Abstract
Background:
Previous studies utilizing glucose data from continuous glucose monitors (CGM) to estimate the Glucose Management Indicator (GMI) have not included young children or determined appropriate GMI formulas for young children with type 1 diabetes (T1D).
Methods:
We extracted CGM data for 215 children with T1D (0-6 years) from a repository. We defined sampling periods ranging from the 3-27 days prior to an HbA1c measurement and compared a previously established GMI formula to a young child-specific GMI equation based on the sample’s CGM data. We examined associations between HbA1c, GMI values, and other CGM metrics for each sampling period.
Results:
The young child-specific GMI formula and the published GMI formula did not evidence significant differences when using 21-27 days of CGM data. The young child-specific GMI formula demonstrated higher correlations to laboratory HbA1c when using 18 or fewer days of CGM data. Overall, the GMI estimate and HbA1c values demonstrate a strong relationship in young children with T1D.
Conclusions:
Future research studies may consider utilizing the young child-specific GMI formula if the data collection period for CGM values is under 18 days. Further, researchers and clinicians may consider changing the default number of days of data used to calculate glycemic metrics in order to maximize validity of CGM-derived metrics.
Introduction
Hemoglobin A1c (HbA1c), a metric that approximates the average glucose values over the previous 8-12 weeks, remains the dominant biomarker for characterizing glycemic levels in type 1 diabetes (T1D). This metric, however, does not provide nuanced information regarding fluctuating daily blood glucose patterns or insulin-dosing decisions, 1 and HbA1c is difficult to obtain when clinical care is delivered remotely via video telehealth. Continuous glucose monitors (CGMs) are devices that are increasingly used to provide helpful information on interstitial glucose concentrations, including detecting trends in average daily glucose, time in range, episodes of hypoglycemia, hyperglycemia, and glucose variability. 2 Since CGM devices collect large amounts of data in real time, they offer alternative and perhaps more immediately actionable ways to assess glycemic levels in persons with T1D.
Several research groups have sought to examine the relationship between HbA1c and average glucose levels using CGM data in older adolescents and adults with T1D. For example, Nathan et al. 3 converted HbA1c to estimate average glucose levels, using an equation that can also be rearranged to convert average glucose levels into estimated HbA1c levels (eA1c). One study evaluated this equation in a pediatric sample, however, wide differences in average HbA1c across samples suggest a need to evaluate young children and older children separately. 4 Offering another approach, Bergenstal et al. 5 suggest calculating the Glucose Management Indicator (GMI) from CGM data to estimate HbA1c levels based on a formula derived from the regression line of mean glucose concentration points and HbA1c values. Both of these studies suggest as few as 12 days of CGM data are adequate to calculate the GMI in adults and older children with T1D, 5 however another study suggests the optimal duration for CGM data to robustly assess glycemic outcomes depends on the population being studied. 6 Indeed, inter-individual differences exist in the mean age (ie, days) of red blood cells. 7 Whether the mean age of red blood cells specifically varies between young children and older populations is less understood, however, theoretically this factor may impact the relationship between estimates of glycemic levels and HbA1c in young children differently than in adults. To date, no study has determined if GMI or eA1c formulas developed in adult samples perform similarly among young children under the age of 6 years. Further, with the absence of GMI-related studies that include young children with T1D, it is not clear if 12 days of CGM data are adequate to achieve a stable estimation of HbA1c in this unique population.
Many clinicians and adult patients find the GMI metric useful when managing T1D remotely since measurement of HbA1c at home is challenging and costly to coordinate, and since CGM data can provide additional information pertaining to a patient’s current glucose management status. 5 Families of young children may particularly benefit from a GMI formula optimized for their needs since managing T1D in young children often involves balancing their heightened insulin sensitivity with their unpredictable eating behaviors, inconsistent physical activity levels, and limited communication skills. 8 Thus, if we were to confirm previous formulas and time intervals are valid for use in young children, or were to alternately provide an optimized young child-specific GMI formula and demonstrate the optimal number of days required to calculate the GMI in young children, then clinicians and families would have a way to gain adequate information regarding a child’s overall glycemic control without the need for a venous blood draw, point-of-care fingerstick, or in-person clinic visits. Ultimately, this would reduce family burden and the number of painful in-clinic procedures performed on children.
The present study sought to determine (1) if the previous GMI formula demonstrates adequate performance characteristics in young children, (2) if an alternate young child-specific GMI formula displays improved performance characteristics in young children, and (3) the number of days of CGM data required to adequately estimate the GMI in young children with T1D. We specifically hypothesized that more days with CGM data would be required than in previous adult studies measuring GMI or estimating glycemic levels since young children exhibit more inter-day glycemic variability than older youth and adults with T1D.
