Abstract
The current lack of continuous glucose monitor (CGM) data integration into the electronic health record (EHR) is holding back the use of this wearable technology for patient-generated health data (PGHD). This failure to integrate with other healthcare data inside the EHR disrupts workflows, removes the data from critical patient context, and overall makes the CGM data less useful than it might otherwise be. Many healthcare organizations (HCOs) are either struggling with or delaying designing and implementing CGM data integrations. In this article, the current status of CGM integration is reviewed, goals for integration are proposed, and a consensus plan to engage key stakeholders to facilitate integration is presented.
Introduction
Medicine is undergoing a digital health revolution. 1 For patients with diabetes, wearable sensors such as continuous glucose monitors (CGMs) are at the heart of this paradigm shift which is presenting massive quantities of physiological, behavioral, and environmental patient-generated health data (PGHD) to the healthcare professional (HCP). 2 This data can be collected, transmitted, presented, stored, and processed in real time to be used in decision support software or algorithms for an HCP to review in real time or asynchronously. 3 To be most useful, the data must integrate into a patient’s electronic health record (EHR). 4
Currently, very few healthcare organizations (HCOs) have successfully integrated continuous glucose monitor (CGM) data directly into the EHR, and most of those have done so with the assistance of third-party integration engines or data aggregators. This integration gap prevents an HCP from reviewing the sensor data in the same way and at the same time as they review other digital data such as clinical lab results, imaging reports, medication records, and physiology test results. 5
The integration of PGHD from CGMs into EHRs will require two steps related to the classification, transfer, and protection of this data. The first step is the creation of CGM-related data standards that provide a common language and a common set of expectations to enable interoperability between devices and systems. These standards will allow data to be organized, combined, and shared between various devices and EHRs. 6 The second step is the development of a basic set of architecture and implementation guidelines that will allow HCOs and CGM manufacturers to standardize integration projects and their various steps, including account linkage and data acquisition (with privacy and cybersecurity protections). This process can use application programming interfaces (APIs) to pull data from the CGM manufacturer’s cloud-hosted database and transfer it to the EHR for display (over any range of dates) and storage. 7 Advanced integrations could support more granular analyses of trends beyond those presented by the manufacturer, including custom metrics that flag particular types of abnormal results or apply composite metrics.8,9 Each step of the process has multiple potential configurations, and exploring each of those possibilities adds time and cost to integration efforts. A standardized and simplified set of implementation policies or guidelines would make these projects faster, more efficient, and less costly for all stakeholders.
In this article, we discuss a pathway to automatic CGM data integration into the EHR. We approach this process from the perspectives of where we are, where we are going, and how we are going to get there.
Where We Are
The Current Status of Interoperability of CGM Data
Interoperability is defined by the Office of the National Coordinator for Health Information Technology (ONC) as “the ability of a system to exchange electronic health information with and use electronic health information from other systems without special effort on the part of the user.” 10 Integration of CGM data into the EHR requires interoperability between CGM systems and EHRs and is intended to leverage that data for improved patient outcomes.
There are currently no data standards or common data management systems widely used by CGM manufacturers. This heterogeneity impacts EHR integration projects, provider workflows, and overall patient care. Further complicating this diversity is the heterogeneity among EHRs. Although the four leading EHRs by hospital market share in 2021 (Epic, 31%; Cerner, 25%; Meditech, 16%; and Allscripts, 5%) account for nearly 90% of the US market, each HCO’s instance is uniquely configured. 11 A unified approach to CGM data integration would need to take into consideration this variability among both data generators (CGM manufacturers) and data consumers (HCO EHRs). Finally, each HCO may have particular specifications for which data is being integrated, how it is being displayed, and where it is being stored.
