Abstract
Background:
Lancet pricks are often poorly received by individuals with diabetes; jet injection may allow lancet-free blood sampling. We examine whether the technique of jet injection can release sufficient blood from the fingertip to enable measurement of blood glucose concentration. In addition, we assess the effect of jet shape and cross-sectional area on fluid release, blood dilution, and perceived pain.
Methods:
A randomized, single-blind, crossover study was conducted on 20 healthy volunteers who received interventions on four fingertips: a lancet prick, and jet injection of a small quantity of saline solution through three differently shaped and sized nozzles. Released fluid volume, blood concentration, and glucose concentration were assessed immediately after the intervention. Pain perception and duration, and any skin reactions, were evaluated both immediately and 24 hours after the intervention.
Results:
Jet injection released sufficient blood from the fingertip to conduct a glucose measurement. A slot-shaped nozzle released the most blood, although less than a lancet, with slightly higher pain. The blood glucose levels estimated from the extracted fluid showed a mean absolute percentage error of 25%. There was no consistent evidence that a jet injection leads to different skin reactions at the intervention site relative to a lancet prick.
Conclusions:
Fingertip penetration by jet injection can release a volume of fluid sufficient for blood glucose measurement. Jet injection with a slot-shaped nozzle and/or a nozzle with larger outlet area helps to release more fluid. This technique may enable blood sampling, glucose concentration measurement, and insulin delivery to be performed in a single device.
Introduction
Individuals with diabetes typically measure their blood glucose concentration several times per day. Blood glucose concentration is usually measured from a drop of capillary blood released by a lancet from a fingertip. Despite its sensitivity, the fingertip has been the standard site for capillary blood sampling due to its high density of blood vessels. However, patient anxiety, pain, local skin damage, and risk of infection have spurred increasing efforts to develop lancet-free methods of capillary blood sampling.1-6
Needle-free jet injection is a well-developed technique for liquid drug delivery into skin: A narrow, high-speed jet of liquid can readily pierce the skin and penetrate many millimeters into the underlying tissue. 7 In a previous study, researchers showed that jet injection into skin released glucose-containing interstitial fluid 8 which was diluted approximately 100-fold by the injected fluid. However, there is no previous study of using a jet injector to deliberately release and collect blood.
An electronically actuated jet injector provides dynamic control over injection pressure and can be used to inject liquid to a controlled depth. 9 Moreover, such injectors are reversible, enabling the applied pressure to be inverted when necessary, creating a vacuum for liquid extraction from an injection site. Any blood released may be diluted by the injectate, which will need to be accounted for if glucose concentration measurements are performed. It is also known that the shape of the nozzle through which the jet is created affects jet shape and tissue penetration. 10 We have previously shown that slot-shaped nozzles with rectangular outlets that resemble the dimensions of a lancet have better potential to release fluid for glucose measurement.11,12
In this work, we conduct a study to investigate the effectiveness of jet injection at releasing blood from human fingertips. On 20 volunteers, we perform four interventions, one lancet and three jet injections, and collect the fluid released from the intervention site in four aliquots, over a period of 60 seconds. The collected fluid samples are analyzed for blood dilution and glucose concentration. The effects of nozzle shape and nozzle outlet area on fluid release are evaluated by jet injection with three differently shaped nozzles.
Methods
Participant Recruitment
This study was prospectively registered (ACTRN12619001362189, anzctr.org.au) and conducted in New Zealand with Health and Disability Ethics Committee approval (19/NTB/168). This study was advertised within the Auckland Bioengineering Institute and Department of Engineering Science at the University of Auckland in New Zealand. The trial consisted of a single visit and a follow-up questionnaire. All participants provided written informed consent before the trial.
Twenty participants aged between 20 and 60 years were required for the study. Key exclusion criteria were insulin-dependent diabetes, hemophilia or other bleeding/clotting disorders, being a carrier of blood-borne infectious agent (eg, human immunodeficiency virus [HIV], hepatitis B virus [HBV]), and fingertip amputations.
Study Design and Procedures
Interventions
Each participant was subjected to four interventions: a standard lancet prick, and jet injections with a cylinder nozzle, a medium slot nozzle, and a small slot nozzle (see Supplemental Material). The lancet prick acted as the control and was performed in accordance with World Health Organization (WHO) guidelines 13 by piercing the skin to a depth of 2.3 mm. The lancing was performed using a standard, commercially available device (ACCU-CHEK Safe-T-Pro Plus, Roche, Basel, Switzerland).
