Abstract
Bariatric surgery is a known and effective treatment for type 2 diabetes mellitus. Patients with type 1 diabetes mellitus and exogenous insulin-requiring type 2 diabetes mellitus require adjusted insulin dosing after surgery to avoid hypoglycemia. This review describes insulin dose adjustments following a variety of bariatric procedures. After searching the available literature and assessing for eligibility, 8 articles were included. The Johns Hopkins Research Evidence Appraisal Tool for literature appraisal was used. The results of this review reveal insulin dose adjustment varies based upon surgical procedure type and time of follow-up from the procedure.
Introduction
With each passing year, the obesity epidemic becomes more pronounced. In 2016, nearly 40% of American adults, approximately 130 million people, were documented as having a body mass index (BMI) of ≥30 kg/m2. 1 The number of bariatric surgeries has increased by 70,000 over the last 7 years, with 228,000 procedures being performed in 2017. 2 In order to qualify for bariatric surgery, standard criteria (BMI ≥ 40 kg/m2, BMI 35-39.9 kg/m2 with a serious weight-related health problem) must be met. Bariatric surgery has been shown to improve outcomes in patients with type 2 diabetes mellitus by inciting an initial remission in over 2/3 of patients with 1/3 maintaining long-term euglycemia without the need for antidiabetic medications. 3 Furthermore, the use of bariatric surgery as a treatment option for type 2 diabetes mellitus is endorsed by various national organizations in their associated treatment guidelines.4,5 When followed over a 5-year timeframe, patients who received bariatric surgery instead of Mingrone et al 6 or in addition to Schauer et al 7 medical therapy were more successful in treating their type 2 diabetes mellitus. 8
The epidemic of obesity has not spared patients with type 1 diabetes mellitus. These patients, whom historically have had a slender body habitus, are now following national trends with 50% of type 1 diabetes mellitus patients currently being obese or overweight, and between 20 and 40% meeting the criteria of metabolic syndrome. 9 While type 1 diabetes mellitus is not caused by obesity, the management of this disease becomes more difficult and the progression of complications is hastened by the presence of morbid obesity. 10
Bariatric surgeries are categorized as restrictive (restricting dietary intake), malabsorptive (limiting absorption of calories), or a combination of both. The 2 most commonly performed operations for morbid obesity include laparoscopic vertical sleeve gastrectomy (LVSG), a restrictive procedure; and Roux-en-Y gastric bypass (RYGB), a restrictive and malabsorptive procedure. Both LVSG and RYGB have shown a dramatic improvement in type 2 diabetes mellitus, resulting in euglycemia and normal insulin levels within days after surgery. 11 Patients with either type 1 or type 2 diabetes mellitus must be monitored carefully in the perioperative period as low caloric intake and rapidly changing insulin sensitivity can lead to hypoglycemia if secretagogues (ie, sulfonylureas) and exogenous insulin are continued at preoperative doses. A recent review by Howard et al 12 reaffirms the lack of specific guideline recommendations to utilize and recognizes inconsistencies in insulin protocols and the outcomes studied thus far in available literature.
Purpose
The following clinical therapy question was asked: for patients with diabetes mellitus, what degree of exogenous insulin reduction can be expected after bariatric surgery? The focus of this review is to discuss the dosing of insulin in the time period following bariatric surgery.
Materials and Methods
PubMed and International Pharmaceutical Abstracts were searched using the key terms: diabetes mellitus, diabetes mellitus type 1, diabetes mellitus type 2, gastric bypass, insulin, bariatric surgery, weight loss, blood glucose, postoperative, Roux-en-Y gastric bypass, biliopancreatic diversion, gastrectomy, and hypoglycemic agents. The search terms used, and their combinations, can be found in Table 1. These search terms were used in various combinations to provide 41 articles. Studies that examined type 1 diabetes mellitus, type 2 diabetes mellitus, or both were included. Studies needed to explicitly mention outcomes associated with insulin doses. To be included in this analysis, studies needed to be retrospective or prospective, single-center or multi-center in nature, and needed to include patients undergoing LVSG or RYGB. Of the 41 results from the search terms, 8 studies qualified for inclusion, after review articles, case studies, meta-analyses, and systematic review articles were excluded. Studies were excluded if the focus was on non-insulin issues, such as polycystic ovarian syndrome (PCOS) commonly seen in bariatric surgery patients, instead of diabetes mellitus. Duplicate results from the searches were also excluded. Additional studies were selected from consulting the references of the qualifying articles. In total, this article summarizes 8 articles. The inclusion and elimination of articles can be seen in Figure 1.
