Abstract
Background:
Hospitalization of persons with diabetes in an inpatient diabetes unit is challenging, notably for patients having different profiles. We aimed to evaluate the feasibility and the benefit of a continuous glucose monitoring (CGM) telemetry system to control glucose excursions in hospitalized patients with diabetes, according to their diabetes type and the reasons for their hospitalization.
Method:
A prospective pilot study was conducted in 53 insulin-requiring diabetes patients hospitalized in the general ward. Glucose was monitored using Guardian Connect (GC, Medtronic) to adopt insulin therapy. The time in range (TIR, target 70-180 mg/dL), the time below range (TBR), and the time above range (TAR) were recorded by GC between the start of hospitalization (SH) and end of hospitalization (EH), and analyzed according to the diabetes type (type 1 diabetes n = 28, type 2 diabetes n = 25) and the reasons for hospitalization (acute complications n = 35, therapeutic education n = 18). Patient and caregiver satisfaction was also assessed.
Results:
In patients with type 2 diabetes and those hospitalized for acute complications, TIR significantly increased between the SH and EH, from 75.7% (95%CI 48.5-84.6) to 82.2% (95%CI 63.2-91.8) P = 0.043 and from 58.3% (95%CI 46.3-69.7) to 66.4% (95%CI 55.6-75.5) P = 0.031, respectively, and TAR significantly decreased, with no change in TBR. In patients with diabetes hospitalized for therapeutic education, TBR significantly decreased from 3.4% (95%CI 0-9.4) to 0% (95%CI 0-3.8) P = 0.037. Finally, 94% of patients and caregivers deemed the GC system useful.
Conclusions:
CGM telemetry system use is feasible and well accepted in patients hospitalized in diabetes care unit and could be useful to improve therapeutic education and metabolic control, especially for specific homogenous populations with diabetes.
Introduction
Hospitalization of patients with diabetes in an inpatient diabetes unit is highly challenging. These patients are indeed admitted for very different reasons, ranging from acute situations to therapeutic education or therapeutic reassessment. In all cases, the objective is to target optimal glycemic control by reducing glycemic variability. It is widely accepted that hyper- and hypoglycemia are associated with an increased risk of complications and mortality, 1 as well as longer hospital stays for patients in intensive care and those admitted to the general ward.2,3
Bedside capillary point-of-care (POC) testing is the current preferred method for glucose monitoring in the hospital setting. However, studies conducted over the past 10 years have reported remarkable consistency between continuous glucose monitoring (CGM) and blood glucose (BG) measurements, yet with a greater number of hypoglycemic events detected at night using a CGM device.2-6 These systems are equipped with alarms and provide real-time information about interstitial glucose levels, including the direction (increasing, decreasing, or stable), and rate of change.
Despite broad-based evidence supporting the sufficient accuracy and reliability of new-generation CGM sensors and their wide use for outpatient follow-up, 7 the technology remains largely investigational in the hospital setting. However, the COVID-19 pandemic is changing practices and more and more diabetes departments use CGM for hospitalized patients.8-10
The aim of the present study was (i) to evaluate the feasibility of using the Guardian Connect (GC, Medtronic) CGM device to analyze the glycemic control of hospitalized patients with diabetes; (ii) to compare the time in range (TIR), the time below range (TBR), and the time above range (TAR) between the start of hospitalization (SH) and the end of hospitalization (EH) period, according to the type of diabetes and reasons for hospitalization; and (iii) to evaluate the patient and the nursing staff satisfaction.
Methods
Participants
Between September 15, 2018 and December 31, 2018, participants were consecutively included in this observational, prospective, open-label, single-center, pilot study if they had insulin-requiring diabetes, were aged ≥18 years, and had been hospitalized in the inpatient diabetes unit of the Strasbourg University Hospital, France. Pregnant women, patients with an intellectual disability, and those already treated with a MiniMed640G® insulin pump with SmartGuard technology (Medtronic) were all excluded from the study. The local ethics committees approved the protocol, and each patient provided a written informed consent.
