Abstract
Background:
Effective exogenous insulin delivery is the cornerstone of insulin dependent diabetes mellitus management. Recent literature indicates that commercial insulin-induced tissue reaction and cellular cytotoxicity may contribute to variability in blood glucose as well as permanent loss of injection or infusion site architecture and function. It is well accepted that insulin formulations are susceptible to mechanical and chemical stresses that lead to insulin fibril formation. This study aims to characterize in vitro and in vivo toxicity, as well as pro-inflammatory activity of insulin fibrils.
Method:
In vitro cell culture evaluated cytotoxicity and fibril uptake by macrophages and our modified murine air-pouch model quantified inflammatory activity. The latter employed FLOW cytometry and histopathology to characterize fibril-induced inflammation in vivo, which included fibril uptake by inflammatory phagocytes.
Results:
These studies demonstrated that insulin derived fibrils are cytotoxic to cells in vitro. Furthermore, inflammation is induced in the murine air-pouch model in vivo and in response, macrophages uptake fibrils both in vitro and in vivo.
Conclusions:
Administration of insulin fibrils can lead to cytotoxicity in macrophages. In vivo data demonstrate insulin fibrils to be pro-inflammatory which over time can lead to cumulative cell/tissue toxicity, inflammation, and destructive wound healing. Long term, these tissue reactions could contribute to loss of insulin injection site architecture and function.
Introduction
Currently, the American Diabetes Association estimates that 7.5 million Americans are insulin dependent and require frequent subcutaneous insulin administration (SIA) for glycemic control. 1 However, patients using multiple daily injections or continuous subcutaneous insulin infusion (CSII) can experience skin irritation, inflammation, and scarring at injection and CSII sites.2-6 Thus, users are encouraged to rotate injection/CSII sites across multiple skin locations to minimize adverse events.5,7,8 Even when following SIA best practices, several undesirable conditions can occur including hyperglycemia and diabetic ketoacidosis.3,4,9 Given that diabetic complications are prevented when euglycemia is achieved, it is critical to understand mechanisms which contribute to unreliable insulin efficacy through the development of skin pathologies.10-12
There are several factors which may contribute to skin inflammation from injection or infusion pump therapy. Catheter material, direction of flow, injection or infused preservatives can all contribute to site inflammation.13-16 In addition, insulin fibril formation can lead to adverse skin pathologies resulting in poor glycemic management.17,18 The formation of insulin derived fibrils (IDF) have been well characterized biochemically and can form as a result of mechanical or chemical stressors.1,19-22 In short, insulin fibrils are a large oligomer characterized by a non-native β-sheet structure rendering the protein inactive.19,20 While biochemical fibril formation has been studied extensively in vitro, data for administration of fibrils in vivo is limited.17,18 Thus, we hypothesize that insulin fibril formation represents another potential contributing factor for tissue reactions at sites of SIA or CSII and as such, it is critical to understand the impacts of fibril administration in vivo. 2
In this report, we utilized streptozotocin (STZ) induced diabetic mice to evaluate tissue response in a modified air pouch which allows for the collection of tissue, cell and fluid contents at injection sites.15,16 Furthermore, the air pouch provides a defined, subcutaneous space for evaluation of injection reagents. Our in vitro studies use murine cell lines found in the subcutaneous area to evaluate cytotoxicity of insulin fibrils and to elucidate a mechanism for fibril uptake. Results are compared to the inflammatory and cytotoxicity of phenolic preservatives (PP) found in insulin formulations as a previously established inflammatory agent.15,16,23 The goal of the present study is to examine the relative contributions to tissue inflammation during repeated SIA with respect to the variables of insulin derived fibrils (IDF) and insulin phenolic preservatives (PP).
Methods
Cell types: Mouse cell lines include macrophages (RAW 264.7) and embryonic fibroblasts (3T3-L1), which were purchased from ATCC bio products (Manassas, VA) and cultured following the supplier’s protocol. 3T3 Fat cells were induced from 3T3-L1 using MDI cocktail (methylisobutylxanthine, dexamethasone and insulin). 24
Insulin fibril generation: Fibril species were generated by incubating Humalog® at 37°C and 300 rpm in a sterile polystyrene culture tube for 24 hours. Following incubation, fibril species were centrifuged and washed 3 times with sterile saline. Subsequently, they were resuspended and diluted in either saline (denoted saline fibril) or diluent (denoted diluent fibril) at a ratio of 1:2. Diluent is commercially available from Eli Lilly and is utilized as a source of pharmaceutical grade phenolic preservative. 15 Diluent and diluent fibrils have a combined PP concentration of 2.25 mg/ml (1.6 mg/ml m-cresol, 0.65 mg/mL phenol). As a comparison, saline fibrils contain no PP.
