Abstract
Maintaining blood glucose levels in the target range during exercise can be onerous for people with type 1 diabetes (T1D). Using evidence-based research and consensus guidelines, we developed an exercise advisor app to reduce some of the burden associated with diabetes management during exercise. The app will guide the user on carbohydrate feeding strategies and insulin management strategies before, during, and after exercise and provide targeted and individualized recommendations. As a basis for the recommendations, the decision trees for the app use various factors including the type of insulin regimen, time of activity, previous insulin boluses, and current glucose level. The app is designed to meet the various needs of people with T1D for different activities to promote safe exercise practices.
Background
Exercise is an integral part of a healthy lifestyle for people with type 1 diabetes (T1D). Regular exercise helps in achieving better glycemic control and decreases the risk of cardiovascular diseases.1-4 Further, it enhances insulin sensitivity and reduces total daily insulin needs.5,6 Despite the benefits of regular physical activity for people with T1D,5,7,8 health-care professionals believe that the lack of formal education on exercise-related metabolism and limited time are common barriers to prescribing guidance around exercise management. 9 Although guidelines for exercise management have been published, they are generally not readily accessible to clinicians or people living with T1D.5,6,10,11 With low activity levels 12 and an increased prevalence of obesity in this population, 13 evidence-based exercise strategies5,6 need to be easily accessible to people with T1D in their everyday lives. A recent study surveying 1542 adults with T1D found that glucose management during exercise was the most difficult task, with 85% of the people desiring application (app)-based technology to help manage glucose levels during this time. 14 The potential value of digital apps for diabetes self-management has recently been highlighted by consensus reports from the joint European Association for the Study of Diabetes (EASD) and the American Diabetes Association (ADA) Diabetes Technology Working Group.15,16 Despite surges in the use of technology by people with T1D, and their potential to improve HbA1c levels, 17 there are still significant gaps between research and the technology in mobile health for T1D management. 18
Here we describe an app, based on the consensus guidelines for exercise management in T1D, 6 which aims to provide personalized guidance on insulin dosing and carbohydrate intake strategies for facilitating glucose control around exercise.
Development of the Exercise Advisor App: Decision Tree Structure
In the software development process, the decision tree from published international consensus guidelines 6 was deconstructed and expanded from a total of eight branches to three separate decision trees for aerobic, mixed, and anaerobic exercise with 11, 11, and four pathways, respectively (Supplemental Figure 1). Additional decision-making around insulin management and/or carbohydrate intake is based on user-input glycemic measures at −90, −60, −15 minutes and the users’ perceptions of the exercise intensity.
Aerobic Exercise
Whether exercise is planned or spontaneous, the initial recommendation for aerobic exercise (Supplemental Figure 1A) is based on whether a bolus insulin dose has been taken or will be taken within three hours of exercise. If so, a bolus reduction is suggested. The degree of reduction depends on the activity type and time between the meal and exercise. Percent basal rate reductions and carbohydrate feeding strategies are outlined depending on the insulin regimen, time of activity, previous boluses, and current glucose level. Starting glucose levels at exercise onset are an important determinant of the suggested guidelines, 19 for example, if a user has hyperglycemia prior to exercise, a bolus reduction or basal rate reduction may not be necessary.
Anaerobic Exercise
For anaerobic exercise (Supplemental Figure 1B), the guidelines are determined by the current glucose level and if the user is on multiple daily injections (MDIs) or continuous subcutaneous insulin infusion (CSII) therapy. Intense anaerobic exercise may result in hyperglycemia,20,21 and therefore, insulin dose reductions are not recommended; however, those on CSII have the ability to increase their basal rate temporarily (by ~20%) in situations of elevated glycemia pre-exercise. Carbohydrate supplementation is only promoted if the glucose level is below the target range (<90 mg/dL).
Mixed Exercise
Mixed exercise (Supplemental Figure 1C) is a combination of the aerobic and anaerobic exercise decision trees, although the order of exercise may be altered. For example, the anaerobic exercise would be prompted first if the user’s glucose level is at or below the target range, but if the glucose level is above the target range, then aerobic activities are prompted first. 22
All three types of activities determine the post-exercise recommendation based on glucose readings.
Uses of the App
The app may allow insulin users to better self-manage their glycemia for different forms of exercise.
The app provides insulin dosing and weight-based carbohydrate supplementation recommendations, as well as preemptive bedtime guidance to help prevent nocturnal hypoglycemia post-exercise.
