Abstract
Background:
Clinical trials often suffer from recruitment barriers and poor adherence, which increases costs and affects trial outcomes.
Objective:
To investigate the feasibility of Decentralized Clinical Trial (DCT) design elements to recruit, enroll, and engage patients with type 2 diabetes mellitus (T2DM).
Methods:
Patients with T2DM were recruited through a pharmacy and online recruitment using advert on Facebook, to 3 weeks monitoring of glucose and behaviometric parameters. Subjects recruited online could either complete an informed consent conversation in the pharmacy or through live video call managed by the study app.
A continuous glucose monitoring (CGM) device to collect glucose data, and a hybrid smartwatch to monitor heart rate, track activity and sleep pattern were delivered by postal service to the participants’ home address. The devices were connected to a study specific app on the participant’s smartphone also capturing GPS data and questionnaire answers.
Results:
Twenty-six subjects (3 pharmacy, 23 online) with T2DM were recruited, 85% preferred online informed consent conversation. All participants were able to self-apply the CGM device, use the smartwatch, and download the app. GPS location was captured more than 100 times for each participant, and more than 90% completed all 3 questionnaires. All the participants felt safe with the informed consent process and they felt confident in participating from home. Three participants dropped-out during the study period leaving a retention rate at 87%.
Conclusions:
Use of DCT design elements to conduct a T2DM study is feasible regarding recruitment, data collection from various electronic devices, and participant engagement.
Keywords
Introduction
Technological advancements have led to increased availability and performance of electronic devices that generate data relevant for understanding and treating several diseases. Decentralized Clinical Trials (DCTs) can facilitate collection of large, high-quality datasets. These are needed to advance the development of methods, algorithms and tools for improved treatment.
Continuous Glucose Monitoring (CGM) devices measure interstitial glucose concentration every 1-15 minutes (depending on the device), and can be used to calculate a range of metrics such as mean glucose, time spent in target glycemic range, hypoglycaemia or hyperglycaemia and glycaemic variability. 1 These measures can be applied to evaluate glycaemic control within a specified timeframe ranging from 2 weeks to several months. CGM data further offers detailed insight into fluctuations occurring over short intervals in response to an event of interest (eg, food or drug intake, acute disease or physical activity). 2
The aim of this prospective, observational, siteless cohort study was to investigate the operational feasibility of using a DCT design elements to recruit, enroll, and engage patients with T2DM.
Methods
In this 3 weeks DCT feasibility study all study data, including questionnaires, was collected through electronic devices connected to a tailored study specific app on the participant’s smartphone. There were no physical visits to the site besides informed consent conversation in the pharmacy.
The participants were recruited through 2 separate channels (pharmacy and online recruitment). In the pharmacy (Glostrup Apotek, Denmark), participants were recruited using a traditional poster, and interested subjects received a flyer with a link to a website including study information and a screening questionnaire. Once the online questionnaire was completed a time slot for informed consent conversation in the pharmacy was scheduled and the subjects were invited to download the clinical study platform app with participant information and education material about electronic devices used in the study. The informed consent process was managed by the study app, including scheduling time slots for the live video call, conducting live video call, and signing the informed consent form by electronic signature (using NemID, a unique identifier for citizens in Denmark). The eligibility was cross checked with medical records (Sundhedsplatformen) through the Civil Personal Registration number, a unique 10 digit number given to all citizens in Denmark. 3
The online recruitment was done with adverts on Facebook that directed the subject to the study website. Qualified subjects had the option to choose between an informed consent conversation in the pharmacy or an online video call with a sub-investigator through the study app. However, all participants were able to contact study personnel, either by phone, email, or video call.
The inclusion criteria were diagnosed T2DM (regardless of current treatment regimen), age>18 years, owner of a suitable smartphone to complete the digital steps of the study (Android operating system: Android 5.0 or higher with activated NFC function, iPhone 7 or higher, operating system: iOS 11 or higher).
Exclusion criteria were, medical treatment that could affect the reading of glucose data (eg, corticosteroids), known sensitivity to medical-grade adhesives, planned magnetic resonance imaging (MRI), computed tomography (CT) scan, or high-frequency electrical heat (diathermy).
Online non-validated questionnaires were completed at baseline (related to set up of devices and current treatment), and at day 7 and 21 (related to their experiences and satisfaction with study devices, the questionnaires were the same on day 7 and 21). Further, a 2-day online diary of meals was completed in week 2.
After enrollment the participants received the study devices (CGM/wearable) by postal mail and guided by education material in the study app the participants set up the devices and synchronized each device with their own smartphone themselves.
Devices
The Abbott FreeStyle Libre and Freestyle Libre Pro® are CE-approved medical devices marketed by Abbott Laboratories (Abbott). The minimally invasive 4 device consists of a sensor patch, an applicator and an app. The patch with an intracutaneous sensor should be applied on the back of the upper arm by the participants themselves, and needs replacement every 14 days. The sensor automatically measures and continuously stores glucose readings for up to 8 hours. To collect and store complete data the participants were prompted to scan the sensor before the end of an 8-hour interval. FreeStyle Libre CGM glucose data were available to the patient through the Freestyle LibreLink app and with Freestyle Libre Pro® the CGM glucose data were blinded to the patient and not available to the patient through the Freestyle LibreLink app. In the study we looked at the number of participants having CGM data reading above 70%.
Participants entering the study through the pharmacy had the Freestyle Libre Pro® the first 14 days before changing to the FreeStyle Libre the last week. Of the participants not entering from the pharmacy, fifty percent had the Freestyle Libre Pro® the first 14 days before changing to the FreeStyle Libre the last week and the other fifty percent had the Freestyle Libre sensor through the entire study period.