The cylinder nozzle was a conventional, commercially available jet injection nozzle (200 µm in diameter, Comfort-InTM ampoule, Mika Medical, Busan, Korea). The medium slot nozzle was constructed with dimensions of 450 by 70 µm—a nozzle outlet area equivalent to that of the cylinder nozzle. The small slot nozzle had the same width but a smaller dimension along the long axis (200 by 70 µm) and hence a smaller outlet area than the medium slot nozzle.
The jet injection system was a custom handheld electronic jet injection device driven by a portable power amplifier and controller. 14 The stainless steel nozzles and ampoules used with the injector were steam sterilized prior to each use. Jet injections, each of less than 25 μL of sterile isotonic saline, were delivered at a speed of approximately 200 m/s into the fingertip. The jet injections were designed to target a similar depth in the dermis as the lancet prick.
Each intervention was performed on a different fingertip. These interventions were performed on the side of the fingertip of the middle (third finger) and ring finger (fourth finger) of each hand. The order of interventions and the identity of fingers that receive these interventions were randomized. The participants were blinded by an opaque barrier that prevented them from observing the procedure but allowed them to communicate with the practitioner.
Sample collection
The site was wiped immediately after each intervention. Squeezing of fingers was performed in accordance with WHO guidelines before each collection, 13 consistently across all interventions. The fluid released was then collected in round capillary tubes (Vitro-TubesTM; VitroCom, New Jersey) at four time periods following the intervention: 15, 30, 45, and 60 seconds. Each intervention was thus associated with four blood samples (a total of 16 blood samples per participant). After each intervention and sample collection, there was a 5-minute pause before the next intervention.
Outcome Variables
Fluid volume
After each collection, the capillary tube containing the sample of extracted fluid was imaged under a digital microscope (Digitech QC3199, Digitech industries, Sheung Wan, Hong Kong) with a transmission light source. The sample volume was calculated from the inner diameter (0.6 mm) of the capillary tube and the length of the tube that was filled with the sample, which was measured by performing image analysis in ImageJ (Fiji). 15 Measurements performed on known volumes indicated that this method provided an accuracy of ±4.3%.
Blood dilution
In this trial, the blood concentration of each fluid sample was determined using colorimetry16-18 and expressed as a percentage with respect to the red color of blood 19 that was discharged during the first 15 seconds after lancing. An estimate of the blood concentration was determined from the mean red pixel value of the image containing the fluid sample. The relationship between red pixel value and blood concentration was precalibrated using pig and human blood (see Supplemental Material) and parameterized for each patient from the first sample of lancet-released blood.
Glucose concentration
Immediately after microscope imaging of each sample-containing tube, fluid was ejected into single-use glucose test strips. The glucose concentration of each sample was measured using a glucometer (CareSens N point of care; i-SENS, Inc, Seoul, Korea). This device has a rated accuracy of ±15.0 mg/dL for glucose concentration <100 mg/dL and ±10% for glucose concentration ≥100 mg/dL, and repeatability within 10.8 mg/dL. The minimum sample volume required to conduct a glucose measurement in a test strip was 0.5 μL. The blood glucose level of any participant was estimated by dividing the glucose concentration of a diluted blood sample by the estimated fraction of blood in that diluted sample.
Perceived pain and skin reaction
After each intervention and blood collection, the participant was asked to assign the pain a score from 0 to 10, with 0 representing no pain and 10 representing extremely severe pain. 20 A microscope image of the intervention site was taken immediately to record any swelling or bruising. The participants were asked to complete a questionnaire 24 hours later and to reassess the level of pain, swelling or bruising, and the duration for which pain or discomfort had remained. A photograph of each intervention site on the fingertips was requested.
Statistical Analysis
Twenty participants were required to achieve 90% power at a significance level of 5% in this study. With an anticipated standard deviation of 0.5 µL in the volume of fluid collected in the study population, 21 this sample size allowed us to observe differences in the mean volume of blood mixture to a lower limit of 0.5 µL. The data across the four interventions were analyzed using the Statistical Product and Service Solutions (SPSS) platform Build 1.0.0.1447 (IBM corporation, Armonk, New York). The linear mixed model coupled with pairwise comparisons was used to compare the means of the volume and blood concentration of fluid samples in pairs from four interventions within each participant. Because each participant was measured in each intervention, the linear mixed model adjusted the within-subject correlation of the measurements.