Insulin derived fibril (IDF) uptake studies using murine macrophages in vitro: Initially, thioglycolate induced macrophages (MQ) were obtained from MaFIA (Macrophage Fas-Induced Apoptosis) or normal C57BL/6 mice, cultured in vitro in RPMI 1640 and 10% fetal calf serum at 37°C and 5% CO2, and validated using anti-F4/80 antibody/immunohistochemistry, and nucleus morphology to confirm the resulting cells identity as MQs. 25 These MQs were cultured with the various dilutions of the non-florescent or Alexa Fluor 594 containing IDF stock solutions (1/10, 1/30 and 1/90) for 24 hours at 37°C and 5% CO2. The cultures were then washed with RPMI 1640 media 3 times, before analysis as described. Both C57BL/6 and MaFIA mice were obtained from Jackson Laboratory (Bar Harbor, Maine). The MaFIA mice express Enhanced Green Fluorescent Protein (EGFP) in 78% of isolated peritoneal macrophages. 26
MTT cytotoxicity assay: Murine cells were plated in a 48-well plate with 2.5 × 105 cells/well in 250 µl of media (DMEM) without phenol red containing 10% fetal bovine serum and 1% penicillin/streptomycin. Following an overnight incubation, cells were treated with test agents from 1:3 to 1:192 then reincubated at 37°C for 3 days. An MTT assay (ATCC bio products Manassas, VA) was performed according to the manufacturer’s protocol as described previously. 15
Mice: Hsd:ICR(CD-1) mice were purchased from Envigo (Somerset, NJ) or bred in-house. Mice were maintained under temperature- and light-controlled conditions (20-24°C, 12-h light-dark cycle) receiving food and water ad libitum. Mice utilized in these studies had an average weight between 30-40 g, 8-10 weeks and were test naïve. A power analysis was performed in order to determine the total number of mice in these studies required to have an 80% chance of detecting a statistically significant difference between these groups at a 95% confidence interval. Each treatment group consisted of 6-9 mice. All studies were conducted with approval from the institutional animal care and use committee (IACUC) at Wayne State University.
Streptozotocin induced diabetes in mice: Diabetes was induced following the protocol developed by Furman. 27 Male mice received a daily intraperitoneal (i.p.) injection of streptozotocin (STZ) (50 mg STZ/kg body weight) for a period of 5 days (Sigma-Aldrich, St. Louis, MO). Blood glucose levels were monitored at least twice weekly following STZ treatment using a Bayer Contour Next EZ Meter (Ascensia Diabetes Care, Parsippany, NJ). Mice with a blood glucose level above 250 mg/dL on 2 sequential blood glucose samples were designated as diabetic.
Murine Air Pouch Model of Tissue Toxicity and inflammation: The classic murine air pouch model was adapted to evaluate the tissue response to injected agents as previously described. 15 Test agents for all studies were injected once daily, (3 days after air pouch initiation), with a volume of 300 µL of saline, diluent (Eli Lilly), saline fibril or diluent fibril into the air pouch. Volume of 300 uL was chosen to effectively cover the surface area of the air pouch to elicit a tissue response, which correlates to injected 0.5 mg insulin equivalents of fibrils. It should be noted that IDF have no effect on blood glucose levels and can be used in both normal and diabetic mice without a hypoglycemic risk. Contents of the air pouch were analyzed using total cell number counts, flow cytometry analysis, and through standard histopathological analysis. Analyses were performed at 3 and 7 days. The initial time point was chosen to reflect FDA approved lifespan for insulin infusion and injection devices whereas 7 days was chosen as an end-stage time point that was more than twice the approved lifespan.2,4
Leukocyte isolation from mouse air pouch and fluorescence-activated cell sorting (FACS): After 3 or 7-days of treatment, mice were euthanized (CO2 inhalation and cervical dislocation) and the air pouch was lavaged with a total of 10 mL of 0.9% saline (Baxter, Deerfield, IL). The collected cell fluid was processed as previously described. 15 The total leukocyte population was then subjected to flow cytometry analysis (FACS) using fluorescent labeled antibodies (1:100) (BioLegend, SanDiego, CA) to detect myeloid cells (CD45+CD11b+), neutrophils (CD45+CD11b+Ly6G+), macrophages (CD45+ CD11b+ Ly6G- Ly6Glow), monocytes (CD45+ CD11b+ Ly6G- Ly6Ghigh), and lymphocytes (SSC-A- CD45high). 24 Fluorescent minus one (FMO) controls were used to differentiate between positively and negatively stained populations. Compensation was performed using BD Comp Beads (BD Biosciences, San Jose, CA) to create single-color controls of each antibody. FACS analyses were performed on a BD-LSRII utilizing the services of the microscopy, imaging, and cytometry core laboratory (MICR), at Wayne State University, Detroit, MI, and the data were analyzed with FlowJo software (FlowJo, LLC).
Statistical analysis: All analyses were conducted using SAS VERSION 9.4 (SAS, INC., Cary, NC). For normative data comparing 2 groups, a 2-tailed t-test was employed to detect a significant difference at a 95% confidence interval. For multiple group comparisons, a one-way ANOVA test was performed to assess for statistical significance at a 95% confidence interval. If significance was detected, then a post-hoc Tukey test was utilized to assess for statistically significant differences among the subgroups assessed in the ANOVA test. Following FACS staining and analysis of the cell populations, all cell numbers were graphed in a boxplot using GraphPad Prism 8 software.