The Inpatient Setting
CGMs have recently become increasingly established in the hospital setting because (1) a CGM can generate hundreds of measured and calculated data points in 24 hours, which far exceeds the number of fingerstick glucose tests that can be performed in a day; (2) a real-time CGM contains a hypoglycemia alarm to warn a patient or a caregiver before an impeding period of hypoglycemia that can precipitate an acute cardiovascular event; and (3) a real-time CGM contains a hyperglycemia alarm to warn a patient or a caregiver before an impeding period of hyperglycemia that can increase the risk of an infection or poor wound healing. 12 For a hospitalized patient with diabetes or stress hyperglycemia, this type of continuous glucose information would be very valuable, just as a variety of continuously monitored vital signs are valuable.13,14 Currently, no CGM is cleared by the United States Food and Drug Administration (FDA) for the inpatient setting where their accuracy and ability to deliver improved outcomes have not been established.15 -17
Since the beginning of the COVID-19 pandemic, hospitals have sought to preserve limited stores of personal protective equipment (PPE), decrease contact between patients and nurses, and save time from donning and doffing PPE. Many hospitals saw the use of CGMs instead of blood glucose monitors as a good substitute to help achieve these goals and to decrease the frequency of hypoglycemic episodes and hyperglycemic episodes.18,19 Interest in introducing CGMs into the hospital setting increased further when two CGM companies announced in the Spring of 2020 that during the pandemic, the FDA had told them that the Agency would not object if they provided devices and technical support to hospitals for this specific off-label use.20,21 Some hospital-based physicians are now prescribing CGMs for investigational or off-label use for patients with COVID-19.
A consensus guideline by 24 international CGM experts was recently published. It contains 78 recommendations for clinical practice, research, and hospital policies in five areas related to using CGMs and automated insulin dosing systems (AIDs) in the hospital. Three of their recommendations pertained to the current problem of clinicians not being able to access their patients’ CGM data in the EHR. These included (1) hospitals need to arrange for CGM results to be automatically uploaded into the EHR; (2) HCPs should develop a set of core data elements and definitions for CGM data for inclusion in common data models and the EHR; and (3) hospitals need to develop a universal platform for their EHRs that can be used by all CGMs to present core data elements and summary glucometrics, consistent formats, and uniform interfaces across all CGM products. 22
Currently, hospitalists establishing inpatient CGM programs typically create a system with factory-calibrated CGMs. These include products by Abbott Diabetes Care (Abbott Laboratories, Abbott Park, IL) and Dexcom (Dexcom, Inc., San Diego, CA). They pair the sensor with the sensor’s reader app (eg, Abbott FreeStyle Libre Link and Dexcom G6 app), the follower app (eg, Dexcom Follow and LibreLinkUp), and software to review patterns on a computer or mobile device (eg, Clarity and LibreView). 23 None of these apps connects directly with the hospital EHR, and the only way the CGM data can be analyzed is by viewing the screen or printing a report. A screenshot image can be saved in the EHR, but the data is not annotated and it will be difficult later to find the screenshot. This is less than ideal because the data is (1) not structured and (2) difficult to search for. Archiving images requires increased capacity for data storage, which could potentially affect system performance at scale. In almost all hospitals or clinics, the data points from the CGM software cannot be searched or analyzed from within the EHR at this time. The two major deficiencies of the current data platforms designed by CGM manufacturers and commercial data aggregators are that (1) the data is presented without clinical context, meaning there is no simultaneous data from the EHR available to review at the same time (such as progress notes or lab reports), and (2) review of the data requires a separate login and workflow in addition to the clinician’s normal EHR workflow.
The Outpatient Setting
The same problem of lack of CGM data integration into the EHR affects outpatients. The data can be viewed and searched to some extent in the sensor manufacturer’s software, but the data cannot be imported into the EHR and analyzed there. During a telehealth visit, a clinician may have multiple application windows open, sometimes on more than one screen—one for the telehealth visit that includes the EHR and a video of the patient, and another for the sensor software to view the data. The most efficient way to unlock the value of CGM data for reviewing, responding to, and storing is to integrate this data into the EHR. 24 Many HCOs deliver care to both outpatients and inpatients and use the same EHR for both settings, so for them a solution that integrates data into the EHR will be useful for clinicians who work both in outpatient and in inpatient settings.