Significance tests were also performed on the perceived levels of pain caused by each intervention and to compare the mean fluid volume and blood concentration in each sample between collection time (15, 30, 45, and 60 seconds) and intervention (four levels). The Shapiro-Wilk test was used to test the normality of the data; a square-root transformation was applied to the volume of fluid samples so that they were normally distributed. 22
The correlation between the total volume of fluid samples and pain score was assessed by Pearson correlation coefficients. The significance of differences among regression lines, or between mean values, of the two groups was tested and declared when P < .05. There was only one fluid sample collected at the third and fourth time period following jet injection with the small slot nozzle. These two samples alone were insufficient to compare the means of volume and blood concentration of fluid samples, collected at these two time periods, with other interventions and were thus excluded in this case.
Results
Twenty healthy participants (five women, 15 men; mean age 33 years, aged 20-51 years; 16 right hand dominant, four left hand dominant) took part in this study. All lancet interventions resulted in blood release. A jet injection was deemed to have penetrated the skin when a wound was observed on inspection under a microscope. Of 60 jet injections, 51 penetrated fingertip skin and released a measurable volume of fluid.
Fluid Release, Blood Concentration, and Pain
The volume of fluid collected during the 60 seconds following each intervention is summarized in Figure 1a. The lancet prick released significantly more fluid than jet injection (P < .05). All 20 lancet pricks, 16 (of 18) jet injections with the medium slot nozzle, and 10 (of 17) jet injections with the cylinder nozzle released at least 0.5 μL of fluid (the minimum volume required for glucose measurement) over 60 seconds; no injections using the small slot nozzle reached this threshold. Jet injection with the medium slot nozzle released a larger fluid volume than jet injection with the cylinder nozzle (P < .05). The small slot nozzle released less fluid than the other three interventions (P < .05).