Histopathologic evaluation: Histological evaluation was followed as previously described by our laboratory. 15 To evaluate tissue responses to the daily air pouch injections, qualitative histopathologic evaluation was performed on mouse tissue samples at 3- and 7-days post-injection. Tissue samples were fixed in 10% buffered formalin (VWR, Radnor, PA) for 24 hours followed by standard tissue preparatory steps, paraffin embedding, and sectioning. Tissue samples were cut into 5 µm sections and stained using Hematoxylin and Eosin stain (H&E). To confirm the presence of macrophages in the air pouch, tissue sections were stained with a mouse macrophage-specific antibody designated anti-mouse F4/80 (Fisher Scientific, catalog # MF48000). Mouse IgG was used as a negative control. Anti-insulin antibody (Invitrogen, PA1-26938) was incubated at 1:100 followed by IgG-HRP conjugate antibody (Invitrogen, A18769) at 1:500. Tissue samples were evaluated using a Nikon microscope and imaging system.
Results
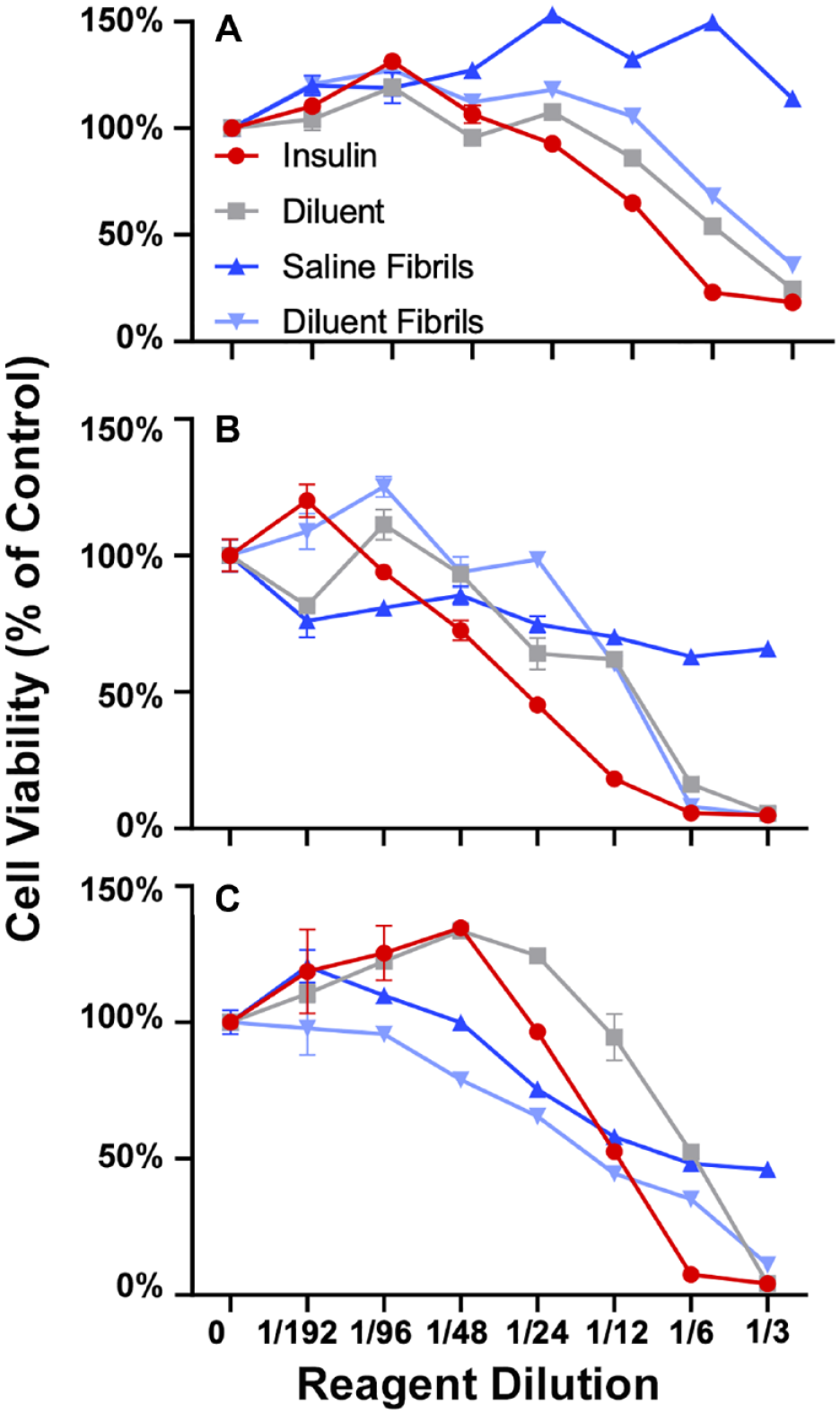
Determination of cellular cytotoxicity of phenolic preservatives and insulin fibrils in murine cell lines and primary cultures in vitro. To determine the in vitro cytotoxicity of phenolic compounds and insulin fibrils in murine cell lines, as well as primary cultures in vitro MTT cytotoxicity assays were used after 3 days of co-culture of cells and test agents. MTT assay was used for 3T3-L1 fibroblast cells (A), 3T3-Fat induced cells (B), raw macrophage (C), following 3-day in vitro incubation with agents. Each panel shows cells treated with diluent (grey) diluent + fibril (light blue), saline + fibril (dark blue), or insulin (red) at increasing concentrations. Cell viability is expressed as percent to saline control. MTT assay (panels A-C) data reflect absorbance at 570 nm. Error bars represent standard deviation. The concentration of all test reagents is based on dilutions in media. Full statistical analysis is presented in supplemental section Table S1.
