Abstract
Background:
Glucommander™ (GM), an electronic glycemic management system, was implemented across a multi-hospital health system as the standard of care for glycemic control. GM provides insulin dosing recommendations based on patient-specific blood glucose (BG) trends after providers select either a custom dose or weight-based multiplier as the initial dosing strategy for the first 24 hours. This study evaluated the impact of initial subcutaneous (SC) GM insulin dosing strategies on glycemic management.
Methods:
Non-intensive care unit patients treated with SC GM using either initial custom (based on provider discretion) or weight-based multiplier settings (0.3, 0.5, or 0.7 units/kg/day) were evaluated in this retrospective chart review. The primary endpoint was time to target BG range defined as time to first two consecutive in range point of care BG. Secondary endpoints included percentage of BG values in target range, percentage of orders following institutional recommendations, length of stay (LOS), average BG, and incidence of hypoglycemia and hyperglycemia.
Results:
A review of 348 patients showed time to target BG was not significantly different between custom and multiplier groups (55 vs 64 hours, P = .07). Target BG was achieved in less than half of patients in both groups (47% vs 44%, respectively). There were no differences in hospital LOS, proportion of BG in target range, rates of hypo/hyperglycemia, and average BG.
Conclusions:
Custom initial SC GM insulin dosing settings showed a nonsignificant decrease in time to target BG range compared to pre-defined multiplier settings. Future studies evaluating the impact of compliance with institutional recommendations on BG control are warranted.
Keywords
Introduction
One of the biggest challenges for many health systems is achieving adequate glycemic control in a safe and effective way. The American Diabetes Association (ADA) defines hyperglycemia in hospitalized patients as blood glucose (BG) greater than 140 mg/dL and hypoglycemia as BG less than 70 mg/dL. 1 The 2020 ADA Standards of Medical Care in Diabetes guidelines recommend initiating insulin when BG rises greater than or equal to 180 mg/dL. Hyperglycemia affects about 38% of hospitalized patients, with approximately 12% of patients having no history of diabetes mellitus (DM) prior to admission. 2 Adverse outcomes, including increased mortality, morbidity, and hospital length of stay (LOS), are associated with hyperglycemia. 1 Hypoglycemia, if untreated, may lead to loss of consciousness, seizure, coma, and death. 3
Basal insulin or basal plus correction insulin (administered in response to BG elevations) is the preferred strategy for correction of persistent hyperglycemia in non-critically ill patients with poor oral intake. 1 Alternatively, a basal, prandial bolus (administered in response to carbohydrate intake), and correction insulin regimen is preferred for patients with adequate oral intake. Once insulin therapy is initiated, a target BG range of 140 to 180 mg/dL is preferred for a majority of critically ill and non-critically ill patients. 1 The ADA recommendations are based on the results of the NICE-SUGAR randomized trial which found a decrease in 90 day all-cause mortality with conventional glucose control (BG less than 180 mg/dL) compared to intensive glucose control (BG of 81-108 mg/dL). 4
Sliding scale insulin (SSI) utilizes short-acting insulin as needed based on patient-specific BG readings. Use of this regimen is reactive to already elevated BG levels and could lead to drastic fluctuations in BG with higher rates of hyperglycemic events. Efficacy and safety of SSI compared to proactive strategies have been evaluated for this reason. 5 The RABBIT 2 trial compared subcutaneous (SC) basal-bolus (insulin glargine and insulin glulisine) dosing regimens with SC SSI (regular insulin with meals) in insulin-naïve medical patients. Results revealed that proactive basal-bolus dosing leads to improved glycemic control in hospitalized patients when compared with SSI. 6
One ADA recommended solution to effectively manage inpatient BG is utilization of decision support systems for titrating insulin doses. 7 Glucommander™ (GM) is a Food and Drug Association-approved electronic glycemic management system (eGMS) developed by the insulin management software company Glytec©, LLC 2020. This software utilizes evidence-based multivariate algorithms to provide computer-guided insulin dosing recommendations which continuously recalculate and adjust to individual BG trends. The GM algorithm limits insulin dose adjustments to a maximum increase or decrease of 30% per day to avoid hypo/hyperglycemia. Upon initiation of SC insulin management via GM, providers must select initial settings of order set type (basal-bolus plus correction or basal plus correction), target BG range, total daily dose (TDD) initial dosing strategy and daily basal percentage. 8 Providers may select from four pre-set BG ranges based on the clinical assessment of the patient (80-120, 100-140, 120-160, and 140-180 mg/dL). Glytec© uses 120 to 160 mg/dL as a standard target which aims for the lower end of the ADA recommended range. 1 The TDD initial dosing strategy allows providers to select a weight-based multiplier (0.3, 0.5, or 0.7 units/kg/day) along with a basal percentage (with the remainder divided equally among breakfast, lunch, and dinner) or a custom basal and bolus order (with individual doses specified by the provider) at their discretion. After the initial 24 hours, insulin requirements and BG readings are analyzed by GM to subsequently provide patient-specific dose adjustments based on its proprietary algorithm, regardless of the initial dosing strategy utilized. Custom orders offer more flexibility when selecting insulin doses to more closely match a home regimen, bolus dose variation or an anticipated insulin need by ordering desired doses at initiation of therapy. For each initial dosing setting, a 20%-50% reduction in home TDD may be considered for concerns of reduced hospital diet, reduced outpatient adherence and acute kidney injury. 8
Alternative eGMS that benefit glycemic control through standardization of dose adjustments similarly to GM include, EndoTool®, GlucoTab®, and GlucoStablizer®. Algorithmic standardization leads to reduced human errors that may result from rapidly changing clinical conditions. Implementation of eGMS in EMR also improves documentation and allows all members of the care team to view necessary clinical information. Differences between each eGMS system are seen in the unique algorithms used to dose insulin. There are also differences in their available platforms and integration capabilities. 9
The efficacy of GM was evaluated in a retrospective review pre- and post-implementation by Newsome et al. Over the first year, 4239 patients treated with GM basal-bolus plus correction insulin dosing strategies had improved BG control with fewer hypoglycemic events (P = .0001), fewer hyperglycemic events (P = .0001) and shorter LOS (P = .0001) compared to non-GM dosing strategies utilizing SSI. After implementation of GM, SSI utilization decreased from 95% to 4%. 10 Aloi and colleagues further compared basal-bolus plus correction SC insulin therapy managed independently by providers vs a nurse-driven eGMS. Investigators in this retrospective crossover study evaluated 993 non-ICU patients before, during, and after GM use, with providers directly managing insulin before and after GM. Compared to before and after, patients managed by GM had more BG readings in target range of 140 to 180 mg/dL (before, P = .002; after, P = .001), fewer hypoglycemic events (P = .001), and fewer hyperglycemic events (P = .001). 11 These studies demonstrate glycemic management using GM results in fewer adverse outcomes than both SSI and provider-driven management strategies.