The Withings Pulse HR is a hybrid smartwatch which is commercially available and features 24 hours heart rate monitoring, activity tracking, connected GPS and sleep tracking. The device requires an initial setup with the study app on the participants’ smartphone, thereafter the participants can track the data. The data were automatically synchronized with study servers.
Ethics
The study was carried out in accordance with the Helsinki II declaration and the national regulations. The study was approved by the Regional Scientific Ethics Committee of the Capital Region of Denmark (H-20008223). The server platform was AWS (Amazon Web Servers). The Withings servers were located in the EU. Data were handled and stored in accordance with GDPR-regulations and Danish regulations for participant data. Encryption was used in transition and in rest, as well as 2 factor authentications. This has been added in the method section.
Results
Recruitment
In total, 26 subjects with T2DM were included, 3 from the pharmacy and 23 from online recruitment. For the recruitment at the pharmacy the unique QR code was scanned 22 times, 6 completed the screening questionnaire (conversion rate 27%) and were qualified, of which 4 downloaded the app and 3 were enrolled.
For the online recruitment there were 549 separate website views, 82 filled out the screening questionnaire (conversion rate 16%), of which 59 were qualified, 35 downloaded the app, and 23 were enrolled. (Figure 1)
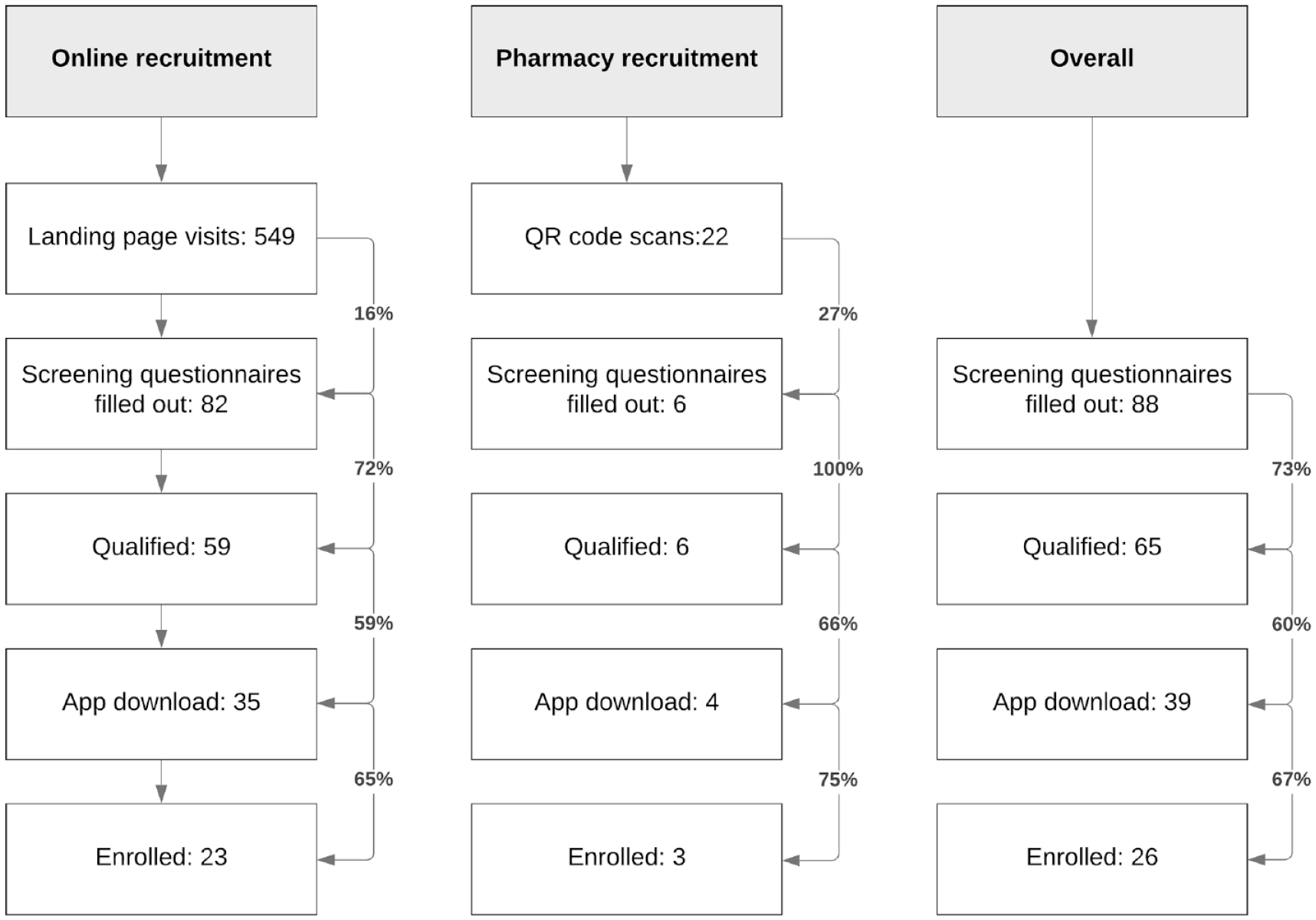
Flowchart of recruitment.
The overall conversion rate from scanned QR code to enrollment was 14% for recruitment at pharmacy, and 4% from website visits to enrollment for online recruitment. The conversion rate for completing the screening questionnaire to enrollment was 50% for recruitment at pharmacy and 28% for online recruitment.
Five of those recruited online chose to have an informed consent conversation at the pharmacy.
Participants were recruited online in just 17 hours whereas it took 3 weeks to recruit participants in the pharmacy. The recruitment was considered a success as the predefined budget for recruitment was not exceeded.