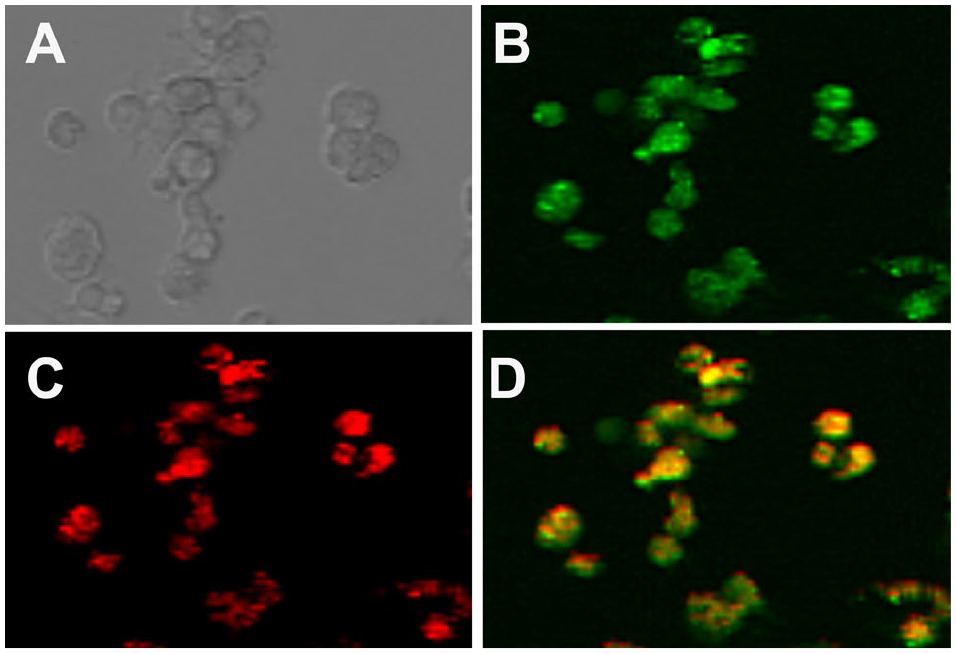
Immunofluorescence of fibril uptake in isolated macrophages. Bright field microscopy of isolated macrophages is shown in (A). MaFIA isolated macrophages expressing GFP (B) and IDF conjugated to alexafluor 594 (C). Overlay of GFP and alexafluor 594 results in yellow after 24 hours of macrophage incubation with IDF (D).
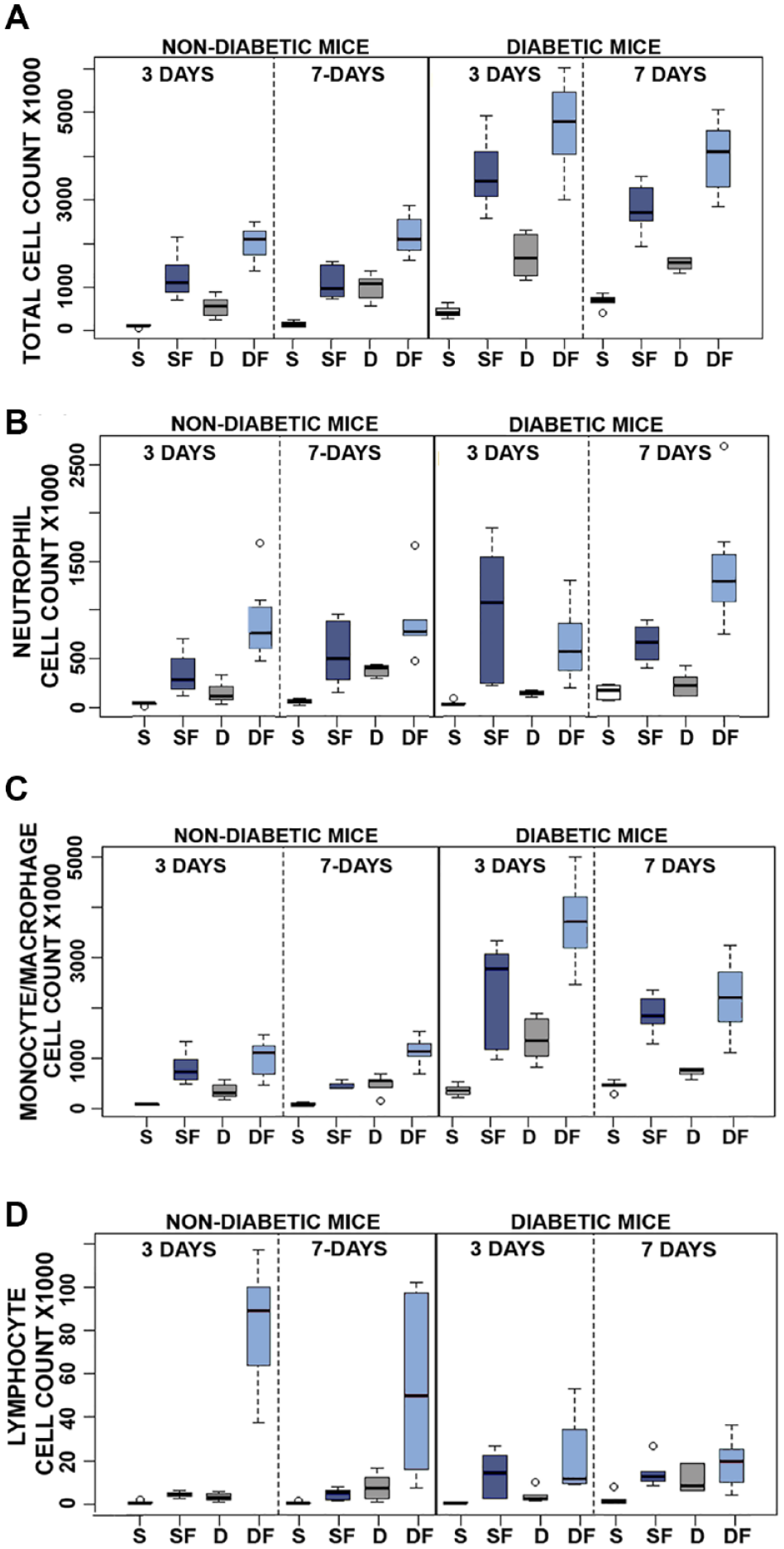
Quantitative analysis of pro-inflammatory reactivity of insulin fibrils and phenolic compounds in vivo using the murine air pouch (MAP) model in non-diabetic and diabetic mice: evaluation of 3 and 7 days of treatment. Results of flow analysis show total cell recruitment (A) and cell type subpopulation for neutrophils (B), monocytes/macrophages (C), and lymphocytes (D), for each treatment: saline (S), diluent (D), saline + fibril (SF) or diluent + fibril (DF). All results are expressed as total cell count X1000, and were obtained by flow cytometry (N = 6–9). Open circles denote outliers. Statistical analysis of this data is presented in Tables S2–5.
