Abstract
The HbA1c value is a well-established parameter used to characterize glucose control. Continuous glucose monitoring (CGM)-derived parameters calculated using daily glucose profiles such as Time-in-Range (TiR) have increasingly been gaining interest for assessing a patient’s current therapy. The question has arisen as to whether TiR could replace HbA1c? Because TiR focuses on the current quality of glucose control during a minimum of 10 to 14 days of CGM use and reflects the variability of glucose concentrations. Time-in-Range could be considered an attractive option for improving diabetes control in patients with diabetes. Due to the lack of established standards for glucose measurements with CGM systems, results from different CGM systems can deviate from each other. Time-in-Range should not be viewed as a replacement for HbA1c, but should be used to deliver valuable additional information.
Introduction and Background
In this commentary, we will discuss the pros and cons of the Time-in-Range (TiR) parameter compared to the role and position of the glycated hemoglobin (HbA1c value). Time-in-Range is a parameter that aids in characterization of the current quality of glucose control when using systems for continuous glucose monitoring (CGM). The TiR is complemented by two important parameters; the TiR is the TbR and TaR (Time below and above target Range).
Over the last decades, the measurement of HbA1c has established itself as a key parameter in diabetology as it provides an estimation of the development risk of microvascular endpoints in patients with diabetes mellitus. Most of the intervention studies performed over the last decades used changes in the HbA1c value as the primary endpoint and correlated such changes to the clinical outcome. Much less data are available on individual blood glucose (BG) values, in particular for individual postprandial glucose values, daily glucose profiles, or the TiR; although the latter has risen in interest in recent years.
We will not discuss any different approaches to calculate an HbA1c value from the measured (blood) glucose values (estimated HbA1c or eHbA1c); as per the FDA, the term “Glucose Management Indicator” is now used for this purpose.1,2 Likewise, we will not discuss the “estimated average glucose (eAG)”, where—contrary to the above—the HbA1c values are converted into eAG values.
There has been discussion as to whether it is even possible for the TiR to replace the HbA1c in everyday clinical practice. In a recent publication by an impressive list of authors, who were members of an International Consensus Group, it is stated that TiR should be regarded as a supplement to HbA1c. 3 While we agree with the International Consensus Group that TiR should be regarded as a meaningful and valuable supplement to the HbA1c measurement, we do also believe that it is worth taking a critical look at the advantages and disadvantages of the TiR in comparison to the HbA1c (Table 1). There are also numerous primary methodological reasons against replacing the HbA1c value with the TiR for risk assessment of microvascular complications. One reason is that a parameter that focuses on the current quality of glucose control provides different information than a parameter that reflects this glucose control over the past months.
Comparison of the Pros and Cons of HbA1c and Time-in-Range.