Subjects, Materials, and Methods
Data Source
The present study extracted data from a research repository at Children’s Mercy on Type One Diabetes in Pediatrics (Mercy on TODP).9,10 This repository contains longitudinal electronic health record data (ie, demographic, clinical, laboratory) of patients with T1D who visited a Pediatric Diabetes Clinic at any clinic location in an 11-site bi-state clinic network. The Institutional Review Board approved the research with a waiver of written informed consent according to the requirements specified in 45 CFR 46.116(d).
Inclusion/exclusion criteria
Inclusion Criteria included children who (1) are aged 0 to 5.99 years, (2) have a diagnosis of T1D, (3) have available CGM data in the repository database collected through the Mercy on TODP repository, and (4) have a historic HbA1c value in the repository database that corresponds temporally to the CGM data collected. Exclusion Criteria included a diagnosis of type 2 or monogenic diabetes, as well as no historic use of a CGM device.
Measures
Continuous Glucose Monitors (CGM)
The present study included data from young children’s personal CGM device; namely, Medtronic Enlite and Dexcom CGM devices (any model). CGM devices continuously measure the glucose concentration in interstitial fluid and report on these integrated glucose concentration signals approximately every 5 minutes over a wear time of 7-14 days. The mean absolute relative difference (MARD) for CGMs in pediatric patients is approximately 10%. 11 CGM data were binned in 3-day increments up to 27 days before the clinic visit when each young child’s most recent HbA1c was measured. We included days with at least 12 hours of available CGM data in the analysis. Further, we only included the CGM data for a given child’s HbA1c if at least 70% of the 27 days were available. These procedures are consistent with previously published studies. 12
HbA1c
The hospital’s Pediatric Diabetes laboratory measured all HbA1c values using either the Tosoh-G8 HPLC assay or the Afinion point-of care instrument, depending on the clinic location. The instruments used to measure each HbA1c were certified by the National Glycohemoglobin Standardization Program and traceable to the Diabetes Control and Complications Trial Reference Method.
Data Analysis
We first identified unique, eligible individuals in the repository with an HbA1c value and available CGM data within a 27-day interval preceding the HbA1c value. We randomly chose a single 27-day period and corresponding HbA1c value for children who had multiple HbA1c in the data repository. For each interval, we defined a sampling period ranging from the 3 days prior to the date HbA1c was measured to the 27 days prior to that date in increments of 3 days (ie, 3, 6, 9, 12, 15, 18, 21, 24, and 27 days). For each child, we computed the following CGM metrics: (1) mean glucose concentration, (2) standard deviation (SD) of glucose concentrations, (3) percentage of time in the target range (70-180 mg/dL), (4) percentage of time in the hyperglycemic range (>180 and >250 mg/dL), (5) percentage of time in the hypoglycemic range (<70 and <54 mg/dL), and (6) coefficient of variation (CV; SD divided by mean glucose concentration).
We first calculated a GMI value based on a previously established formula by Bergenstal et al. 5 We then computed an equation from the regression line of mean glucose concentrations plotted on the x-axis and HbA1c values plotted on the y-axis to calculate a young child-specific GMI formula. We used the equation derived from the regression line to compute all young child-specific GMI values for the young children in the sample. See Table 1 for each equation used in the analysis. Next, we calculated the difference between each GMI value and the laboratory-based HbA1c value and evaluated for significant differences between each GMI value and HbA1c across each sampling period. We assessed the association between HbA1c and the young child-specific GMI value using the squared value of the Spearman correlation coefficient (R2). To examine when correlations plateaued, we examined how each of the core CGM metrics correlated with 27 days of the same measure. For example, we examined how the standard deviation of 27 days of glucose data correlated with 3 days, 6 days, 9 days, and so on. and we repeated this process for time below range (ie, <70 mg/dL), time above range (ie, >180 mg/dL), time in range, and CV. We used SPSS version 26 for all data analyses and considered a P-value <.05 as our threshold for statistical significance. Lastly, we assessed the degree of agreement between both GMI formulas and laboratory HbA1c values through Bland-Altman plots created using the R statistical platform.
Equations for GMI Evaluated in Sample of Young Children with T1D.

Results
We included data from 215 young children in the present analyses. The average age for the sample was 3.87 ± 1.02 years, 57% were female, and 96% were non-Hispanic white. The average HbA1c for the sample was 8.53 ± 1.64%, with a range of 4.9-13.2%. Regarding CGM type, 91% wore a Dexcom sensor, 5% wore a Freestyle Libre sensor, and 4% wore a Medtronic sensor. For all 27-day sampling periods analyzed, the average glucose level was 183 mg/dL, children’s mean percent time in range (71-180 mg/dL) was 56%, children’s mean percent time above range (>180 mg/dL) was 35%, children’s mean percent time below range (<70 mg/dL) was 9%. Further, children’s mean percent time in the very low range (<54 mg/dL) was 0.4%, children’s mean percent time in the very high range (>250 mg/dL) was 16%, and children’s average CV was 32%. The regression line of mean glucose concentrations plotted on the x-axis and HbA1c values plotted on the y-axis revealed the following equation = 4.824 + 0.017 [mean glucose in mg/dL]. See Figure 1 for the plot of CGM-measured mean glucose concentrations and laboratory HbA1c values used to estimate the young child-specific GMI formula. Of note, the Bergenstal et al. 5 GMI formula only included Dexcom sensor data, thus we computed a similar young child-specific GMI formula using only the Dexcom data in the event the GMI formula significantly differed based on sensor type. The regression line of mean glucose concentrations from Dexcom sensors and HbA1c values revealed the following equation = 4.874 + 0.016 [mean glucose in mg/dL]. We retained the GMI formula using all sensor data for the remaining analysis as the formulas were very similar. The average Bergenstal et al. 5 GMI value for the sample was 8.82 ± 1.11%, with a range of 6.2-12.5%, and the average young child-specific GMI value for the sample was 8.74 ± 0.79%, with a range of 7.1-11.8%.