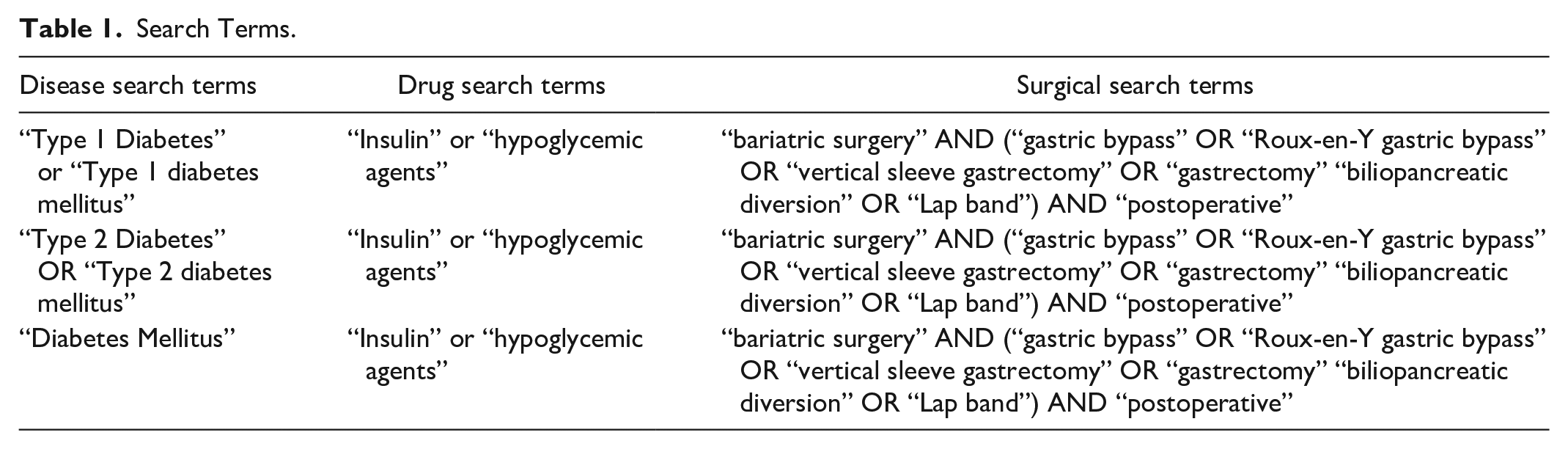
Search Terms.