Twenty-three were excluded; 6 did not have a modern smartphone, 4 used corticosteroids or had a planned MRI/CT/diathermy, 3 already used continuous glucose monitoring, 3 had known allergy for adhesive plaster, 2 were participating in another trial, T2DM could not be confirmed in two, 2 were not willing to use continuous glucose monitoring, and the last one was exceeding the study deadline. Baseline characteristics for enrolled subjects are shown in Table 1.
Baseline Characteristics of the Included Participants.
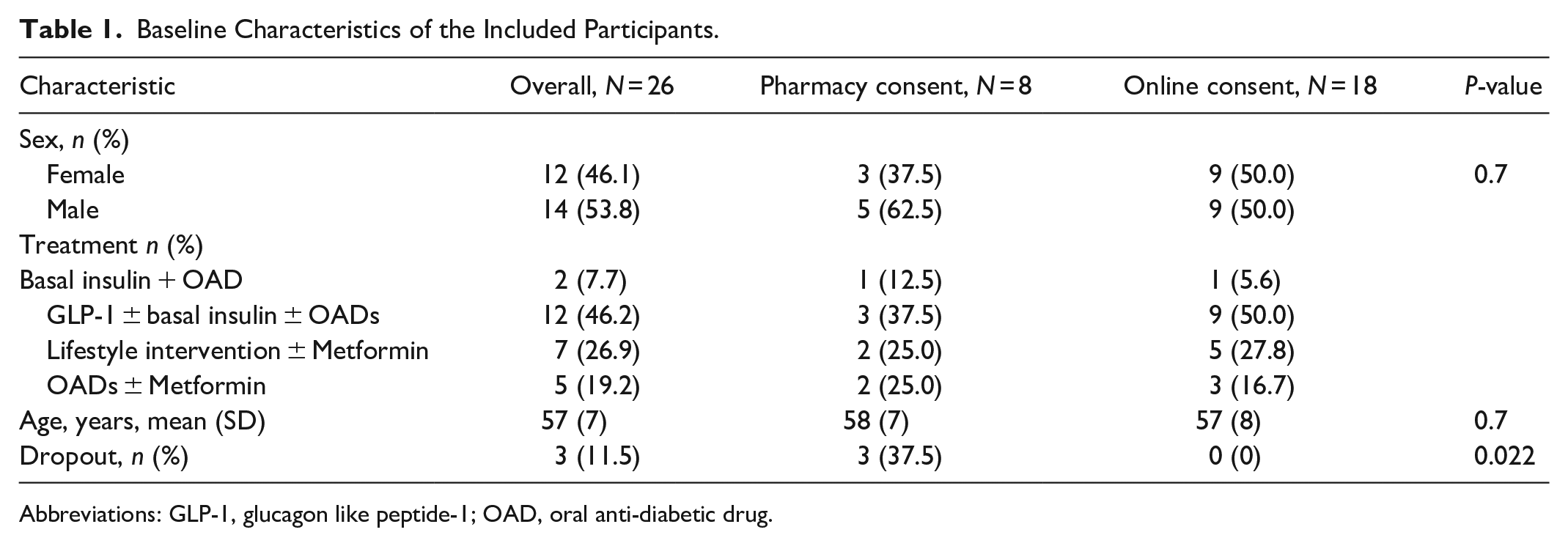
Abbreviations: GLP-1, glucagon like peptide-1; OAD, oral anti-diabetic drug.
During the study 3 participants dropped out, one recruited from the pharmacy and 2 recruited online but completed their informed consent conversation in the pharmacy. Two participants dropped out as they found the study too overwhelming and one was confused by the regular and pro sensor. All 3 drop-outs were participants with Freestyle Libre Pro® sensor the first 2 weeks and Freestyle Libre sensor the last week.
After the start of recruitment, the first patient’s first visit (FPFV) was after 1 day. The last patient’s first visit (LPFV) was 3 weeks after the start of the recruitment (Figure 2). The last patient’s last visit (LPLV) was 2 months after the start of the recruitment. The average days between app download and completing the consent was 7.9 days (SD 6.3). The study personnel were contacted 189 times during the study period.