Continuous Glucose Monitoring System
The GC device (Medtronic) is composed of two parts. The first part is the Enlite subcutaneous glucose sensor, whose transmitter is placed on the patient’s abdomen to record glucose values in the interstitial fluid every 5 minutes for a maximum of 6 days (sensor lifespan). The second component is the GC application, which is connected via Bluetooth to an iPad/iPhone in the patient’s room. This hand-held GC receiver displays the most recent sensor interstitial glucose reading, glucose reading graph, and trend information, all of which are sent from the sensor unit. Simultaneously, information is wirelessly transmitted from the patient’s room to a central computer at the nursing station. GC also provides the opportunity to program alarms. Sensor data were calibrated with capillary BG measurements only twice a day (every 12 hours).
Study Design
We conducted a pilot study under real-life conditions in a French specialized medical care unit in diabetology. For participants included in the study, insulin treatment, that is, insulin injections or external insulin pump, was chosen based on the reasons for hospitalization. The acute complication group was composed of patients who had been hospitalized for diabetes onset, diabetic foot ulcer, or uncontrolled diabetes with glycated hemoglobin (HbA1c) greater than 8%. The therapeutic education group was composed of patients with HbA1c < 8% enrolled in a therapeutic education program who had been hospitalized for flexible therapy or insulin pump initiation.
The Enlite glucose sensor was fitted on the patient’s first day of hospitalization. During the first 24 hours, we performed the traditional bedside capillary POC testing to determine the daily insulin dose, due to the known lack of reliability of the sensors during the hours immediately following sensor installation. From the second day, capillary POC tests were realized every 12 hours for sensor calibration (Accu Chek meter, Roche Diagnostics). Calibrations were performed away from meals to avoid problems related to rapid rates of change. Physicians adapted the insulin doses twice a day based on the sensor glucose readings from the preceding 24 hours, and patients were familiarized with dose adjustments by visualizing the curve of their glucose results on the iPad that was installed in the patient’s room.
The system’s low glucose alert was set at 70 mg/dL (3.8 mmol/L), and the high glucose alert was set at 250 mg/dL (14 mmol/L). As soon as interstitial BG lower limit was reached, the glucose telemetry system alerted the nursing staff, which could then initiate actions to prevent impending hypoglycemia. Nurses responded to either low or high alerts with carbohydrates or a correction bolus, as appropriate, after obtaining a confirmatory BG measurement. They adapt their behaviors in accordance with a protocol defined for each patient according to their characteristics in advance by the physician. Adverse effects and technical problems were documented.
At the EH period, the equipment was removed. The interstitial glucose values of the 24 hours of the second day and the 24 hours of the last day of hospitalization were downloaded anonymously using the Care Link software. This represents approximately 280 BG values per day, which were ranked according to the target ranges defined.
Metabolic and Satisfaction Outcomes
For data analysis, we defined glucose target ranges as those recently recommended by international consensus11,12: TIR in target 70 to 180 mg/dL (3.9-10 mmol/l), hypoglycemia for glucose values below 70 mg/dL (TBR), and hyperglycemia above 180 mg/dL (TAR).
Glycemic control was assessed by the analysis of CGM between the SH (interstitial glucose values of the 24 hours of the second day) and EH (interstitial glucose values of the 24 hours of the last day), according to the type of diabetes and the reasons for hospitalization.
Severe hypoglycemic events and ketosis decompensation were documented.
All patients filled in a questionnaire on the last day, as did the caregiver who removed each patient’s equipment.
Statistical Analysis
Demographic values and biological characteristics of participants are expressed as means and standard error of the means, and percentages for the categorical variables. Statistical analysis between two groups was performed using Mann–Whitney test.
The dataset consisted of the CGM readings from the second and last full 24 hours of treatment are presented as median and 95% CI of median. Wilcoxon matched-pairs signed rank test was applied to compare each other.
Data analysis was carried out using GraphPad Prism8® (La Jolla, USA). The threshold of significance was set at 5%.