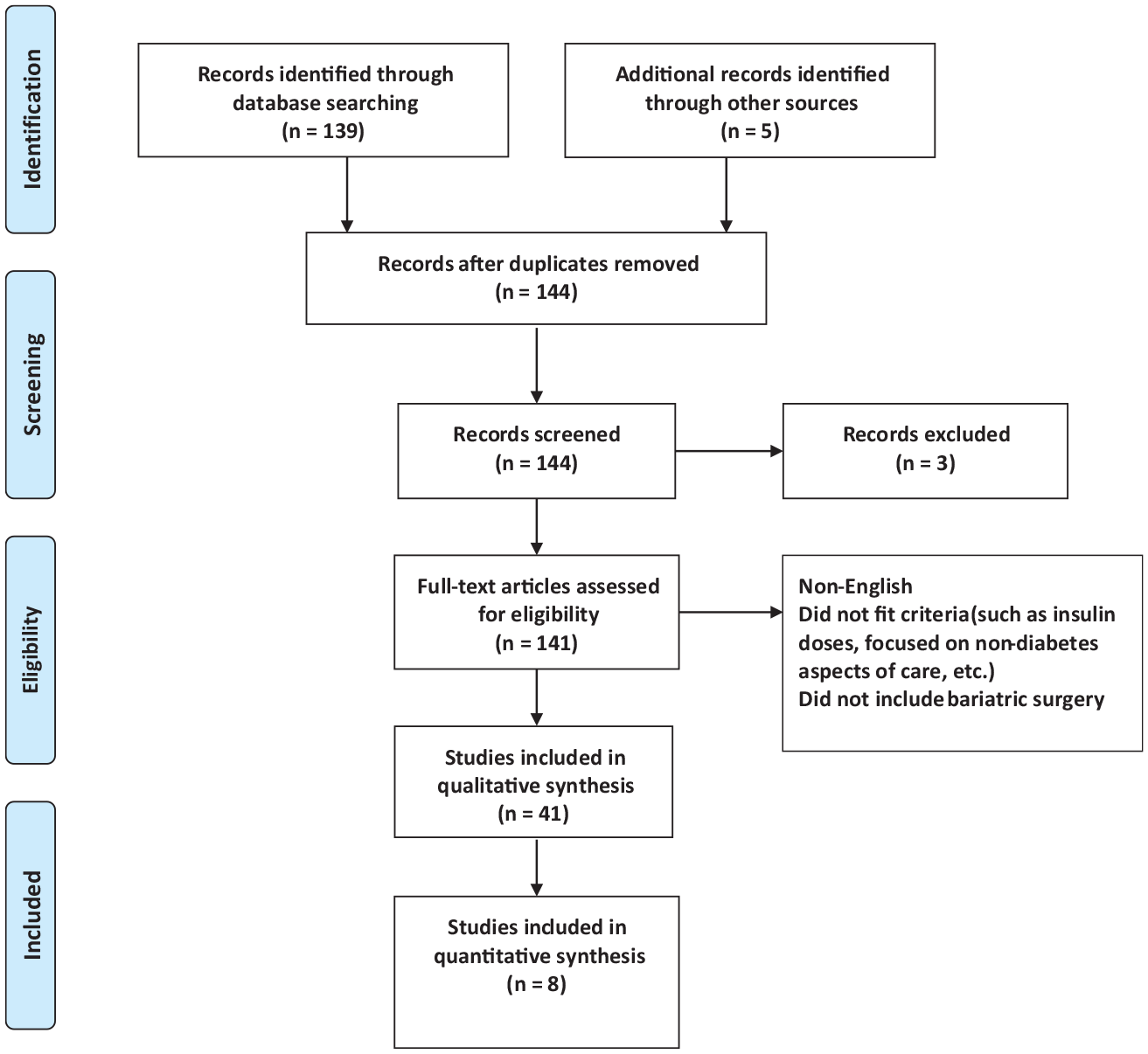
PRISMA flow diagram for inclusion of articles.
Articles were reviewed by 2 independent reviewers to ensure inclusion criteria were met. The Johns Hopkins Evidence Appraisal Method was used to grade the evidence of the included articles. This copyrighted tool is a part of the Johns Hopkins Nursing Evidence-Based Practice Model tools developed by Johns Hopkins Medicine. Literature is assessed to fit into differing levels (ie, I, II, III, IV, V) based on study design. A quantitative assessment is also assigned (A: high quality, B: good quality, C: low quality of major flaws) based upon study design, generalizability of results, sample size, limitations, presence (or absence) of a control arm, and literature reviews for the study in question. 13
Results
Studies were divided into categories for review, based upon the type of diabetes present within the studied population. Surgical techniques varied among the studies, which are further delineated in the synopses found in Table 2.
Synopsis of Available Literature.
