Abstract
The LabPatch glucose sensing device (sensor) consists of a flexible chip that is hardwired into a multichannel potentiostat and connected to a laptop to record results. The flexible chip is placed directly on the subject’s skin, and the potentiostat outputs precise voltages to the chip to trigger interstitial fluid (IF) extraction from the subject’s skin. The chip continues fluid extraction until the analysis chamber detects a sufficient volume of fluid, which then triggers the fluid analysis inside the chamber. IF analysis is based on the current output from the chip due to glucose oxidation, which is reported on the laptop as glucose concentration. This process takes around 30 seconds per test, and each chip can be used to conduct 4 tests.
We evaluated sensor accuracy using the median absolute relative difference (MARD) and Clarke Error Grid (CEG) analysis, compared to 3 references: YSI 2300 STAT Plus (YSI Incorporated, Yellow Springs, Ohio), OneTouch Verio (LifeScan, Inc., Milpitas, California) [SMBG 1], and Freestyle lite (Abbott Diabetes Care Inc, California) [SMBG 2]. We enrolled 30 subjects with type 1 diabetes (T1D) or insulin-treated type 2 diabetes. At 15- or 30-minute intervals over ~7-hour visit, measurements for sensor and references were taken. The study was powered to detect a small difference of 2% in MARD with 80% power and a 1-sided significance level of 0.0125. At least 469 independent paired glucose values were required for each comparison based on a 1-sample
A total of 1,701 (sensor-reference) pairs were captured. There was no difference between MARDs for sensor using YSI (29.4% interquartile range [IQR]: [14%-46%]), SMBG 1 (29.5% [14.3%-48.8%]), and SMBG 2 (29.8% [14.3%-50.3%]) as reference measurements (
MARD for sensor was significantly higher than those for SMBG 1 and SMBG 2 using YSI as reference (29.4% [14%-46%] vs 8.3% [4.9%-13.4%], and vs 9.1% [4.5%-13.4%], respectively,
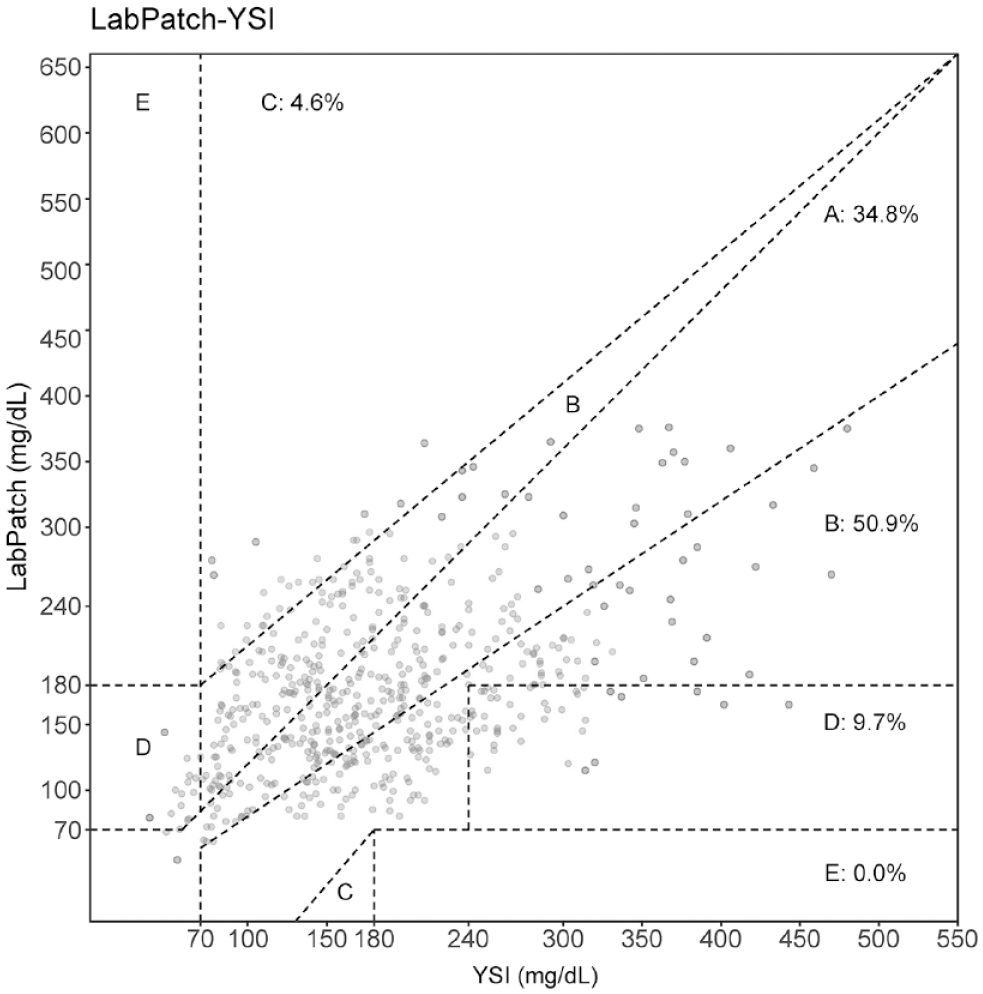
Clarke error grid analysis of LabPatch glucose sensor using Yellow Spring Instrument (YSI) as reference measurement. N = 567 paired glucose measurements. y = 0.37x + 106; Root mean square error (RMSE) = 54.
