Abstract
Introduction:
INSULIA is an insulin-titration app developed for patients with type 2 diabetes treated with basal insulin as part of a basal insulin-supported oral therapy (BOT). The app uses patient-logged fasting blood glucose (FBG) values and a titration plan defined by the treating physician to provide basal insulin dosing recommendations. Physicians use the web portal to monitor their patients’ therapy progress and, if necessary, adjust therapy. The aim of this study was to assess the app, specifically its features, handling and impact on diabetes treatment and self-management in Germany.
Methods:
This German retrospective pilot study included physicians (diabetologists, general practitioners, and internists) and patients with type 2 diabetes who either receive or start BOT using the app. Both groups completed group-specific questionnaires between December 2018 and June 2019.
Results:
Overall, 10 physicians and 34 patients with type 2 diabetes completed their respective questionnaires. Physicians perceived their app-using patients to be more involved and more confident in managing their insulin therapy than patients not using the app. The majority of patients considered the app as a tool that assists with safer insulin treatment. The physicians perceived that due to the app use, FBG and HbA1c target values were achieved more often than patients not using the app and the number and severity of hypoglycemic episodes was reduced.
Conclusion:
The titration app seems to have a positive impact on BOT patients’ FBG and HbA1c target achievement and was highly appreciated by both physicians and patients alike.
Introduction
Type 2 diabetes mellitus is a progressive disease which is initially commonly treated with metformin either alone or in combination with other antidiabetic drugs. 1 From a specific disease stage, insulin supplementation is needed. According to guidelines, a long-acting basal insulin is added to the existing antidiabetic treatment, an escalation which is known as basal insulin-supported oral therapy (BOT). 2 Despite treatment intensification, glycemic control still remains challenging due to various factors including hesitant insulin titration and patient compliance.3-5
The titration period—where the insulin dose is step-wise adapted to reach a fasting blood glucose (FBG) target—is crucial for optimizing diabetes treatment, which, in turn, minimizes long-term complications. 3 ,6-10 However, there are no structured recommendations/guidelines for insulin titration, as multiple factors demand patient-specific, individualized algorithms.
INSULIA is an insulin-titration app developed by Voluntis and tested in a pilot study in Germany by Sanofi. It is a Conformité Européenne (CE)-certified medical device for patients with type 2 diabetes who receive long-acting (basal) insulin as part of their BOT. This app is indicated for use with insulin detemir (Levemir 100 Units [U]) once- or twice-daily, insulin degludec (Tresiba 100 [U]) once-daily and insulin glargine (Lantus 100 U, Toujeo 300 U, and Basaglar 100 U) once-daily. The app should not be used for basal dose recommendations with intermediate-acting insulin (Neutral Protamine Hagedorn) nor with premixed insulin.
The benefit of applications on the efficacy of managing diabetes parameters has been previously reported. 11 This study collected data from physicians and patients on the app’s features, handling, and impact on diabetes treatment.
Methods
Study Design
The study was a retrospective pilot study (October 2018 to September 2019), which included an online evaluation of the insulin-titration app by physicians and patients completing specific questionnaires between December 2018 and June 2019. Patients were from Hessen and Brandenburg, Germany, and were included in the study if they had type 2 diabetes that was being treated with BOT and had email access (from a mobile, personal computer, or other mobile device). Participating physicians comprised diabetologists, general practitioners, and internists experienced in treating patients with type 2 diabetes. The study complied with the Declaration of Helsinki and written informed consent was obtained from all participating patients.
App Overview
The basal insulin self-titration app can be used with any once-daily basal insulin. It is available as a mobile app or via a web portal, 12 and is a prescription-only medical device that is CE marked (risk class IIb).
The app is based on an integrated basal insulin value calculator and provides real-time insulin dose recommendations by taking into account the patients’ data and the treatment plan. Personal data (patient’s name, date of birth, gender, and contact details), disease-related data (eg, patient weight and last HbA1c reading), and the prescribed insulin therapy (including the dose per injection) are collected for each patient.
The app was activated during a patient clinic visit by the treating physician through the web portal with either a standard titration algorithm or an individualized treatment plan. The physician determines how often the insulin dose should be adjusted (eg, every 7 days), the maximum increase per adjustment (eg, 2 units [U]), the maximum daily dose (eg, 0.5 U/kg), and an automatic reduction of 20% in the insulin dose if the blood glucose (BG) level drops below 70 mg/dL. No limits are coded into the app’s software; the prescribing physician can combine the appropriate titration increments and frequency as needed for the patient. After logging these settings, the patient receives an automated email with login credentials to access the app.