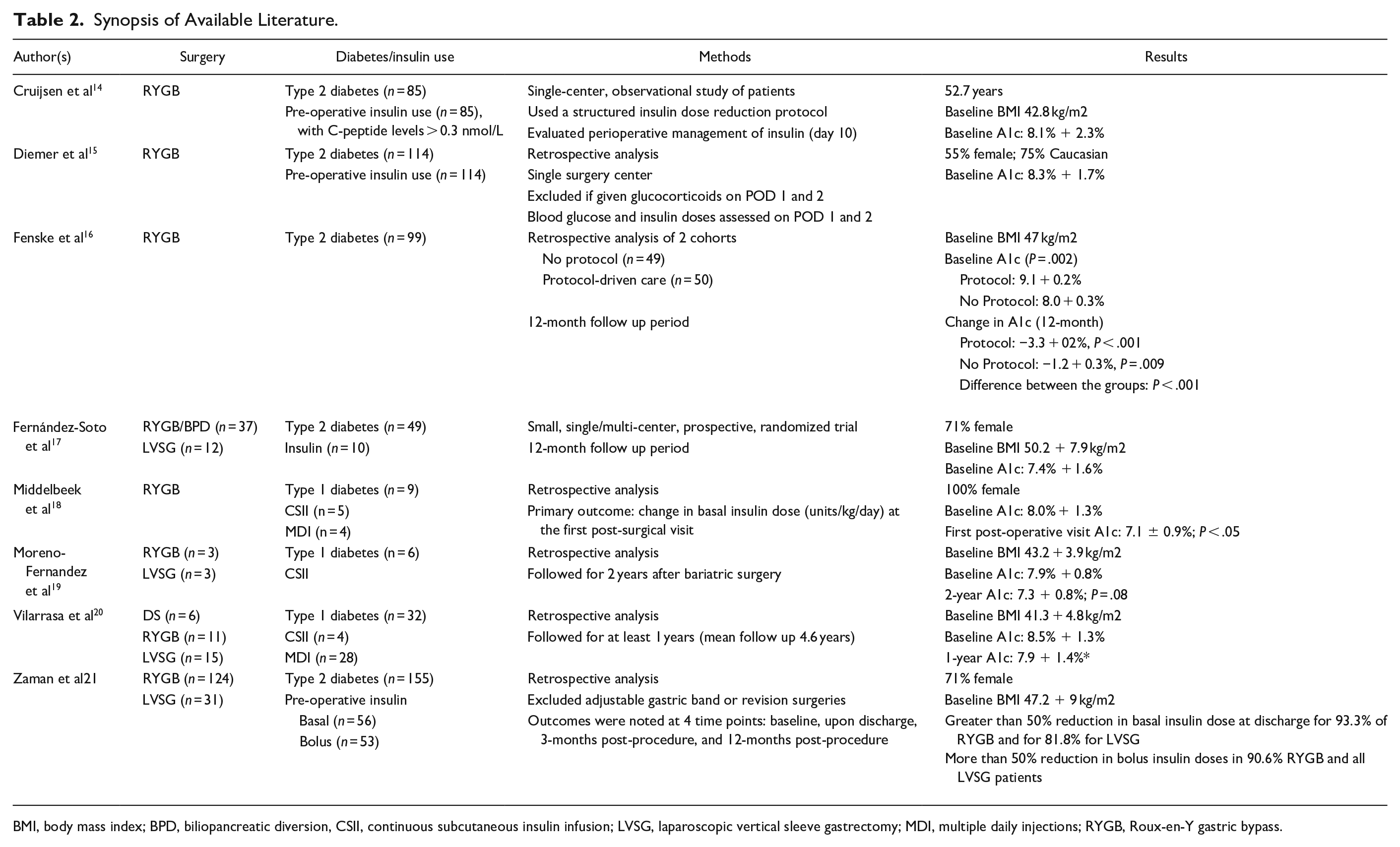
BMI, body mass index; BPD, biliopancreatic diversion, CSII, continuous subcutaneous insulin infusion; LVSG, laparoscopic vertical sleeve gastrectomy; MDI, multiple daily injections; RYGB, Roux-en-Y gastric bypass.