Results
Characteristics of the Population
A total of 65 patients were enrolled for participation in the study, 12 of whom were excluded because of computer signal loss (n = 2), mechanical problems (sensor detachment) (n = 2), or missing data (non-extractable data from Medtronic sensors, due to a computer problem) (n = 8). Therefore, 53 patients were analyzed. The average duration under CGM was 4.6 ± 0.2 days. The detailed patient characteristics at baseline are presented in Table 1.
Characteristics of the Study Participants.
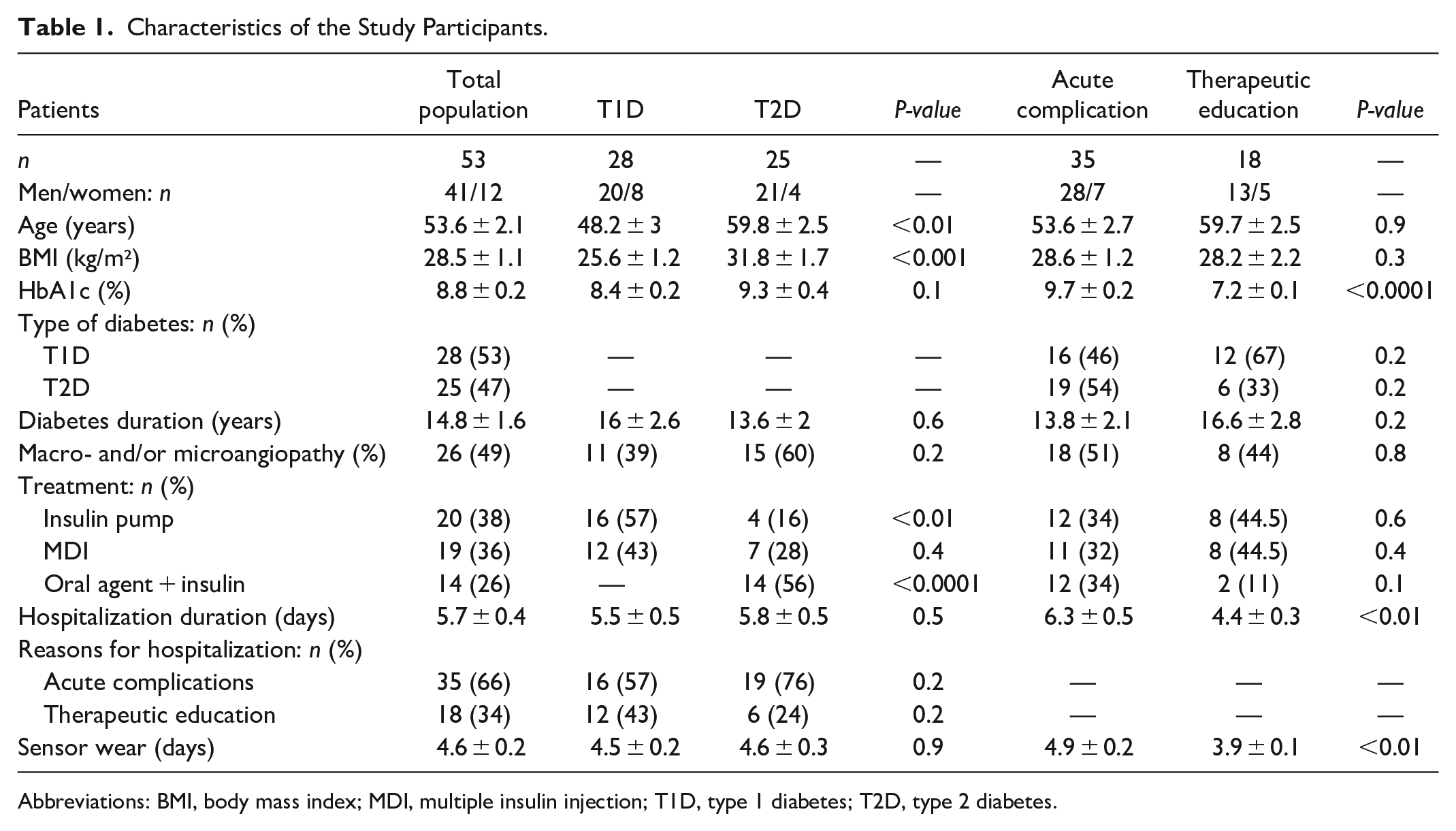
Abbreviations: BMI, body mass index; MDI, multiple insulin injection; T1D, type 1 diabetes; T2D, type 2 diabetes.
