europe and US in 2005 to POCT-01-A2. 83 These standards are now mandatory by regulatory authorities in many countries.
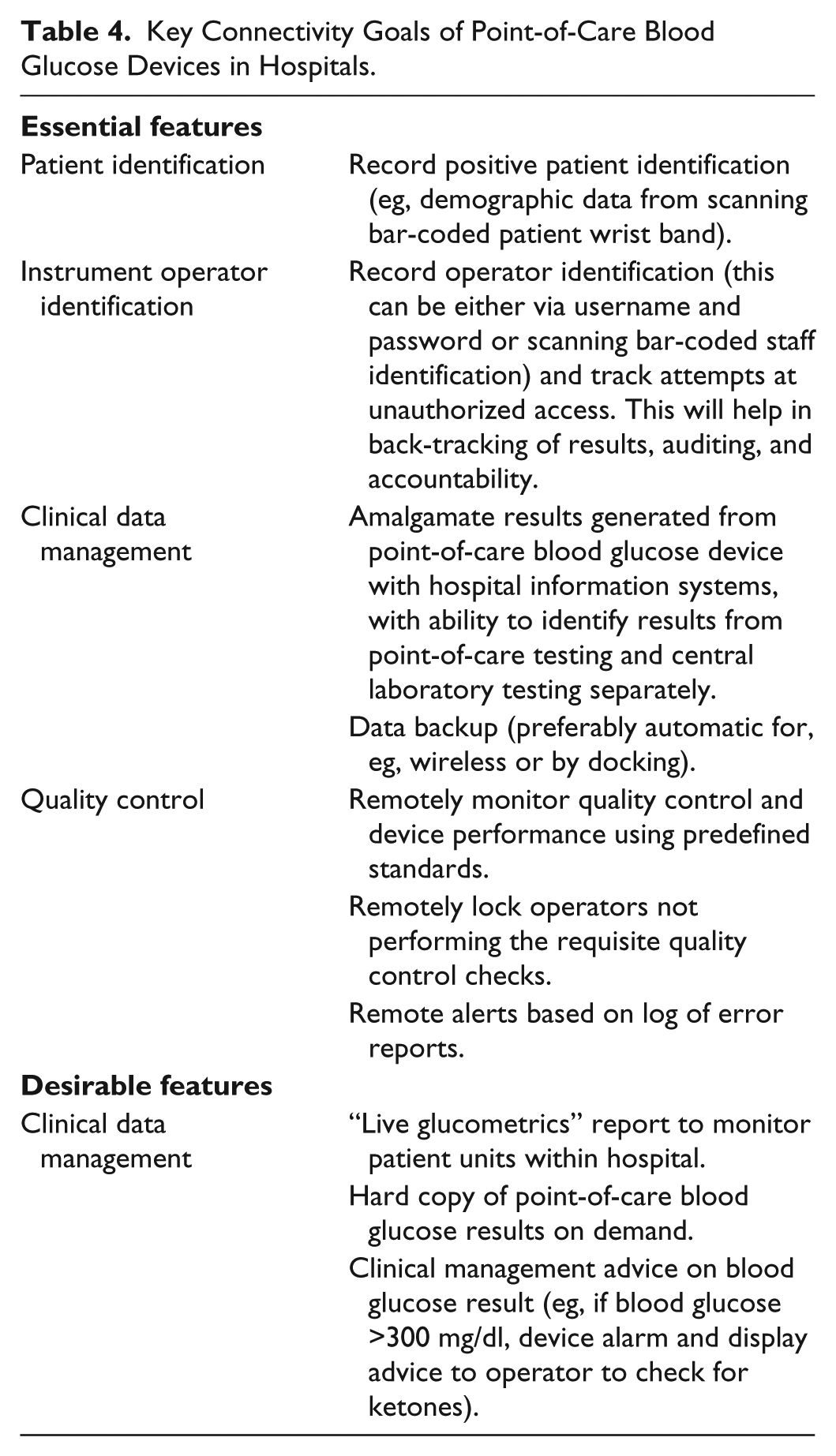
The key connectivity goals are summarized in Table 4. Connectivity is the key to hospital point-of-care BG management programs including aspects such as quality control, identification of testing personnel, and postanalytical transfer of results to HIS. It can help in reducing medical errors, improving staff compliance, showing that regulatory requirements are met and track wasted supplies.32,82 Importantly, it can also be cost effective to the institution. Salka et al showed that by simply electronically linking point-of-care BG meter data to HIS, it resulted in a total annual saving of $119 095; the greatest cost saving was achieved by eliminating the time required for nursing staff to manually enter results into HIS. 84 Currently, fourth-generation point-of-care BG data management systems are available that connect seamless and bidirectionally with wireless enabled point-of-care BG meters. 85
Key Connectivity Goals of Point-of-Care Blood Glucose Devices in Hospitals.