The app recommends basal insulin doses according to this plan and to the logged blood glucose values. The patients enter their FBG and information on any hypoglycemic events on a daily basis. For each insulin application, an individual dose is calculated and the patient decides whether or not to use the recommended dose. The insulin dose that was injected needs to be documented in the app. Images of the app and associated screens are available from www.insulia.com. The app identifies missing data entries and/or missed insulin injections and will notify the physician who can contact the patient if necessary. Furthermore, a premature adaptation of the treatment plan is possible and the dose-adjustment function can be blocked by the physician if required.
Data Collection and Analysis
Patients and physicians were asked to complete two different electronic questionnaires concerning their demographic data and their experiences with the app (questionnaires: Supplementary file 1 and 2). Physicians filled out the questionnaire based on their experience of patient treatment. The collected data were analyzed descriptively. Values are stated as absolute values and percentages.
Results
Study Participants: Physicians and Patients
Overall, 10 physicians and 34 patients with type 2 diabetes mellitus on BOT completed study questionnaires. Most physicians were men (80%), half were 50 to 59 years old, 60% were diabetologists (n = 6), 30% internists (n = 3), and 10% general practitioners (n = 1). Physicians reported that they treat 150 to 4000 patients with diabetes in one quarter of a year. Assessments of the 10 participating physicians were based on their experience with a total of 112 patients who were supported with the app. Three physicians supervised >59% (n = 20) of the patients using the app. In six practices additional staff (physicians [n = 2; 20%], diabetes consultants [specially trained, non-physician staff that guide patients with their treatment; n = 3; 30%], and study nurse [n = 1, 10%]) managed the web portal.
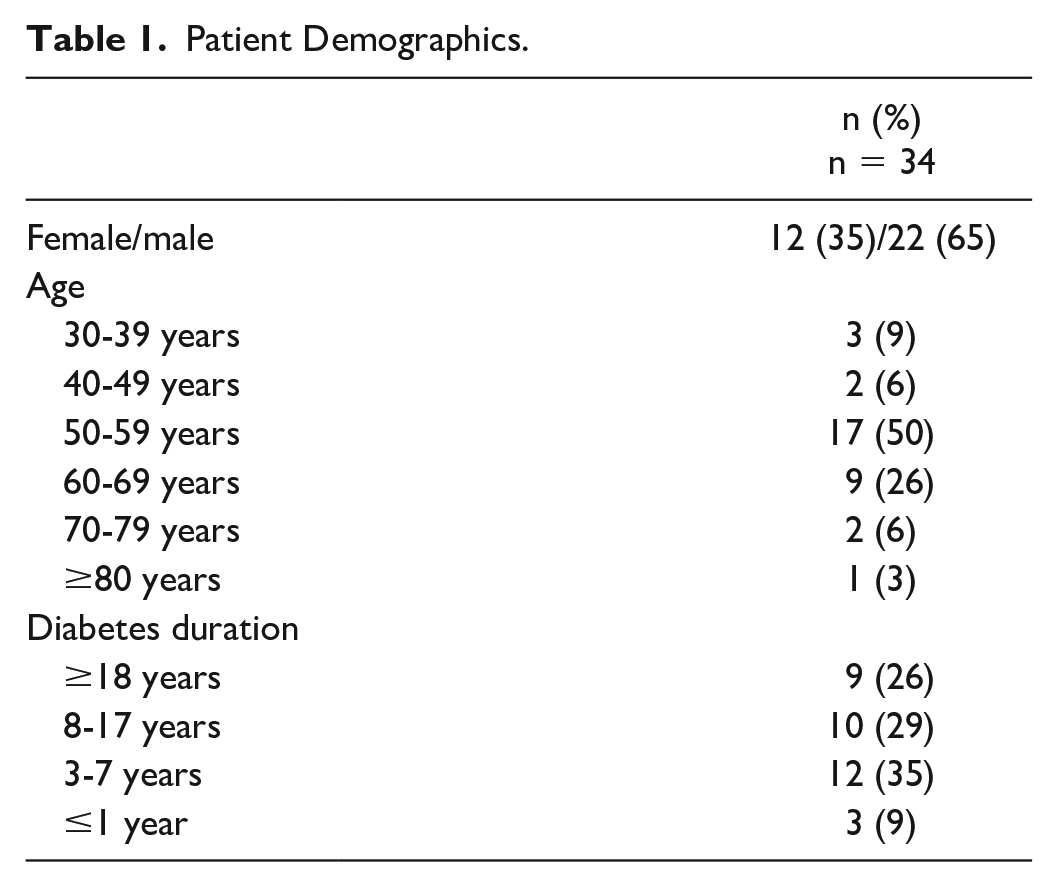
Patient demographic data are presented in Table 1. Two-thirds of patients were male (65%), half were aged 50 to 59 years (n = 17), and 35% had a diabetes duration of 3 to 7 years (n = 12). Overall, 59% of patients received insulin before initiation the app.
Patient Demographics.