Design and Functioning of Exercise Advisor App
During the user setup, the type of insulin regimen (CSII or MDI) and type of glucose management (continuous glucose monitoring [CGM] or blood glucose monitoring) are initially identified during the user “onboarding” process. This process automatically adjusts the decision tree algorithm. After setting exercise start and end time, activities are selected from a list provided, already categorized internally as aerobic, anaerobic, or mixed; however, users can change this category for some of the activities by selecting from the terms “steady”, “sprint”, or “mixed”. Exercise prompts prior to anticipated start time are initially based on whether the exercise will occur within 60 minutes of the current time or later. Planning more than 60 minutes ahead (ideally 90 minutes) allows for more advanced planning strategies for basal (and potentially bolus) insulin delivery such that reductions in circulating insulin levels are achievable for aerobic exercise. 23 However, if exercise is within 60 minutes, different strategies are initiated, primarily focusing on carbohydrate feeding. Alternatively, if the user wants to avoid carbohydrate feeding, the app can also promote activities that tend not to promote a large drop in glycemia. If scheduling allows, pre-exercise prompts alert users at specific times (ie, meal before exercise, T–90 minutes, T–60 minutes, T–15 minutes) to adjust the recommendations according to their glucose level and estimated active/on-board insulin. The app sets a recommended target blood glucose level for each activity type, visually showing the user whether they are within, below, or above their target glucose level; basic insulin and/or starting point strategies; and a summary learnings screen (Figure 1A-1D). For example, during the 15-minutes pre-activity glucose check-in, if the blood glucose level is between 151 and 250 mg/dL, the app will alert the user by displaying the message “consider aerobic warmup.”
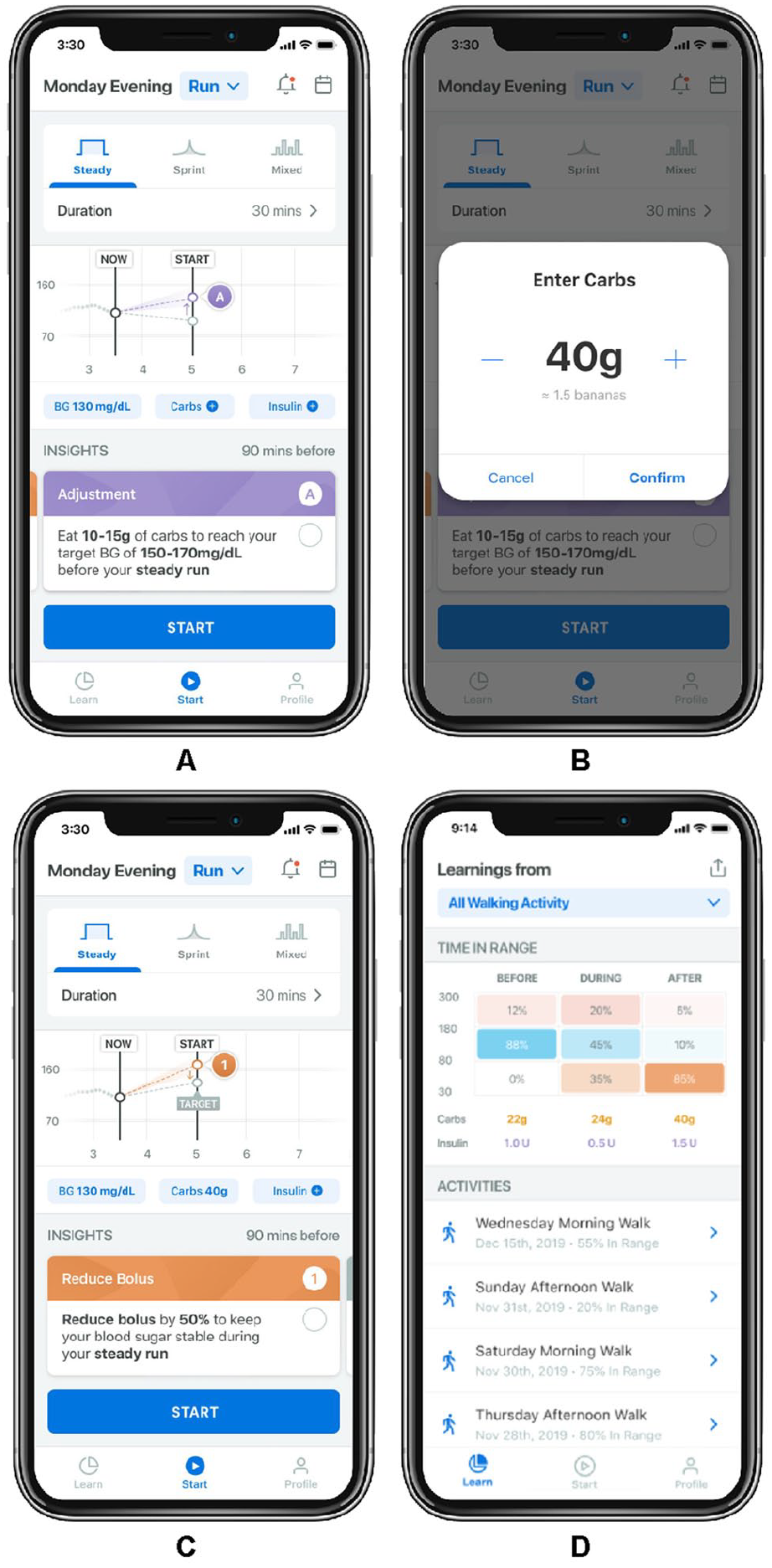
Example screenshots of the prototype exercise application. (A) Example: pre-exercise glucose target and carbohydrate intake recommendation 90 minutes before exercise for an individual below target glucose level prior to a steady state run. (B) Example: carbohydrate intake manual data capture screen. (C) Example: meal bolus reduction prior to a steady state run. (D) Example: learning screen.