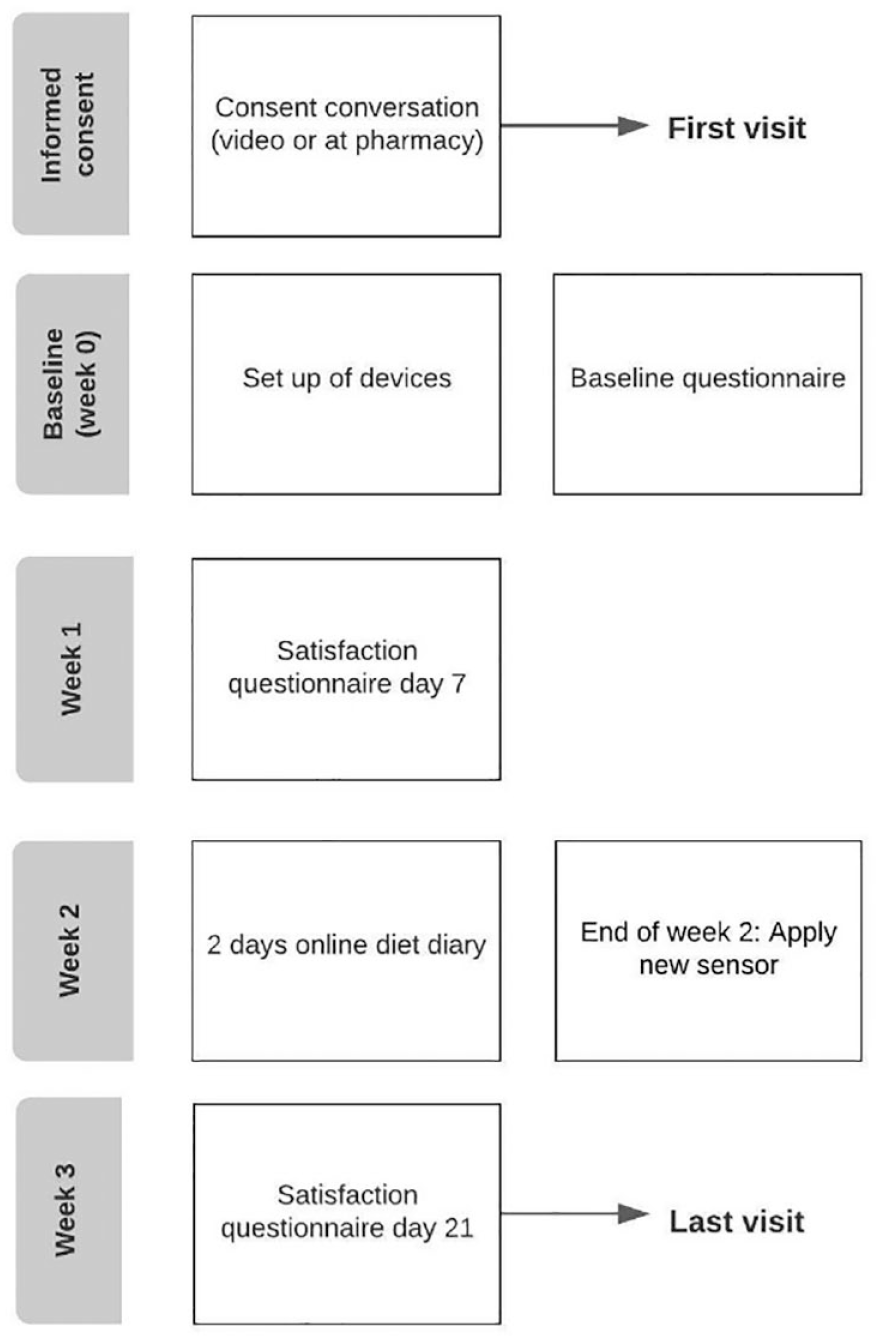
Flowchart of study tasks and visits.
Continuous Glucose Monitoring
The average duration (including shipment) from consent until first glucose measurement data was 11.8 days (SD 9.2) for Freestyle Libre Pro® and 6.5 days (SD 1.1) for Freestyle Libre. A total of 78% of participants had CGM data reading above 70% in the Freestyle Libre group and 29% of participants had CGM data reading above 70% in the Freestyle Libre Pro® group (Figure 3). Glucose readings from Freestyle Libre Pro® and Freestyle Libre are given in Table 2. Seven from the Freestyle Libre Pro® group (three withdrew) and one from Freestyle Libre group did not return the sensor, as the sensor fell off earlier than expected and couldn’t be replaced in the study period.
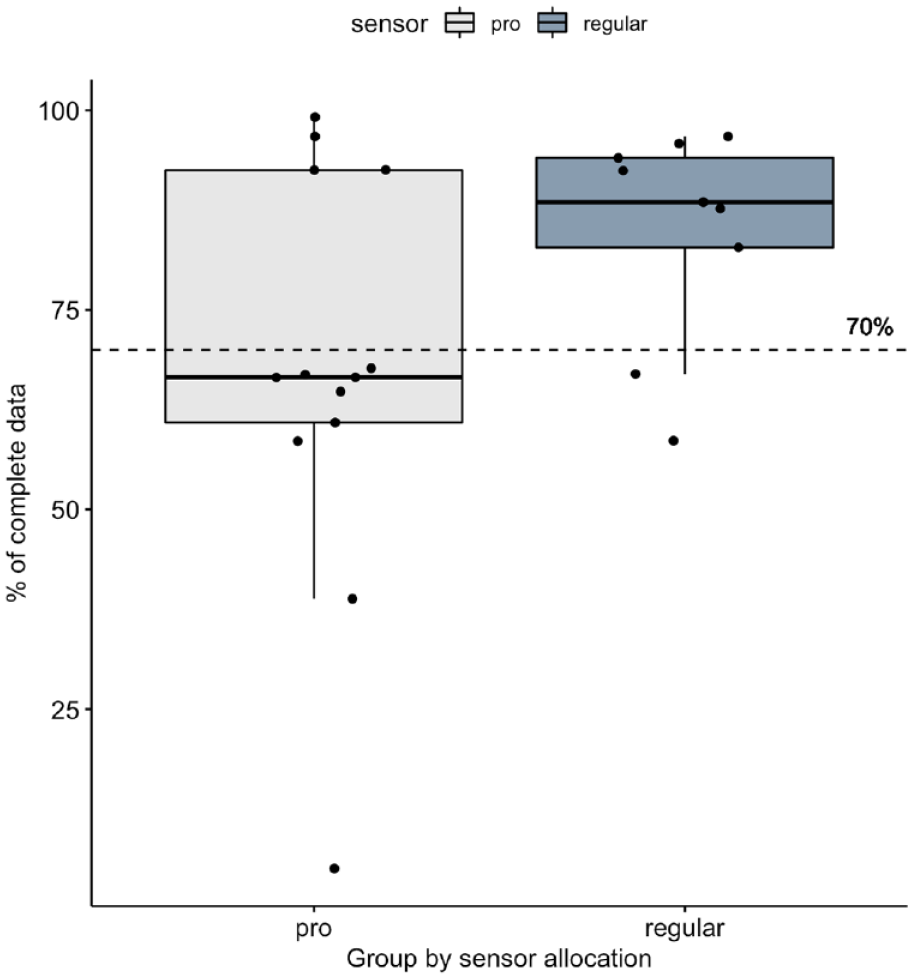
Percentage of completed glucose data.