Type 2 Diabetes Mellitus
Five studies14-17,21 met the inclusion criteria for patients with type 2 diabetes mellitus, though the time frame of follow-up varied by study. Fernández-Soto et al 17 and Zaman et al, 21 included patients with multiple types of surgeries and followed them for 12 months. Fernández-Soto et al17 found that at the one-year post-operative time period, over 75% of patients, regardless of group, were in remission for their type 2 diabetes mellitus, and the insulin needs for those who remained on insulin (n = 4) decreased by 84% and 94% for RYGB/BPD and LVSG, respectively (Table 3). One major limitation of this study was that only 10 patients were on insulin at the beginning of the study. Zaman et al 21 included more patients who were on insulin at baseline, but separated them into basal and bolus groups for analysis separately. By 12 months, basal insulin doses decreased by approximately 85%, whereas, bolus insulin doses decreased by over 90%. While the authors differentiated insulin doses between RYGB and LVSG, the insulin reductions between the surgeries were not clinically meaningful. Neither of these studies described how the insulin adjustments were made or if they were standardized.
Reported Mean Insulin Dosing per Day at Specific Time Points for Type 2 Diabetes Studies.
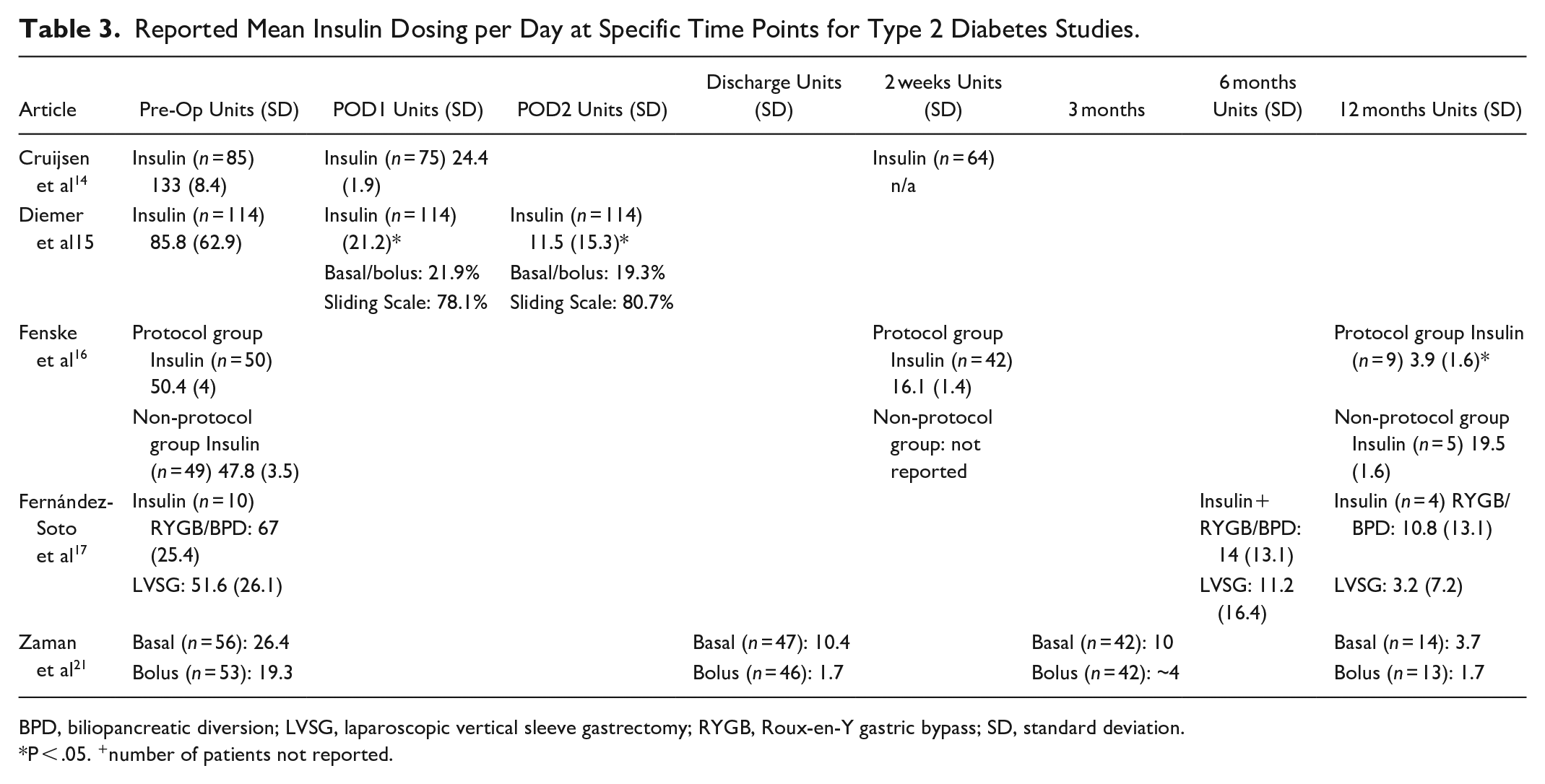
BPD, biliopancreatic diversion; LVSG, laparoscopic vertical sleeve gastrectomy; RYGB, Roux-en-Y gastric bypass; SD, standard deviation.