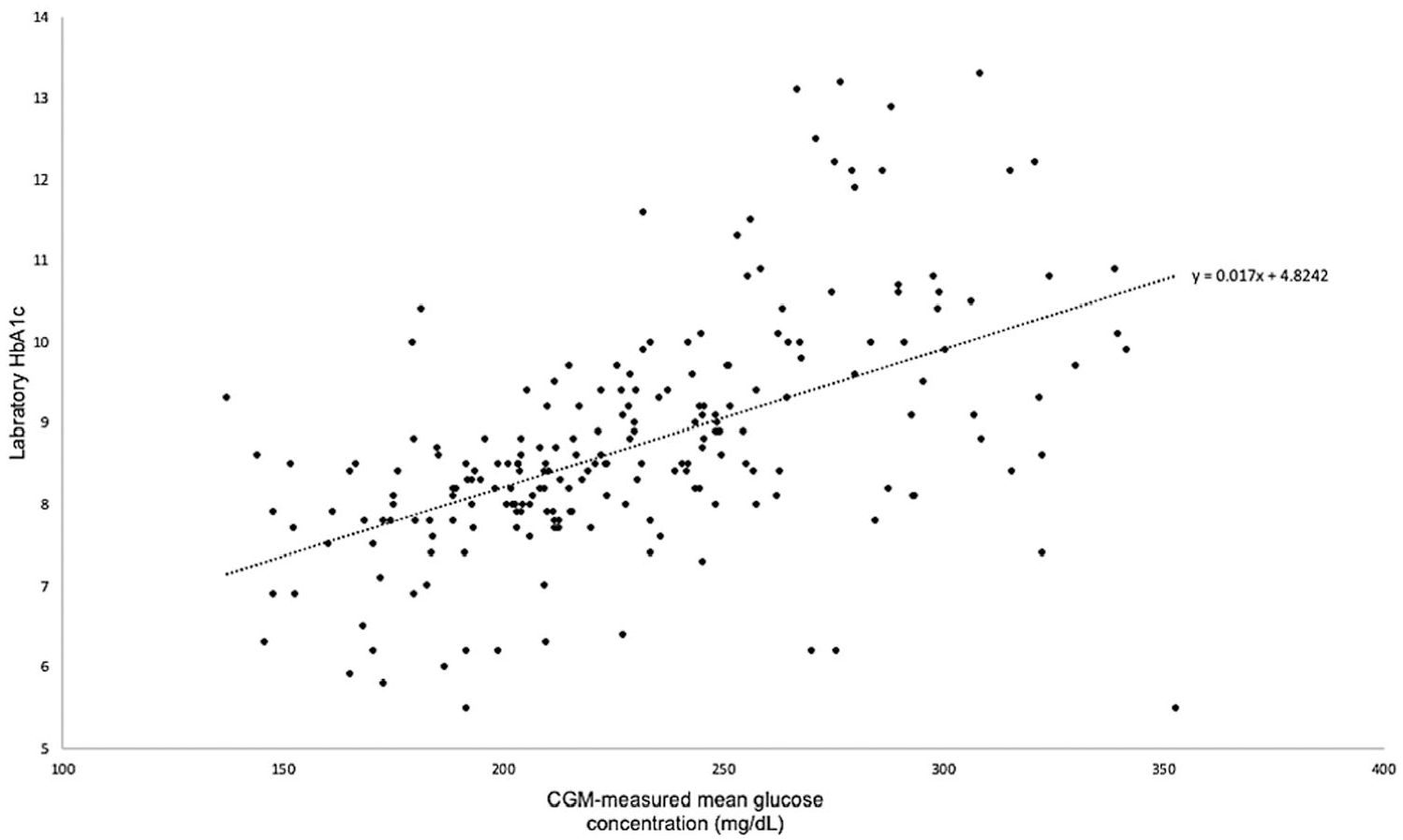
Plot of CGM-measured mean glucose concentrations and laboratory HbA1c values used to estimate the GMI formula.
