Abstract

Keywords
Interest in antioxidant therapy for a variety of diseases including Cancer and Alzheimer’s is increasing. Vitamin C is among the most commonly used antioxidant dietary supplements. These compounds are electron donors and can potentially interfere with measurements by an electrochemistry blood glucose monitor (BGM) by increasing the current at the electrode and causing a falsely elevated measured glucose concentration called pseudohyperglycemia. We noted 3 cases reported by Katzman et al. 1 of interference by high concentrations of the antioxidant ascorbic acid on the readings of 3 point of care (POC) BGMs intended for testing blood glucose in a hospital setting. BGMs can respond to ascorbic acid in addition to glucose, which results in an electrical current that can be incorrectly attributed to the presence of glucose and can result in a falsely elevated reported glucose concentration (see Figure 1). The authors tested the effect of high concentrations of ascorbic on 3 widely used hospital POC BGMs. All 3 systems used maltose-independent enzyme-cofactor electrochemistry. The BGMs were (1) Roche Accu-Chek Inform II system (Roche Diagnostics, Mannheim, Germany), (2) Abbott Precision Xceed Pro (Abbott Laboratories, Abbott Park, IL, USA), and (3) Nova StatStrip Glucose Hospital Mete, (Nova Biomedical, Waltham, MA, USA). These BGMs reported, respectively, spuriously elevated values (interpreted as pseudohyperglycemia), spuriously elevated values, and an error message. We agree with the authors’ conclusion that manufacturers and users of some BGM should be aware of potential interference by ascorbic acid because a falsely elevated glucose concentration can promote excessive insulin therapy. Antioxidants other than ascorbic acid, such as N-acetylcysteine, and glutathione, can also be interferents detected by BGMs. 2
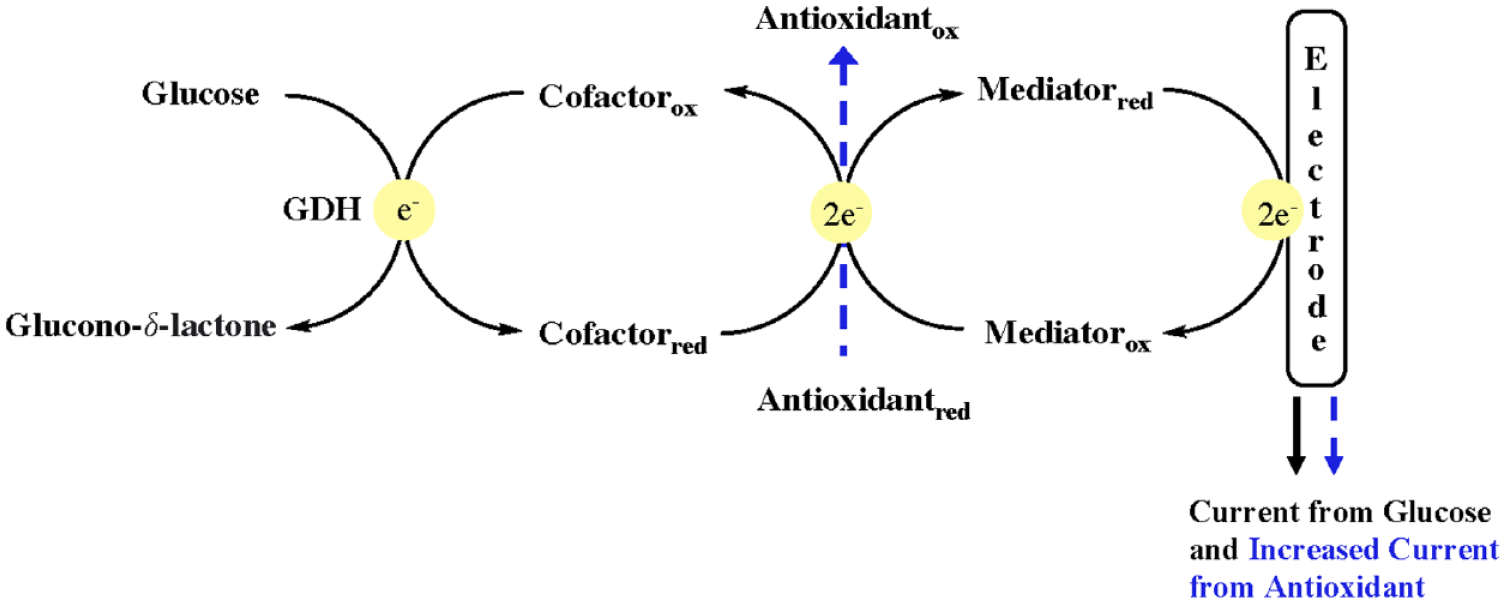
Schematic representation of the effect of antioxidants on enzyme-cofactor-mediator electrochemistry of blood glucose monitors.
We also noted another recent case report this year of pseudohyperglycemia secondary to high-dose intravenous ascorbic acid therapy by Orija and Zahid. 3 The authors stated that the Inform II system was used by every patient at their hospital. They stated that this system utilizes the enzyme-cofactor combination of glucose dehydrogenase (GDH) and pyrroloquinolinequinone (PQQ). The article reviewed the risk of pseudohyperglycemia from BGMs containing GDH-PQQ, most commonly when using (1) icodextrin-containing peritoneal dialysate solutions and (2) maltose-containing intravenous immunoglobulins. 4 However, the Inform II system does not use GDH-PQQ. In 2012, Roche Diagnostics replaced the Inform system with the Inform II system, which uses the enzyme-cofactor combination of GDH with a mutant variant of quinoprotein glucose dehydrogenase (together known as Mut. Q-GDH).5,6 The switch was made mainly because of cases of pseudohyperglycemia during peritoneal dialysis and intravenous globulin therapy. Most of the reported causes of pseudohyperglycemia associated with the enzyme-cofactor contained in the old Inform are no longer a problem in the Inform II. The Inform II can show interference from high doses of ascorbic acid, which the patient in this report had received. According to the Inform II user’s manual, “Intravenous administration of ascorbic acid that results in blood concentrations of ascorbic acid greater than 3 mg/dL will cause overestimation of blood glucose results.” 6 Thus, interference affecting BGM results must be considered in patients on antioxidant therapy.
Footnotes
Acknowledgements
We thank Annamarie Sucher-Jones for her expert editorial assistance.
Abbreviations
BGM, blood glucose monitor; GDH, glucose dehydrogenase; POC, point of care; PQQ, pyrroloquinolinequinone
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: NYX, KTN, and CM have nothing relevant to disclose. DCK is a consultant for EOFlow, Fractyl, Lifecare, Novo Nordisk, Roche Diagnostics, Samsung, and Thirdwayv.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