P < .05. +number of patients not reported.
Fenske et al 16 compared a protocol-driven approach versus no protocol for type 2 diabetes management following RYGB. Protocol-driven patients used metformin 1 gram twice daily and sliding scale insulin while inpatient, and patients were discharged on the equivalent glargine dose of their previous 24-hours use of insulin. Those not in this group had glucose management through the primary care provider and the care was not standardized. There was a significant reduction in A1c in both groups, though the protocol-driven group had a larger reduction. Every patient required a dose reduction in the first post-operative week. By the end of 2 weeks the protocol group had mean insulin doses decreased by 68%. Overall at 12 months, the study had a 85% discontinuation of insulin and a dose reduction of 60-90%.
The studies above examined a longer-term follow-up, whereas Diemer et al 15 and Cruijsen et al 14 studied insulin during the peri-operative time period for the RYGB surgery. Diemer et al 15 found significant reductions of 85-95% in the post-operative insulin doses on POD 1 and 2 compared to pre-operative doses. The mean insulin doses for POD1 and 2 were between 0.05 and 0.15 units per kg body weight. Cruijsen et al 14 used a prespecified protocol to handle perioperative glucose management. This included reducing the basal insulin by 50% the day prior to the surgery, then switching to bolus insulin 3 times daily at a dose that was 75% lower than the preoperative dose on POD1. This insulin was combined with glimepiride, which replaced the basal insulin, and metformin. Insulin doses fell by 82% between pre-op and POD 1. Both studies showed reductions in insulin doses perioperatively after bariatric surgery and could be utilized to develop protocols during this time period; however, they offer little insight into long term outcomes of the patients after discharge.
Type 1 Diabetes Mellitus
Three studies18-20 were included for patients with type 1 diabetes mellitus. The studies also represented 3 surgical types: RYGB, duodenal switch (DS), and LVSG. Middelbeek et al 18 followed 9 patients to the first post-surgical follow up visit (7.7 weeks post-bariatric surgery) and evaluated change in insulin dose, with the primary outcome being change in basal dose (Table 4). This was done because the bolus dose history was not always found for those on continuous subcutaneous insulin infusions (CSII). A significant reduction in basal and total daily dose of insulin (of those able to be calculated) was found. Approximately a 50% reduction in both was found from pre-operative to the first post-surgical follow-up, but there was no correlation between insulin requirements and weight loss indicating other physiological changes may also affect insulin doses (r = 0.34, P = .37). Vilarrasa et al 20 found that A1c was reduced between baseline and the 1-year post-operative follow-up period, but this did not correlate with weight loss and did not differ with surgery types. At the end of the first year, the insulin requirements had significantly decreased by 50%, and then did not change greatly through the five-year follow-up plan, indicating that most changes resulting in insulin dose reduction occur during the first year following a surgery. The decreases were similar for basal and bolus requirements, 50.2% and 54%, respectively. Moreno-Fernandez et al 19 conducted a small study in those on CSII only (n = 6) where they found no difference in A1c between baseline and 2-years post-operative, though the insulin dose significantly decreased. Although limited by the number of patients, it is the only study available that only follows CSII patients after bariatric surgery and does show a decrease in overall insulin need after surgery.