A retrospective review conducted by Ullal and colleagues analyzed patients with DKA and compared IV glycemic control with GM to conventional insulin regimens. Treatment with GM resulted in lower rates of hypoglycemia (12.9% vs 35%, respectively; P = .001), faster time to glucose normalization (9.7 ± 8.9 hours vs 10.97 ± 10.2 hours, respectively; P = .0001), and shorter hospital LOS (3.2 ± 2.9 days vs 4.5 ± 4.8 days, respectively; P = .01). Subgroup analysis of preset IV insulin dosing multipliers (0.01, 0.02, and 0.03) demonstrated that a multiplier of 0.01 with a target BG range of 120 to 160 mg/dL resulted in the most optimal treatment outcomes with a rate of hypoglycemia of 7.9% and time to BG less than 250 mg/dL of 7.5 hours. In comparison, a multiplier of 0.02 and BG range of 120 to 160 mg/dL resulted in a hypoglycemia rate of 15.1% and a time to target BG of 7.5 hours. 12 Based on these findings, choice of initial multiplier selection may have an impact on clinical outcomes and assessing strategies to optimize setting selection would be worthwhile. Furthermore, while this study evaluated the impact of initial multiplier settings for IV dosing, there are no previously published studies that evaluate SC GM initial dosing multiplier settings. By extrapolating the results of this study, it is reasonable to hypothesize that a similar difference may be seen if a similar review were conducted for SC management.
GM was universally implemented across the health system in 2018 as the standard of care for glycemic control. An internally conducted medication use evaluation assessing 328 patients found a 14.6 hour improvement in time to therapeutic BG range during the first three months after GM was implemented compared to the three months prior (87.5 hours vs 72.9 hours; P = .015). 13 As part of our institution’s commitment to continuous quality improvement, it is important to evaluate GM utilization and implementation in order to improve time to target BG range, decrease hospital LOS, and decrease hypo/hyperglycemia. The goal of this study was to evaluate the impact of initial GM insulin dosing strategies on SC glycemic management through retrospective chart review at several institutions within the health system.
Methods
This multi-center, retrospective chart review evaluated the impact of initial GM insulin dosing strategies on SC glycemic management. The initial dosing strategies compared custom insulin doses vs weight-based multipliers 0.3, 0.5, and 0.7 units/kg/day. GM setting selection, including target BG range, was ordered by providers on a case-by-case basis. All home DM insulin regimens were converted to basal-bolus strategies, ideally following institutional clinical decision tool for insulin dosing recommendations (Table 1). Home non-insulin anti-hyperglycemic medications, including sulfonylureas, were paused with the exception of metformin. Thiazolidinediones, dipeptidyl peptidase-4 inhibitors, and alpha glucosidase inhibitors were recommended to be discontinued, however, may be continued at provider’s discretion. GM version 3.4.4.3 (Glytec©, LLC 2020) was utilized for this study.
Institutional Recommendations for Initial SC Insulin Dosing to Determine Appropriateness of Ordered Settings.
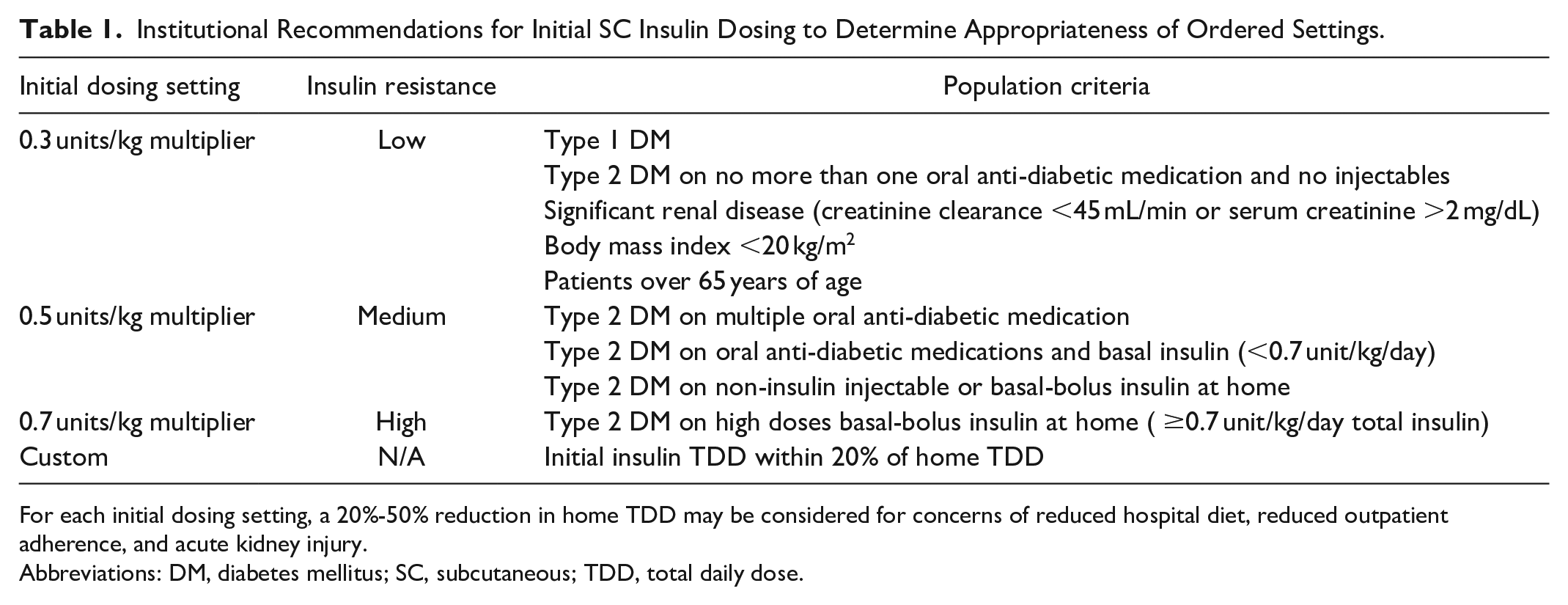
For each initial dosing setting, a 20%-50% reduction in home TDD may be considered for concerns of reduced hospital diet, reduced outpatient adherence, and acute kidney injury.