Where We Are Going
Current Successful Integrations of CGM Data Into the EHR
Three main approaches to data integration into the EHR have been described: (1) using intermediary software, (2) connecting directly to the manufacturer, and (3) relying on a data aggregation platform. In 2016, Kumar et al. described a successful integration of Dexcom data into an Epic EHR (Epic, Verona, WI) through the use of an intermediate product, the Apple Health Kit (Apple, Cupertino, CA). This project allowed patients to push CGM data into their EHR and view it in the Epic MyChart patient portal. 25 In the past year, several hospitals have provided access to limited health data sets to patients through mobile apps on iPhones, although no CGM data has been available to review in these programs. 26 The Apple Health Kit is not a universal solution because it does not interact with all EHRs and it excludes patients who have Android and other smartphones. Many HCOs are reluctant to partner with consumer data and technology companies such as Apple, Facebook, and Google, because of privacy and security concerns. Finally, this approach adds the complexity of managing an additional set of integrations and data handoffs.
The process of connecting directly to the CGM manufacturer has been reported in two settings. The first was at Children’s Hospital Los Angeles where a scalable data integration project using HL7 standards allowed clinicians to request and review Dexcom CGM data from within their EHR workflow. The second was at the International Diabetes Center, which partnered with Abbott Diabetes Care on a project to make Abbott CGM data available in a group of endocrine and primary care clinics. 27 One possible downside of automatic data uploads is that clinicians might become overwhelmed with data (hundreds of data points per day) and might miss an out-of-range alarm if they are expected to review results in real time. Integration of sensor data into the EHR can support HCPs’ workflow. 28 However, introducing a new type of data into the HCP’s workflow must also be carried out carefully to avoid data overload resulting in errors due to alarm fatigue and burnout.2,29,30
Third-Party Data Aggregators
Third-party data aggregators are those entities that act as intermediaries between the CGM manufacturers and HCPs to aggregate device data. 31 Companies like Tidepool (Palo Alto, CA) and Glooko (Palo Alto, California) collect and integrate data from multiple devices and provide digital diabetes information from CGMs, blood glucose monitors, insulin pumps, insulin pens, fitness trackers, and scales. 32 These platforms do not automatically integrate into the EHR. Reviewing data often requires a separate workflow and a separate login from the EHR. Some (like Glooko) are capable of integrating with the EHR, but this can require additional contracting and data agreements, as well as significant internal information technology (IT) resources for configuration and additional fees.
Benefits of Integrating CGM Data Into the EHR
By standardizing and simplifying CGM data integrations, the barriers to entry will be lowered and more HCOs will take on these projects. As more HCOs integrate CGM data, several benefits should become clear: (1) patients will have their data in one place, leading to better care, better healthcare experiences, and higher ratings in satisfaction surveys 33 ; (2) clinicians can work faster and more efficiently when all the information they need is in one place 34 ; (3) medical billers can more readily identify documentation to bill for interpretation of CGM data and therefore bring in more revenue 35 ; and (4) HCOs can develop innovative care models that leverage data in new ways to enable smarter, more cost-effective care.
How We Will Get There
Data Standardization and Workflow Barriers to Overcome
The ONC (Office of the National Coordinator for Health Information Technology) Roadmap for Nationwide Interoperability specifies twelve policy and technical items that must be in place to achieve interoperability for a particular type of data. 10 These items are listed in Table 1. The six technical items (numbers 7-12 in Table 1) can be thought of as establishing a set of standards and the six policy items (numbers 1-6 in Table 1) can be thought of as establishing a set of workflow policies.
Policy and Technical Components of an Interoperability Program.

The iCoDE Project
In this article, we propose that a consortium of stakeholders who are interested in seeing CGM data uploaded efficiently into the EHR convene as the Integration of Continuous Glucose Monitoring Data into the Electronic Health Record (iCoDE) Project (Figure 1). The two goals of the iCoDE project will be to (1) develop standards for the exchange, classification, and mapping of CGM data and (2) develop policies to guide the integration of CGM data into the EHR. Standards will be developed to define, authenticate, extract, exchange, store, visualize, and integrate CGM data according to the ONC roadmap. Policies will be developed to guide contracting, workflows, privacy and security safeguards, and patient onboarding strategies, according to the ONC roadmap. All these standards and policies will be intended to facilitate CGM data integration. Considerations for developing data standards and implementation policies to integrate CGM data into the EHR are presented in Table 2. Building from the twelve items in the ONC roadmap for health data interoperability and the key considerations presented in Table 2, we have developed a practical framework of eight critical concepts for CGM data integration. Each concept and its component factors are presented in Figure 2. The iCoDE project will address all of the elements and subelements in that figure.