Reported Mean Insulin Dosing per Day at Specific Time Points for Type 1 Diabetes Studies.
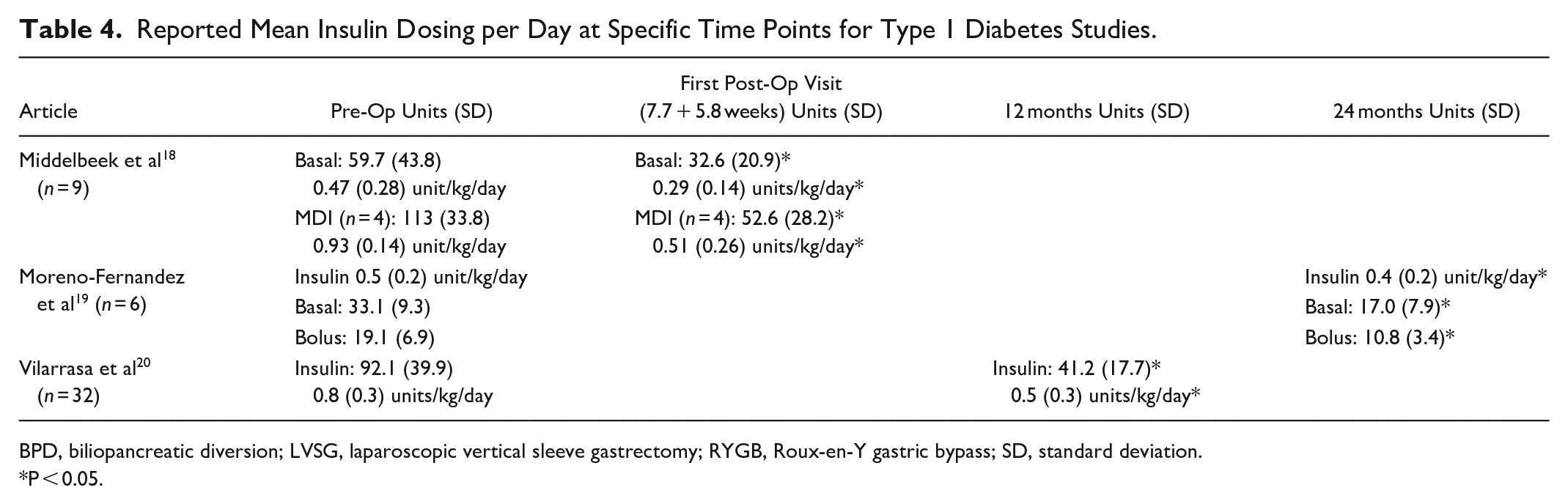
BPD, biliopancreatic diversion; LVSG, laparoscopic vertical sleeve gastrectomy; RYGB, Roux-en-Y gastric bypass; SD, standard deviation.
P < 0.05.
Discussion
It is well-documented that bariatric surgery results in resolution of diabetes and other comorbid conditions.4-8 The doses of insulin are drastically reduced following bariatric surgery, with approximately a 75-90% reduction in patients with type 2 diabetes14-17,21 and approximately a 50% reduction in patients with type 1 diabetes.18-20
Previous studies that investigated in-hospital doses and the resultant reductions indicate a reduced dose of insulin while admitted for bariatric surgery.14,15 It is unknown how or if these dose reductions are maintained following discharge, as there are no long-term follow up studies for these specific patients. A link between the pre-operative, inpatient, and then post-operative dosing schemes would allow prescribers to anticipate dose reductions are specific time points following the bariatric surgery. The decrease is evident in the trials presented here; however, the ability to predict specific dose reductions at specific timepoints post-operatively remains unknown.
An insulin dose reduction is expected for patients with type 1 diabetes following bariatric surgery, without the goal of completely removing insulin therapy. Interestingly, the 3 included studies of patients with type 1 diabetes indicate that weight loss is not correlated with the insulin dose required.18-20 This could be due to the small number of patients included, or there may be other physiological principles that are not accounted for when comparing weight loss and insulin dose. Moreno-Fernandez et al 19 showed a significant reduction in insulin dose throughout their study period; however, it is important to note that their starting total doses were lower when compared with the other trials of patients with type 1 diabetes.
