Abstract

Keywords
Introduction
The annual size of the global wearable technology market was valued at 40.7 billion dollars as of 2020, and this market is expected to expand at a compound annual growth rate of 23.8% between 2021 and 2026. 1 The wearable device industry includes products for a variety of purposes, including the treatment of diabetes. Wearable products for diabetes can continuously monitor concentrations of analytes like glucose 2 (and potentially ketones3,4) in the interstitial fluid of the subcutaneous compartment and deliver drugs like insulin, 5 glucagon, 6 or incretins 7 into this space. All these devices are shrinking in size with each new generation, and they can look quite similar to each other. To a non-expert, each of these wearable devices is an unidentified wearable object (UWO) that appears to be a white plastic patch or case attached, for example, to the arm or abdomen of a person with diabetes (PwD).
The aim of this editorial is to raise awareness to a potential problem with wearable devices used for treating diabetes that are not or cannot be identified by emergency personnel. A UWO on a PwD who cannot communicate, for example, after an accident, is potentially dangerous when mismanaged by emergency personnel.
Misidentification of Unidentified Wearable Devices For Diabetes
A recent National Health Service (NHS) National Services Scotland Safety Action Notice on September 30, 2021, reported that people using wearable medical devices for monitoring and treatment were at risk of severe prolonged hypoglycemia as a consequence of misidentification of their devices. 8 The report stated that failure to correctly differentiate an insulin patch pump from a continuous glucose monitor (CGM) in PwD may result in severe prolonged hypoglycemia, leading to serious injuries and even death. The report recommended that (1) each patient with a UWO, who requires care but cannot communicate, should be identified; (2) an assessment of any device should be carried out, including a review of any device markings, to ensure that an insulin patch pump is correctly distinguished from a CGM; and (3) if a device cannot be positively identified, then a local team of diabetes clinicians should be consulted for advice. The report expressed concern that if a patient with diabetes requires emergency care and is unable to communicate, then the clinical staff might incorrectly identify an insulin patch pump as a CGM sensor and leave it in place. In such a scenario, a PwD admitted for hypoglycemia might continue to receive insulin inappropriately, which could result in delayed recovery and prolonged hypoglycemia. These are the recommendations given in the notice mentioned above for emergency response in Scotland; however, the United States (US) and many other countries have a different emergency response and thus would need tailored recommendations.
Emergency Medical Services
Emergency Medical Services (EMS) are frequently called to evaluate and treat an unconscious patient. Given the dilemma of removing or retaining a UWO, emergency personnel might have a difficult time deciding what to do when confronted with such a device. Unidentified wearable objects may be for diabetes or other diseases. There currently is no reliable way to ascertain a UWO’s function from the appearance. Professionals must make life-or-death decisions with minimal pertinent history, limited light, hectic environments, and difficult-to-assess patients for which immediate reactions are often needed.
In view of the rapid increase in the use of wearables / medical products by PwD, we expect that EMS personnel will increasingly be confronted with unconscious patients with UWOs attached to them. There should be clear information on the wearable device to paramedics or Emergency Medical Technicians (EMTs) regarding the function of the device. This would remove the UWO status from the device. If patients are using a UWO for the treatment of other diseases or if they are monitoring health biomarkers, then this could lead to less certainty about the patient’s medical history and correct course of action.
The recommendation by NHS National Services Scotland to have a diabetes clinician consultation in the field is currently not an option in the American EMS system and as stated above is unlikely to happen. The American EMS system uses paramedics and EMTs to deliver care in the prehospital setting, and as such, physicians would not be present. There is a need for a better way to identify these devices quickly and accurately on scene in most countries.
Consequences of Misidentification
If a PwD is wearing an insulin patch pump and found to be hypoglycemic after an assisted monitoring of blood glucose (BG) measurement by EMS, then it is possible that the pump is continuing to deliver an inappropriate dose of insulin. 9 On the other hand, for hyperglycemia in diabetes, removing an insulin pump and administering glucose increases a patient’s risk of ketoacidosis. The EMS training is unable to keep up with the fast-changing world of wearable diabetes devices. Furthermore, finding the pump controller, determining the insulin delivery rate, and adjusting the rate of insulin delivery to the current needs of the PwD are beyond their scope of practice. In such a situation, pump removal is therefore advised. On the other hand, if the pump is not delivering insulin, then there would be no need to remove the pump.