Glycemic Control Outcomes
With regard to the results for the total population (n = 53), no significant differences in TIR, TBR, or TAR were observed between the SH and the EH.
In contrast, in the T2D group (n = 25), the TIR (target 70-180 mg/dL) significantly increased between the SH and the EH (P = 0.043) and the TAR significantly decreased (P = 0.049), with no change in the TBR (Table 2).
Metabolic Evaluation of Subsets Groups with Continuous Glucose Monitoring–Guardian Connect.
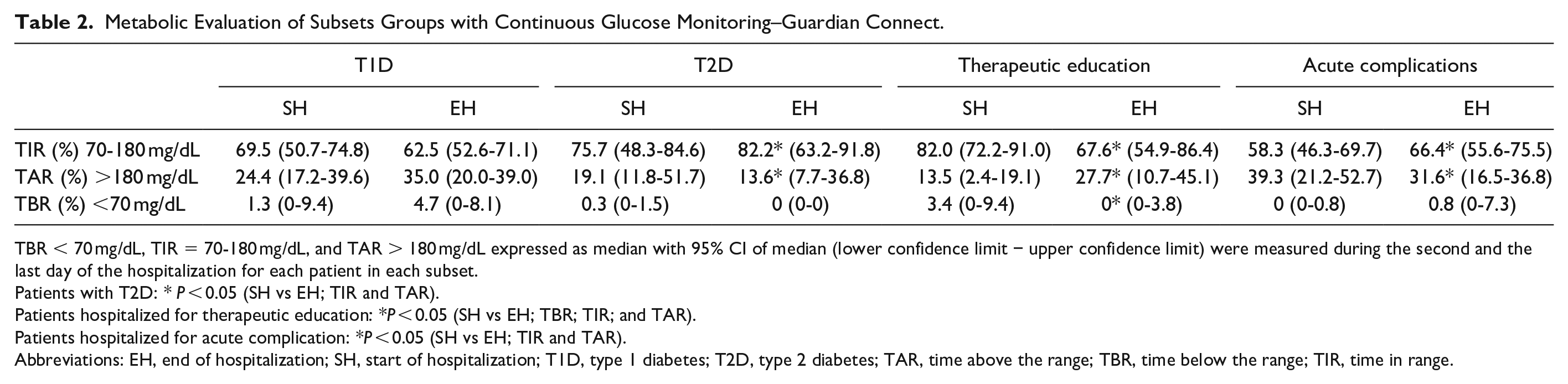
TBR < 70 mg/dL, TIR = 70-180 mg/dL, and TAR > 180 mg/dL expressed as median with 95% CI of median (lower confidence limit − upper confidence limit) were measured during the second and the last day of the hospitalization for each patient in each subset.
Patients with T2D: * P < 0.05 (SH vs EH; TIR and TAR).
Patients hospitalized for therapeutic education: *P < 0.05 (SH vs EH; TBR; TIR; and TAR).
Patients hospitalized for acute complication: *P < 0.05 (SH vs EH; TIR and TAR).
Abbreviations: EH, end of hospitalization; SH, start of hospitalization; T1D, type 1 diabetes; T2D, type 2 diabetes; TAR, time above the range; TBR, time below the range; TIR, time in range.
In the group of patients hospitalized for therapeutic education (n = 18), a significant decrease in TBR was observed between the SH and the EH (P = 0.037), but this was associated with an increase in TAR (P = 0.013) and a decrease in TIR (P = 0.044).