The main screen of the app shows the timing of the expected activity, current, and target glucose levels, active carbohydrates, as well as insights of what measures should be taken for safe exercise to occur. If these suggestions are not confirmed in the app, the recommendations are changed. Furthermore, depending on the pre-exercise glucose level, the app automatically recommends several activities that generally minimize the need for carbohydrate snacking if blood glucose is in the target range (90-124 mg/dL) or below target (<90 mg/dL). Once the activity commences, a timer starts, allowing the activity to be stopped or extended, if required. After the exercise, personal notes can be recorded to indicate how the user felt about their workout and glycemia (ie, satisfied, dissatisfied). Prompts occur 15 minutes after exercise to provide the user with strategies to support glycemic control in recovery. All logged exercise events are recorded and can be reviewed later. Future app iterations may incorporate advanced onboarding features such as individualized insulin settings (e.g., basal rates, insulin to carbohydrate ratio, etc.), integrated CGM, wearable technologies for exercise intensities, and perhaps machine learning technology.
Summary
This prototype personalized exercise advisor app is designed to meet the various needs of people with T1D and addresses an important therapeutic need for the facilitation of exercise. It has been created for people with T1D and requires multiple user inputs; therefore, it might not be an ideal user experience for everyone with diabetes. However, we believe that it is the first step toward better understanding the real-world challenges of managing insulin administration and carbohydrate intake for exercise. With this knowledge and information, there will be opportunities in the future to create more automated activity guidance for everyone with diabetes.
Supplemental Material
sj-png-1-dst-10.1177_1932296820979811 – Supplemental material for The Development of an Exercise Advisor App for Type 1 Diabetes: Digitization Facilitates More Individualized Guidance
Supplemental material, sj-png-1-dst-10.1177_1932296820979811 for The Development of an Exercise Advisor App for Type 1 Diabetes: Digitization Facilitates More Individualized Guidance by Sarah M. McGaugh, Stephanie Edwards, Howard Wolpert, Dessi P. Zaharieva, Nany Gulati and Michael C. Riddell in Journal of Diabetes Science and Technology
Supplemental Material
sj-png-2-dst-10.1177_1932296820979811 – Supplemental material for The Development of an Exercise Advisor App for Type 1 Diabetes: Digitization Facilitates More Individualized Guidance
Supplemental material, sj-png-2-dst-10.1177_1932296820979811 for The Development of an Exercise Advisor App for Type 1 Diabetes: Digitization Facilitates More Individualized Guidance by Sarah M. McGaugh, Stephanie Edwards, Howard Wolpert, Dessi P. Zaharieva, Nany Gulati and Michael C. Riddell in Journal of Diabetes Science and Technology
Supplemental Material
sj-png-3-dst-10.1177_1932296820979811 – Supplemental material for The Development of an Exercise Advisor App for Type 1 Diabetes: Digitization Facilitates More Individualized Guidance
Supplemental material, sj-png-3-dst-10.1177_1932296820979811 for The Development of an Exercise Advisor App for Type 1 Diabetes: Digitization Facilitates More Individualized Guidance by Sarah M. McGaugh, Stephanie Edwards, Howard Wolpert, Dessi P. Zaharieva, Nany Gulati and Michael C. Riddell in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors would like to thank Healthmade for their design ideas for the current version of the smart phone application.
Abbreviations
CGM, continuous glucose monitoring; CSII, continuous subcutaneous insulin infusion; MDI, multiple daily injection; T1D, type 1 diabetes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SMM has no conflict of interest. HW and SSE are employees and stockholders of Eli Lilly and Company. DPZ has received speaking honoraria from Medtronic, Ascensia Diabetes Care, and Insulet. NG is an employee of Eli Lilly Services India Pvt. Ltd. MCR has received speakers’ fees from Eli Lilly, Novo Nordisk, Medtronic, Dexcom, and Insulet.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Lilly Innovation.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