Abbreviations: DM, diabetes mellitus; SC, subcutaneous; TDD, total daily dose.
The study included 348 patients across three hospitals within the health system. Patients were included if they were initiated on SC GM for at least 24 hours between October 1, 2018 and September 30, 2019, were at least 18 years old, and had an initial BG greater than or equal to 180 mg/dL. Patients were excluded if they were obstetric or ICU patients, on IV insulin, post-cardiovascular surgery, or had an exclusion to GM use (Table 2).
Inclusion and Exclusion Criteria.
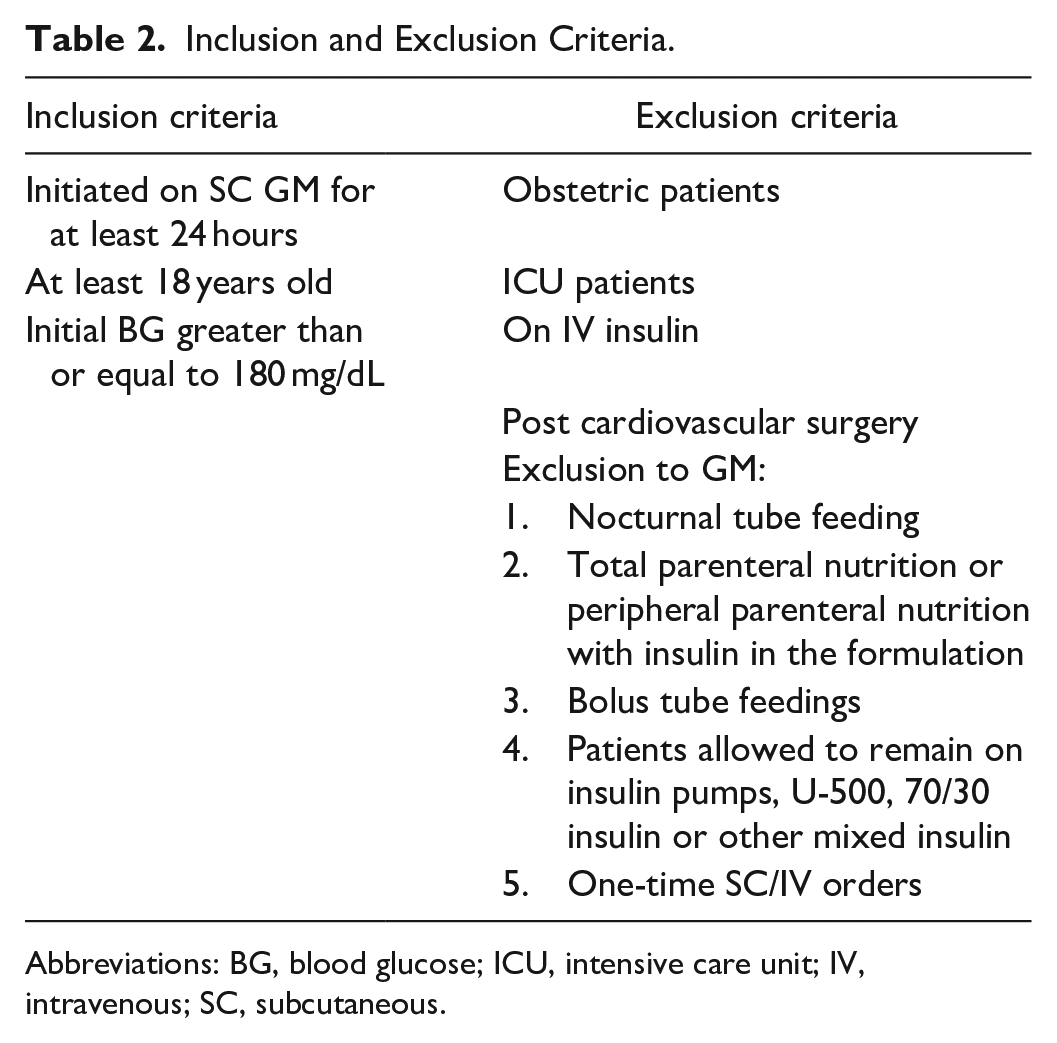
Abbreviations: BG, blood glucose; ICU, intensive care unit; IV, intravenous; SC, subcutaneous.
The primary endpoint was time to target BG range, defined as time to first two consecutive point of care (POCT) BG readings within the preselected BG range. POCT BG were drawn three times daily before meals and at bedtime with an option for a 0200 BG to be ordered at the provider’s discretion for patients with an oral diet and every four or six hours for patients on continuous tube feedings or nothing by mouth. Freestyle precision pro devices were used to collect POCT BG samples using capillary, venous or arterial blood. Typically, capillary blood was utilized unless the patient had poor vasculature, or a specific site was specified by an ordered protocol. If patient did not reach target BG range, total time on GM was used in its place. Secondary endpoints included percentage of BG values in target range, percentage of initial orders following the institutional clinical decision tool for insulin dosing recommendations (Table 1), mean LOS, incidence of hypoglycemia less than 70 mg/dL, incidence of severe hypoglycemia less than 40 mg/dL, incidence of hyperglycemia greater than 180 mg/dL, and average BG. At least one criterion from the institutional insulin dosing recommendation (Table 1), needed to be met for a regimen to be considered appropriate.