Needle-free glucose sensors could become convenient alternatives to invasive methods; however, peer-reviewed clinical data are scarce. A previous biographer (GlucoWatch [Cygnus, Redwood City, California, discontinued in 2007]) utilized reverse iontophoresis by applying a negative charge directly on the skin to attract glucose ions to an electrode. 1 In a pilot study among 28 people with T1D, GlucoWatch had a MARD of 19% SD (16.4%) and up to 41% in the hypoglycemic range.2,3 Moreover, there were reports of painful skin reactions and inaccurate calibrations. 4 Unlike the current sensor, GlucoWatch required a 3-hour “warm-up” period.
In this pilot study, a needle-free glucose sensor had comparable yet sub-optimal accuracy against YSI and 2 glucometers. LabPatch has potential to identify glucose trends in situations that require close monitoring of diabetes over shorter intervals (eg, gestational diabetes, pre-pregnancy planning, pre-elective surgery, and diabetes screening). Larger studies using standardized sensors are needed to determine if accuracy could be optimized.
Footnotes
Acknowledgements
Data from this work were presented at the American Diabetes Association’s 80th Scientific Sessions, June 2020 (Virtual).
Abbreviations
MARD, median absolute relative difference; CEG, Clark Error Grid; IQR, interquartile range; CGMs, continuous glucose monitors; FDA, Food and drug administration; YSI, Yellow spring instrument.
Author Contributions
S.T. collected data, conducted statistical analysis, and drafted the manuscript. A.H.E. and M.W.T collected data and reviewed and edited the manuscript. V.B. provided sensor technical support, contributed to the methods section, and reviewed and edited the manuscript. O.H. designed the study, supervised the work, and reviewed and edited the manuscript. S.T. and O.H. are guarantors of this work and take responsibility for the integrity of the data and the accuracy of the statistical analysis.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: S.T. reports consulting for Research America, inc. and shares in Amarin corp outside of the submitted work; V.B. is an employee of Cambridge Medical Technologies, LLC; O.H. reports consultation to Abbott Nutrition, Merck Serono and Sanofi Aventis, received research support from Novo-Nordisk, Eli Lilly, National Dairy Council, and Gilead, and is on the advisory board of L-Nutra and Twin Inc. He is also shareholder of Heathimation Inc. A.H.E., and M.W.T. have no disclosures relevant to this work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Cambridge Medical Technologies, LLC. S.T. and O.H. are supported by a National Institutes of Health (NIH)/National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) Diabetes Research Center grant (P30DK036836) awarded to Joslin Diabetes Center.