The iCoDE project logo.
Considerations for Developing Data Standards and Implementation Policies to Integrate CGM Data Into the EHR.

Abbreviations: CGM, continuous glucose monitor; EHR, electronic health record; HCO, healthcare organization.
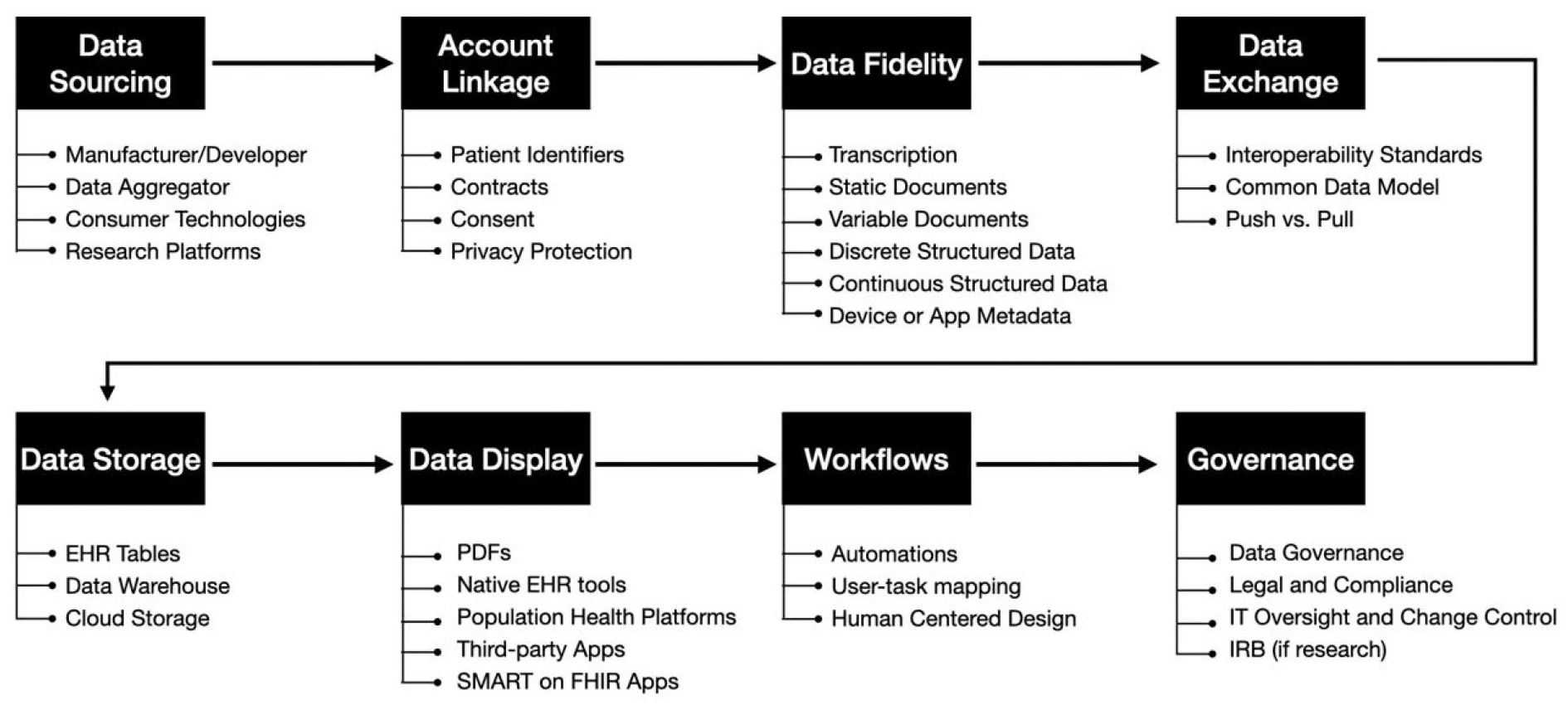
Eight critical concepts in a practical framework for continuous glucose monitor data integration into the electronic health record.