Use and Assessment of the App by Physicians
All physicians chose participating patients for the app based on the patients’ affinity to use digital apps. For 70% (n = 7) of physicians, another important patient selection criterion was a high level of self-motivation and willingness to cooperate (including those patients were openly discussing their disease, documenting discussions and/or asking relevant questions). The patient’s age only mattered for 40% (n = 4) of physicians. Half of physicians (50% [n = 5]) revisited the web portal weekly or more frequently, while the remaining physicians revisited it two-to-three times a month (Figures 1a and b). The homepage and detailed patient diary were most frequently used (10% several times a week and 30% once a week, respectively; Figure 1a). Thirty percent of physicians took a look at the notifications their patients received once a week. The physicians used the app’s web portal mostly either during or directly before patient visits (Figure 1b). Overall, 78% of physicians assessed the individual treatment plan and all physicians used the detailed diary before or at the time of the patient visit. At least occasionally, all physicians changed the treatment plan, especially the FBG target, based on patient-logged information (Figure 1c).
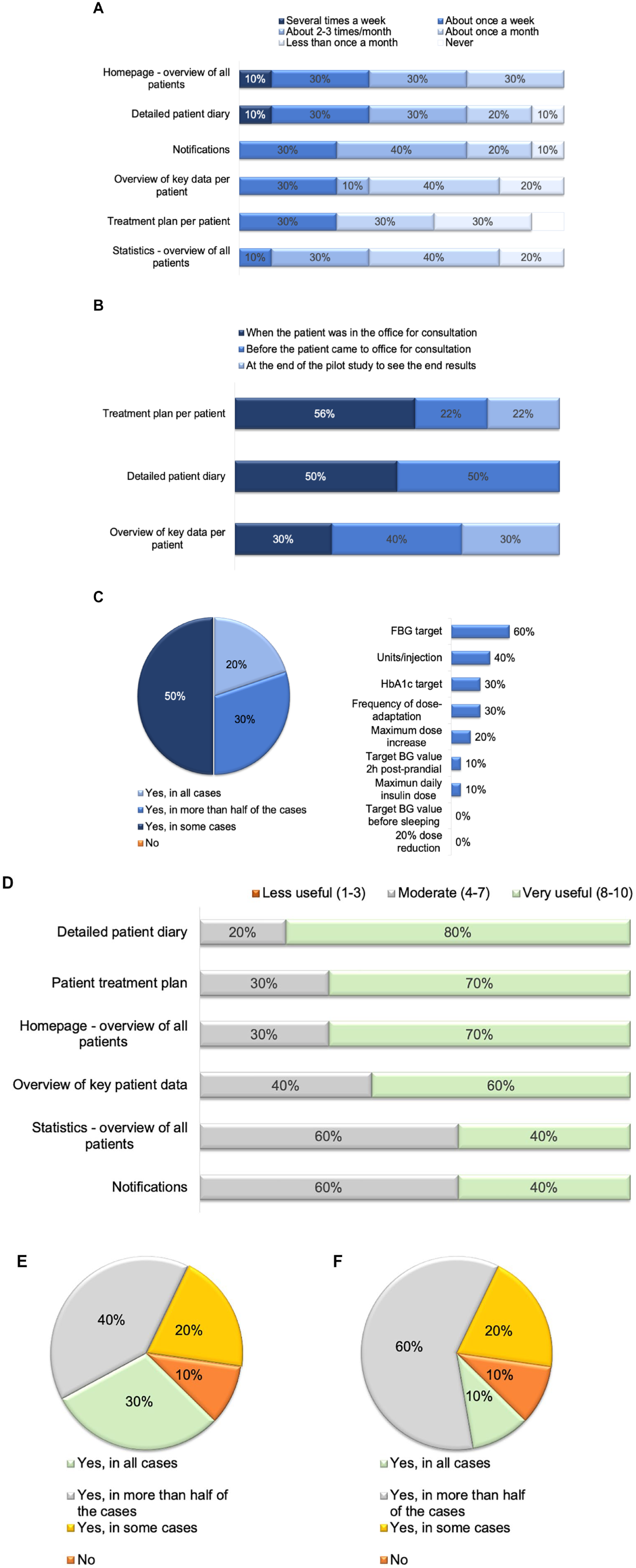
Use and assessment of the app by physicians. (a) Frequency of use. (b) Common time point to use the web portal. (c) Change in therapy plans by physicians based on patient input. (d) Physicians’ opinion on the web portal. (e) Easy to explain to the patients. (f) Easy handling for patients. Abbreviations: BG, blood glucose; FBG, fasting blood glucose; HbA1c, hemoglobin A1c.