In addition, wait times can be substantial in some EMS systems with some wait times or delays before securing a bed in an emergency department (ED) being hours in length. Furthermore, the handoff from EMS to ED staff may lose valuable information about the device that may have been known in the prehospital setting, but becomes a UWO in the hospital if there is a lack of communication.
The plastic device (or in the future there might be two devices) could be part of a system for automated insulin delivery (AID) using wearable devices (ie, a CGM system and an insulin pump, which might be a patch pump with future AID systems). In that case, removal of these devices would cause disruption of the function of the AID system. Current generation patch pumps are typically controlled by mobile apps running on the smartphones of the PwD. Often, EMS have no access to these devices when a PwD is unable to login to the dedicated app to check their function or regulate them. Some smartphones offer the option that such apps can be accessed by EMS in an emergency situation without a regular login, but as mentioned controlling drug rates is beyond their scope of practice.
The first thing that EMS must do for an unconscious hypoglycemic PwD is administer intravenous glucose. If a PwD is wearing a CGM system with a glucose sensor attached to the arm or abdomen that communicates with an external receiver or smartphone for data transfer, then the EMS personnel removing the sensor from the body of the PwD could cause harm through the loss of continuous glucose data. If the PwD receives an infusion of intravenous glucose, then the CGM system might fail to display the rapid increase in glucose levels because of the lag time between the increase in BG levels and the increase in interstitial fluid glucose levels. If the rescue staff use the CGM system (rather than fingerstick BG measurements) to monitor the glucose response to the dose of glucose delivered intravenously, then they might miss seeing an immediate rise in the glucose concentration. They might then go on to administer extra unneeded glucose, which could result in severe hyperglycemia. Our recommendation is that EMS should not access the CGM data and rather follow the policies and procedures established by their Medical Director for treating hypoglycemia in the field.
Wearable Patch Pumps
Examples of US Food and Drug Administration (FDA)-cleared wearable patch pumps include the CeQur Simplicity (CeQur, Lucerne, Switzerland) and the Omnipod (Insulet, Acton, Massachusetts). The V-Go patch pump (Valeritas, Bridgewater Township, New Jersey) was FDA-cleared, but the product is no longer on the market. Non-FDA-cleared patch pumps available in some countries outside the US include the Accu-Chek Solo (Roche Diabetes Care, Mannheim, Germany) and the EOPatch (EOFlow, Seongnam-si, South Korea). Images of these patch pumps are presented in Figure 1.
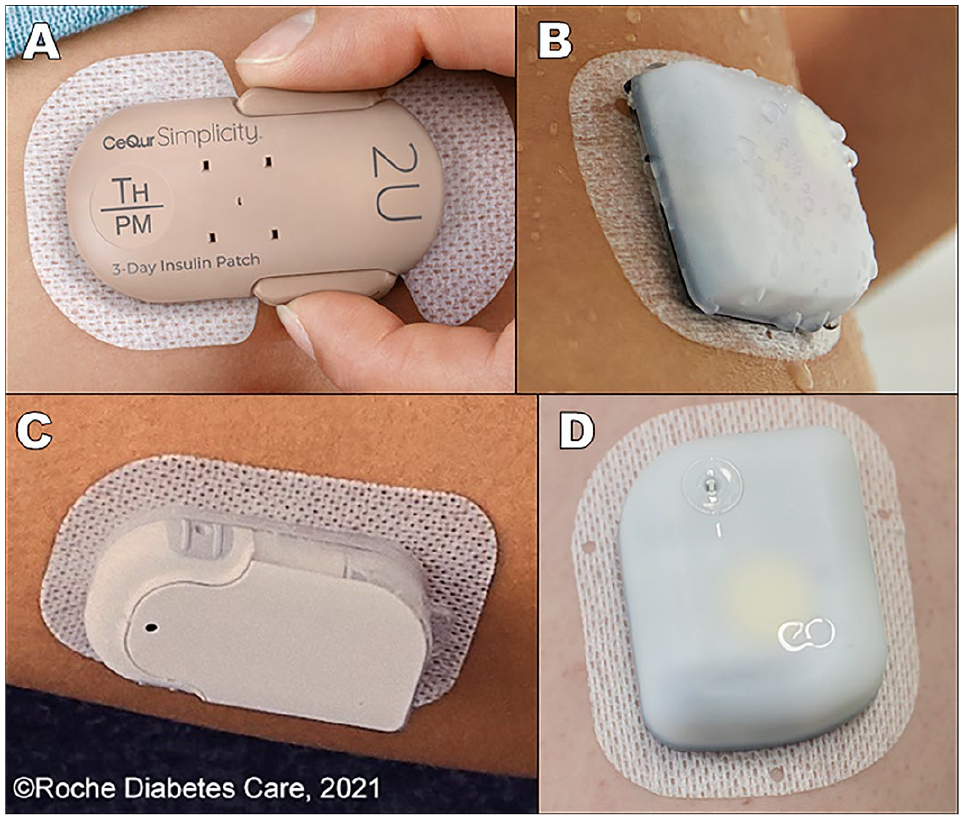
Wearable patch pumps: (a) CeQur Simplicity, (b) Insulet Omnipod DASH, (c) Roche Diabetes Care Accu-Chek Solo, and (d) EOFlow EOPatch.