Data Interoperability Standards
Standards provide a common language and a common set of expectations that enable interoperability between systems and devices. 36 The five unique types of health IT standards for data interoperability specified by ONC along with their functions are illustrated in Figure 3. A sixth type of standard (healthcare directories and resource location) specified by ONC is actually a summary of the first five. In the following five sections, the five unique types of CGM data interoperability standards that iCoDE will develop are discussed.
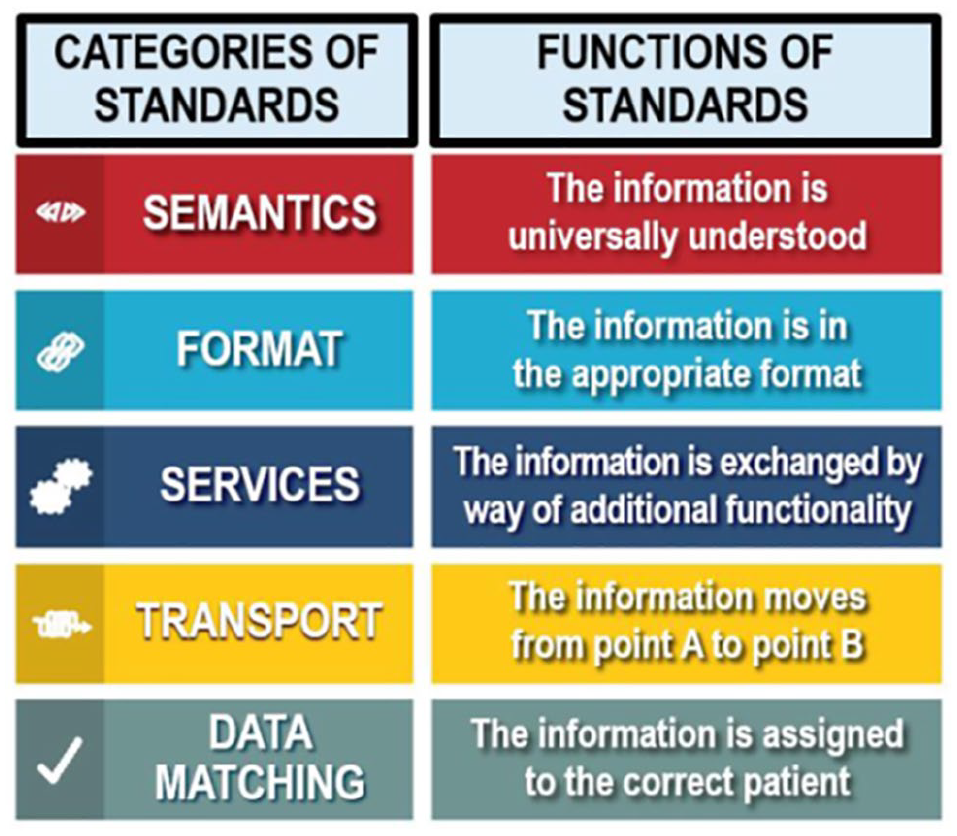
Categories and functions of five unique types of continuous glucose monitor data interoperability standards.
Consistent Data Semantics
Semantics organizes data into categories. An ontology is a formal specification of the meaning of a term or category and its relationship with other terms so that data from various sources can be identified, extracted, and aggregated. For CGM data to be interoperable, a consistent meaning for vocabularies is needed to convey clinical information. Core data elements should be mapped to existing medical data classes and data elements using applicable vocabulary standards as specified by the United States Core Data for Interoperability (USCDI) standard. 37 Differences between CGM vendors in data definitions or methods for calculating metrics increase complexity, create the possibility of clinical and technical errors, and require the maintenance of multiple data dictionaries.