Six out of ten physicians (60%) appreciated the manufacturer’s training, the handling and design of the web portal. The majority (80%) of physicians found the detailed patient diary very useful and 70% appreciated the treatment plan and the homepage offering an overview of all patients (Figure 1d). Most physicians found it easy to explain the app to their patients (40% in all cases and 30% in more than half of the cases; Figure 1e) due to detailed information material/self-explanatory app properties. The app was easier to explain to cognitively competent patients (for example, patients without signs of dementia) and those who regularly use their smartphone or mobile device (for example, those patients who bought their smartphones to their appointment). The most frequently identified hurdles by physicians were attributed to the additional time needed to implement management of insulin therapy via the app, as well as the time to explain about data protection, app handling, app setup on mobile devices, and additional email correspondence. The physicians’ impression was that the app was easy to use for their patients (Figure 1f), with 60% of physicians sharing this opinion in all cases and only one physician (10%) rating the handling not to be easy. Physicians reported that mostly patients with lower digital affinity had difficulties in handling the app. In contrast, they stated that downloading and installing the app from the App Store was simple, including following the menu navigation and entering data. Half of physicians (50%) reported that they were able to implement their treatment plan completely and that they used patient-appropriate pre-set treatment targets (HbA1c and FBG values) for their patients (based on information such as patient condition, age, and comorbidities).
Use of the App by Patients
Over three-quarters (76%) of patients received recommendations for using the app from their diabetologists and 24% from their general practitioners. Most patients (59%) already received insulin prior to using the app, 41% of patients started the basal insulin treatment together with app use. Most patients preferred to use the app on their smartphone (85%), 12% on a personal computer/laptop, and only 3% on a tablet. Most patients used the app daily (94%), while a few patients (6%) used it several times a week. The majority of patients entered their FBG values and insulin doses daily into the app (85% and 76%, respectively) (Figure 2a). The hypoglycemia reporting tool was used by 35% of patients on a daily basis and by 12% on a weekly basis. In general, data entries were performed at home (97% for FBG values and 80% for information about hypoglycemic episodes). No patient needed help from staff in the physician’s office to enter data. Most patients (82%) received recommendations concerning changes in insulin dose several times (Figure 2b). Overall, the vast majority of the patients (82%) always followed these suggestions for dose changes, 11% of patients followed the suggestions sometimes, and 7% never followed the suggestions (Figure 2c). Patients who did not adjust the dose reported that they were afraid that the recommended insulin dose might be too high or that they wanted to consult the treating physician first.
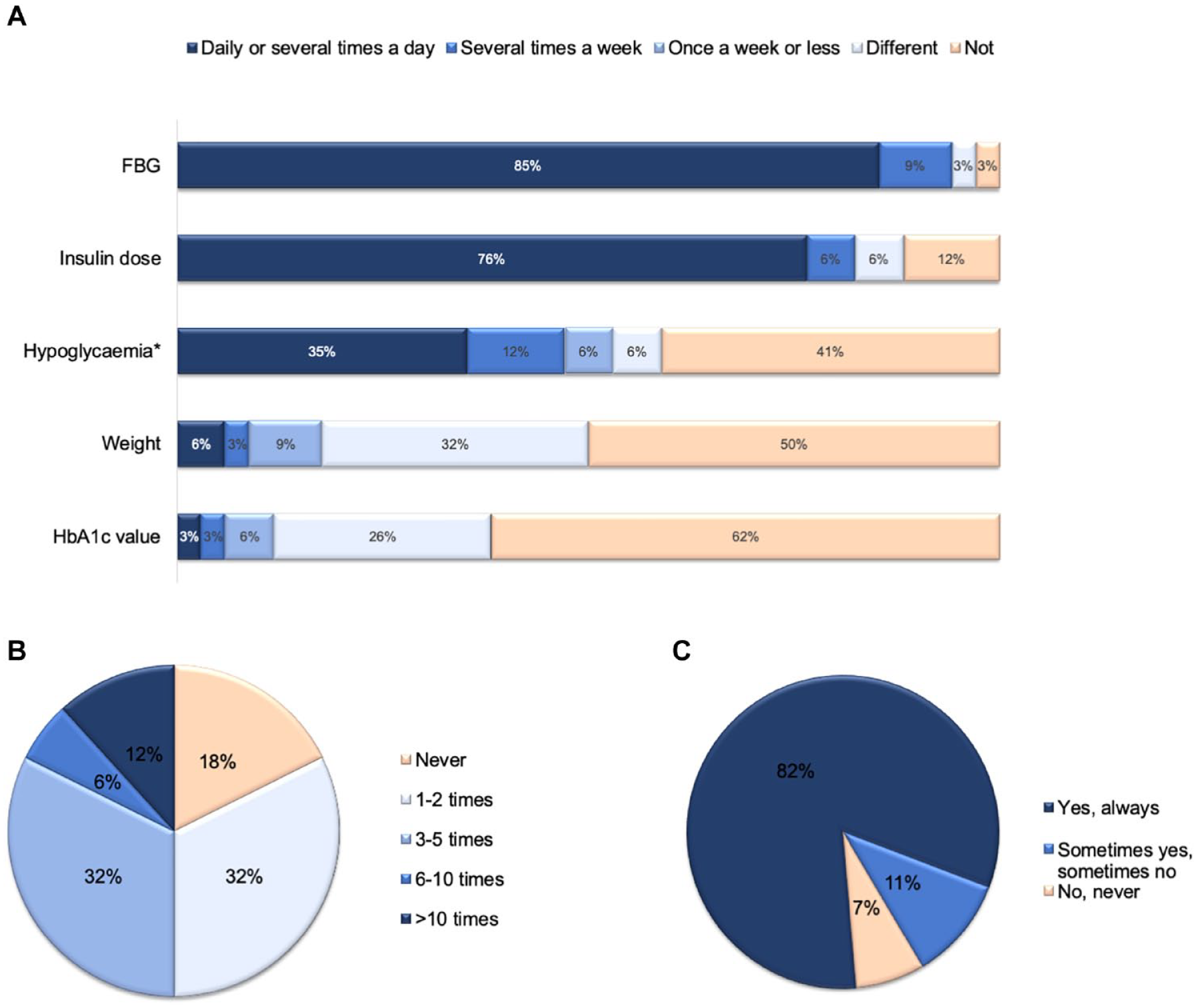
Use and assessment of the app by patients. (a) Frequency of data entries by patients. (b) Frequency of recommendations to change insulin dose. (c) Patients’ adherence to insulin dose recommendations. Abbreviation: FBG, fasting blood glucose; HbA1c, hemoglobin A1c.