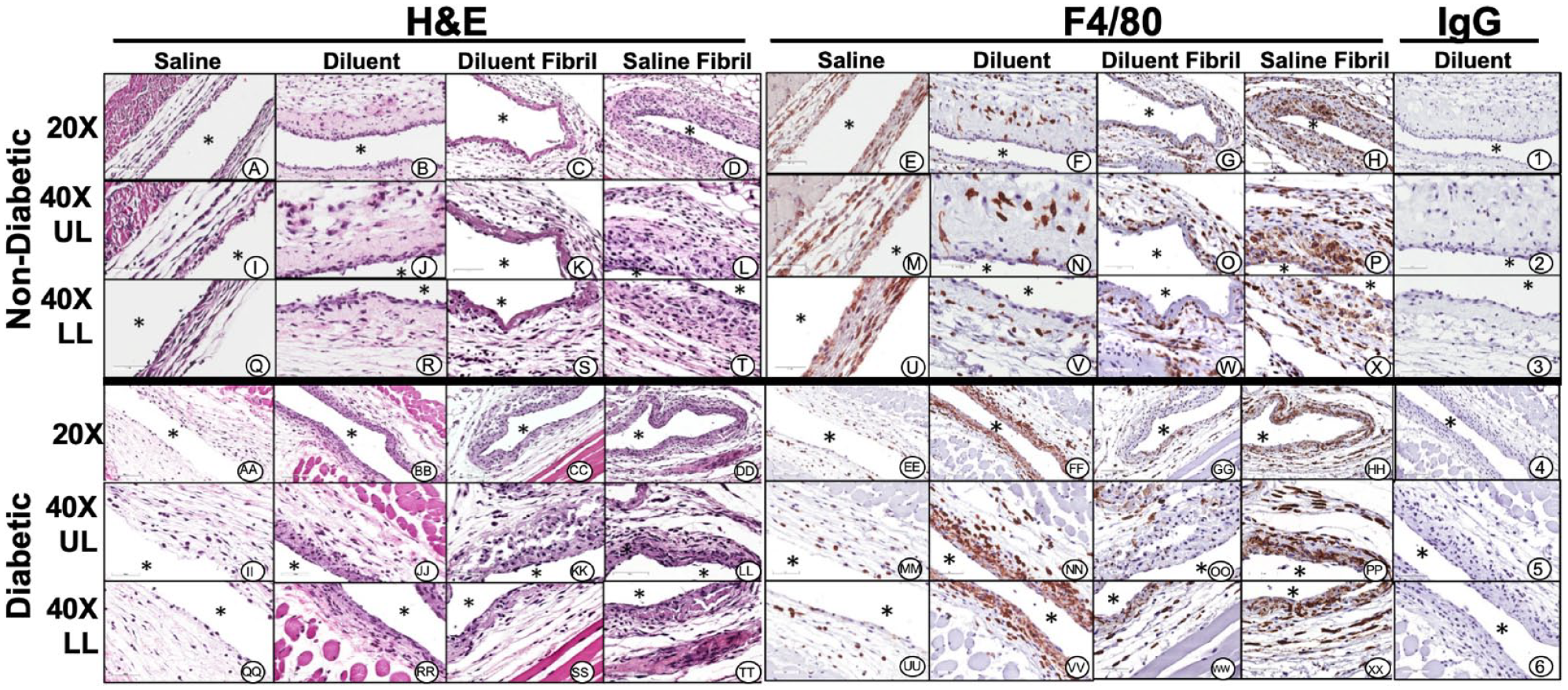
Histopathologic and immunohistochemical evaluation of mouse air-pouch tissue (post lavage) from non-diabetic and diabetic mice after 3 days treatment with insulin fibrils and phenolic diluent. Representative photo-micrographs of tissue reactions from 3-day treated non-diabetic and diabetic mice (post lavage) including: saline, diluent, saline fibril and diluent fibril treated non-diabetic and diabetic mice. Matched tissue sections were stained for general histopathology using H&E staining, and for macrophages using Anti-F4/40 antibody or matched non-immune IgG. All photo-micrographs were taken at 20× or 40× as designated. Each area of the MAP tissue evaluated in the study is designated as: UL (upper layer of MAP tissue), and LL (lower level MAP tissue), respectively. White area containing (*) indicates air space location of murine air pouch.