The acute complication group (n = 35) showed a significant increase in TIR between the SH and the EH (P = 0.031) and a significant decrease in TAR (P = 0.022), while TBR remained unchanged.
No sensor-related adverse effects (redness, itching, allergy) were reported. In terms of technical problems, we recorded two connection losses, which were related to a sensor malfunction, and two sensor detachments, but there were no calibration errors.
Severe hypoglycemia or severe ketosis were not observed during the trial.
Patients and Nursing Team Satisfaction
In total, 94% of patients reported that the GC was useful and that they did not find it very restrictive (Figure 1). Although GC was also reported to be useful for 94% of nurses, only 64% of nurses reported that it saved time. Specifically, the GC allowed to anticipate hypoglycemia and hyperglycemia excursions for 85% of patients and 64% of the nursing team. For more than half of the patients (68.5%), GC facilitated therapeutic education about diabetes and adaptation of insulin treatment, compared with only 45% of caregivers. Finally, the majority of patients (87%) expressed their interest in using the GC for other periods of hospitalization compared with 60% of the nursing team.
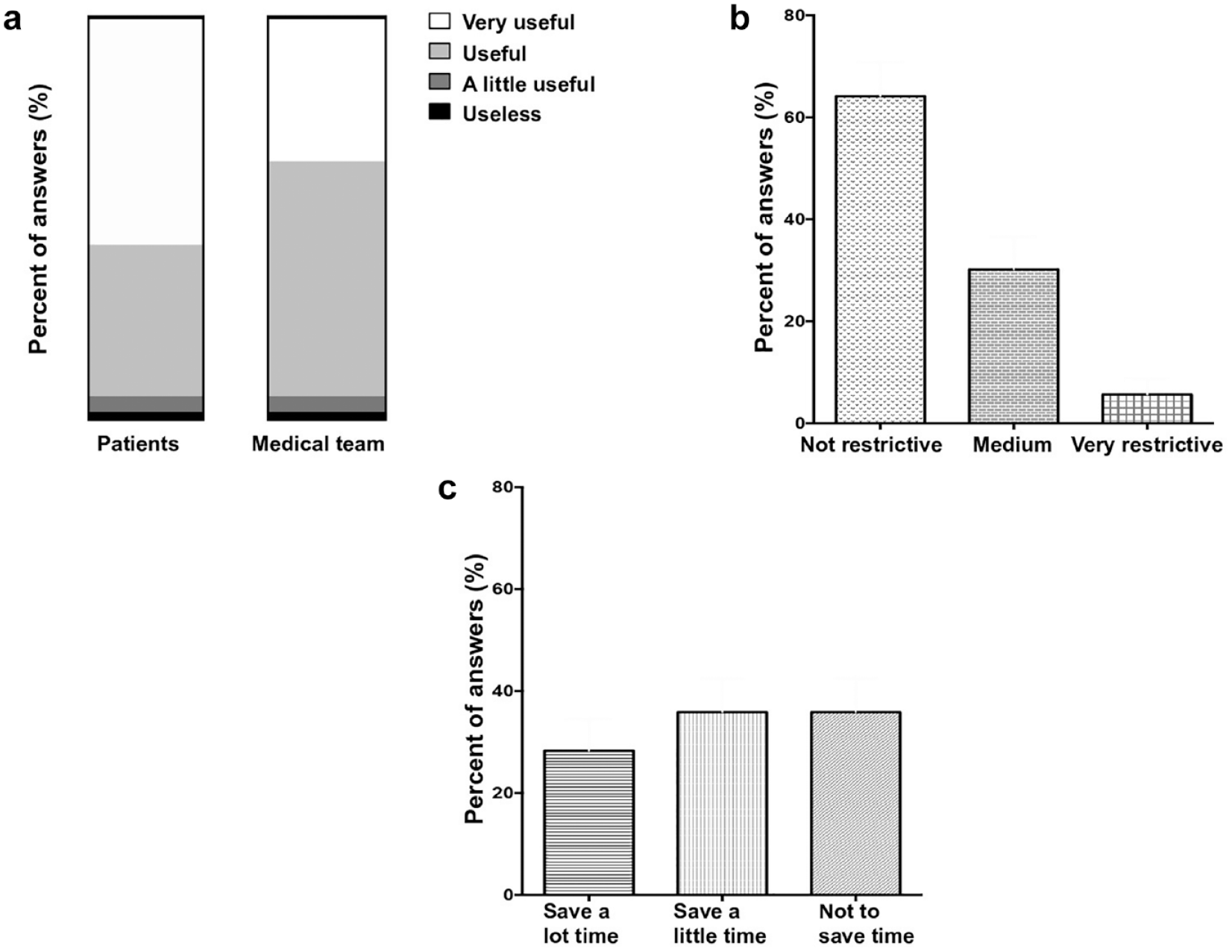
Patient and nursing team satisfaction. (a) Usefulness of Guardian Connect (GC) for patients and nursing team. (b) Restrictive nature of GC for patients. (c) Time saved by GC for nursing team.
