Abstract
A new insulin patch pump for continuous subcutaneous insulin infusion was developed. The pump is composed of reusable and disposable parts and operates with a stepping motor. This pump was compared to a patch pump and a durable pump regarding basal rate and bolus accuracy. Using a microgravimetric method, boluses of 0.2 U, 1 U and 7 U, and a basal rate of 1 U/h were tested. For all pumps, bolus accuracy was higher when larger volumes were delivered. While median deviations were similar for all pumps, there were differences in the precision of individual boluses and when regarding basal rate delivery divided into 1-h windows.
Introduction
Continuous subcutaneous insulin infusion (CSII) has been commonly used for intensively-managed insulin therapy as an alternative to multiple daily injections. 1 CSII requires an insulin pump that delivers fast-acting insulin according to a preset basal rate and boluses on demand. There is a variety of insulin pumps on the market; however, while there are many different durable pumps, only a few tubeless patch pumps are available.
The newly developed MEDISAFE WITH™ (MW) insulin patch pump (Terumo Corporation, Tokyo, Japan) consists of separate pump and controller units, with the controller and display components integrated in a wireless remote control that allows users to adjust settings for insulin delivery and enter operation commands on an LCD touch panel, so that the pump can be controlled via wireless communication.
All components of the pump, including the pump body, tubing, cannula and cartridge, are combined to a single unit, which can be adhered directly to the user’s skin. This insulin patch pump can be removed before bathing to avoid decreased potency of insulin due to thermal denaturation. The disposable parts of the pump (cannula and cartridge) are consumable supplies while the driving part (pump body) is reusable (Figure 1).
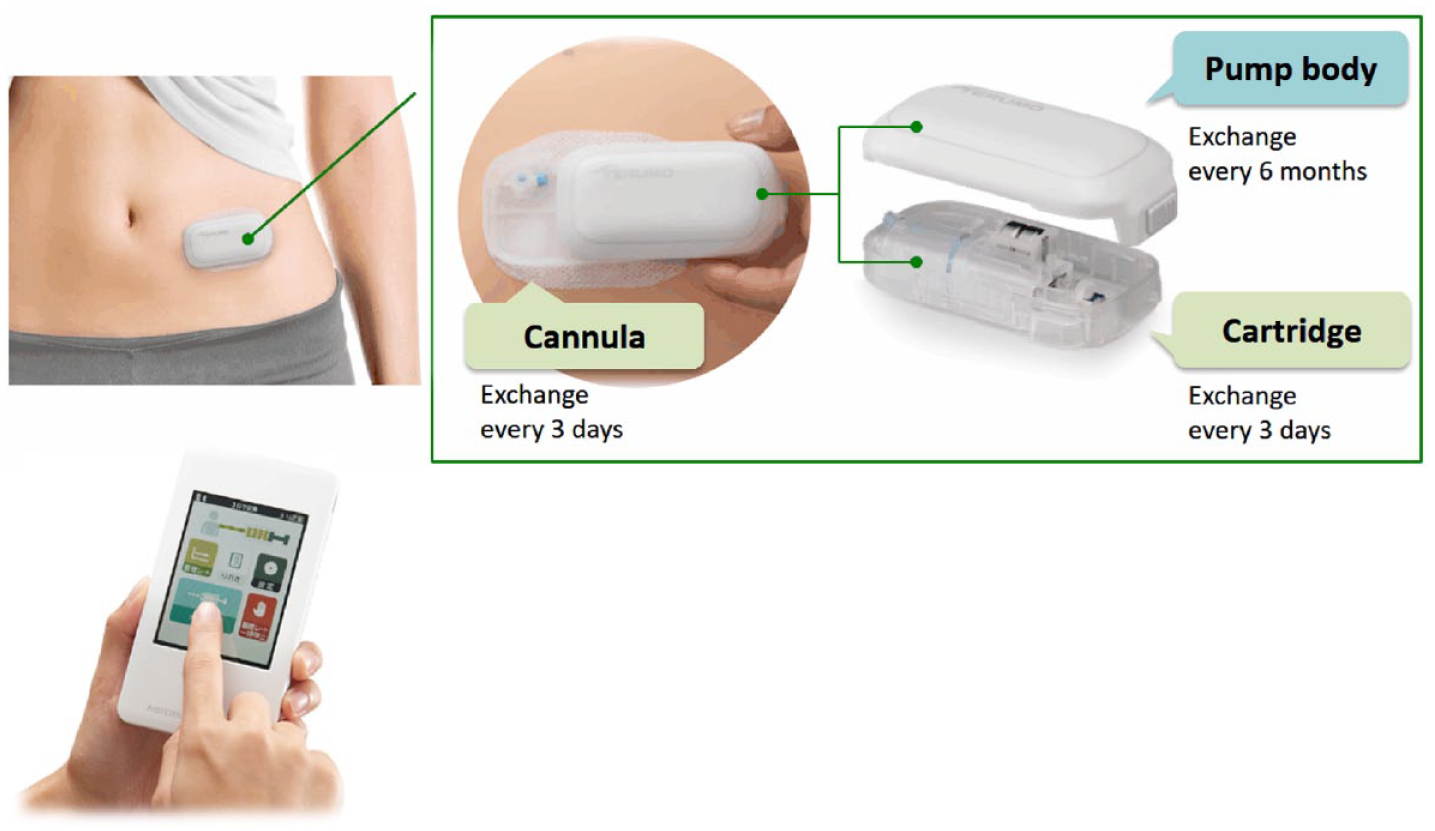
Components of the pump system: pump (above) and controller (below).