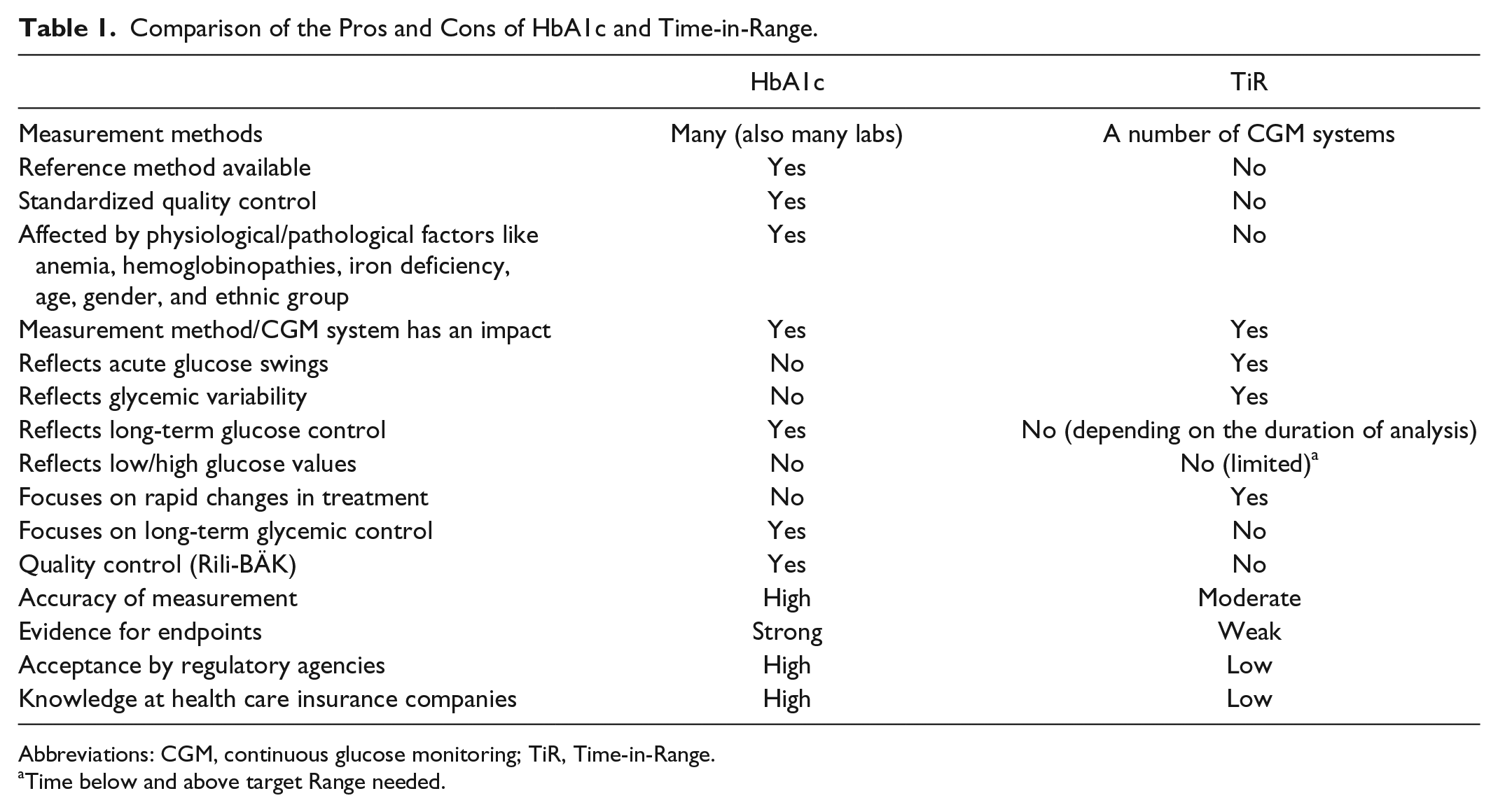
Abbreviations: CGM, continuous glucose monitoring; TiR, Time-in-Range.
Time below and above target Range needed.
Time-in-Range and Further CGM-Derived Parameters
Under physiological conditions, BG levels in healthy subjects range between 70 and 110 mg/dL (3.9-6.1 mmol/L) at night and between meals; postprandial levels may increase briefly to higher levels; after prolonged fasting or during periods of intense physical exercise, they may decrease to <70 mg/dL. Continuous glucose monitoring systems allow patients with diabetes to track the course of glucose levels in the interstitial fluid in subcutaneous fatty tissue over periods of time. The data collected using different CGM systems on the market can be analyzed using software packages provided by the manufacturers. One widely used method for presenting CGM data over a period of days is the ambulatory glucose profile (AGP).