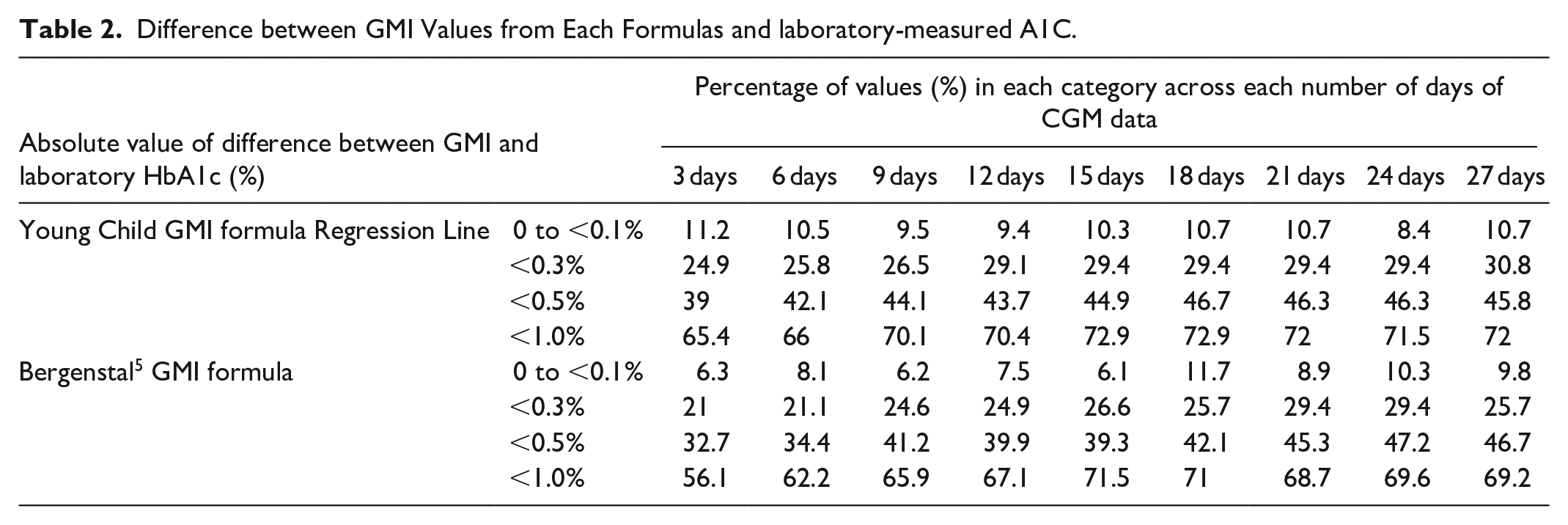
Once we calculated each GMI value based on the 2 equations (see Table 1) we evaluated the difference between each GMI value and the laboratory-based HbA1c value across each sampling period. The results of these differences indicate that between 8.4 and 11.2% of the time the young child-specific GMI value and laboratory HbA1c have an identical value, while 24.9-30.8% of the time they differed by at least 0.3% HbA1c points, and 39.0-46.7% of the time they differed by at least 0.5% HbA1c points across all sampling periods (see Table 2). For the Bergenstal et al. 5 GMI formula, 6.1-11.7% of the time the GMI value and laboratory HbA1c had an identical value, while 21.0-29.4% of the time they differed by at least 0.3% HbA1c points, and 32.7-47.2% of the time they differed by at least 0.5% HbA1c points across all sampling periods. Interestingly, we did not observe significant differences between our young child-specific GMI formula and the Bergenstal et al. 5 GMI formula for values calculated from 21-27 days of CGM data. We did however observe our young child-specific GMI formula demonstrate higher accuracy when 18 or fewer days of CGM data were used to calculate the GMI value (P < .05).
Difference between GMI Values from Each Formulas and laboratory-measured A1C.