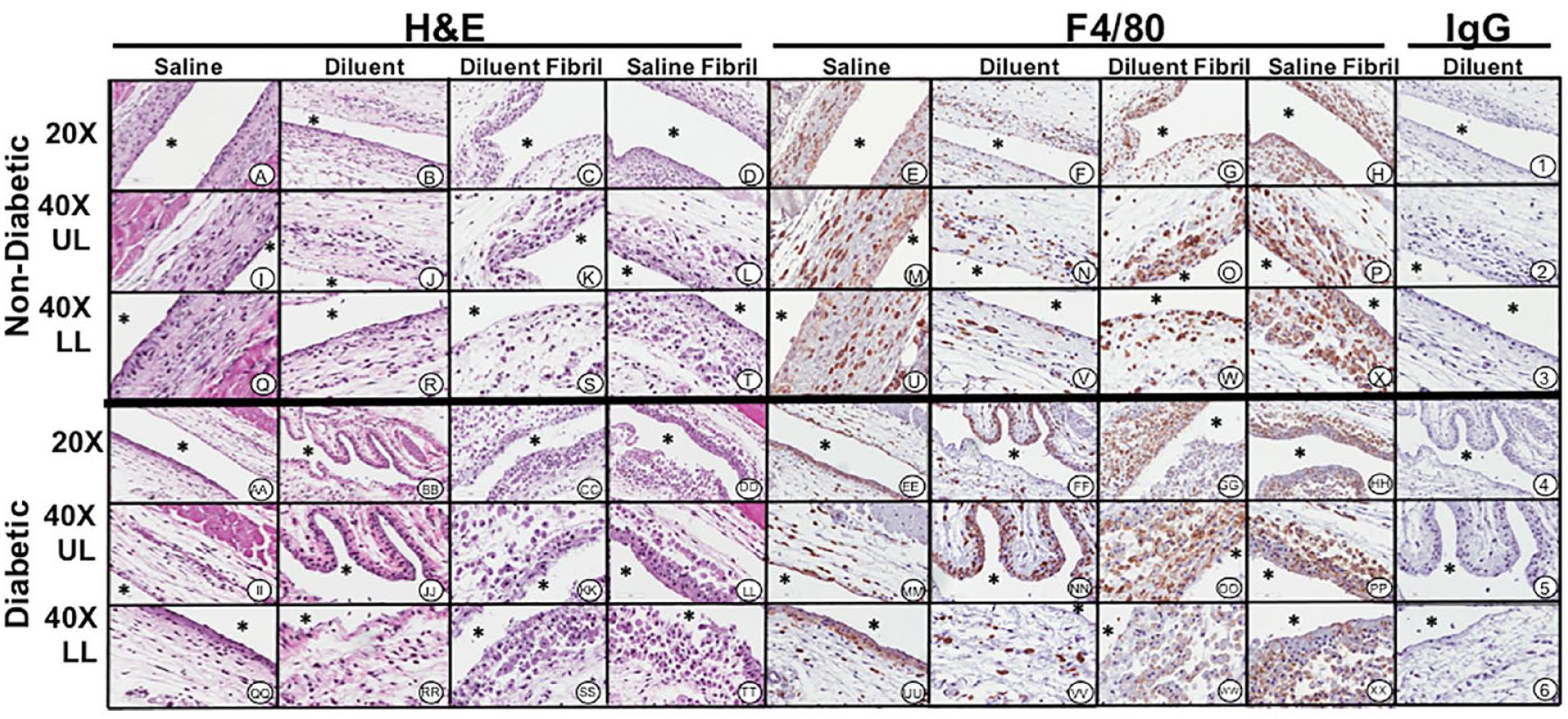
Histopathologic and immunohistochemical evaluation of mouse air-pouch tissue (post lavage) from non-diabetic and diabetic mice after 7 days treatment with insulin fibrils and phenolic diluent. Representative photo-micrographs of tissue reactions from 7-day treated non-diabetic and diabetic mice (post lavage) including: saline, diluent, saline fibril and diluent fibril treated non-diabetic and diabetic mice. Matched tissue sections were stained for general histopathology using H&E staining, and for macrophages using Anti-F4/40 antibody or matched non-immune IgG. All photo-micrographs were taken at 20× or 40× as designated. Each area of the MAP tissue evaluated in the study is designated as: UL (upper layer of MAP tissue), and LL (lower level MAP tissue), respectively. White area containing (*) indicates air space location of murine air pouch.