Time to target BG range and LOS were reported as medians with interquartile range (IQR) and analyzed using Mann-Whitney U and Kruskal Wallis for subgroup analysis of multiplier settings. Continuous normalized data were reported as means with standard deviation (SD) and analyzed using a two-sample t-test and ANOVA. For data represented as incidence of events, Fischer’s exact test and Chi-Square were used. A sample size of 348 patients (174 in each group) was required to meet power of 0.80 with a two-sided α of 0.05 to detect a 15 hour difference in time to target BG between the multiplier and custom groups.
Results
A total of 348 patients were randomly selected from 13,311 patients who received SC GM during the study period (Figure 1). At baseline, demographic and clinical characteristics were similar between the multiplier and custom groups in all aspects except TDD of home insulin (Table 3). Patients in the custom group had a significantly higher TDD of home insulin (62 ± 61 units) compared to the multiplier group (32 ± 38 units; P = .001). Additionally, baseline use of home non-insulin anti-hyperglycemic medications trended higher in the multiplier group (57%) compared to the custom group (47%; P = .053). Use of sulfonylureas, in particular, were higher in the multiplier group (19%) compared to the custom group (12%).
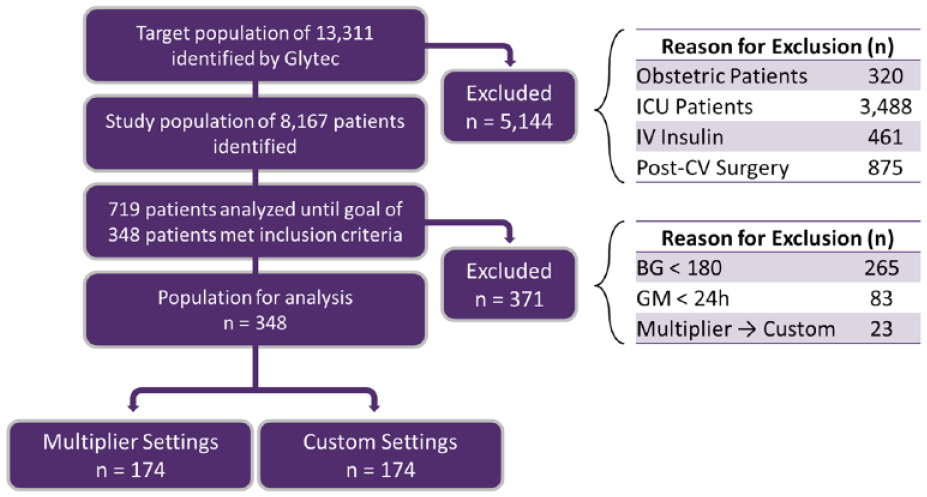
Patient allocation and exclusion.
Characteristics of the Patients at Baseline.
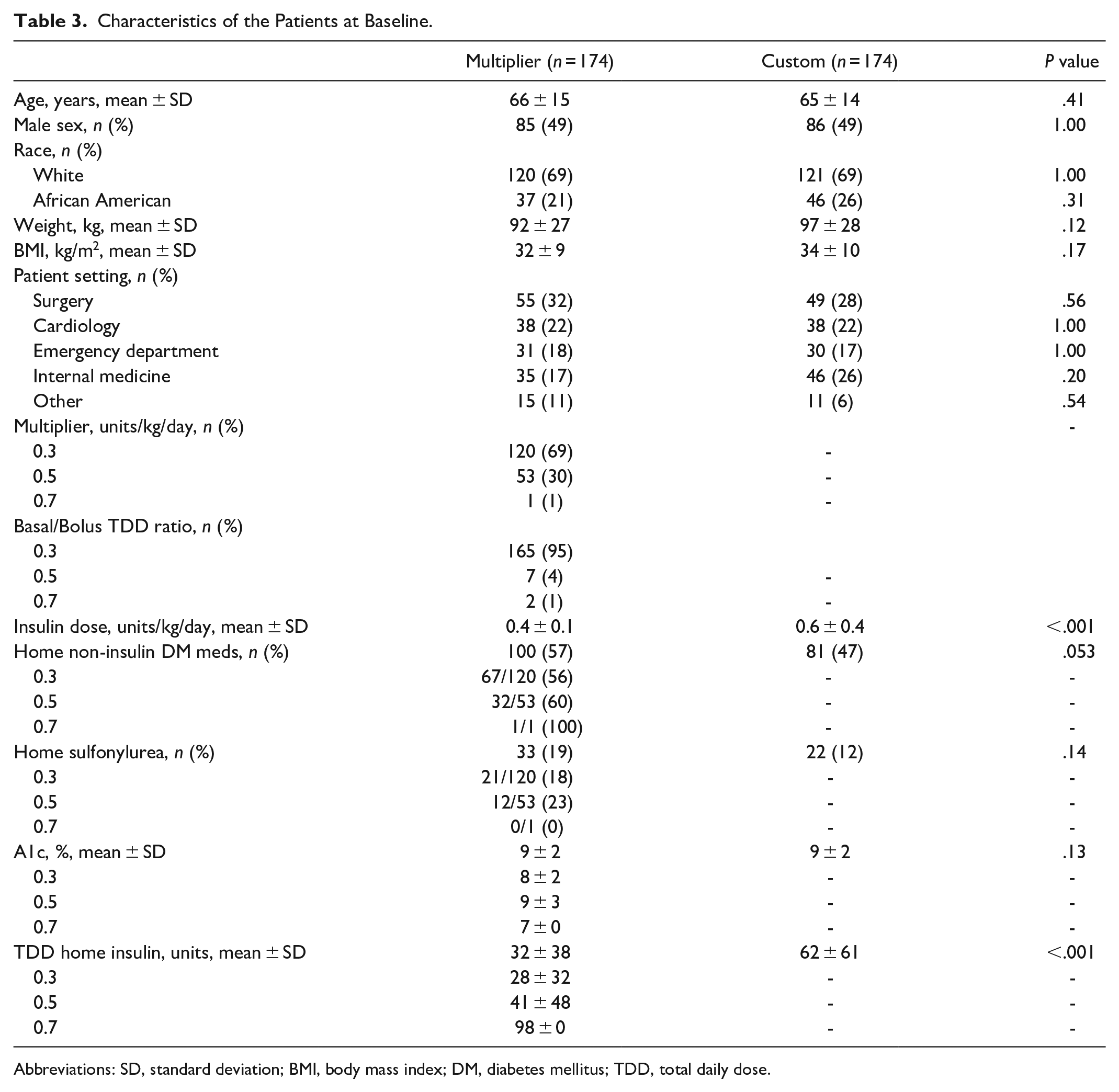
Abbreviations: SD, standard deviation; BMI, body mass index; DM, diabetes mellitus; TDD, total daily dose.