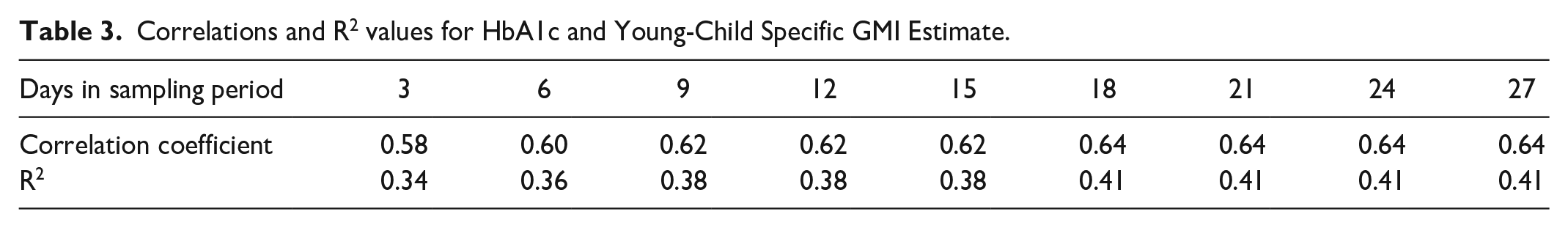
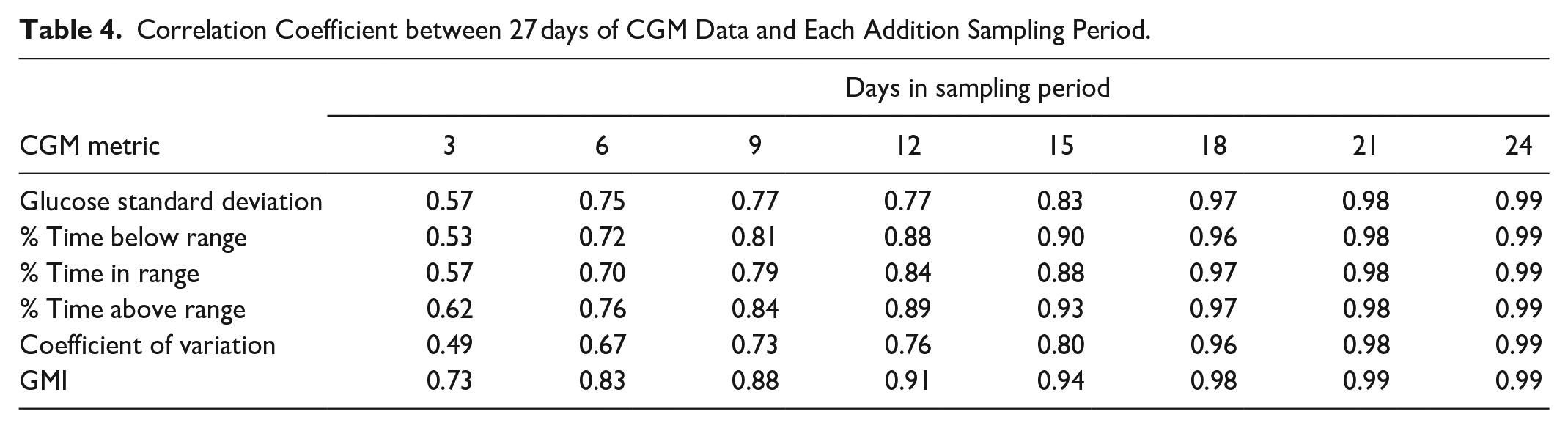
Next, we examined the correlations between our young child-specific GMI value, HbA1c, and other CGM-derived glycemic metrics. Overall, we observed that our young child-specific GMI estimate and HbA1c values demonstrated strong associations across sampling periods (see Table 3). However, as hypothesized, we observed a R2 peak at 18 days of CGM data when we examined HbA1c values (see Table 3). Therefore, to confirm this peak, we examined the correlations between 27 days of CGM data and each shorter sampling period for each of our CGM metrics and our young child-specific GMI value. The results of these correlations suggest a plateau begins at 15-18 days of CGM data (see Table 4). As a final measure of concordance between each GMI formula and the laboratory HbA1c values, we present Bland-Altman plots in Figures 2 and 3. These plots demonstrate that approximately 95% of the data points fall within 2 standards deviations of the mean difference between HbA1c and GMI values. We include Bland-Altman plots for both GMI formula across each sampling period in the supplemental materials.
Correlations and R2 values for HbA1c and Young-Child Specific GMI Estimate.
Correlation Coefficient between 27 days of CGM Data and Each Addition Sampling Period.