Novel Applications of Point-of-Care Connectivity
In 2006, Goldberg et al were the first to report hospital glucometrics (standardized glucose performance metrics to assess quality of inpatient diabetes care) using electronic point-of-care BG data (EPGD) that aimed to objectively compare hospitals and patient care units within hospital. 86 Subsequently, other glucometrics have been reported using EPGD, some of which were used to demonstrate national benchmarking and compare year-on-year trends.87-90 Boaz et al used EPGD to assess the impact of their institution wide intervention program for patients with diabetes. 89 A recent systematic analysis of clinical decision support systems (CDSS) that included EPGD concluded that there was consistent evidence that it improved glucose control in inpatients. 91 The authors however acknowledged that most of the studies were of suboptimal quality and that it was difficult to attribute the effect solely to CDSS alone. 91 In our institution, EPGD was used to demonstrate diurnal patterns of hypoglycemia in hospitalized patients with diabetes. 92
Cost-Effectiveness
The most controversial issue in POCT for BG in hospitals is its cost-effectiveness which to date has not been demonstrated, probably because there is no effective way to assess this. In 2004, the Q-Probes study (that enrolled 445 institutions from across the United States) by the College of American Pathologists compared the analytical costs of CLT versus POCT for BG. 30 The median costs per BG test were $1.18, $1.96, and $4.66 for CLT, high-volume POCT, and low-volume POCT sites, respectively. The largest percentage of the cost per test was for labor (59.3%, 72.7%, and 85.8%). It was concluded that the analytical cost per BG test were lower for CLT, with the cost of POCT being highly variable and dependent on volume. Other studies have also shown that costs of POCT for BG can exceed CLT costs from 1.1 to 4.6 times.93-96 It is expected that when POCT for BG is introduced, CLT for BG would decline. However, 2 different studies reported an increase of 10% 93 and 18% 97 in the number of BG tests performed in central laboratories after a hospital wide POCT program was established despite similar patient demographics. The reasons for this were unclear.
Caution must however be exercised when interpreting studies comparing the cost effectiveness of POCT versus CLT. While the cost of equipment, supplies and initial training are more accurately factored, others like cost of labor, changes in productivity, impact of reduced sample volume for POCT, impact of turn-around time and accuracy of results and costs of ongoing training have not been well accounted.30,98 In 2006, The National Academy of Clinical Biochemistry, the Academy of the AACC concluded that there was insufficient evidence on whether POCT for BG in secondary care setting leads to an improvement in clinical outcomes compared to CLT and therefore were unable to recommend for/against routinely using POCT for BG in hospitals from the clinical point-of-view. From a purely economic point of view, since there was no published evidence that strongly favored economic benefit of POCT for BG compared to CLT, it recommended against its routine use in the hospital setting. 99
From all the published studies, it appears that as the number of point-of-care tests for BG increases it becomes more cost effective. In this regard, in today’s hospitals the volumes of POCT has increased tremendously compared with the volumes reported in the Q-Probes study, with a medium sized 500 bedded NHS hospital such as ours performing approximately 120 000-150 000 bedside BG tests annually; 16-20 times higher than the median of the high volume POCT sites in the Q-Probes study. 30 In this situation, the costs of POCT versus CLT may be quite different. Data used for financial rationalization of point-of-care BG meters in hospital are widely aberrant and more robust studies are needed before judging its cost-effectiveness.