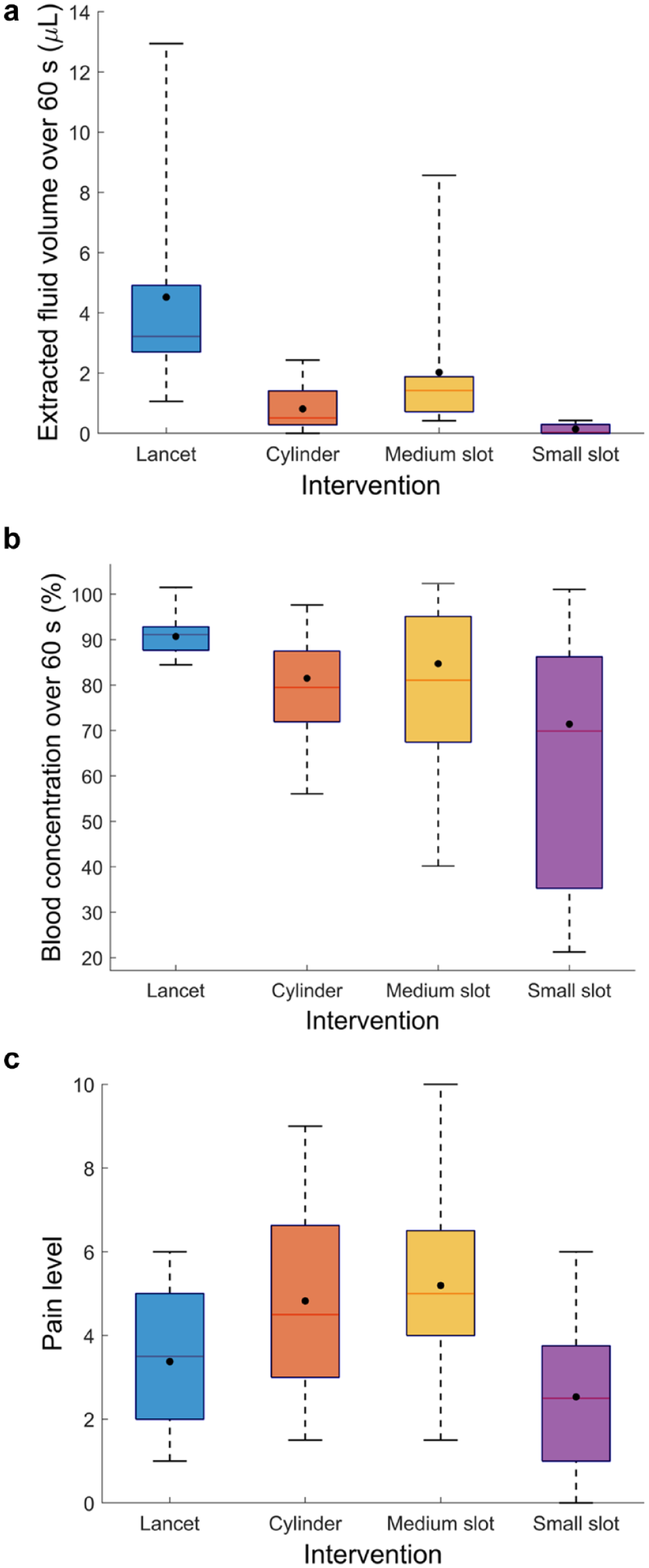
Boxplots showing the effect of intervention on the (a) total volume of fluid collected, (b) blood concentration, and (c) pain score perceived. The box spans the interquartile range (25th to 75th percentiles). The marker inside the box indicates the mean value. The line inside the box denotes the median value. Whiskers extend from the group minimum to maximum values.
The fluid samples released over 60 seconds by lancet pricking were slightly diluted, but more concentrated than those collected following jet injection (P < .05) (Figure 1b). There was no significant difference in the blood concentration of fluid samples collected over 60 seconds across jet injection with three nozzles. Fluid collected after jet injection with the cylinder nozzle and the medium slot nozzle exhibited a mean blood concentration of approximately 80%. There was a wide variation in the blood concentration released by the small slot nozzle compared with the cylinder and the medium slot nozzles.
The participants reported a pain level of 5.2 ± 2.0 (mean ± standard deviation) and 4.8 ± 2.1 from jet injection with the medium slot nozzle and cylinder nozzle, respectively (Figure 1c). Both the medium slot nozzle and cylinder nozzle caused a higher level of pain than the lancet prick (P < .05), whereas jet injection with the small slot nozzle caused less pain (P < .05). Only one participant reported pain levels of above 7, which occurred following jet injection with the circular jet and the medium slot-shaped jet. There was no correlation between the total fluid volume collected and pain score for any interventions (P > .05).
Fluid Release and Blood Concentration Over Time