There was a nonsignificant difference in time to target BG between the multiplier (64 hours) and custom group (55 hours; P = .07) (Figure 2). Additionally, all secondary endpoints were found to be comparable between groups (Table 4). It is important to note that target BG was achieved in less than half of the study patients in both the multiplier (44%) and custom (47%) groups (Figure 3). Custom orders were more adherent to the institutional insulin dosing recommendations (60%) compared to the multiplier group (54%). Furthermore, 30% of patients in the multiplier group and 19% of patients in the custom group had a dose reduction of greater than 50% from their home insulin regimen (Figure 4).
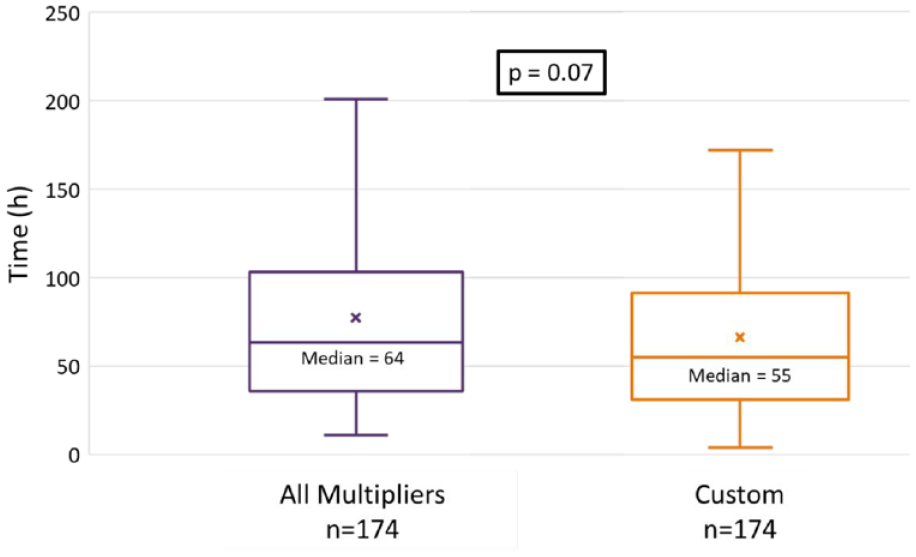
Primary endpoint: Time to first two consecutive in range blood glucose.
Secondary Outcomes.
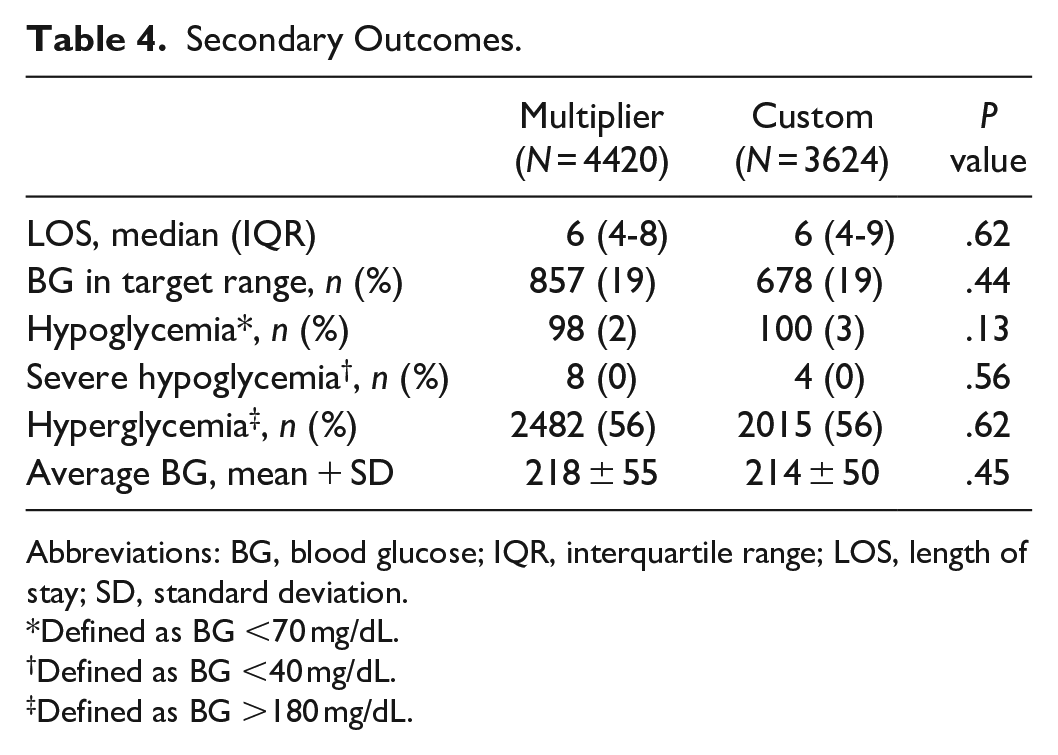
Abbreviations: BG, blood glucose; IQR, interquartile range; LOS, length of stay; SD, standard deviation.
Defined as BG <70 mg/dL.
Defined as BG <40 mg/dL.
Defined as BG >180 mg/dL.