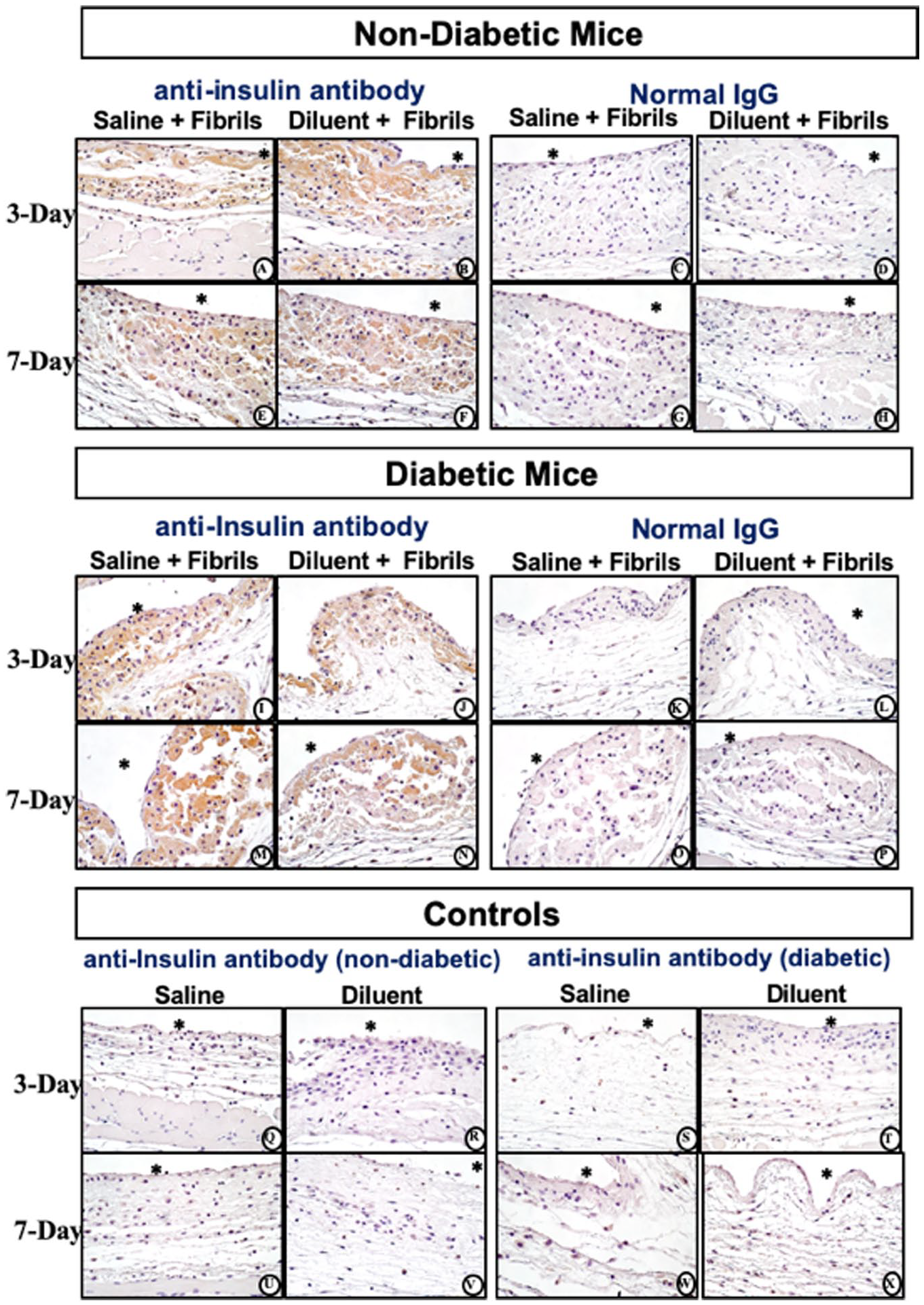
Histopathologic and Immunohistochemical evaluation of mouse air-pouch tissue (post lavage) from non-diabetic and diabetic mice after 3-and 7-days treatment with insulin fibrils. Matched tissue sections were stained for anti-insulin or non-immune IgG. Representative photo-micrographs of tissue reactions from 3 and 7 day treated with insulin fibrils for non-diabetic mice (top) and diabetic mice (middle). Background controls for anti-insulin stain for non-diabetic and diabetic mice treated with saline and diluent are shown in bottom panel. All photo-micrographs were taken at 40×. White area containing (*) indicates air space location of the murine air pouch.