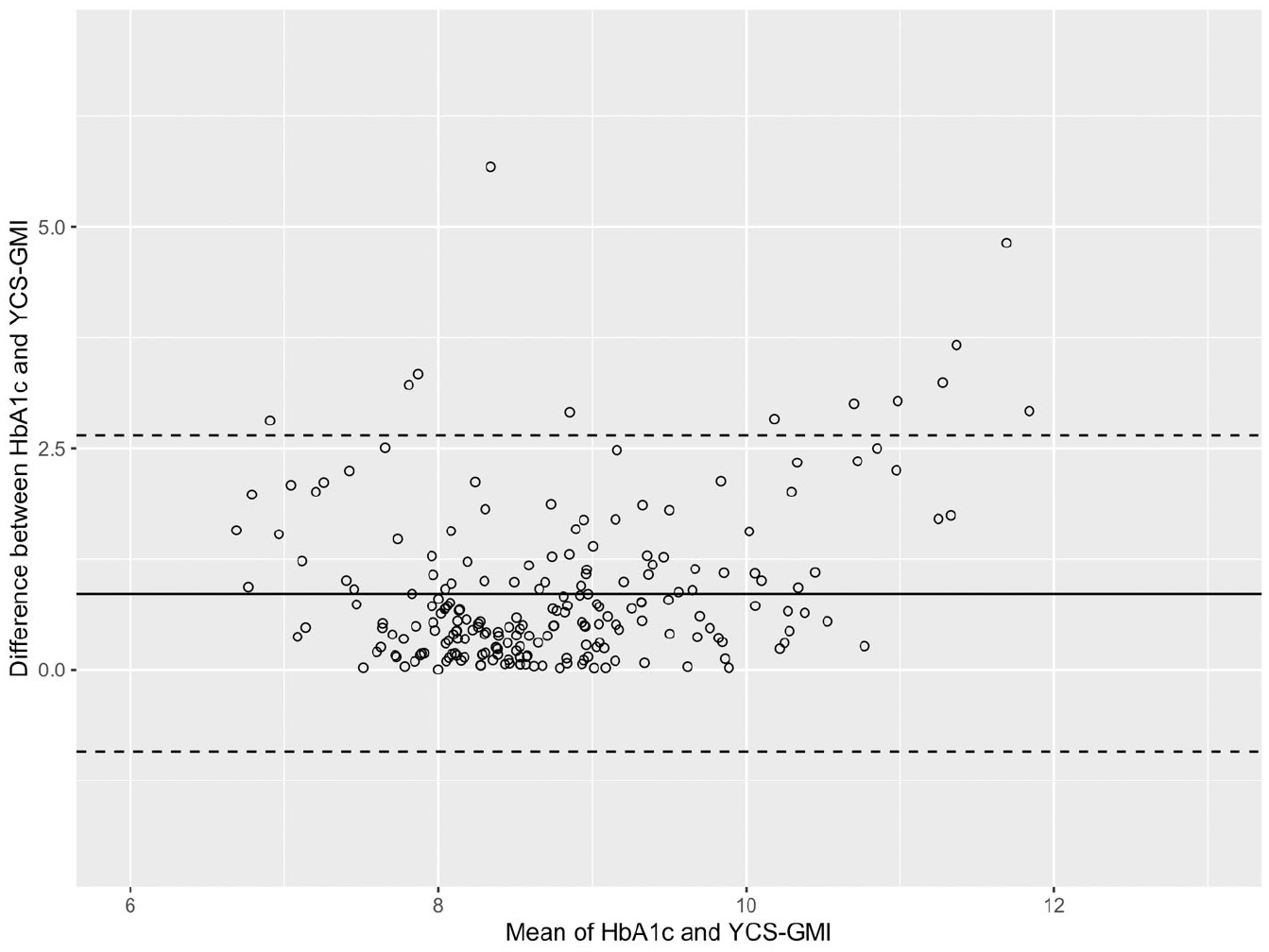
Bland-Altman plot displaying the agreement between the laboratory HbA1c value and young child specific GMI (YCS-GMI) value using 18 days of CGM data. The Y axis indicates the difference between the HbA1c and YCS-GMI values while the X axis indicates the average of these values. The solid line represents the average of the differences between values. The dotted line represents 2 standard deviations above and below the mean difference.
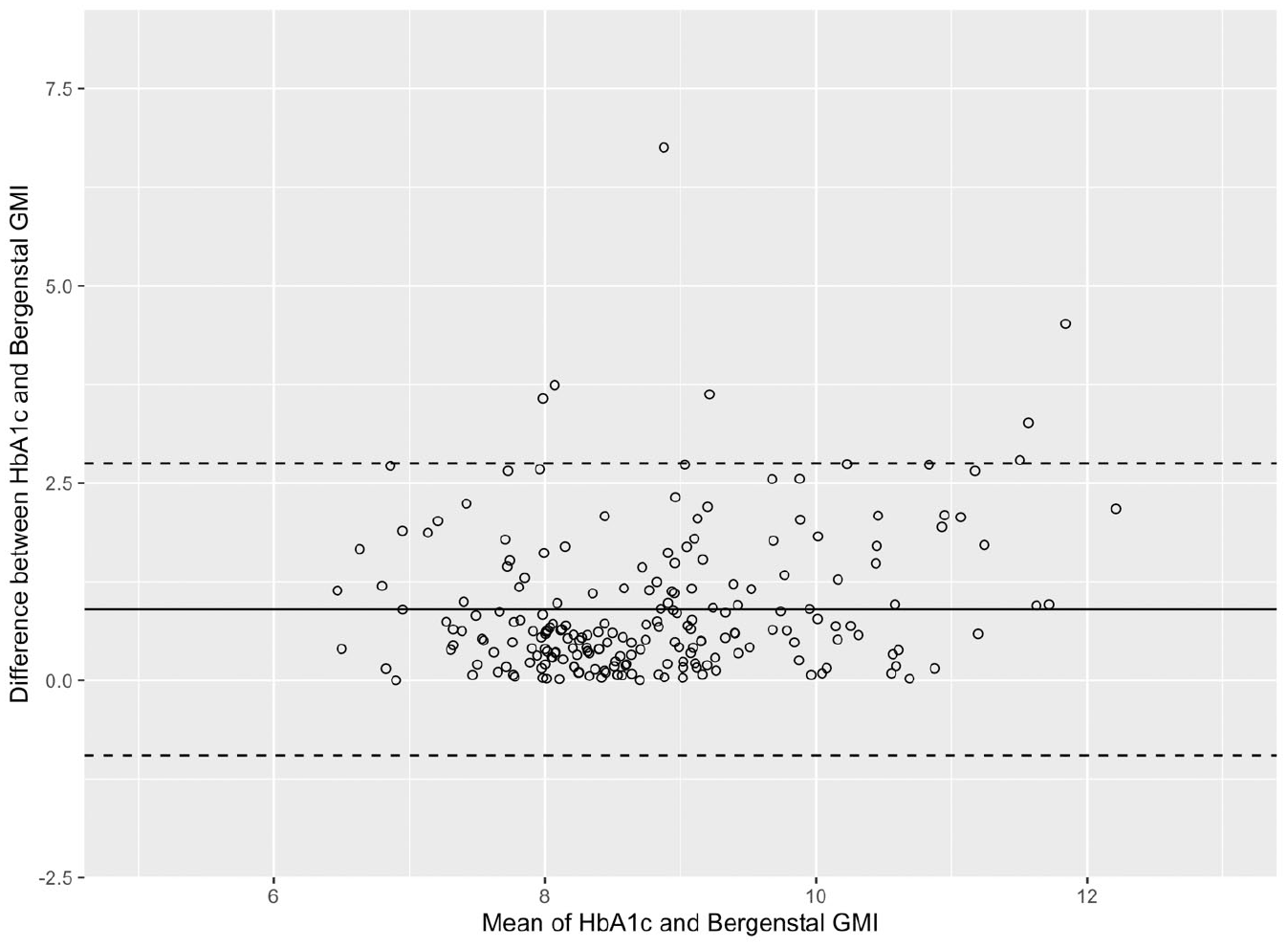
Bland-Altman plot displaying the agreement between the laboratory HbA1c value and Bergenstal GMI value using 18 days of CGM data. The Y axis indicates the difference between the HbA1c and Bergenstal GMI values while the X axis indicates the average of these values. The solid line represents the average of the differences between values. The dotted line represents 2 standard deviations above and below the mean difference.
Discussion
Our results suggest that a young child-specific GMI formula may be more precise if using 18 or fewer days of CGM data, but if using 21-27 days of CGM data, either our young child-specific formula or the Bergenstal et al. 5 formula may be acceptable to use in children under 6 years with T1D. They further indicate that ≥18 and ≥21 days of CGM data may be required to generate the most stable estimate of HbA1c using the young child-specific and Bergenstal et al. 5 GMI formulas, respectively. Further, our results build upon previous research estimating glucose metrics in children with T1D broadly by providing options for estimating GMI in young children specifically. 13 Additionally, consistent with our hypothesis, we found that more days of CGM data are necessary in young children compared to adults to provide a stable estimate of 90-day average glycemic levels, an effect which may be due to the level of increased insulin sensitivity and glycemic variability typically observed in younger children with T1D versus adults and older adolescents. 14 One might also hypothesize that age-related differences in either red blood cell lifespan or glycation of hemoglobin could theoretically contribute to age-related differences in the relationship between mean CGM-derived glucose values and HbA1c, but age-related differences do not appear to have been documented in the literature. These factors may account for the correlation between mean glucose and HbA1c being lower than the same correlation reported for adolescents and adults with T1D.