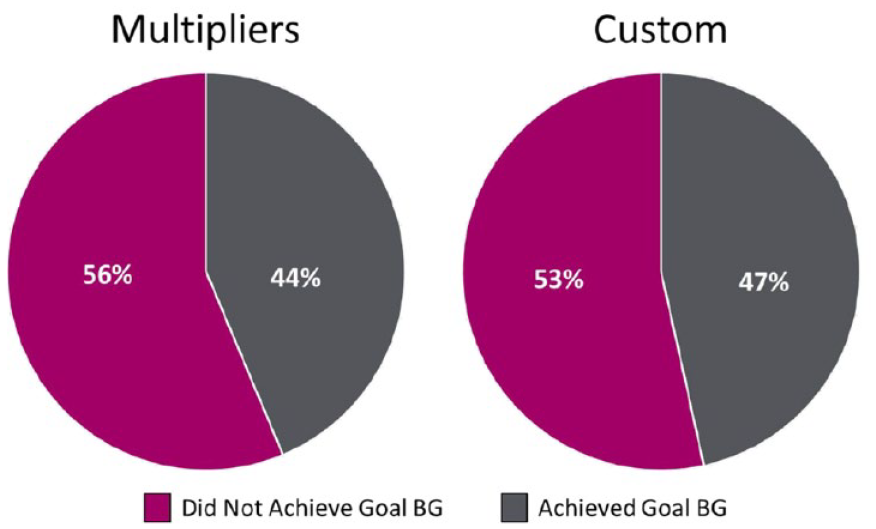
Percentage of patients achieving target blood glucose.
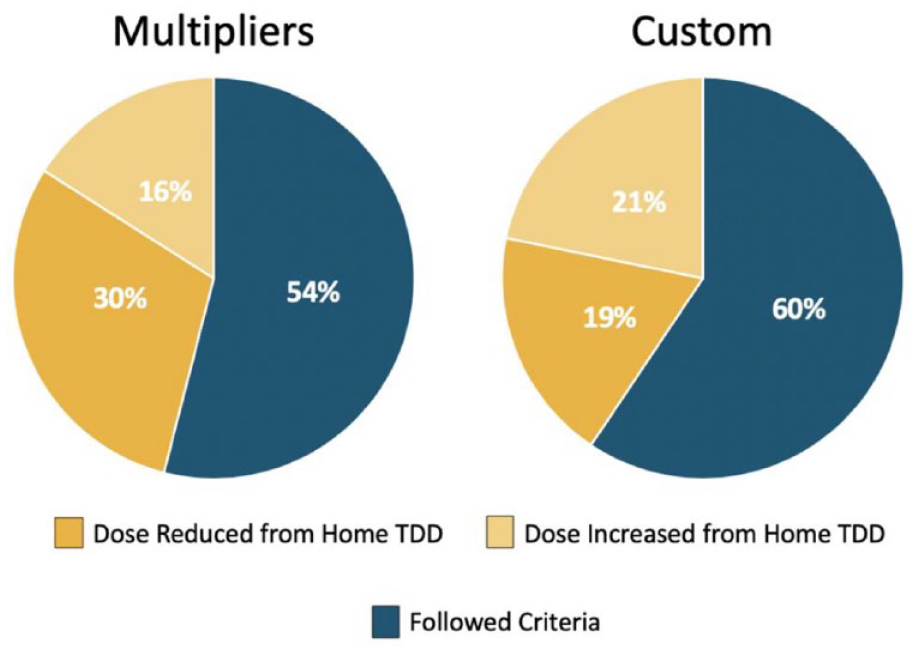
Percentage of GM orders following institutional initial insulin dosing recommendations.
In subgroup analyses of 0.3 and 0.5 units/kg/day (excluding 0.7 units/kg/day due to limited sample size), there was no difference in time to target BG, hospital LOS, proportion of BG in target range, rates of hypo/hyperglycemia, and average BG (Figure 5 and Table 5). There was also no meaningful difference between multipliers in rate of target BG achievement (Figure 6); however, the 0.3 unit/kg/day group had a lower rate of orders following institutional dosing recommendations (49%) compared to the 0.5 units/kg/day (64%) group (Figure 7).
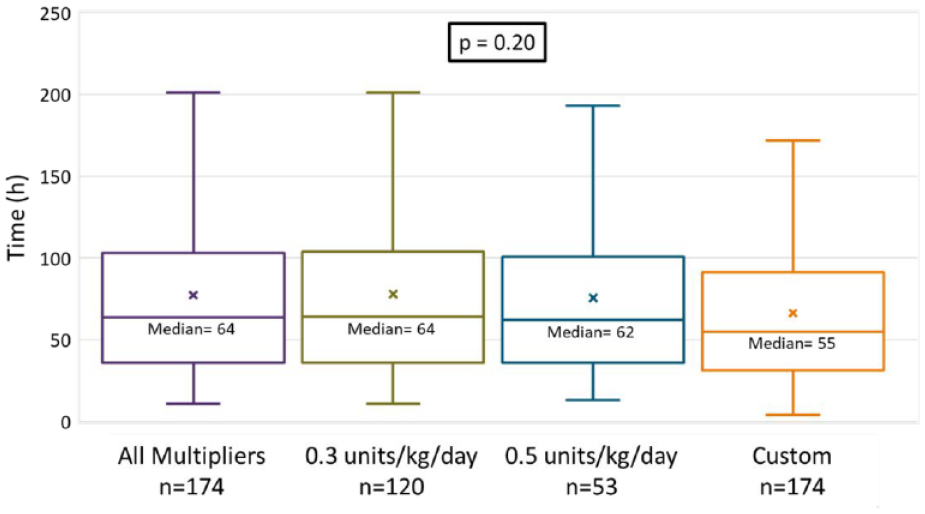
Time to first two consecutive in range blood glucose across multiplier groups.
Secondary Outcomes Across Multiplier Groups.