The drive mechanism of MW, iCURADRIVE™, uses a small stepping motor. The operating principle and structure of a stepping motor are shown in Figure 2. For simplicity, an example motor in which the rotor is a diametrical two-pole magnet with four electromagnets arranged around it is shown. On insulin delivery, pulsed currents triggered by the remote control are applied to the electromagnets 1 to 4. These currents generate attractive and repulsive forces between the permanent magnet and electromagnets, driving the rotation. In this example, feeding a single pulse current will result in a quarter of a revolution. The motor’s rotation is transmitted to the gears that drive a piston within the cartridge and initiate insulin delivery. The pulse drive method employed in the patch pump prevents the motor from rotating by itself even in the event of hardware failure, such as a failure in the electric circuit. To further ensure safety, the motor’s rotation status is constantly monitored by a rotation sensor installed on a gear directly connected to the motor axis.
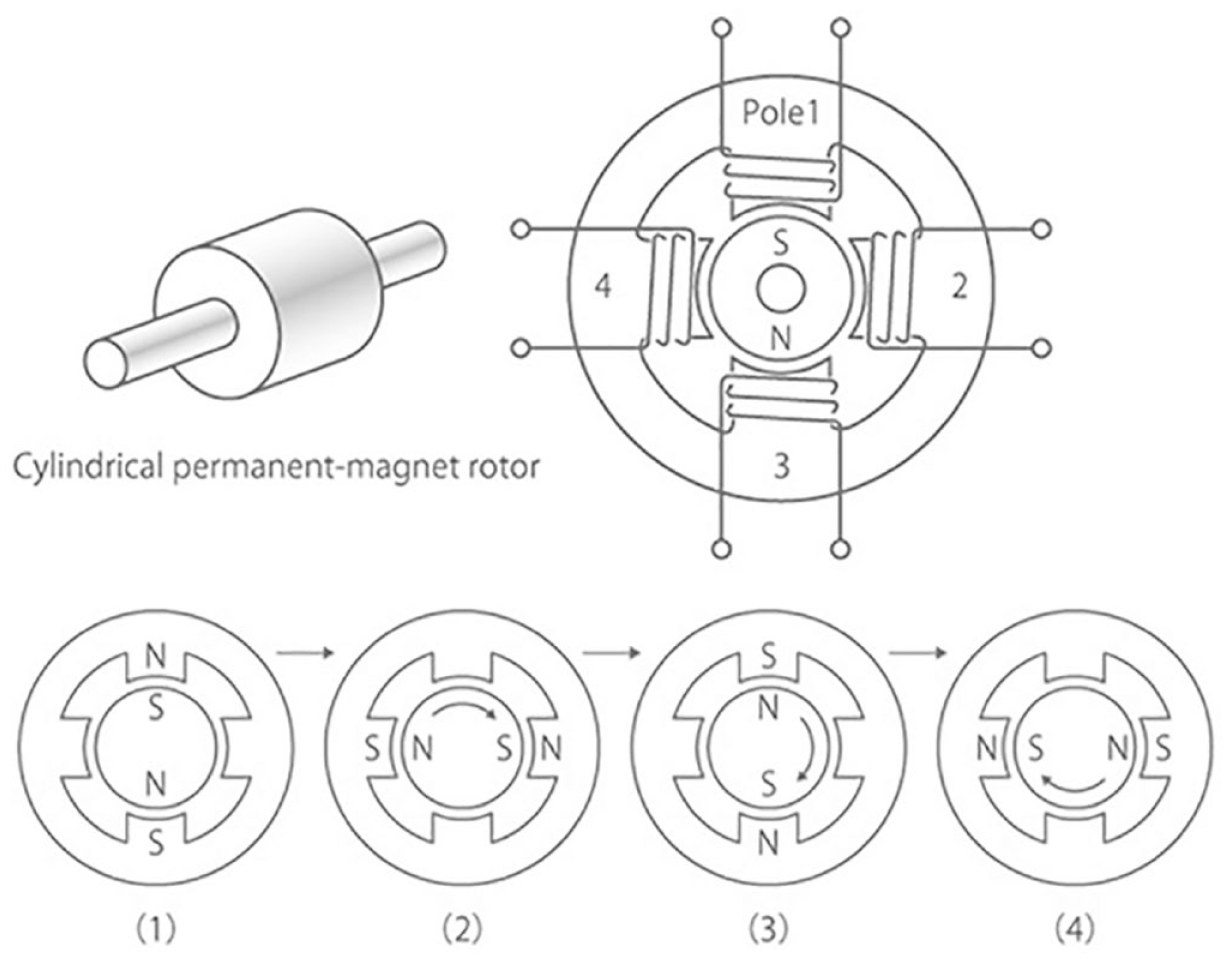
Stepping motor structure and 4-step rotation principle.
MW is already available in Japan and recently received a CE mark.
Patch pumps as well as durable pumps are expected to accurately deliver programmed basal rates and insulin doses to ensure a safe therapy and achieve adequate glycemic control. In this investigation, insulin delivery accuracy of the new patch pump was assessed and compared to another patch pump and a durable pump prior to its global market introduction.
Methods
Accuracy of MW was compared to a patch pump (OmniPod® (OP), Insulet Corporation, Billerica, MA, USA) and a durable pump (MiniMed™ 640G (MM) with MiniMed® Quick-set® infusion set, Medtronic MiniMed, Northridge, CA, USA). All tests were performed in a laboratory setting at the Institut für Diabetes-Technologie, Forschungs- und Entwicklungsgesellschaft mbH an der Universität Ulm. Insulin pumps were not tested in humans; no ethical approval was required. Procedures were based on IEC 60601-2-24 2 with some modifications. Test setups, procedures and data analysis were previously described in detail for patch pumps and durable pumps. 