In general, patients were satisfied with the handling of the app (88%), the training received from the treating physician (79%), the ability to download the app (71%), the design/presentation of the app (68%), and the registration process (65%) (Figure 2d). The presentation of the diary and insulin dose recommendations in the app were much appreciated by the patients (68% and 79%, respectively) (Figure 2e). Advantages of the app from patient perspective included ease of use (41%), recommendation and adaptation of insulin doses (21%), and the clear design (18%). Patients considered missing the possibility to enter additional data (12%) and difficulties in synchronizing the app (12%) to be disadvantages of the app. Approximately one-third of patients (32%) did not see any need for app improvements (Figure 2f).
Impact of Using the App
The 10 physicians filled out the questionnaire based on their experience with a cohort of 112 patients with diabetes.
The physicians perceived positive effects of the app mainly on patient management. Overall, 71% of physicians stated that the frequency of telephone calls was reduced, the titration phase was shortened (70%), and their overview of the patient had improved (70%) (Figure 3a). The majority (70%) of physicians thought that their patients were more involved in their diabetes therapy and 60% perceived their patients using the app to be more self-confident with disease management compared with BOT patients not using the app (Figure 3b).
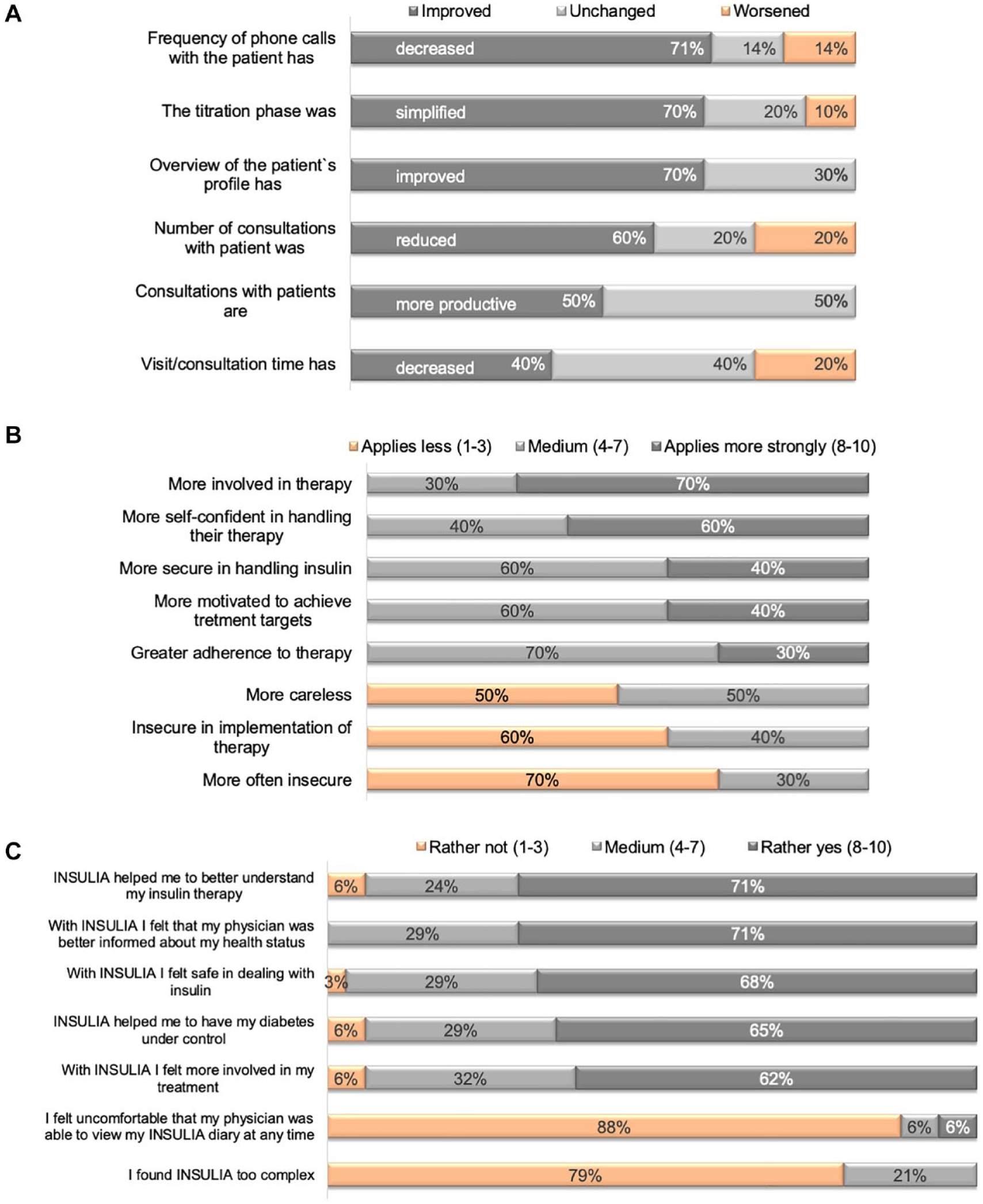
Effect of the app on physician-patient relationship and on patients’ attitude to their diabetes. (a) Effects of INSULIA on patients management as perceived by physicians. (b) Effects of INSULIA on patients in comparison to patients not using INSULIA as perceived by physicians. (c) Effects of INSULIA on patients as perceived by patients.
Overall, 71% of patients stated that the app helped to get a better understanding of their insulin therapy. The same percentage of patients had the impression that their treating physicians were better informed about their health status. Approximately two-thirds (68%) of patients felt more secure in handling insulin and 65% patients felt that the app helped them to get their diabetes under control (Figure 3c). Most of the patients would continue using the app and would recommend using it (88%). Five diabetologists and two internists (70% of physicians) saw a high positive impact value and would continue to use the app after the test phase.