Discussion
In this pilot study, we highlighted the feasibility and the potential benefit of the GC device in improving metabolic control in specific populations hospitalized in an inpatient diabetes unit. The main advantage of the GC device is that it provides access to 280 glucose measurements per day, compared with the 12 glucose values that are usually provided by POC capillary BG monitoring, and it also integrates telemetry technology with alarms.
The lack of statistical significance of glycemic control in the total population between the SH and the EH might be related to the short period of CGM recording. Indeed, in most cases, there were only 3 days between data registration on the second day and on the last day of hospitalization (the mean sensor wear time was 4.6 days). In Gómez et al’s 13 study, involving 34 T2D patients hospitalized in the general ward, a significant difference in the time spent in the target of 140 to 180 mg/dL (7.8-10.0 mmol/l) was also only observed from the 5th day (71.6% on the 1st day vs 87.2% on the 5th day). In agreement with these results, a trial conducted by Schaupp et al 14 involving 84 well-controlled T2D inpatients with CGM, also found a significant improvement in the time spent in the glycemic target of 70 to 180 mg/dL (3.9-10.0 mmol/L) over time, from 67.7% on the 1st day to 77.5% on the 7th day (P < 0.04).
It should be noted that glucose values for the total population were similar to those of previous studies, including the above-mentioned Schaupp et al’s 14 study: the overall time spent within the BG target of 70 to 180 mg/dL was 67.7% compared with 65.5% in this present trial.
Interestingly, we evaluated glycemic control in subgroups, and our results suggest that a CGM device could be more beneficial in specific populations.
Impact of CGM could be different between type 1 diabetes (T1D) and type 2 diabetes (T2D) because of their specific pathophysiology that leads to different glycemic variability profiles. Gómez et al 13 showed that glycemic variability and mean glucose level are independent factors associated with hypoglycemic events of <70 mg/dL. Thus, the well-controlled patients who had been hospitalized for therapeutic education in our trial, and who had a TIR reaching 82%, had a higher risk of hypoglycemia. In this population, we showed that the GC is beneficial to reduce the time spent in hypoglycemia.
In the IMPACT trial, Bolinder et al 15 also reported the benefit of using Freestyle Libre (Abbott®) to minimize hypoglycemia in patients with well-controlled T1D (HbA1c < 7.5%) and for intact awareness of hypoglycemia (38% reduction in mean time in hypoglycemia <70 mg/dL). However, among T2D patients, the Cowart et al’s 16 review asserted that the evidence is mixed regarding the benefits of CGM on hypoglycemia.
Indeed, poor glycemic control at the time of hospital admission in T2D or acute complication groups forced us to set broad glycemic targets to avoid a too-rapid drop in BG. Thus, the risk of hypoglycemia in these two groups was low, which probably explains why the time spent in hypoglycemia did not change between the beginning and end of the trial.