3 In brief, pumps delivered insulin (NovoRapid® Insulin Aspart, Novo Nordisk A/S, Bagsværd, Denmark) into a water-filled vessel which was placed in a balance and weight gain was recorded. Boluses of 0.2 U, 1.0 U and 7.0 U were tested by delivering 25 (0.2 U and 1.0 U) or 24 (7.0 U) boluses per run and recording weight gain after each bolus. From each pump model, three pumps or reusable pump components were used and tested three times using new disposables for each run. In total, nine runs were performed with each pump model. A basal rate of 1.0 U/h was tested for 72 h, recording weight gain every 5 minutes in 9 runs as well. The three pumps were tested in parallel. Deviations from target were determined from weight changes during insulin delivery. For each pump, median and 2.5th to 97.5th percentile from all measurements of all runs were calculated. For the basal rate evaluation, delivery during each 1-h windows (72 per run) was calculated. Deviations less than or equal to ±15% from target delivery were considered acceptable.
Results
Accuracy of bolus delivery was evaluated based on 9 runs with 25/24 consecutive bolus deliveries each resulting in 225 (0.2 U and 1 U) or 216 (7 U) values per pump. For all pumps, accuracy was higher when larger volumes were delivered, that is, the median deviation from target was smaller (Figure 3). Median deviations ranged from +4.7% to +7.0% for the 0.2 U bolus, from +2.1% to +3.2% for the 1 U bolus and from −0.2% to +1.5% for the 7 U bolus without marked differences between the three pumps (Table 1). Precision of delivery of individual boluses was high for MW and MM with at least 97% of boluses within ±15% from the target and lower for OP.