The majority of patients (79/112; 71%) treated by the participating physicians achieved their FBG- and the same number of patients (n = 79; 71%) achieved their HbA1c target values. Most physicians believed that these values were reached in a shorter timeframe than patients who did not use the app (Figure 4a). From the patients’ perspective, an improvement was often perceived (FBG 68%; HbA1c44%) (Figure 4b), but only 39% of patients (FBG) and 33% (HbA1c), respectively, stated that the treatment targets had been achieved. Slightly more than half of the patients (53%) thought that their treating physicians were satisfied with their values; 18% of the patients thought that they were not.
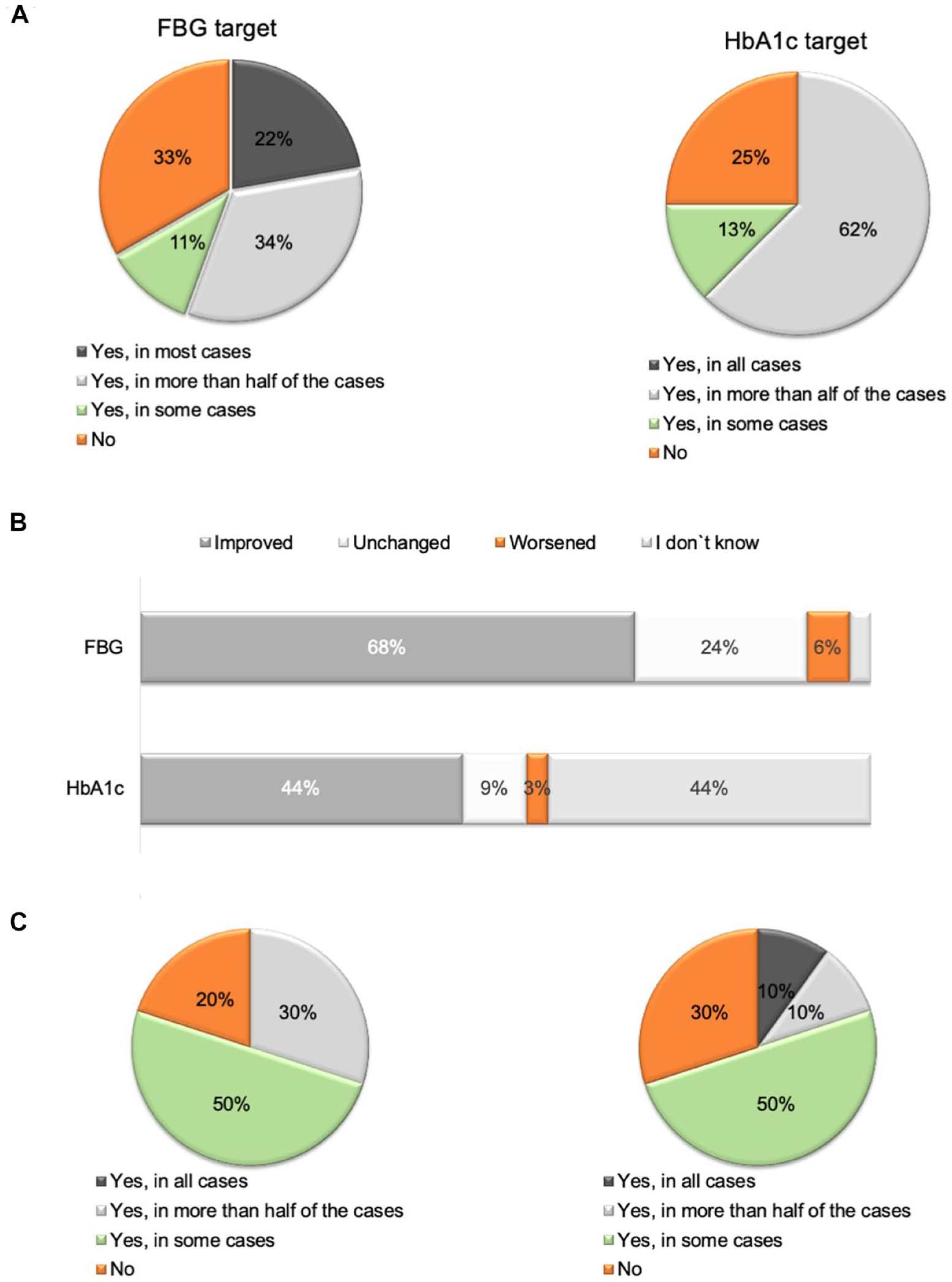
Impact of the app on diabetes target values and hypoglycemia. (a) Earlier target-value achievement in participating patients compared with other patients as perceived by physicians. (b) Target-value achievement in participating patients as perceived by patients. (c) Positive improvement on number of hypoglycemia (left) and severe hypoglycemia (right) events as perceived by physicians. Abbreviation: FBG, fasting blood glucose; HbA1c, hemoglobin A1c
Most physicians saw a positive impact of the app on the number and severity of hypoglycemic episodes (80% and 70%, respectively) (Figure 4c). Furthermore, from the patients’ perspective, the app had a positive effect on the number of hypoglycemic episodes: with 45% stating they experienced hypoglycemia less often.
Discussion
In this study, the app was highly appreciated by physicians and patients alike and they would continue to use the app after the test phase was completed. The app was perceived to have many benefits. The app had a perceived positive impact on the BOT patients’ HbA1c and FBG target achievement (Figure 4). The patients showed a better understanding of their therapy (this is subjective but could be based on the patient’s ease if making decisions and how comfortable they felt making app-recommended adjustments to their treatment), felt