Discussion
These data indicate that insulin derived fibrils contribute to tissue inflammation during SIA. Our study assessed both in vitro cell culture studies and a mouse air-pouch model designed to analyze cellular and tissue reactions following repeated injections with saline, PP, diluent, and fibrils suspended in either saline or diluent. The in vitro studies investigating cytotoxicity of PP and insulin fibrils suspended in saline or diluent demonstrated a macrophage specific cytotoxicity for the fibril addition (Figure 1). Mouse fibroblast and fat cells exhibited cytotoxicity towards the PP diluent, to insulin and to diluent fibril but not to saline fibril. This indicates that PP is cytotoxic to these cell lines whereas, the MQ cell line demonstrated cytotoxicity towards PP as well as fibrils. This suggests that fibril cytotoxicity is highly dependent on the cell type. Future studies should elucidate MQ death mechanisms following fibril phagocytosis.
Our in vivo data supplemented the in vitro findings. Specifically, FACS data revealed that MQ are recruited in vivo to the injection site when treated with diluent or saline fibril species (Figure 3). Total cell recruitment supports the hypothesis that fibril species and PP-containing diluent are pro-inflammatory. Influx of inflammatory cells is increased in the presence of insulin-derived fibril. Notably, saline versus diluent did not show any significant differences in inflammatory cell numbers for either 3-day in non-diabetic or 7-day in diabetic CD-1 mice, which is in contrast to our previously published results investigating infusion of these agents. 15 We attribute these differences to the different amounts of fluids administered between these 2 studies and duration of exposure. In the published infusion studies, 15 a total of 1.2 mL/day were administered continuously whereas the current study utilized a total of 0.3 mL once per day, suggesting a toxicity threshold of the diluent.
Histopathological evaluations of the air pouch tissue post lavage demonstrated that saline fibril and diluent fibril are equally capable of inducing inflammation characterized by an influx of PMNs and monocytes/macrophages (Figures 4 and 5). Notably the concentration of fibril solution injected into the air pouch is unlikely to be encountered clinically. Future follow-up studies should be designed to characterize a dose-response curve. Notwithstanding, it is likely that even small amounts of fibril could induce severe IDF that would alter tissue architecture and function rendering this injection or infusion site unusable. Irrespective of the saline or diluent agent utilized for fibril injections, the post lavage air pouch tissue site demonstrated neutrophils and macrophages phagocytizing insulin fibrils. An amorphous pink staining material was observed both external and internal to the PMN and MQ (Figures 4 and 5). The impact of fibril uptake by phagocytes on cell function and proinflammatory activity including the expression of proinflammatory cytokines will need to be investigated to determine the mediators and mechanisms of fibril-induced inflammation. Moreover, the potential contribution exhibited by ongoing leukocyte mediated fibril uptake and possible cell necrosis needs investigation. As diabetes is a chronic illness, one of the cornerstones of diabetic management is to maximize the availability of useful injection or infusion sites. Any strategy designed to optimize exogenous insulin administration and efficacy must mitigate pro-inflammatory factors including IDF.
Conclusions
Administration of insulin fibrils can lead to cytotoxicity, specifically in macrophages. In vivo data demonstrate insulin fibrils to be pro-inflammatory which over time can lead to cumulative cell/tissue toxicity, inflammation, and destructive wound healing. Long term, these tissue reactions could contribute to loss of insulin injection site architecture and function.
Supplemental Material
sj-pdf-1-dst-10.1177_19322968211033868 – Supplemental material for Insulin Derived Fibrils Induce Cytotoxicity in vitro and Trigger Inflammation in Murine Models
Supplemental material, sj-pdf-1-dst-10.1177_19322968211033868 for Insulin Derived Fibrils Induce Cytotoxicity in vitro and Trigger Inflammation in Murine Models by Brianne E. Lewis, Adam Mulka, Li Mao, Roshanak Sharafieh, Yi Qiao, Shereen Kesserwan, Rong Wu, Don Kreutzer and Ulrike Klueh in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
BL, LM, and YQ collected and interpreted the data and contributed to discussions. BL, DK, and UK drafted the manuscript. AM, RS, and SK contributed to discussion and data interpretation. RW completed statistical analysis. UK is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. The Microscopy, Imaging and Cytometry Resources Core is supported, in part, by NIH Center grants P30 CA22453 to the Karmanos Cancer Institute and R50 CA251068-01 to Dr. Moin, Wayne State University, and the Perinatology Research Branch of the National Institutes of Child Health and Development. This work was supported by The Leona M. and Harry B. Helmsley Charitable Trust (2017PG-T1D008).
Abbreviations
CSII, continuous subcutaneous insulin infusion; FACS, fluorescence-activated cell sorting; IDF, insulin derived fibril; MQ, macrophage; PMN, polymorphonuclear leukocytes; PP, phenolic preservatives; SIA, subcutaneous insulin administration; STZ, streptozotocin.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Ulrike Klueh, PhD and Don Kreutzer, PhD are co-founders and co- owners of the small business Cell and Molecular Tissue Engineering, LLC, Avon CT. No other potential conflicts of interest relevant to this article are present.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Leona M. and Harry B. Helmsley Charitable Trust supported these studies (2017PG-T1D008).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