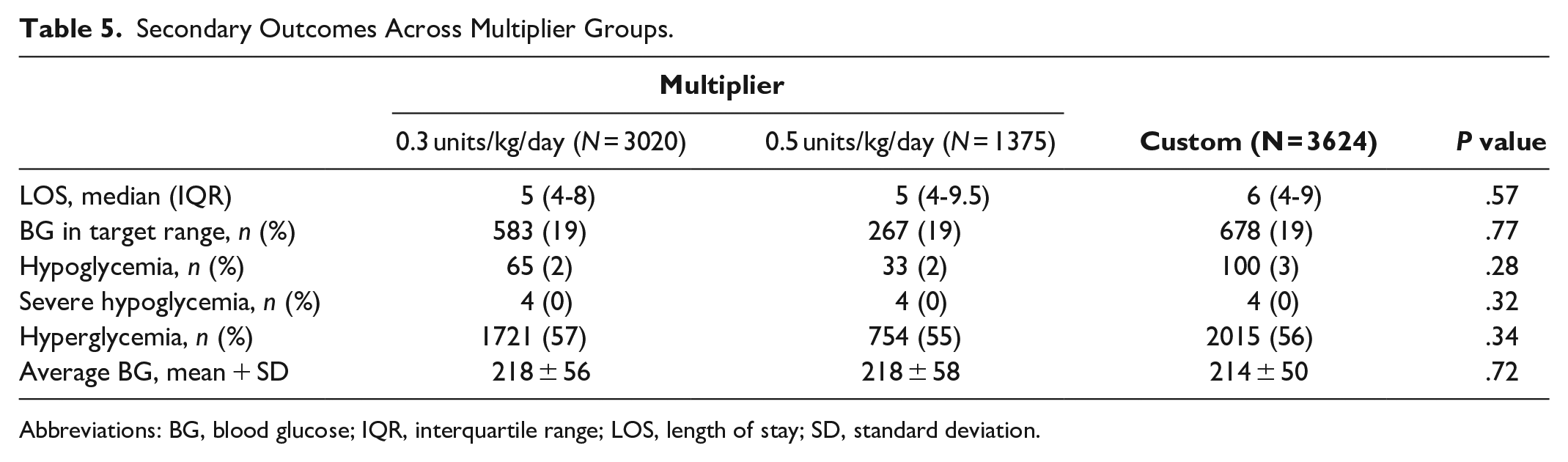
Abbreviations: BG, blood glucose; IQR, interquartile range; LOS, length of stay; SD, standard deviation.
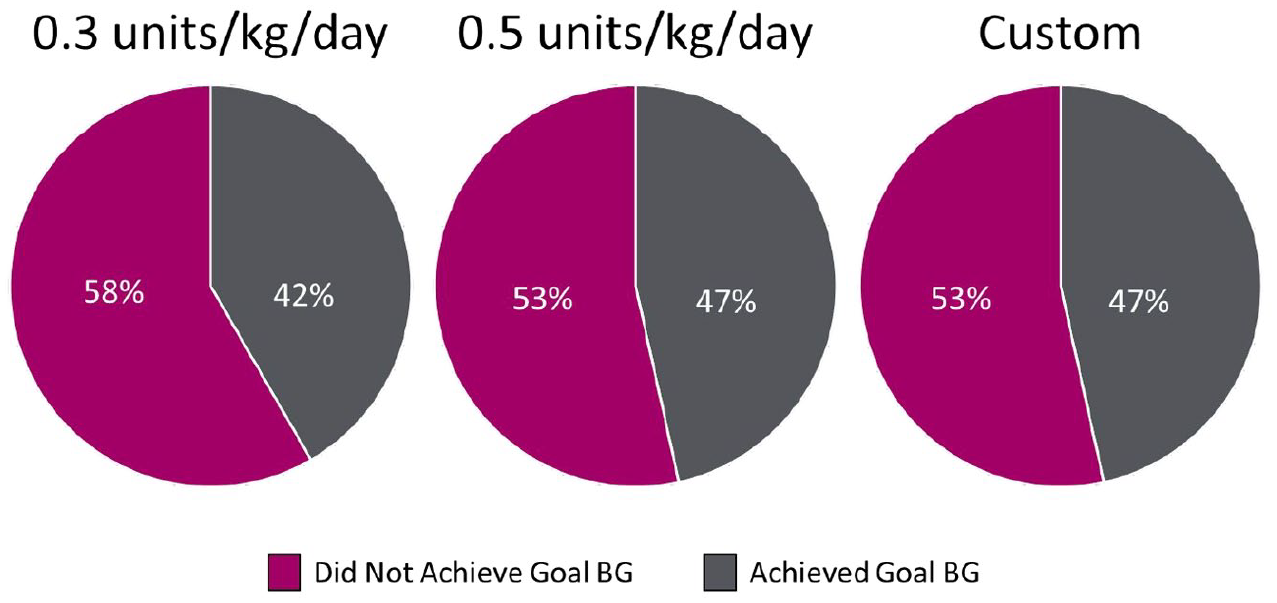
Percentage of patients achieving target blood glucose across multiplier groups.
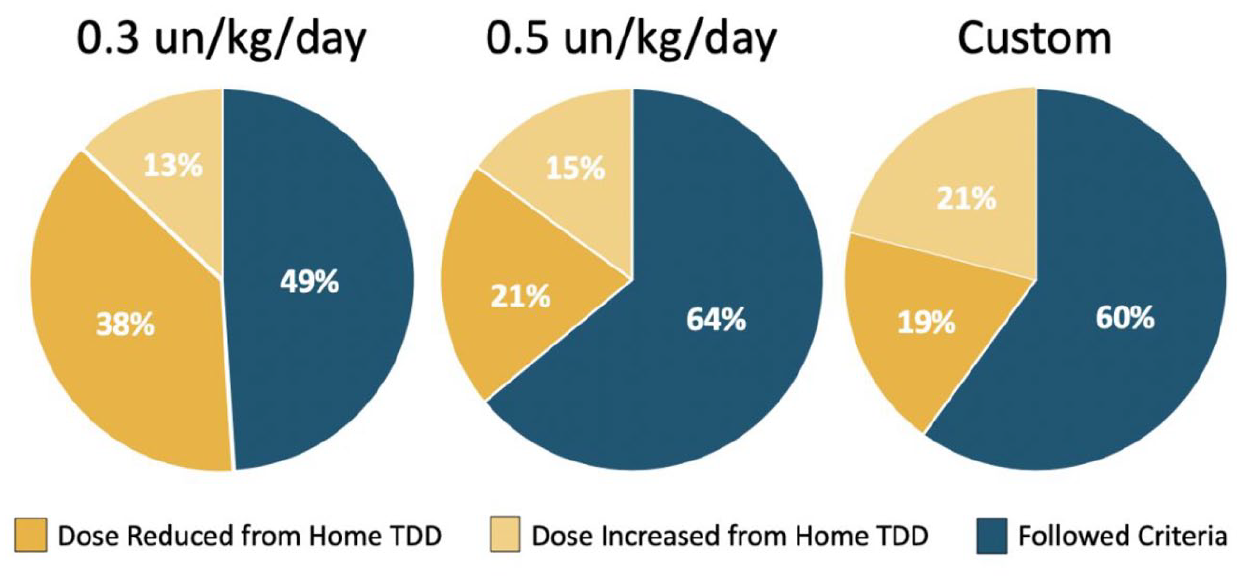
Percentage of GM orders following institutional initial insulin dosing recommendations across multiplier groups.