safer during the titration phase, and had better overall therapy management and thus showed higher compliance, which in turn was associated with a greater therapy success. These findings are in line with the results from a systematic review and meta-analysis on the use of app-assisted self-care interventions for improving outcomes in patients with type 2 diabetes and hypertension. 13 In addition, the number of telephone calls with the practice, visits, and their duration were reduced—providing relief for physicians and their staff. This is an important consideration as direct costs associated with diabetes treatment/management are spiraling. 14 Overall, the use of the app suggests that the insulin dose titration phase may have been simplified.
The use of diabetes apps by patients to self-manage their condition is positively associated with self-care behavior. 15 Studies have shown that diabetes apps can be safely used by patients aged ≥50 years, although apps with multiple functions have been used less. 16 The key factors affecting the acceptance of apps in patients aged ≥50 years were the perceived additional impact and user friendliness. While using the app, it was evident that the possibilities for individual adaptations were important to patients and their treating physicians. Personal contact, especially in the initial phase of app use, is of utmost importance to patients. This can significantly decrease the fear of data loss or incorrect data entries and increase acceptance among the target group aged ≥50 years. 17
In this pilot study of the app, 85% of patients were ≥50 years old and they appreciated many benefits of the app. Patients found the app easy to use and 82% followed the suggestions to change the insulin dose, and only 18% remained skeptical regarding recommended dose changes and were more cautious. Patients typically felt safer in their therapy because their treating physician was able constantly to monitor their therapy. Patients needed most support during the insulin dose titration phase. For patients, fear of hypoglycemic episodes was reduced due to clear instructions. Patients were perceived to have a higher level of trust in the therapy, because of the knowledge they had gained from the initial detailed instructions in the app.