The volume of fluid samples collected at every time period following each intervention is illustrated in Figure 2a. The lancet prick released significantly more fluid than jet injection with the cylinder and small slot nozzles at every time period (P < .05). There was no difference between the fluid volumes obtained from a lancet prick and jet injection with the medium slot nozzle at the first time period. Jet injection with the medium slot nozzle resulted in a higher volume of fluid samples than jet injection with other nozzles during any collection time period (P < .05).
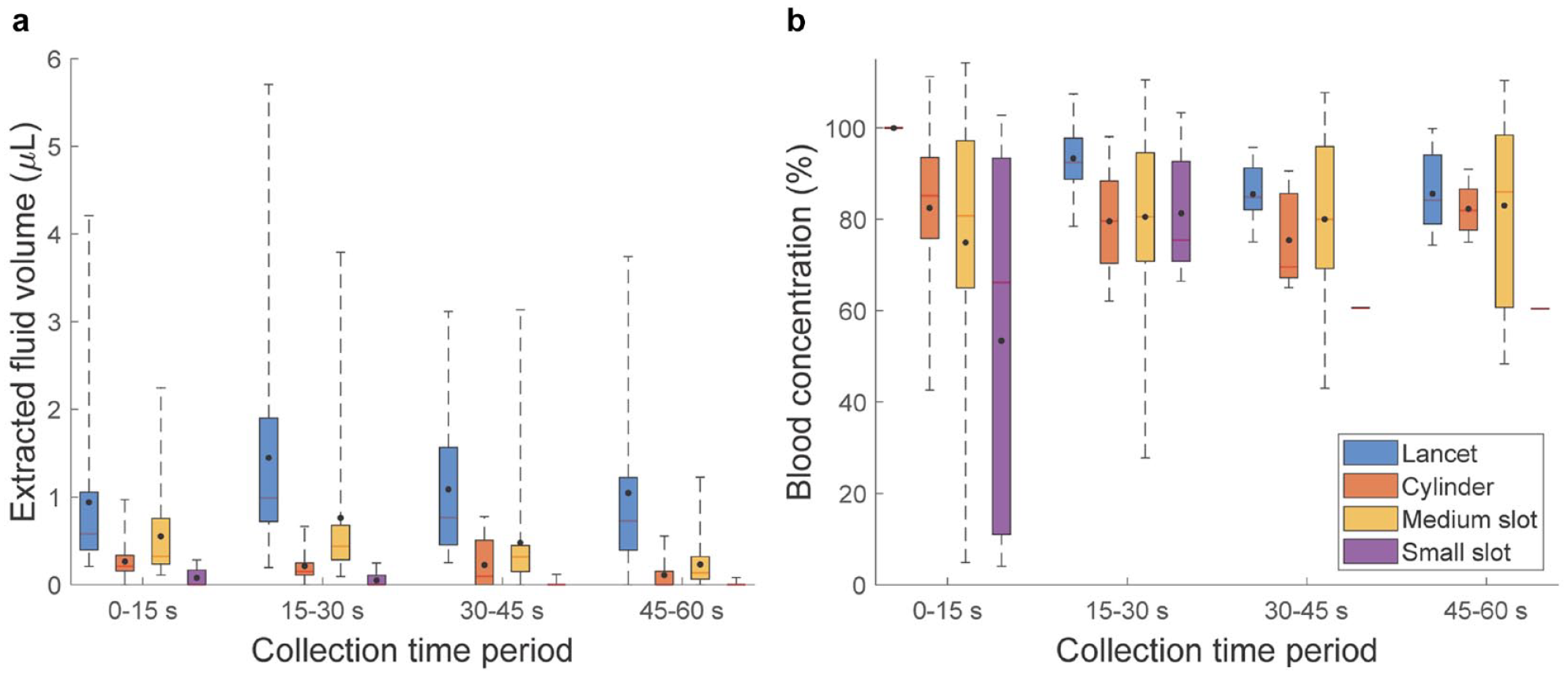
Boxplots showing the effect of intervention and collection time period on the (a) fluid volume and (b) blood concentration.
Fluid samples resulting from the lancet prick were more concentrated than those arising from jet injection at the first time period and those arising from jet injection with the medium slot and cylinder nozzles at the second time period (P < .05) (Figure 2b). There was no difference in blood concentration between fluid samples obtained at the third time period between lancet prick and jet injection with the medium slot nozzle. No difference in blood concentration of fluid samples obtained at the last time period was observed across the lancet prick and jet injection with the medium slot and cylinder nozzles.
Blood Glucose Concentration
The glucose concentration of lancet-released samples showed little change with time. Nine jet-released fluid volumes (from 15-second collection periods) were sufficient to enable glucose measurement in the glucometer (Figure 3). The blood glucose concentration estimated from these samples had a mean absolute percentage error of 25% when compared with measurements from lancet-released blood. The root mean squared error across all estimates of blood glucose was 34.6 mg/dL. The best-fit linear relationship between predicted and actual blood glucose concentration was 0.94 ± 1.00 (mean ± standard error). The offset of the fitted line was 2.81 ± 102 mg/dL.