Discussion
Patients dosed using custom settings had a significantly higher baseline home insulin TDD than the multiplier group, potentially indicating that providers were more likely to utilize custom settings for patients with greater insulin resistance. This is a major confounding variable as insulin resistant patients may inherently take longer to achieve target BG. Despite meeting power, the treatment arms being unmatched in regard to baseline insulin dosing makes it difficult to definitively interpret the findings of this study. A larger study with matched cohorts at baseline would be ideal to support the results without the influence of initial patient complexity.
Despite having an increased baseline insulin regimen, the custom group demonstrated a nonsignificant decrease in time to target BG compared to orders utilizing initial weight-based multipliers. The nine hour reduction in BG normalization, while not statistically significant, may yield a substantial clinical improvement in glycemic management. Improved glycemic control may decrease hospital LOS, promote wound healing, and reduce the risk of infections and death. 14
The GM algorithm’s 30% daily maximum insulin adjustment limit may contribute to the difference between groups. With the algorithm’s limited ability to make substantial adjustments per day, it may take a significant amount of time for GM to catch up to the patient’s insulin needs if they differ drastically from the patient’s home requirements. Theoretically, mirroring the patient’s home regimen using custom dosing should minimize the need for algorithm adjustments and drastically reduce the time to target BG.
Another confounding variable identified in this study is the discrepancy in use of non-insulin anti-hyperglycemic medication use prior to admission, with greater use in patients in the multiplier group. These medications, including long acting glucagon-like peptide-1 receptor agonists use prior to admission, may have a substantial effect on BG independent of the patient’s insulin regimen. The degree of impact on glycemic control, may be difficult for providers to predict and translate to an inpatient insulin dose. Use of sulfonylureas prior to admission may additionally pose a special challenge to providers. Typically, sulfonylureas are held on inpatient admission due to their high incidence of hypoglycemia, especially in combination with insulin. 15 Deterioration of glycemic control is reported in multiple studies when sulfonylureas are withdrawn from therapy due to impacted insulin secretion.16-18 As such, the multiplier group may have been disproportionately impacted by this factor due to the slightly greater baseline use of sulfonylureas. Accurate medication reconciliation and critical evaluation of anti-hyperglycemic medications prior to admission, including sulfonylureas, and appropriate adjustment of insulin in response would be beneficial to improve inpatient glycemic control.
In comparison to the insulin dose prior to admission, the rate of under-dosing in the multiplier group was higher than the custom group. One factor that may contribute to this is that the 0.3 units/kg/day multiplier is the preselected setting on the institutional order set. This may result in more patients receiving lower insulin doses than their home regimens. Removing 0.3 units/kg/day as the automatic predefined multiplier and requiring providers to intentionally select the desired initial dosing setting may result in more patients receiving insulin doses closer to their home regimen and, in turn, improve the time to target BG.
While many patients did receive lower doses of insulin in comparison to their home regimen, it is interesting to note that hospital LOS, number of BG readings in target range, rates of hypo/hyperglycemia, and average BG were all similar between groups. This could be due to the consistent way GM adjusts insulin with the same algorithm, regardless of initial settings. GM’s set algorithm constantly evaluates the patient’s BG and insulin sensitivity in order to provide nurses with adjusted insulin regimens to keep patients within target range. As observed in this study, the GM algorithm is able to uniformly adjust and normalize BG with no significant difference in hypo/hyperglycemia between multiplier and custom groups.
Limitations of the study include the inability to assess home SSI regimens due to a high degree of variability and limited reporting of home regimens. For this reason, SSI doses were excluded in the calculated home insulin doses. A future prospective study would be able to account for this and provide a better estimate of SSI utilization. Additionally, due to the nature of GM, the chance of transcription errors is an unknown limitation. When providers order GM in the electronic medical record, the nurse must manually input the settings into GM. Custom orders may have a higher risk of transcription errors as nurses must manually enter the specified insulin doses as opposed to clicking a preset multiplier button when programming. The data used in this study were pulled directly by Glytec© from the settings input into GM and it is possible some degree of patients in the multiplier group were actually initially ordered as custom by the provider.
Furthermore, the fact that mean BG in both groups was elevated above recommended target and many patients did not reach target BG limits some of the conclusions that may be drawn from this study. Total time on GM was utilized in lieu of time to target BG for patients that did not achieve two consecutive in range BG levels. In doing so, the actual time needed to reach target BG may be underestimated in this study. Results would be more robust if the primary endpoint was achieved in a higher degree of patients prior to discharge.
The high percentage of non-adherence to the institutional dosing recommendations highlights that initial insulin dosing is an area of improvement for the health system. GM was implemented roughly one year prior to the start of the present study which may have impacted protocol familiarity. Additional education for providers, nurses, and pharmacists is likely warranted to increase visibility and adherence to the health system clinical decision tool for insulin dosing recommendations. Development of a new pharmacist monitoring template for use at point of verification may also be beneficial to emphasize evaluation of home insulin and anti-hyperglycemic medications. These methods will facilitate critical reviews of the patient to improve inpatient glycemic management. A future study comparing outcomes with initial dosing strategies adherent vs non-adherent to institutional guidance would also be critical to further review the impact of insulin dosing recommendations.
Conclusion
When utilizing SC GM eGMS, custom initial insulin dosing settings showed a nonsignificant decrease in time to target BG range compared to pre-defined multiplier settings. Future studies evaluating the impact of compliance with institutional recommendations on BG control are warranted.
Footnotes
Acknowledgements
Glytec for assisting in data retrieval.
Abbreviations
GM, Glucommander; BG, blood glucose; SC, subcutaneous; ADA, American Diabetes Association; DM, diabetes mellitus; ICU, intensive care unit; SSI, sliding scale insulin; eGMS, electronic glycemic management system; TDD, total daily dose; DKA, diabetic ketoacidosis; IV, intravenous; BMI, body mass index; CV, cardiovascular.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