Summary of Glucose Data.
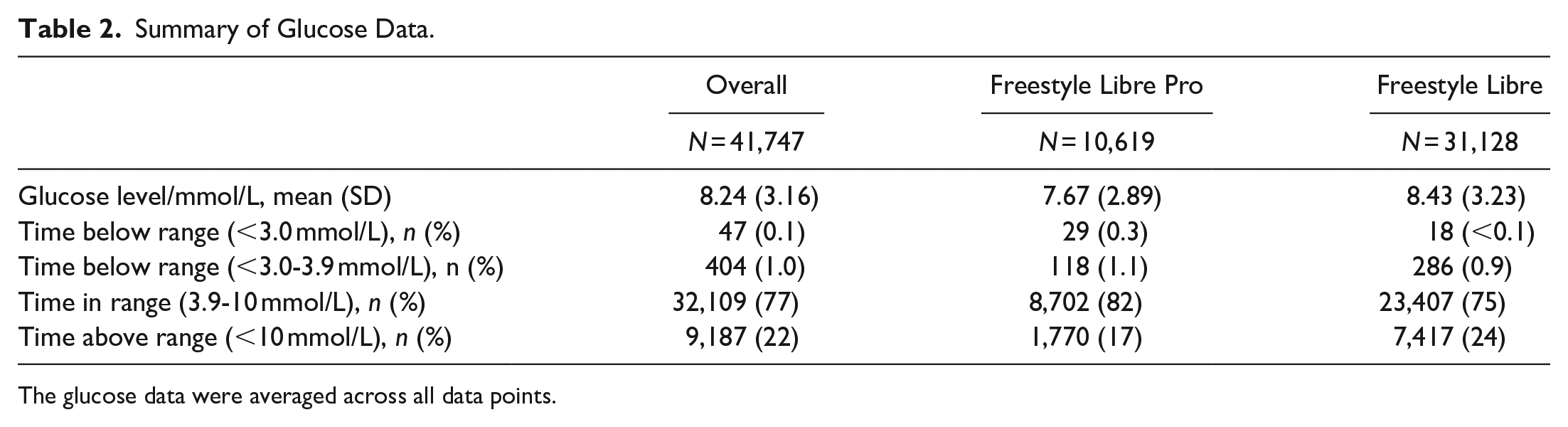
The glucose data were averaged across all data points.
Health and GPS Data
The average time from consent until first recording from smart watch was 7.7 days (SD 3.4 days).
Heart rate was measured when the Withings watch was worn, and for 11 (42%) participants more than 70% of heart rate data was captured (Figure 4). For 17 (65%) more than 70% of step data was captured, and for sleep data it was 11 (42%). An example of 1 week glucose data, heart rate, and sleep data for a single participant is given in Figure 5.
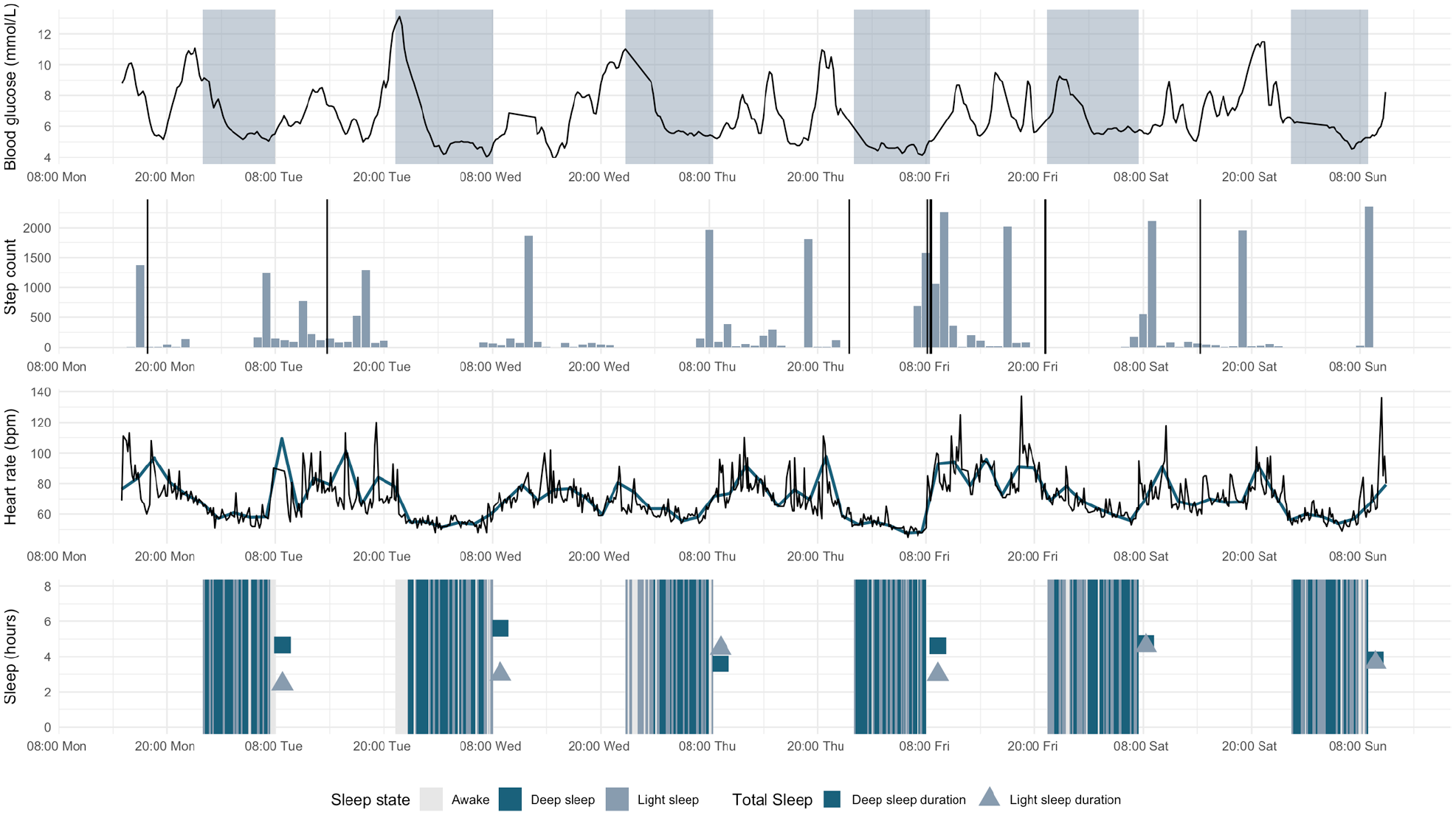
An example of one-week glucose, heart rate, and sleep data for a single participant.