In the T1D cohort, glycemic control worsened without significant difference. This trend in glycemic control degradation should be interpreted in the context of the high variability of T1D.
Most patients were satisfied with the GC. The GC application, which was connected to an iPad via Bluetooth, allowed to look at the patients’ glycemic curves at patient’s bedside. This gave us the opportunity to introduce patients to the principles of anticipatory adaptation of insulin doses, instead of the corrective adaptation that is currently implemented by most patients with diabetes.
Moreover, 85% of patients were reassured by alarms. Other studies,17,18 including that conducted by Ryan et al 19 , have already reported that real-time CGM can minimize the risk of moderate hypoglycemia and eliminate the risk of severe and prolonged hypoglycemia, especially at night, which would be ignored by intermittent capillary BG monitoring. With hindsight we also think it would have been smarter to choose a higher threshold glucose value such as 80 mg/dL rather than 70 mg/dL to prevent more hypoglycemia.
However, concerning hyperglycemia, soon after the beginning of our study we had to disable the upper-limit alarms because of near-continuous alarms that required too much nursing time. The results of our trial may have been different if we had used alarms for hyperglycemia. A higher glucose alert as 300 mg/dL would have been more suitable in hospitalization but requires validation of an inpatient diabetes unit.
Finally, 64% of nurses estimated that the CGM device saves time, despite the time needed for regular calibration to address sensor drift, for following upward/downward trends in CGM glucose measurements, and for responding to hyper-/hypoglycemia alarms. This parameter was also probably minimized because of the learning curve and computer bugs.
Data about inpatient satisfaction with CGM devices are scarce. However, our results confirm data from studies with ambulatory patients. All randomized controlled trials20-23 evaluating patient satisfaction with CGM devices that have been included in the recent Cowart et al’s 16 systematic review found improvements in questionnaire scores related to diabetes-treatment satisfaction.
The main limitation of our study is the absence of a control group preventing us from asserting the CGM accuracy or reliability. The use of CGM has not been validated in the inpatient setting and it is very difficult to know if times in ranges are accurate. However, calibrations required with the GC device were realized to reduce the mistakes of CGM sensors and during the first 24 hours, we performed the traditional bedside capillary POC testing due to the known lack of reliability of the sensors during the hours immediately following sensor installation. Besides, the improvement in glycemic control may have been attenuated by the use of data from the second day of hospitalization rather than data from the first. Indeed, during the first 24 hours, patients already benefited from diabetological expertise to adjust insulin doses, thus probably allowing improved glycemic control.
Recently, in the context of the COVID-19 pandemic, the US Food and Drug Administration (FDA) issued a policy in March 2020 to expand the availability and capability of noninvasive remote monitoring devices to reduce patient and health care providers’ exposure. This allowed their wide use in the inpatient setting with satisfactory results regarding feasibility, reliability, and precision,8,9 such as in the Sadhu et al’s 10 study despite the small samples of patients hospitalized in the intensive care unit. Our study assesses the feasibility on a relatively large number of patients hospitalized in diabetology.
Another limitation of our study is the short observation period of 5 days. However, this corresponds to the average length of stay for patients hospitalized in an inpatient diabetes unit in our center.
Conclusions
Our study shows that real-time CGM telemetry system use is feasible and well accepted in patients hospitalized in a diabetes care unit. This technology could be useful to improve therapeutic education and metabolic control, especially for specific homogenous populations with diabetes. These results provide a basis for further control investigations that aim to determine the best situations and most appropriate candidates for CGM during hospitalization.
Footnotes
Acknowledgements
We thank Laura Forest, Alban De Courville, and Mounira Delye for their help with the statistical analysis and for proofreading the manuscript.
Abbreviations
BG, blood glucose; CGM, continuous glucose monitoring; EH, end of hospitalization; GC, Guardian Connect; POC, point-of-care; SH, start of hospitalization; TAR, time above the range; TBR, time below the range; TIR, time in the range; T1D, type 1 diabetes; T2D, type 2 diabetes.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