The results of the present study are particularly important for families and providers to consider when aiming to estimate average glucose concentrations and their variance among very young children with T1D. While HbA1c values are a helpful measure to consider when providing care to guide glucose management, this value unfortunately does not afford much information regarding glycemic variability. A GMI estimate, rather, incorporates CGM data in its calculation, and offers an alternative to the HbA1c as a summary metric of longitudinal glycemic control. The CGM data used in the calculation also allows one to calculate multiple additional metrics to characterize the quality of glycemic control. A GMI estimate may be of use to families or providers who wish to monitor the trend in a child’s average glucose concentrations in between clinic visits, or who wish to monitor other clinical indicators that relate to variability in glucose concentrations. Some difference between the GMI and HbA1c values may be expected since the HbA1c laboratory value reflects glucose levels over the previous 2-3 months, while the GMI calculation reflects glucose levels over the prior 14-27 days. Further, the results of the present study show a wide range of HbA1c values for a given mean glucose value, as demonstrated in Figure 1. Even under stable conditions, GMI and HbA1c values may differ due to nonglycemic factors such as generic differences in red blood cell lifespan. In addition, GMI values may be higher or lower than contemporaneous HbA1c values due to changes in treatment regimen or lifestyle, or due to white-coat adherence, which occurs when individuals escalate their self-management behaviors immediately prior to a visit with their healthcare professional. 15
Future research studies may consider alternate ways CGM data can routinely provide estimates of glycemic control in addition to a GMI estimate. For example, Fabris et al. 16 demonstrate the utility of using time in target range to approximate HbA1c in adults with T1D. In addition, estimated HbA1c or GMI values from CGM data generally involve some discrepancy to a laboratory-measured HbA1c value, which can limit clinical use. 17 To address this, a previous study used adult CGM data to derive a personalized kinetic model that takes into account red blood cell turnover, cross-membrane glucose transport, and hemoglobin glycation processes. 18 This new glycemic marker, termed calculated HbA1c (cHbA1c), more accurately reflects a laboratory HbA1c value and may be of use to clinicians who wish to replicate a more exact HbA1c value specific to an individual. 19 The practicality of using a GMI estimate, however, may hold value for clinicians who aim to replicate an HbA1c value that is traceable to an average glucose metric and can be compared to historical databases. Both approaches are valuable, however different, and the utility of a personal model or population-based calculation will likely depend on the clinical or research question.