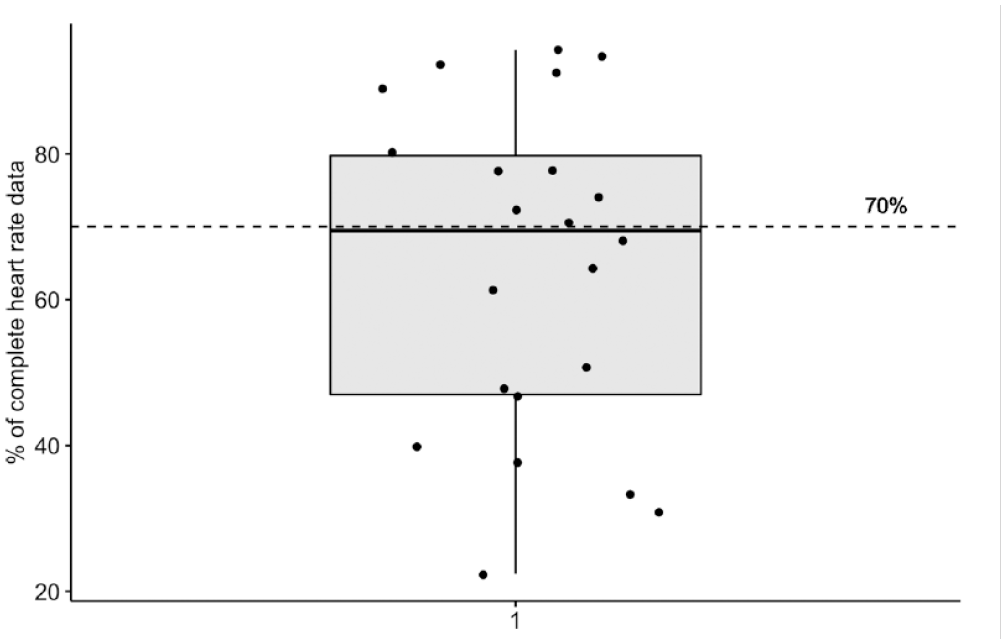
Percentage of completed heart rate data.
All participants gave access to their GPS data. GPS was captured every time a participant opened the main menu in the app (no instruction on opening the app on regular intervals was given to the participants). On average GPS points were gathered 5 times daily for each participant, and more than 100 times for each participant over the study period. Number of visits to the main menu was highest in the beginning of the study and peaked between day 4 and day 8, where the participant received the devices (Figure 6).
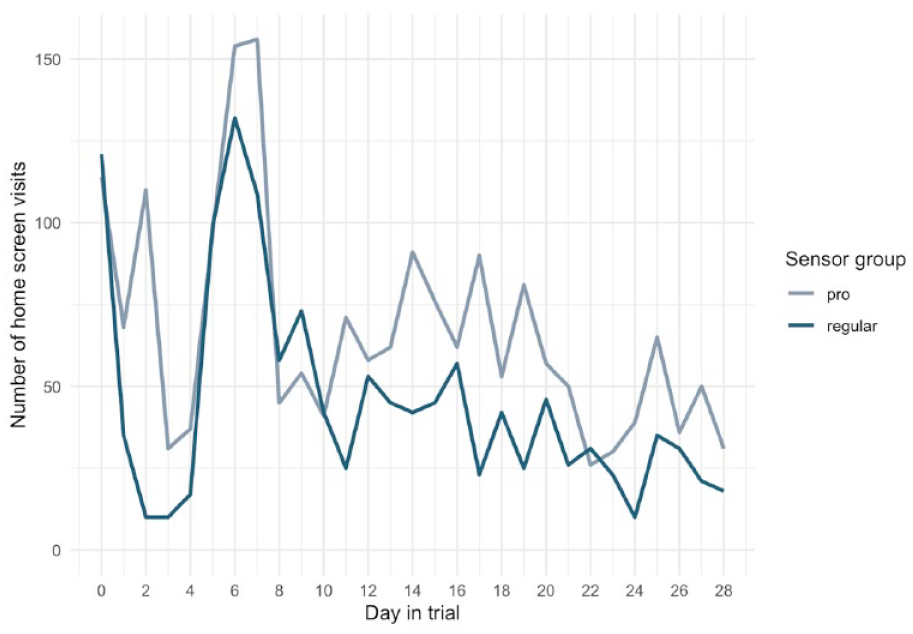
Overall number of visits to app home screen for all participants.
