Abstract

The OneTouch Verio Reflect (LifeScan Global Corp., Malvern, PA) blood glucose (BG) meter is cleared for self-monitoring of blood glucose in the E.U., United States, and other regions worldwide. In the E.U., ISO15197:2015 accuracy criteria requires at least 95% of values to be within ±15 mg/dL or ±15% of a reference comparator at <100 mg/dL and ≥100 mg/dL, respectively, across the claimed system operating range. 1 U.S. clearance requires at least 95% of all results to be within ±15% of comparator and 99% of all results to be within 20% of the comparator method. 2
Although glucose meters must meet these minimum accuracy requirements to obtain market clearance, there is a growing desire to express meter accuracy beyond these limits. The current analysis examines results obtained in the OneTouch Verio Reflect clinical accuracy studies for FDA clearance 2 in a manner that permits additional accuracy comparisons. These studies were performed on a meter version which features an improved blood glucose algorithm and is currently available only in the United States. The studies included a lay-user evaluation of ≥350 intended users and an extreme glucose evaluation based on 50 capillary blood samples <80 mg/dL and 50 samples >250 mg/dL, tested in triplicate in unique subjects. Samples in the extreme glucose study were manipulated by spiking or allowed to glycolyze to obtain the appropriate glucose concentrations. Data from the lay-user study were combined with data from the extreme glucose study to ensure that data from the entire glucose range were more equally represented.
The accuracy results from this combined analysis are shown in Table 1. 97.9% of the BG results were within 10 mg/dL of the reference method at glucose concentrations <100 mg/dL; 96.5% of the results were within 10% of the reference method at glucose concentrations ≥100 mg/dL; and 96.9% of the results met these criteria across the entire glucose range. Additionally, 95.3% of the BG results were within 10% of the reference method across the entire glucose range. All BG results were within 20% of the comparator method. Looking at the data somewhat differently, 95% of all BG results were within 8.8 mg/dL or 8.8% of the reference method at glucose concentrations <100 mg/dL and ≥100 mg/dL, respectively. The calculated mean absolute relative difference (MARD) was 4.21% (3.97-4.46; 95% confidence limits) across the entire glucose range (n = 654). These values are similar to those reported by other leading blood glucose meter companies including Ascensia Diabetes Care (Contour Next One) 3 and Roche Diabetes Care (Accu-Chek Guide) glucose meters. 4
Accuracy of OneTouch Verio Reflect Glucose Meter in Capillary Whole Blood Samples.
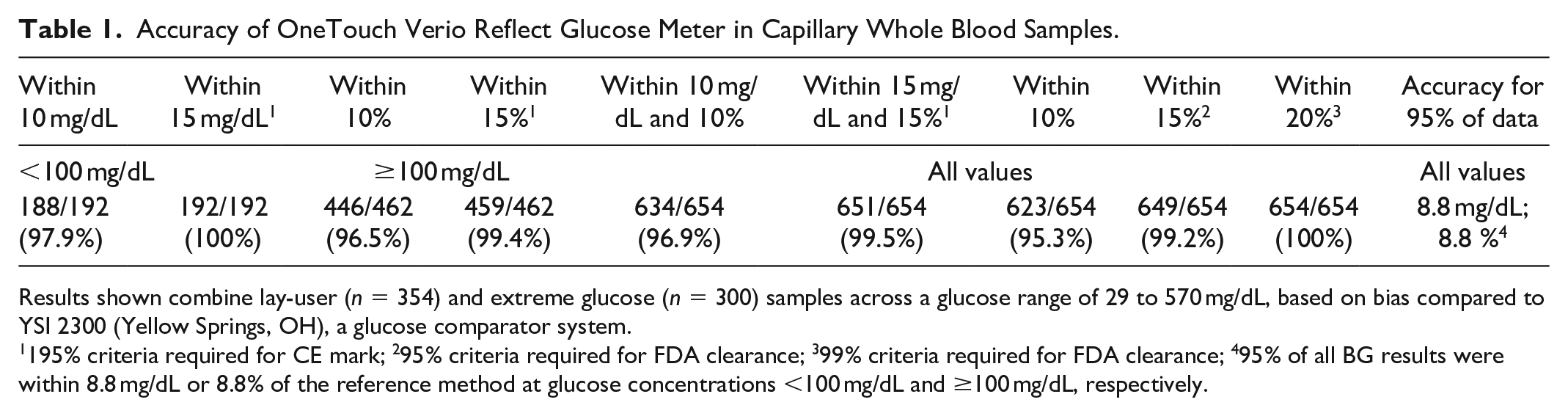
Results shown combine lay-user (n = 354) and extreme glucose (n = 300) samples across a glucose range of 29 to 570 mg/dL, based on bias compared to YSI 2300 (Yellow Springs, OH), a glucose comparator system.
195% criteria required for CE mark; 295% criteria required for FDA clearance; 399% criteria required for FDA clearance; 495% of all BG results were within 8.8 mg/dL or 8.8% of the reference method at glucose concentrations <100 mg/dL and ≥100 mg/dL, respectively.
LifeScan meters using OneTouch Verio glucose strips have been subject to an on-going clinical surveillance program by the manufacturer since 2009. Over this time, randomly sampled strip batches have exhibited excellent clinical accuracy across all ranges of subject blood glucose in over 74 000 individual glucose results.5,6
This analysis confirms that the OneTouch Verio Reflect glucose meter with an improved blood glucose algorithm has a level of accuracy beyond the minimum requirements of ISO15197:2015 and FDA guidance and compares similarly to reported data of other leading meter manufacturers.
Footnotes
Abbreviations
BG, blood glucose; MARD, mean absolute relative difference.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: LK, AS, HC, SS and MK were all full-time employees of LifeScan Global Corporation at the time of the work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by LifeScan Global Corporation.