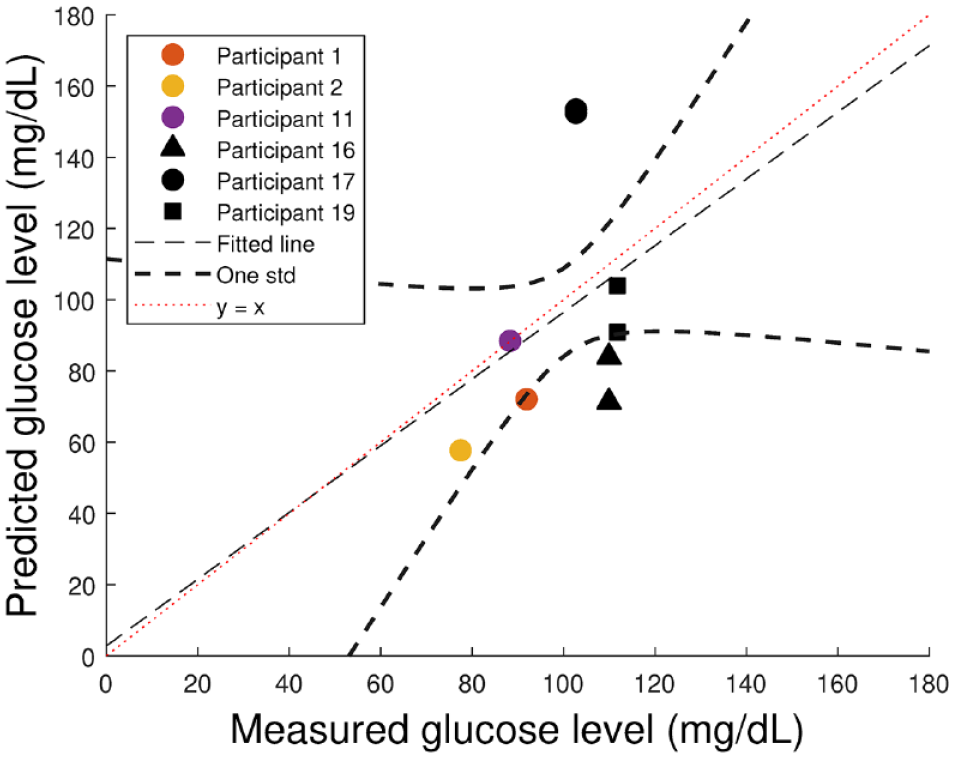
A scatter plot with line of best fit showing the predicted blood glucose levels of samples collected after jet injection plotted against the measured glucose levels of blood samples following a lancet prick. The dashed curves indicate the boundaries of data within one standard deviation of the mean.
Skin Reaction
The lancet-pricked fingertip site consistently exhibited a slit-shaped wound immediately after the intervention (Figure 4a). Jet injection with the cylinder nozzle and the medium slot nozzle resulted in a shorter but slightly wider wound than lancet prick (Figure 4b & c). The wound induced by successful jet injection with the small slot nozzle was the smallest among the four interventions, although infrequently observed (Figure 4d).

Examples of images of fingertip intervention sites taken soon after receiving (a) lancet prick, participant X; (b) jet injection with the cylinder nozzle, participant X; (c) jet injection with the medium slot nozzle, participant Y; and (d) jet injection with the small slot nozzle, participant Z.
Seven participants self-reported 10 cases of bruising on fingertips 24 hours postintervention: five from lancet pricks, two from jet injection with the cylinder nozzle, and three from jet injection with the medium slot nozzle. Five cases of swelling at the intervention site were reported 24 hours postintervention: one from lancet prick and two each from jet injection with the cylinder and the medium slot nozzle. Of the five cases, one participant experienced swelling at the intervention sites from three interventions. There was no difference between the time for which pain or discomfort remained around the penetration sites following any interventions.
Discussion
The measurements of fluid volume in the samples demonstrate that jet injection can release blood from human fingertips. Most jet injections with the medium slot and cylinder nozzles released sufficient fluid (≥0.5 μL) for conducting a glucose measurement. The medium slot nozzle released more fluid than the cylinder nozzle with no difference in blood dilution or pain. This finding suggests that the slot shape may be a better design to obtain a fluid sample for glucose measurement.
Jet injection with the small slot nozzle was less likely to penetrate skin and released less fluid than the other two nozzles, at lower levels of discomfort. However, the total volume of fluid released using this nozzle was insufficient for glucose measurement. This finding suggests that the area of the nozzle outlet has an influence on skin penetration, the resulting fluid volume, and the perceived pain.
Capillary blood sampling with jet injection required a small volume (<25 μL) of sterile saline to be injected into the skin. Jet injection typically causes residual injectate to remain on the surface of skin. An even smaller volume of saline would have been delivered in the dermis; such a small volume should be safe for patients. Patients using this technique would need to wipe away any excess injectate before collecting a sample to avoid contamination and minimize blood dilution. However, patients with diabetes should be familiar with the practice of wiping away the first drop of blood to avoid contamination, in accordance with the WHO guidelines on drawing blood. 13
Injections with the slot nozzles in this trial used sterilizable stainless steel ampoules and nozzles. It is unlikely that using items requiring steam-based resterilization would allow a system to be conveniently and safely used by diabetes. Instead, single-use solutions such as the polycarbonate ampoules used in many commercial jet injection devices may be more appropriate. Consideration will need to be given to how these devices can be conveniently and consistently loaded or preloaded with small volumes of sterile fluid for skin penetration.