Adherence
All participants completed the baseline questionnaire, 91% (n = 21) completed the second questionnaire (day 7), and 96% (n = 22) completed the last questionnaire (day 21). The retention rate was 87%, hence 3 dropped-out. In total 12 (46%) participants had complete, analyzable data on all variables.
All the participants felt safe in the way they received the study information regardless of the method of informed consent (pharmacy vs. online video call). Despite the decentralized design of the study, participants felt confident in participating from home, found it easy to fit the study participation into daily routines, and the majority did not miss contact with study staff (Figure 7). Further, the majority of the participants preferred having the informed consent conversation on a live-video call (22 out of 26 participants) and 50% (11/22) of them would have been willing to sign the informed consent statement without a consent conversation in a video call with study staff.
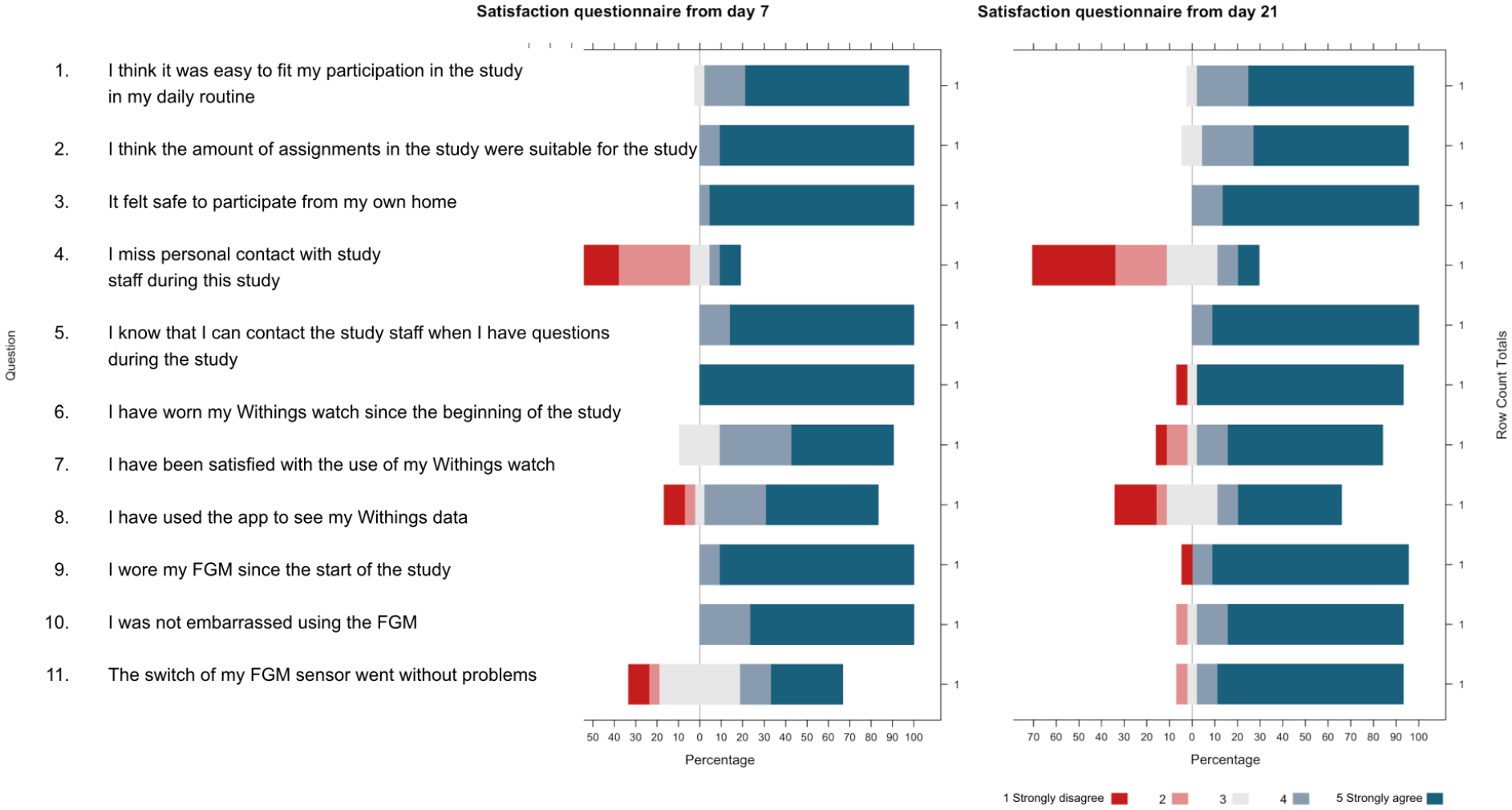
Responses from 22 participants at day 7 and 21 into the study.
Discussion
We were able to recruit 26 subjects with T2DM, 3 from the pharmacy and 23 from online recruitment, during 3 weeks in a DCT study design. The conversion rate was 50% for recruitment at pharmacy and 28% for online recruitment. Further, we were able to remotely collect glucose, GPS, and activity data from various electronic devices. Above 90% of the participants completed all 3 questionnaires, and 3 participants dropped-out during the study.