The results of the present study have further implications for clinical care. Providers are increasingly implementing and testing the efficacy of telehealth video conferencing to provide patient care in pediatric populations.20,21 While these services are particularly useful for families who live far from clinic or who do not have access to reliable transportation, these services do limit the healthcare team’s ability to obtain an in-clinic HbA1c value. Indeed, options exist for families to complete HbA1c kits and return their samples via mail, however, families may not accurately record information for the HbA1c sample (ie, forget to include the collection date and time), they may fail to return the kit via mail, or the kit may be exposed to extreme temperatures or extended travel times that make it an unreliable measure. The results of the present study suggest that a GMI estimate data may provide a helpful alternative to HbA1c for summarizing average daily glucose in a format that is recognizable to clinicians but that minimizes the variance in the relationship between the summary metric and true average daily glucose, especially when an HbA1c value cannot be obtained by the healthcare team.
The GMI value further provides a glycemic metric in units comparable to those of a lab-measured HbA1c, which does not require providers or patients to alter their understanding of glycemic control terms. The GMI offers a useful steppingstone toward utilizing purely CGM metrics for assessing glycemic control and moving away from solely measuring HbA1c. Of note, HbA1c is still a historically important biomarker. Thus, while there may be some effort to migrate away from the use of HbA1c, it is important to have a measure that’s related to HbA1c so that researchers can compare future outcomes to those in the historical research literature. In addition, many young children do not use a CGM device, so continued use of the HbA1c in select populations still allows researchers and clinicians the ability to compare glycemic control across CGM users and CGM non-users.
The present study included a large sample of young children who demonstrated a range of glycemic control. There are, however, a few limitations to note. First, the sample was largely homogeneous with respect to racial/ethnic groups. Previous research suggests that racial differences exist in the relationship between HbA1c and mean glucose concentration.22,23 While the demographics of the present sample are representative of the patients typically seen in the recruiting clinics,9,10 the results may not fully generalize to underrepresented groups at the national level. 24 Second, data from the present study come from children using their personal CGM as part of their standard of clinical care. Future studies may consider utilizing a standard device to control for variations in device use. Lastly, as we previously mentioned, other research groups have demonstrated that the new glycemic marker of cHbA1c is more accurate when compared to a laboratory HbA1c values.18,19 The analyses in the present study did not include person-specific models and future researchers and clinicians may consider using kinetic models if aiming to calculate an individual-specific HbA1c value.
Conclusions
As CGM devices increasingly become part of diabetes care, families and providers may rely more heavily on their data to inform T1D management decisions and to provide more information regarding glycemic control. Young children with T1D may experience more variability in their glucose levels day-to-day than observed in adults, and thus additional days of CGM data may be needed to estimate GMI in this unique population. The results of the present study suggest a benefit of using a young child-specific GMI formula if calculating based on 18 or fewer days of CGM data. In addition, if a study aims to collect an alternative metric to HbA1c using CGM data in young children, then the results of this study suggest a data collection period of at least 18 days is required to calculate a young child’s GMI. Further, most systems currently default to 14 days of CGM data when uploading glucose data. Researchers and clinicians may consider changing the number of days automatically displayed from devices in order to maximize glucose estimates. Lastly, companies responsible for producing data aggregating software may consider altering their software to default to 18 days of CGM data in order to calculate GMI metrics in young children under 6 years.
Footnotes
Acknowledgements
None
Abbreviations
CGM, continuous glucose monitor; eA1c, estimated HbA1c; GMI, glucose management indicator; HbA1c, hemoglobin A1c; MARD, mean absolute relative difference; Mercy on TODP, Children’s Mercy on Type One Diabetes in Pediatrics; T1D, Type 1 diabetes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ADM and SRP have no conflicts of interest to disclose. MAC is the chief medical officer for Glooko, has consulted with Medtronic Diabetes, Eli Lilly, and receives research support from Abbott Diabetes.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