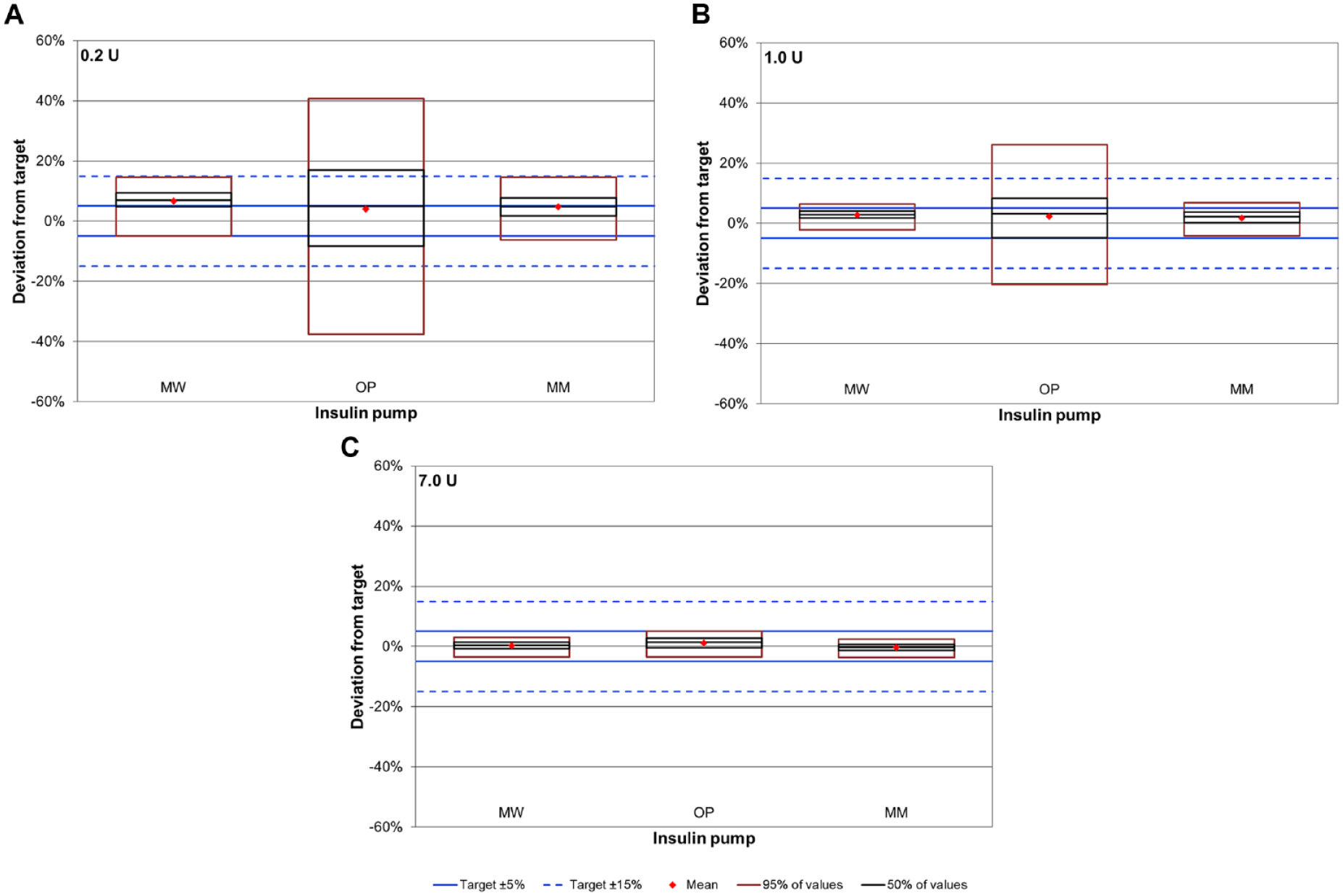
Bolus accuracy for 0.2 U (A), 1.0 U (B) and 7.0 U (C) boluses. Boxes represent 9 runs with 25 (A, B) and 24 boluses (C) each (n = 225 for A and B, n = 216 for C).
Bolus and Basal Rate Accuracy.