Blood dilution was observed in all interventions, including the lancet pricks. The colorimetric method of blood concentration measurement used in this study compares the “redness” of extracted fluid with an initial sample of pure blood. The decreased red color intensity of samples produced by a lancet puncture suggests the presence of body fluids other than blood in the samples. The decreased blood concentration, yet unchanged glucose concentration, in these samples suggests that the released blood might be diluted by glucose-containing interstitial fluid and/or plasma. This is consistent with other studies that have shown no difference in the blood glucose concentration between the first and the second drops of blood released by a lancet. 23
The blood concentration of jet-released samples collected during the first 15 seconds for each participant was lower than of lancet-released samples. Some of this dilution is likely caused by the injectate, in addition to interstitial fluid. Further work is required to determine the influence of the composition (blood, plasma, and interstitial fluid) 24 of the extracellular fluid on the prediction of dilution and hence of glucose concentration. The fluid samples obtained during the last time period might be the best with which to measure blood glucose concentration, if blood is diluted by the same volume and type of extracellular fluid as lancet-released samples, and sufficient fluid can be collected.
The blood glucose concentration predicted using our technique was, on average, within 25% of the value measured by the same glucometer from undiluted blood from the same participant. This difference cannot be explained by the precision of the glucometer (~3.1%) alone and underscores a limitation in the approach used to estimate dilution. It will be important in future investigations to distinguish between the transparent body fluids (interstitial fluid and plasma) and the injected saline present in the collected sample. In future, we plan to add a biocompatible extrinsic marker (eg, a fluorophore) to the injectate and measure its concentration in the extracted fluid as an explicit measure of blood dilution. 25
Another limitation of this study is variability in skin penetration depth from jet injection on the fingertips, which was not measured nor explicitly controlled, and may influence the perceived levels of pain and released fluid volume. In addition, we did not control the contact force between the device and skin, which may affect the penetration depth. These issues can be addressed through further refinement of the device design for clinical use.
In the future, we plan to use the reversibility of our motor to apply techniques such as vacuum and skin vibration to enhance the volume of blood released and thereby minimize the extent of blood dilution. By marking our injectate with a biocompatible fluorophore, we plan to determine the dilution of blood by injectate separately from dilution by interstitial fluid. Finally, by applying this technique on alternative sites of the body (ie, not the fingertip), we hope to increase the volume and concentration of blood samples while minimizing pain.
Conclusions
This study is the first to demonstrate the potential of jet injection for releasing blood from the human fingertip and is another step toward lancet-free blood release and sampling for diabetes management. The reversibility of our device will allow us, in future, to apply vacuum to the skin penetration site and extract blood into an integrated glucose sensor. We anticipate that in the future a single electronically controlled jet injection device could be used to perform capillary blood sampling, provide glucose measurement, and then deliver an appropriate volume of insulin, needle-free, for people with diabetes.
Supplemental Material
sj-docx-1-dst-10.1177_19322968211053895 – Supplemental material for Jet-Induced Blood Release From Human Fingertips: A Single-Blind, Randomized, Crossover Trial
Supplemental material, sj-docx-1-dst-10.1177_19322968211053895 for Jet-Induced Blood Release From Human Fingertips: A Single-Blind, Randomized, Crossover Trial by Jiali Xu, James W. McKeage, Bryan P. Ruddy, Poul M. F. Nielsen and Andrew J. Taberner in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
We would like to thank Dr Nandoun Abeysekera for his guidance in study planning and acknowledge the assistance of phlebotomist Farina Nor Hashimi who oversaw the blood sampling procedures in this study.
Abbreviations
ABI, Auckland Bioengineering Institute; FDA, Food and Drug Administration; SfTI, Science for Technological Innovation; SPSS, Statistical Product and Service Solutions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science for Technological Innovation National Science Challenge (SfTI) of New Zealand, and the Auckland Bioengineering Institute (ABI) at the University of Auckland, New Zealand. A.T. is supported by a James Cook Research Fellowship from the Royal Society of New Zealand.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