Consistent Data Format
Data interoperability requires consistent representation of electronic health information across different systems. The format of shared electronic health information, which is how the information is packaged, must be consistent and machine-readable to retain the meaning of the information when it is shared.
Standard Secure Services
Data integration functions should be modular, secure, and standards-based if possible. Architecture can be defined as the collective components of a software system that interact in specified ways and across specified interfaces to ensure specified functionality. 38 Service-oriented architecture (SOA) integrates distributed software components, including CGM APIs. With SOA, diverse APIs can share algorithms and capabilities by using shared services, rather than requiring unique functionality each time they are needed. 39 The result is less complexity and less cost to the system.
Consistent Secure Transport Techniques
Transport techniques are the means by which data gets from one place to another securely. Interoperability requires that transport techniques have three characteristics. They are (1) non-proprietary, (2) easy to configure, and (3) widely used. Any transport standard for CGM data exchange should be secure and require the fewest number of necessary protocols.
Accurate Individual Data Matching
CGM data from various sources must be accurately matched to prevent information fragmentation and incorrect merging or duplication of records. Algorithms must be developed to identify individuals by using the minimum number of unique identifiers possible. During matching and linkage, it is important to be aware of and avoid certain types of patient record errors: (1) duplicate records (a patient has two or more assigned medical record numbers), (2) overlap records (the patient has different medical record numbers in separate facilities), and (3) overlay records (one medical record contains information of two separate individuals).
ICoDE Deliverables
iCoDE will consist of key stakeholders who will meet via videoconferencing. The members of this consortium will have the technical and clinical experience to develop data standards and implementation policies to integrate CGM data into the EHR. The goal will be to create a consensus set of standards and policies that will address the current barriers that impede the flow of CGM data from siloed software and mobile apps into the EHR. Once there, the liberated data can be combined with other health information to develop better treatment plans for managing the health and well-being of people with diabetes.
Conclusions
CGM data is a powerful tool for the clinical management of diabetes, and it will only become more useful as it becomes integrated into the clinician’s main EHR workflow and is contextualized with the rest of the patient’s clinical data. This integration will be one more component in healthcare’s paradigm shift from episodic care to continuous care that generates large amounts of data with increasing volume, velocity, variety and value, and veracity. 40 Integration of CGM data into the EHR will require the establishment of data standards and implementation policies. Now is the time for key stakeholders from the healthcare, industry, academic, IT, and regulatory sectors to come together and develop needed standards and policies for this purpose. The iCoDE project is our proposed approach to create consensus, advance the field, and make this technology more accessible to all patients and healthcare professionals.
Footnotes
Acknowledgements
We thank Annamarie Sucher-Jones for her expert editorial assistance.
Abbreviations
AID, automated insulin dosing system; API, application programming interface; CGM, continuous glucose monitor; EHR, electronic health record; FDA, Food and Drug Administration; HCO, healthcare organization; HCP, healthcare professional; iCoDE, Integration of Continuous Glucose Monitoring Data into the Electronic Health Record; IT, information technology; ONC, Office of the National Coordinator for Health Information Technology; PGHD, patient-generated health data; PPE, personal protective equipment; SOA, service-oriented architecture; USCDI, United States Core Data for Interoperability
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JE’s efforts were supported by the Food and Drug Administration under award number P50FD006425 for The West Coast Consortium for Technology & Innovation in Pediatrics, and grants UL1TR001855 and UL1TR000130 from the National Center for Advancing Translational Science (NCATS) of the US National Institutes of Health. The funding sources had no involvement in the development of this manuscript or in the decision to submit the paper for publication. The content is solely the responsibility of the authors and does not necessarily represent the official views of the FDA or the NIH. JE is a consultant for AI Health. NYX and KTN have nothing relevant to disclose. DCK is a consultant for Abbott, Dexcom, EOFlow, Integrity, Lifecare, Medtronic, Novo Nordisk, Roche Diagnostics, and Thirdwayv.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