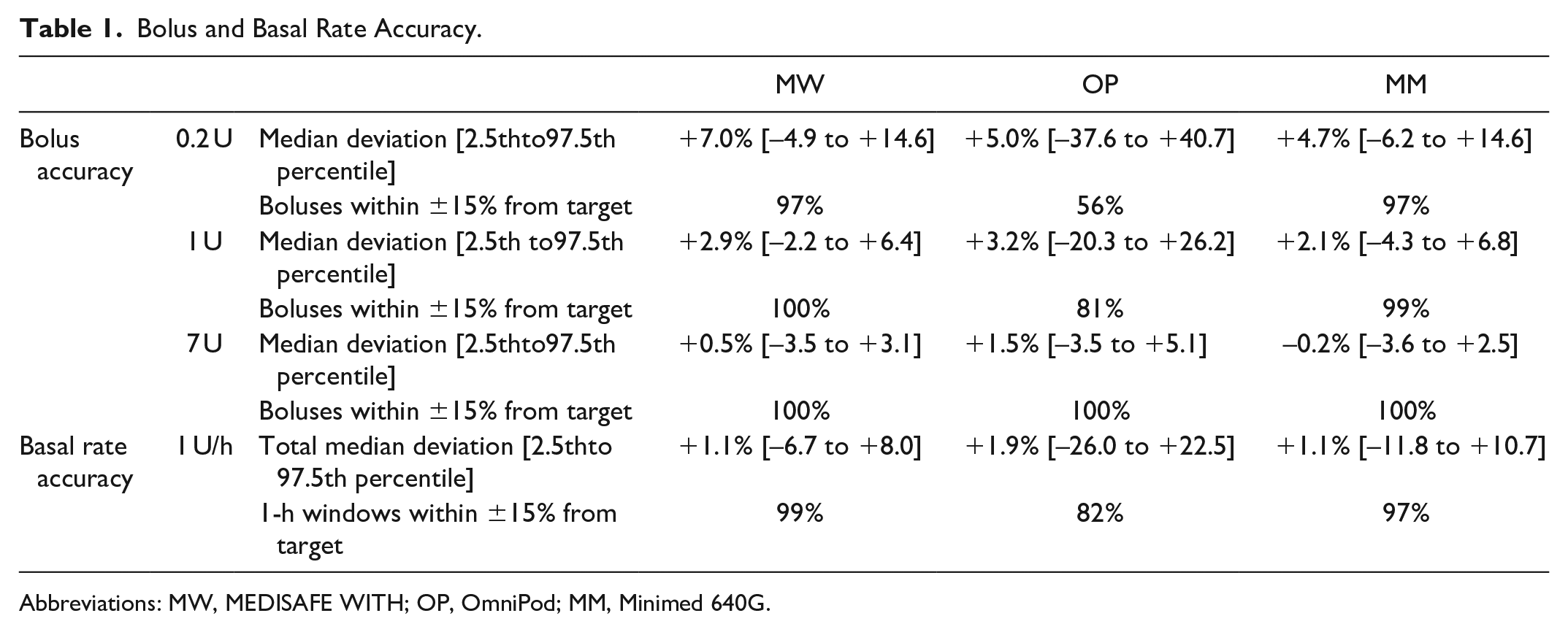
Abbreviations: MW, MEDISAFE WITH; OP, OmniPod; MM, Minimed 640G.
Basal rate delivery is displayed for each 1-h window during the 72-h test period (Figure 4). MW and MM showed a similar delivery pattern with an initial overdelivery during the first approx. 12 hours, followed by a median delivery within ±5% for the remainder of the experiment. By contrast, OP showed larger deviations between individual 1-h-windows throughout the whole observation period. Accordingly, OP had 82% of 1-h windows within ±15% from target compared to 99% and 97% for MW and MM, respectively. Total deviation after 72 hours was comparable between the three pumps.
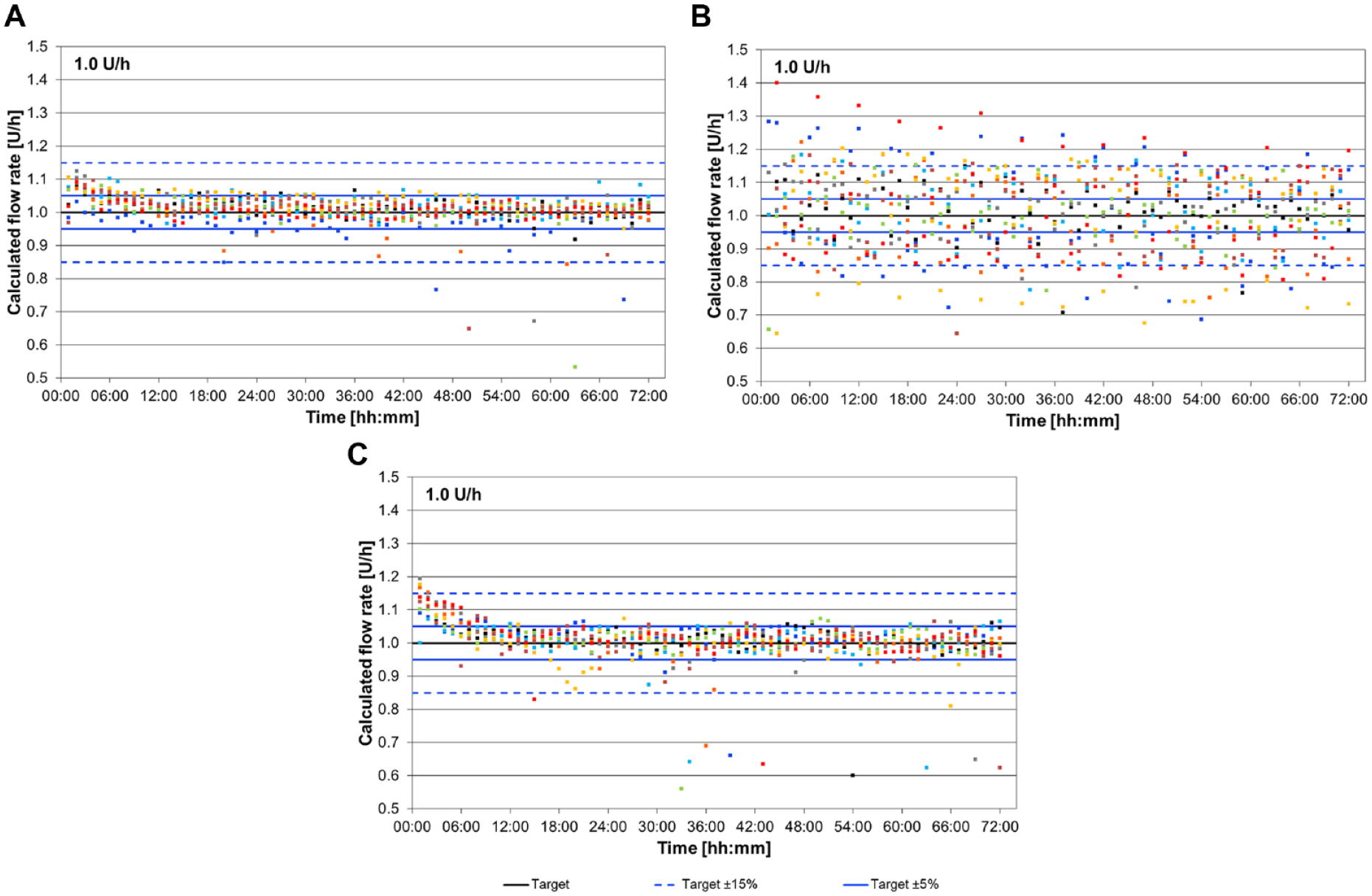
Basal rate delivery calculated for 1-h windows of 9 runs (colored dots) during 72 hours delivery for MW (A), OP (B) and MM (C).
Conclusions
MW is a novel insulin pump for patients that prefer a tubeless but also detachable insulin pump. This accuracy evaluation showed a high level of accuracy regarding the tested basal rate and boluses. Deviations from the target were similar to those observed for the durable pump and tended to be lower compared to the other patch pump. While the other two pumps were already tested before,4,5 these are the first results presented for MW. Because MW will also be integrated into an automated insulin delivery system, 6 reliability of insulin delivery, particularly considering small doses, is important to ensure a safe and effective insulin therapy.
Footnotes
Acknowledgements
The authors would like to thank Delia Waldenmaier (IfDT) for medical writing support, Stefan Pleus (IfDT) for his scientific input, and Martina Tesar and Natalie Neuburger (IfDT) for performance of the measurements.
Abbreviations
CSII, continuous subcutaneous insulin infusion; MM, Minimed 640G; MW, MEDISAFE WITH;OP, OmniPod.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: GF is general manager and medical director of the Institut für Diabetes-Technologie Forschungs- und Entwicklungsgesellschaft mbH an der Universität Ulm, Ulm, Germany (IfDT), which carries out clinical studies on the evaluation of BG meters, with CGM systems and medical devices for diabetes therapy on its own initiative and on behalf of various companies. GF/IfDT have received speakers’ honoraria or consulting fees from Abbott, Ascensia, Dexcom, i-SENS, LifeScan, Lilly, Menarini Diagnostics, Metronom Health, Novo Nordisk, PharmaSense, Roche, Sanofi, Sensile and Ypsomed.
BA, JM and CH are employees of the IfDT. JU is employee of Terumo Corporation, Tokyo, Japan.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Performance of the study and writing of the manuscript were funded by Terumo Europe NV.