Wearable CGMs With Visible Patchlike Transmitters
Examples of FDA-cleared wearable CGMs with visible patchlike transmitters include Dexcom G6 (and G7 is expected to be on the US market soon; Dexcom, San Diego, California), Freestyle Libre 14 Day System and Freestyle Libre 2 (and Freestyle Libre 3 is expected to be on the US market soon; Abbott Diabetes Care, Alameda, California), and Guardian Sensor 3 (and Guardian Sensor 4 is expected to be on the US market soon; Medtronic, Northridge, California). A wearable CGM with a patchlike transmitter that has a Conformité Européenne (CE) Mark, but is not FDA-cleared, is the Cascade (WaveForm Diabetes, Wilsonville, Oregon). Other CGM systems with similar form factors are under development. Images of these CGMs with a patchlike appearance are presented in Figure 2.
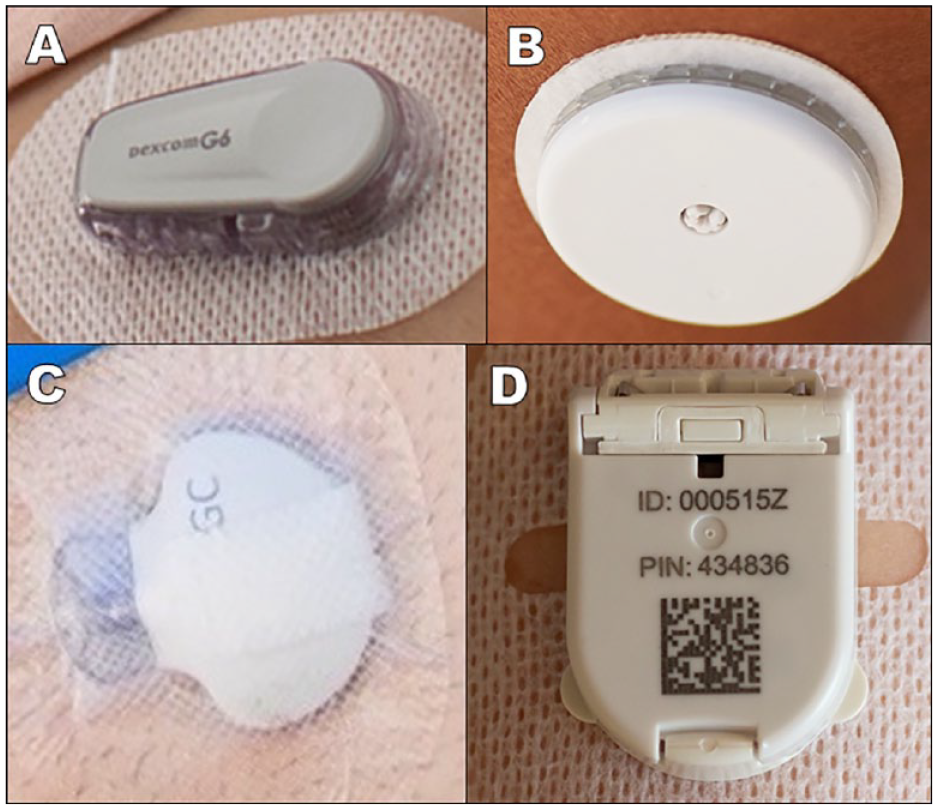
Wearable continuous glucose monitoring systems: (a) Dexcom G6, (b) Abbott Freestyle Libre 2, (c) Medtronic Guardian Sensor 3, and (d) WaveForm Cascade.