The treating diabetologist can also analyze the AGP to draw conclusions regarding changes to the current diabetes therapy. Patients can optimize their diabetes therapy in a clear and problem-oriented manner using the visualization of glucose changes over time. In order to meaningfully calculate parameters such as the TiR, CGM data from at least 10 to 14 days of CGM use are required. 4
In addition to calculating the TiR values, additional descriptive analyses of CGM data provide information on how long the glucose values were in different glucose ranges:
- In the target range (70-180 mg/dL; 3.9-10.0 mmol/L; Time-in-Range—TiR)
- In the low glucose range (<70 mg/dL; <3.9 mmol/L; Time below target Range—TbR)
- In the hyperglycemic range (>180 mg/dL; >10.0 mmol/L; Time above target Range—TaR)
The calculated values for these parameters are expressed either as time spent in different ranges (in minutes or hours per day) or as a percentage of glucose measurements obtained per day. 5 In order to correctly estimate the hypoglycemic risk and to recommend proper therapeutic changes, it is important to know the TbR, at minimum, in addition to the TiR.
Ideally, the glucose levels of all patients with diabetes would always remain within the target range (TiR = 100%); however, with the therapeutic options currently available (see below), a TiR of up to ~70% is realistic. Recent individual reports indicate that avoiding hypo- or severe hyperglycemic events by primarily using systems that perform Automatic Insulin Delivery (AID) can achieve a TiR of 80% or even 90% under everyday conditions.
A retrospective analysis of older study data showed that high TiR values are correlated with good long-term glucose control, ie, low HbA1c values (see below). As a result, both the AGP and the TiR are considered valuable options for improving diabetes control in patients with diabetes.6-8 One last side note is that, however, another option for using TiR helps patients visualize and more deeply understand their glucose patterns is patient’s education.
HbA1c Measurement
In measuring the HbA1c value, the percentage of glycated hemoglobin circulating in the blood is determined; the glycation of proteins is associated with the development of diabetes-related complications. It has taken several years to develop a reference method for the measurement of HbA1c and to standardize and establish this method so that reliable and reproducible measurement results can be obtained. 9 Tight quality control limits for HbA1c are already implemented in many countries. In Germany, efforts are underway to tighten the current limits for internal and external quality control according to the guidelines of the German Medical Association for interlaboratory comparisons of HbA1c measurements. This is expected to lead to a further improvement in measurement quality.10-12
HbA1c measurement can be used for diagnosing diabetes although it is not widely used as such. In practice, HbA1c is most often used to evaluate the level of glycemic control over longer periods of time; however, not all HbA1c measurements provide results that match the BG values measured by the patients themselves. One possible explanation for such discrepancies could be diseases that affect erythrocyte life (such as anemia). 13 While the correlation between the HbA1c values and the mean BG value of the last three months might not be particularly close in many patients, the HbA1c remains is a good parameter for the patients to gage their overall glycemic control. One could argue that the accuracy of the TiR measurement is approximately as good as the HbA1c value: The range of HbA1c observed is usually from 6% to 10% with a measurement error of 0.1% to 0.2%. This error equals approximately 5% of the total (usual) working range. For TiR, the measurement error is approximately 2% (for the 14-day period with 70% of values present, 10 days of data minimum). The working range for TiR is about 40% to 90%. Hence, the measurement error is approximately 4% as compared to 5% for HbA1c. Additional analyses of the existing data from clinical studies are needed to try to resolve the comparison between TiR and HbA1c.
The HbA1c value is not only a well-established parameter for the characterization of glucose control, but it is also used to assess the success of new drugs for improving BG control in clinical studies. HbA1c is also regarded as a surrogate for the incidence and progression probability of microvascular complications, and its significance for macrovascular endpoints is controversial.