The conversion rate at each recruitment step was better at the pharmacy compared to the online recruitment. This is in line with the literature showcasing that traditional recruitments are known for outperforming online recruitment regarding conversion rates. 5 Recruitment at the pharmacy was effective in the sense of screening the right participants, as many screened participants went on to participate in the study. When the patients are recruited at the hospitals, potential participants to be screened are often selected from a pre-qualified group of patients. In this way fewer but qualified patients, that most likely will be included, are screened, which reduces the time consumption and improves the conversion rates. It is likely that the pharmacy also has chosen the same approach to be effective, as traditional recruitment requires time commitment by the study staff. The pharmacy most likely does not have a pre-qualified group of patients; however, a pre-screening could have been done over the desk and most involved participants were invited to informed consent conversation. Online recruitment does not require the cost of personnel time in the same way as traditional recruitment. For online recruitment it is an advantage that the advert reaches out to many potential participants and the screening questionnaires are completed by many, resulting in a low conversion rate. In that way participants passing the screening questionnaire can move on to online video-call requiring personnel time. However, online recruitment is superior in regard to time efficiency and cost-effectiveness compared with traditional recruitment. 5 The pharmacy was not able to recruit, screen, or include the same number as online recruitment. To include pharmacies as supplementary to online recruitment has some advantages. First, it gives an opportunity to recruit more and a different type of patients than the usual patient population seen in the hospital. Second, it is a good prospect to patients that prefer to have informed consent conversation in-person. Further, if they are a slightly unfamiliar with technology, they can get to know the study-devices in-person by trained study staff. However, it can be a barrier as numerous pharmacies must be involved if patients must have a real chance of opting for a nearby pharmacy to complete an informed consent conversation in global or nationwide conducted studies.
Further, the patient engagement reflected as adherence to the study procedures was higher in the online recruited group probably explained by the fact that the patients themselves have sought participation in the study and have not been encouraged to participate by the study staff.
Lastly, some participants experienced that their Freestyle Libre sensors fell off before they should have been changed which resulted in only 29% of participants having CGM data reading above 70% in the Freestyle Libre Pro® group. This is indeed an important limitation that could be avoided by supplying new sensors when needed; however, this was not anticipated, and due to the short study duration no replacement sensors were provided in this study.
The use of CGM in clinical trials has many benefits. Besides getting the continuous glucose level readings it can also provide data to understand exactly how the trial drug is affecting the health of the patient, improve patient compliance and reduce attrition from trials by making the glucose readings much easier for patients, and reduce data loss and human error by automatically collecting data. The CGM data quality in the present study was regarded high enough to guide potential treatment intensification. Open and masked CGM was used in the present study to explore if it was feasible in a DCT setting to get participants to both apply and wear masked sensors, we observed that the duration from consent to first measurement was shorter for opened compared to masked CGM probably due to the motivation factor as the participants in the open CGM have access to their glucose data. Experience with using digital diabetes technologies in studies has been good so far.
Franc et al. 6 found a clinical and statistically significant reduction in blood glucose levels in a randomized study investigating a telemedicine solution combining a mobile app for the patients and a web portal for health care providers allowing real-time monitoring and adjustment of therapy. In another multicenter, crossover, randomized, controlled study, conducted under free-living home conditions, Thabit et al. 7 found improved glucose control and reduced hypoglycemia in patients using a closed-loop insulin delivery system. Besides continuous glucose data collection GPS tracking in clinical research is also valuable as information on weather conditions, water hardness, activity tracking and socialization can be explored in relation to the investigated objectives.
The study has some important limitations that need to be addressed. First, online recruitment may lead to selection bias. To avoid this bias, recruitment at the pharmacy was included; however, there were not enough participants to make a comparison between the 2 groups to investigate whether participants recruited online resembled those recruited from the pharmacy. Further, only targeting smartphone users may also lead to bias which can be prevented by including pharmacies that can help the participants in person. Second, missing participants from the entire treatment spectrum may add some bias, as no patients on multiple daily insulin injections were included.
Third, the short duration may have an impact on compliance and retention. Studies with longer duration should also investigate potential impact of app fatigue and how to prevent it with e.g push notification. Fourth, a control arm representing the traditional clinical trial design with physical study was not included, making it difficult to estimate the feasibility. Including a control arm will also make the investigation of retention better as the drop-out can be compared in the 2 arms. The feasibility can most likely also be attributed to the study specific app which gathered all the study tasks in one place making it easier for the participants. Fifth, this was an observational study without any experimental intervention. Future studies should investigate the DCT design in interventional studies preferably randomized to investigate if DCT is also feasible when an intervention is part of the study design.
In conclusion, online recruitment outperformed pharmacies in recruitment; however, pharmacies are a good option to provide physical informed conversations to participants requiring it. Participants were able to download the study app, and self-apply devices and activate them with guidance through the study app. Further, glucose, GPS, and activity data can be collected from various electronic devices by the participants with no physical meeting or guidance from the study staff. Future studies should investigate whether it is cost-effective to include pharmacies as an option to conduct the informed consent conversation in-person for patients preferring a face-to-face conversation in nationwide DCT studies. Other feasible alternatives to conduct the in-person informed consent conversations as an option in DCT design should also be investigated.
Footnotes
Acknowledgements
We would like to thank Maria Alexaki, Olena Budnik, Simone Schaadt from Studies&Me with help to online recruitment, and Hanne Høje Jacobsen, Julie Krømann, Kristine Hallberg Friis Aarup, Kristian Østergaard Nielsen from Glostrup Apotek to help with the recruitment from the pharmacy. Charlotte AP Laugesen, Signe Havsager, and Lotte Klim for trial support.
Abbreviations
CGM, Continuous glucose monitoring; DCT, Decentralized clinical trial; SD, Standard deviation; T2DM, Type 2 diabetes mellitus.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Zarqa Ali, Simon Francis Thomsen: None related to this project
Teske Jacqueline Valk, Theis Bjerre-Christensen, Sigurd Brandt, Ari Pall Isberg, Anders Daniel Andersen, John Robert Zibert: Studies&Me employee.
Morten Lind Jensen, Lise Sylvest Helledi, Anne Kaas: Novo Nordisk employees and shareholders.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Studies&Me