Future Prospects in Point-of-Care Blood Glucose Monitoring of Hospitalized Patients With Diabetes
POCT for BG has become synonymous with tests performed with BG meters at the bedside. We provide a brief outline of other techniques that can potentially provide point-of-care BG results in the inpatient setting. A detailed review is beyond the scope of this article.
Continuous Glucose Monitoring
The usefulness of continuous glucose monitoring (CGM) systems in inpatients, especially in ICU where TGC is targeted has been a subject of considerable debate due to conflicting reports on its impact in hospitalized patients. Using CGM, Holzinger et al reported an absolute risk reduction of severe hypoglycemia in patients receiving assisted ventilation by 9.9% with a number needed to treat of 10.1 compared to point-of-care BG meters with no difference to mean glucose control. 100 Despite some reports that CGM is accurate and reliable in critically ill patients,101-103 this has not been consistently demonstrated.104-106 Besides, issues such as calibration, electrode wetting and signal loss have been reported of which calibration is a major source of error.104,105,107 There are limited data in hospitals and ICU on the effects of interference from common clinical conditions such as hypoxia, hypotension, anemia, and others on the accuracy of CGM, especially when calibrated with point-of-care BG meters. Therefore, both the Endocrine Society and the American Association of Clinical Endocrinologists have currently recommended against the use of CGM in hospitals and ICU, especially as the sole BG monitoring system.108,109
Noninvasive Glucose Monitoring
In 1982, Rabinovitch et al reported the first noninvasive glucose monitoring (NGM) device by determining BG levels from optical rotation of aqueous humor with sclera lens. 110 However, 30 years on, there is still no device in clinical practice in any setting, hospital or outpatient use. Devices such as Pendra© and OrSense NBM-200G, although CE approved, have not been commercialized so far. 111 A number of challenges lie ahead in the development of NGM such as cost, sensitivity, analytical time, accuracy, precision, signal-to-noise ratio, calibration, and skin tolerability.111,112 It is likely to be several years before commercially viable NGM devices are used in hospitalized patients with diabetes.
Conclusions
Pascal’s Wager is an argument based on probability and game theory devised by the 17th-century French philosopher, mathematician and physicist Blaise Pascal. It suggests that even though the existence of God cannot be determined through reason, a person should gamble that God exists because he/she has everything to gain and nothing to lose. This can be applied to the existing point-of-care BG meters in hospitals. While POCT for BG has become the mainstay for monitoring and decision making in the management of diabetes in inpatients, it cannot replace CLT for precision and accuracy. Point-of-care glucose testing is user friendly with rapid turn-around times and requires only a small sample volume. Advanced connectivity features now make it possible to define hospital glucometrics for benchmarking and assess impact of intervention programs. However, its cost-effectiveness remains to be proved. Newer technologies such as CGM hold promise in the near future but further research is needed in this area.
Footnotes
Abbreviations
AACC, American Association for Clinical Chemistry; ABG, arterial blood glucose; ADA, American Diabetes Association; BG, blood glucose; CDC, Centers for Disease Control and Prevention; CDSS, clinical decision support systems; CE, Communauté Européenne; CGM, continuous glucose monitoring; CIC, Connectivity Industry Consortium; CLSI, Clinical and Laboratory Standards Institute; CLT, central laboratory testing; DTS, Diabetes Technology Society; EASD, European Association for the Study of Diabetes; EMR, electronic medical records; EPGD, electronic point-of-care blood glucose data; FAD, flavin adenine dinucleotide; FDA, Food and Drug Administration; GDH, glucose-1-dehydrogenase; GO, glucose oxidase; HIS, hospital information systems; HK, hexokinase; ICU, intensive care units; IFCC, International Federation of Clinical Chemistry and Laboratory Medicine; ISO, International Organization for Standardization; LIS, laboratory information systems; NAD, nicotine adenine dinucleotide; NGM, noninvasive glucose monitoring POCT, point-of-care testing; PQQ, pyrroloquinolinequinone; TGC, tight glycemic control.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