In agreement with other studies and meta-analyses regarding the clinical impact of diabetes apps,18-21 this analysis showed a perceived improvement in FBG and HbA1c values in patients using the app. Overall, physicians stated that 79% of their app-using patients achieved their individual treatment targets (FBG and HbA1c). Most physicians had the impression that these targets were achieved more quickly by patients supported by the app compared with patients not using the app.
The need for additional support from a health care professional or coach when using a mobile app to improve target HbA1c values has been previously reported. 22 Diabetes teams play a key role in guiding the patient toward more self-management when they are faced with major challenges. Patients learn a lot during this guidance. Consequently, they transfer their knowledge and are able to respond to potential complications arising during insulin therapy in a timely manner. Apps can improve diabetes self-management and confidence in therapy. 15 Patients with diabetes often require support for practical aspects of their diabetes self-management. They need this to reduce the cognitive load by means of automated, personal coaching, reminders, and alarms, as long as this is continuously encouraged and motivated, and interconnectivity with the treating physicians exists.23,24 Within the web portal, physicians can take a look at patient notifications and they have the possibility to give them feedback on their therapy.
Another important point is that according to the majority of the physicians, the number and severity of hypoglycemic episodes decreased in some of the patients (50%) or in more than half of the cases (hypoglycemia 30%; severe hypoglycemia 10%). Similarly, 45% of the patients using the app supported this finding and stated that they experienced fewer hypoglycemic events. The goal of diabetes self-management is to improve glycemic control with fewer blood glucose fluctuations and to avoid hypoglycemic events.
While insulin-titration apps have been shown to improve HbA1c levels, 19 several other factors also need to be considered including avoiding hypo- and hyperglycemic episodes, 25 and maintaining a user friendliness for both patients with diabetes and their physicians. 26 This analysis supports the assumption that the app addresses these factors and appears to be a useful tool to improve diabetes self-management in patients with type 2 diabetes receiving BOT.
Limitations
The purpose of the study was to determine the ease of use of a diabetes app from the perspective of patients and physicians alike. The study was not designed to collect or assess specific treatment targets. This study had a limited number of participants—10 physicians and 34 patients—who completed their respective questionnaires. In particular, patients were selected by their affinity for digital solutions. Although participants’ affinity for digital solutions could be considered a limitation of the study, we see it rather as a key selection criterium to leverage the full potential of a health application. Furthermore, the inclusion criterion for patients was being on BOT. This included both patients starting treatment with insulin, as well as patients who were already used to insulin treatment. The former group of patients certainly needs more support from physicians or the app in this initial phase. Subgroup analyses could be performed to determine the impact of these limitations but are outside the scope of this initial study.
Conclusions
Overall, the app was highly appreciated by physicians and patients alike in this pilot study. Due to the app, FBG and HbA1c targets were perceived by treating physicians to be achieved in a shorter timeframe. In addition, the app use was perceived to have a positive impact on the number and severity of hypoglycemic episodes. Patients were more involved in therapy management through use of the app and felt more confident in dealing with insulin therapy by following recommendations received from the app. Most physicians and patients would continue to use the app and recommend using it. This pilot study, therefore, gives confidence in the potential role that health applications might play in everyday life of people with chronic diseases.
Supplemental Material
sj-docx-1-dst-10.1177_19322968221090521 – Supplemental material for A Smartphone-Based Application to Assist Insulin Titration in Patients Undergoing Basal Insulin-Supported Oral Antidiabetic Treatment
Supplemental material, sj-docx-1-dst-10.1177_19322968221090521 for A Smartphone-Based Application to Assist Insulin Titration in Patients Undergoing Basal Insulin-Supported Oral Antidiabetic Treatment by Dietrich Tews, Evanthia Gouveri, Jörg Simon and Cornelia Marck in Journal of Diabetes Science and Technology
Supplemental Material
sj-docx-2-dst-10.1177_19322968221090521 – Supplemental material for A Smartphone-Based Application to Assist Insulin Titration in Patients Undergoing Basal Insulin-Supported Oral Antidiabetic Treatment
Supplemental material, sj-docx-2-dst-10.1177_19322968221090521 for A Smartphone-Based Application to Assist Insulin Titration in Patients Undergoing Basal Insulin-Supported Oral Antidiabetic Treatment by Dietrich Tews, Evanthia Gouveri, Jörg Simon and Cornelia Marck in Journal of Diabetes Science and Technology
Footnotes
Abbreviations
BOT, basal insulin-supported oral therapy; CE, Conformité Européenne; FBG, fasting blood glucose; HbA1c, hemoglobin A1c.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dietrich Tews was involved in the conduct of the study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was sponsored by Sanofi Aventis Germany. Medical writing was provided by the Institute for Pharmacology and Preventive Medicine, Cloppenburg and paid for by Sanofi.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