Time-in-Range—Measurement Aspects of CGM
Since TiR is not a measurement in itself but a parameter calculated from CGM data, there are a number of aspects to consider in CGM systems that are important for the reliable, ie, reproducible, determination of TiR, other parameters, and graphical representations derived from CGM data, such as AGP. As there is no established standard for glucose measurement with CGM systems, measurement results obtained with one manufacturer’s CGM system may systematically deviate from the results with the CGM system of another manufacturer. If the CGM systems have a different bias, the glucose profiles are then shifted in the concentration/y-axis against each other—with the same glucose course in a head-to-head comparison (ie, simultaneous measurement in one test person). This can result in clear differences in, for example, the number of measured values and/or the times <70 mg/dL (3.89 mmol/L).14,15
There are few studies that have systematically evaluated the measurement quality of current generations of CGM systems focusing on the glucose range <100 mg/dL (6.12 mmol/L). In virtually all CGM studies, measurement results <100 mg/dL are included, but most often only a relatively small number of such values are included, only studies with a focus on this could properly quantify the problem. There are current efforts to establish a CGM standard, which will hopefully adequately consider the methodological aspects of CGM measurement, ie, enable good traceability of glucose determination. 16
Systematic shifts in the glucose concourse can also result from other factors, such as errors in calibration by the user (eg, caused by the BG meter used) or “factory calibration” in different CGM systems. The algorithms implemented in the CGM systems have a significant influence on the calculated and displayed glucose values which means that the current software version must also be considered. Under hospital conditions, it is also to consider the influence of comedication, infections, and disturbances in circulation on the glucose measurement.
If the parameters determined in patients, such as TiR, deviate systematically from each other due to any of the differences mentioned above, this could lead to different therapeutic recommendations which, in turn, could have potential negative consequences for the quality of glucose control that can be achieved.
Time-in-Range Determination
In order to calculate the TiR, a sufficient data basis in the form of sufficient CGM data should be available. In concrete terms, this means CGM measurements over 14 days should be available in order to be able to fall back on data of at least 10 days with a usage duration of 70% to 80%. In addition, a sufficient number of data points per day should be available to achieve an acceptable description of the “typical” CGM profile in an individual patient.
The CGM system software, used for analyzing CGM data, provides setting options that can also influence the glucose profiles and the parameters calculated therefrom. To our knowledge, there are no published statements as to how much the results of calculating TiR value differ when identical data sets are analyzed with different evaluation programs.
In contrast to the HbA1c, the Rili-BÄK (Guidelines of the German Medical Association; https://www.bundesaerztekammer.de/suche/?L=0&id=6022&tx_solr%5Bq%5D=guidelines) does not provide any quality requirements for CGM systems, as this is not an in vitro diagnostic; for derived parameters, basically no specifications are made. It remains to be seen if regulatory authorities do regard TiR—vs HbA1c—as a relevant parameter in the clinical development of new antidiabetic drug products. Regardless, the TiR can provide additional information about the properties of these products.
Many factors result in more or less pronounced fluctuations in glycemia during the course of the day and over time. While the HbA1c value provides practically no information on these fluctuations, the TiR can be used to deliver this important information. It is also possible to describe the extent of these fluctuations by specifying the coefficient of variation (CV). Coefficient of variation values <36% are considered acceptable. 7 The time below 70 mg/dL should only be a small percentage.
Correlation Between TiR and HbA1c
The TiR is only moderately correlated with the HbA1c.17,18 This means that for given TiR values, the individual HbA1c values can vary considerably. If 50% of the values obtained by conventional BG measurement are within the TiR, the HbA1c is about 8%, and if about 70% are in TiR, the HbA1c corresponds to 7%.5,17,18 This means that the TiR alone does not provide a satisfactory description of the overall glycemic control. An increase in TiR or a decrease in TaR of 10% (equivalent to 2.4 hours per day) is associated with a decrease in HbA1c of approximately 0.6% (15). A recent study by Beck et al showed a strong correlation between TiR and the development of microvascular complications. 19 For each 10% lower TiR, the risk rate of retinopathy progression and microalbuminuria development increased by 64% and 40%, respectively. The positive correlation shown between retinopathy and TiR is also reflected in HbA1c. This could be viewed as a lack of additional information provided by TiR for this area; nevertheless, believers in TiR might insist that both parameters provide similar information. Another recent study found an association between TiR and diabetic retinopathy in type 2 subjects. 20
Correlation of TiR With Clinical and Patient-Relevant Endpoints
In addition to the (methodological) technical aspects of CGM systems and their evaluation in the form of TiR, the question arises as to what clinically relevant additional benefits the information on TiR offers for both practitioners and patients. First, it should be mentioned that the definition of TiR, as stated above, is essentially oriented toward the glucose fluctuation range in healthy individuals. It seems pathophysiologically plausible but has not yet been sufficiently tested as a clinically relevant parameter for therapeutic interventions, in contrast to HbA1c. It can be assumed that as CGM systems become more widespread, more data on the correlation of TiR and clinically relevant endpoints will become available in the future. Whether TiR can challenge the priority of HbA1c in certain patient groups will have to be proven in large prospective studies.