The Omnipod 5 AID system was recently designated by the FDA as the first cleared AID system using a patch pump (the Omnipod), along with a wearable patchlike CGM system (the Dexcom G6). 10 In addition, two FDA-cleared AID systems use a wearable patchlike CGM system (Dexcom G6) along with a standard insulin pump that is carried. These include insulin pumps from Medtronic and Tandem (Tandem Diabetes Care, San Diego, California). However, if an unconscious PwD is wearing an FDA-cleared AID system that uses a CGM system or an investigational AID system consisting of a CGM system and a patch pump, then this system might counteract a rapid rise in glucose (induced by intravenous glucose administration). The AID system might tamp down the rapidly rising glucose concentration from the intravenous infusion by increasing the subcutaneous insulin infusion rate. The result would be a less than expected rise in the glucose concentration. Even when paramedics and medical staff in the ED recognize a CGM system, they are not necessarily familiar with an AID system. They might not know why a person is wearing two patchlike devices. Would they follow the principle that in an emergency, the safest response is to tear off the device(s)? Or would they follow the principle that in an emergency, everything should be left as is in the absence of instructions to remove?
Wearable Patchlike Diabetes Devices Under Development
A wearable subcutaneous Bluetooth-enabled triage health care monitoring system that measures cardiac and respiratory function for creating a picture of a person’s health status for emergency personnel has been proposed. The visible patchlike part of this worn device resembles that of a transmitter of a CGM system. 11 Subcutaneous delivery of high-dose/volume biologics containing more than 3 mL of fluid can be accomplished with wearable drug delivery devices. 12 We expect that the number of wearable patchlike sensors or drug delivery devices that resemble CGM systems or patch pumps for many purposes even besides diabetes will increase substantially in the next decade. Although in the future it is likely that other types of devices for monitoring or delivering drugs for diseases other than diabetes might become widely used, at this time, no such wearable patchlike products are as widely used as CGM systems and insulin pumps.
Labeling of Wearable Devices
The EMS and emergency medical physicians must be prepared to see more and more patchlike wearable devices. We see the need to avoid potential risks (like the ones mentioned in the Safety Action Notice from Scotland above) by taking appropriate measures. Our recommendations for manufacturers and regulators of wearable devices are presented in Table 1. The risk of wearable devices becoming perceived as UWOs in an emergency situation extends beyond diabetes devices to wearable devices for all diseases. We advocate a standardized process for labeling wearable diabetes devices (and wearable devices for other purposes) to eliminate the UWO phenomenon. A consensus selection process for determining the types of markings for identifying wearable diabetes devices would be helpful for PwD and EMS in emergency situations.
Recommendations for Labeling of Wearable Devices.

Abbreviation: EMT, Emergency Medical Technician.
Conclusion
Unidentified wearable objects present risk in an emergency. The recommendations from a consensus group could become a voluntary guideline, a necessary standard, or a regulatory requirement mandated by a regulatory agency. Now is a good time to plan ahead for a process to eliminate the confusion and risk to patients associated with UWOs.
Footnotes
Acknowledgements
The authors thank Tejaswi Kompala, MD, for her helpful advice and Annamarie Sucher-Jones for her expert editorial assistance.
Abbreviations
AID, automated insulin delivery; BG, blood glucose; CE, Conformité Européenne; CGM, continuous glucose monitor; ED, emergency department; EMS, emergency medical services; EMT, Emergency Medical Technician; FDA, US Food and Drug Administration; NHS, National Health Service; PwD, person with diabetes; QR, quick response; UDI, unique device identifier; US, United States; UWO, unidentified wearable object.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: LH is a consultant for a number of companies that are developing novel diagnostic and therapeutic options for diabetes treatment. He is a shareholder of the Profil Institut für Stoffwechselforschung GmbH, Neuss, Germany. KTN and NYX are consultants for Abbott Diabetes Care. GHG has nothing relevant to disclose. DCK is a consultant to AI Health, Dexcom, Eli Lilly, EOFlow, Integrity, Lifecare, Medtronic, Novo, Roche Diagnostics, Rockley Photonics, and Thirdwayv.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