Bariatric surgery is a recommended therapy for patients with type 2 diabetes. 5 The landmark trials7,22 indicate achievement of glycemic control in patients who underwent bariatric surgery plus intensive medication therapy. Though even in these patients who were enrolled in longitudinal studies to determine the efficacy of bariatric surgery (RYGB and LVSG) on glycemic control and hyperglycemia resolution, their success waivered, depending upon the point in time from surgery. Almost 40% of patients who underwent RYGB had achieved an A1c < 6.0% at 3-years, but this number decreased to 29% at 5-years. Mean weight loss followed a similar, though not as drastic pattern: −24.5 + 9.1% at 3-years and approximately −23% at 5-years. Patients who underwent LVSG had similar rates of achieving A1c < 6.0% at 3- and 5-years: 24% and 23%, respectively. Mean weight loss was higher at the 3-year mark (−21.1 + 8.9%) compared with the 5-year timepoint (−19%) for patients who had received LVSG. The results of the STAMPEDE trial indicate that bariatric surgery facilitates achievement of glycemic control more than intensive medical therapy alone; however, they demonstrate that the patients did not maintain as tight of A1c control or weight loss the further out from surgery.
Another study of 50 patients who underwent LVSG followed patients post-operatively 23 showed that individuals who were categorized as percent estimated weight loss (%EWL) > 50% were more likely to have A1c < 7.0% and be off of insulin therapy at the 5-year timepoint. Fewer patients in this study were on insulin (n = 11, 21.5%) and had better A1c control compared with the STAMPEDE trial cohort (7.9 + 1.6% at baseline). At the 5-year point, only 3 patients (5.8%) remained on insulin therapy: 2 of whom had %EWL < 50% and one of whom had %EWL > 50%. This study did not assess the post-operative outcomes are various timepoints follow surgery and only did so at the 5-year mark, thus providing excellent information for long-term follow-up. It does lack in providing specific dosing of insulin for the patients as well as evidence at which timepoint insulin dosing is predictably reduced following bariatric surgery.
These findings are echoed in a retrospective analysis of a single center by Barry et al 24 It followed 223 patients who had LVSG, many of these were patients with type 2 diabetes (n = 137) on insulin (n = 69). Although this study showed that only 24.8% of patients with type 2 diabetes remained on insulin, no specific information on insulin dosing prior to or after the study was mentioned, so no analysis on how and when the reductions happen could be made.
Limitations
While the data presented here contribute to the growing body of literature surrounding bariatric surgery, it is not all-inclusive for how to best manage a patient’s insulin dosing following bariatric surgery. Most of the studies included in this review are small and are of retrospective and single-center design. Larger studies may not be applicable to all patients, such as the Veterans Affairs population inclusion of many more males than females. While RYGB is the second most-performed surgery in the world, it has fallen behind LVSG. Most studies presented here include RYGB and BPD, which may not accurately reflect contemporary and evolving surgical practices.
Conclusions
Bariatric surgery consistently leads to weight loss; however, the impact on insulin doses and glycemic management varies based upon type of diabetes as well as procedure type. Additional physiological factors may impact insulin doses, particularly after the first year following surgery, making it difficult to define a predictable pattern of insulin use after bariatric surgery. The included studies focus on either short-term or long-term follow-up, limiting the clinician’s ability to preemptively adjust the insulin doses to avoid hypoglycemic events. Additional studies are likely needed to describe the impact on insulin dosing and glycemic control in patients with type 1 diabetes mellitus or insulin resistant type 2 diabetes mellitus following laparoscopic bariatric surgery procedures.
Footnotes
Abbreviations
%EWL, percent estimated weight loss; BMI, body mass index; BPD, biliopancreatic diversion; CSII, Continuous subcutaneous insulin infusion; LVSG, laparoscopic vertical sleeve gastrectomy; MDI, multiple daily injections; PCOS, polycystic ovarian syndrome; POD, Post-operative day; RYGB, Roux-en-Y gastric bypass.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