It is unlikely now or in the foreseeable future that all people with diabetes will use CGM systems, therefore, data on TiR and clinically relevant endpoints are likely to be expected in patients with more intensive insulin therapies and in pregnant women with diabetes, as these are also classic target groups for CGM systems.
Regardless of this, TiR is already a useful parameter for all patients using CGM systems for the evaluation of metabolic control. For example, a patient whose values are in the target range (TiR) for longer has less hypo- and/or hyperglycemia, which is clear by the definition of TiR itself. However, more TiR does not necessarily mean less hypoglycemia. A number of clinical studies show either no21-24 or only a weak25,26 correlation.
Time-in-Range (TbR and TaR) can also provide valuable additional information when new therapies are being changed or discontinued, or when lifestyle advice is given. Patient groups that benefit in particular from the additional information TiR provides are pregnant women with diabetes who should achieve glucose levels as close to normal as possible as well as patients with pronounced glucose fluctuations (often with insulin therapy). Time-in-Range can also prove to be a very useful tool in structured diabetes training. Patients with type 2 diabetes could therefore temporarily use a CGM system and TiR as part of training and thereby glean important information on the influence their lifestyle has on glucose fluctuations. This, in turn, could facilitate behavioral changes. With the use of CGM, the healthcare professional could also receive valuable information about the therapy to be applied and about possible escalation and de-escalation strategies in people with type 2 diabetes. However, routine use of CGM systems and thus also TiR in patients with type 2 diabetes without insulin therapy is currently neither planned nor adequately established by evidence.
After a lengthy process, the HbA1c value for the assessment of glucose control has established itself not only in clinical patient management, but also in different regulatory processes in health policies. A change to a different parameter would require evidence that this parameter—similar to the HbA1c value—is simple and feasible in a very broad patient population according to standardized criteria and that it correlates to clinical and patient-relevant endpoints.
Outlook
In answering outstanding questions on the value of CGM for clinically relevant endpoints, aspects of improved quality of life and patient-reported outcomes (PRO) should not be ignored. In our view, all options should be used to provide patients with the information they need to perform safe and efficient diabetes therapy. Established and proven parameters should not be abandoned without good reason simply because new parameters are available. Since the TiR represents a different parameter of glucose metabolism than the HbA1c and can also only be meaningfully measured in CGM users, it should be used as a complement and not an alternative to HbA1c. It would also be worth defining the TiR value to be achieved for the corresponding patient groups, the individualized therapy objective, and the therapy form(s) on the basis of scientific knowledge. Since there is no internationally accepted standardization of CGM measurements for TiR evaluation to date, such standardization is necessary in order to ensure that future studies can be properly compared with each other.
In the future, the additional significance of increased glucose variability (which is only measurable using CGM) in the form of TiR (with comparable HbA1c values) will need to be examined in depth. There will be much to be clarified in how TiR can support therapy decisions in lowering the risks of severe hypoglycemia, acute and chronic cardiovascular and renal events, microvascular complications and excess mortality in people with diabetes.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
